Abstract
The interrelationship between endothelial function and arterial stiffness may be different for central and peripheral arteries due to their structural and functional differences. The study aims to assess the interrelationship between central and peripheral vascular function and haemodynamics in metabolic syndrome. Thirty-seven patients [63.0 (57.5–66.0) years, 68.4% males] of metabolic syndrome (National Cholesterol Education Program – Adult Treatment Panel-III criteria) were studied. Carotid-femoral, carotid-radial pulse wave velocity and augmentation index (AIx@75) were assessed using applanation tonometry. Endothelial function was evaluated by brachial flow-mediated dilation using B-mode ultrasonography. Central and peripheral pressures were measured by radial tonometry and sphygmomanometer, respectively. Carotid-radial pulse wave velocity correlated significantly with peripheral diastolic blood pressure (r = 0.33, p = 0.04) and inversely with flow-mediated dilation (r = –0.61, p = 0.0001). AIx@75 correlated significantly with carotid-femoral pulse wave velocity (r = 0.35, p = 0.03) and with aortic pulse pressure (r = 0.43, p = 0.01). In principal component analysis, an inverse relationship was observed between flow-mediated dilation and carotid-radial pulse wave velocity but not with carotid-femoral pulse wave velocity. Regional arterial stiffness assessed by pulse wave velocity in central-elastic and peripheral-muscular arteries differentially relates to endothelial dysfunction. The central arteries might be predominantly influenced by endothelial dysfunction-induced structural changes, while the peripheral arteries are majorly affected by functional alterations.
Keywords
Introduction
Vascular stiffening is an important risk factor for development of cardiovascular diseases (CVDs). In particular, increased arterial stiffness of the central-elastic vessels would result in an increased cardiac workload and decreased coronary perfusion. Additionally, central arterial stiffness is known to increase with ageing and as a consequence of insulin resistance, diabetes, hypertension and atherosclerosis.1,2 This increase in vascular stiffness has been attributed to structural alterations in the elastin–collagen ratio leading to stiffening of the extracellular matrix, medial calcification and collagen linkage to advanced-glycation end products.3,4 In addition to these structural modifications, arterial stiffness can also be increased by a functional increase in vascular smooth muscle tone, which can be influenced by a number of vasoactive mediators released by the endothelium, for example, endothelin-1 (ET-1), nitric oxide (NO) and endothelium-derived hyperpolarizing factor (EDHF). 5
The sub-clinical event preceding CVD is endothelial dysfunction, which is closely associated with arterial stiffness. Endothelial dysfunction plays an important role in the development of atherosclerosis, 6 which can in turn lead to changes in the vessel intima and an increase in arterial stiffness.7,8 Additionally, endothelial dysfunction in the presence of hyperglycaemia can lead to vascular calcification,9,10 which would further result in an increase in arterial stiffness. Moreover, an increase in arterial stiffness can alter endothelial function and possibly act as a mechanism for endothelial dysfunction due to ageing. 11 Increased arterial stiffness can also lead to higher central systolic and pulse pressures (PPs), resulting in an increased circumferential wall stress, which can cause breakdown of vessel wall elastin and endothelial dysfunction progressing to atherosclerosis. A mutually interdependent and intricate relationship exists between the endothelial dysfunction and arterial stiffness.
While stiffness in the peripheral-muscular arteries is of potential clinical importance, its role in prediction of CVD is not well established. Peripheral arterial stiffness may be specifically associated with peripheral vascular disease, which is linked to hypertension and diabetes, both of which are risk factors for CVD. Different arterial segments may be differentially influenced by the structural and functional changes involved in the development of arterial stiffness due to the inherent differences in the vessel wall constitution. Due to a higher smooth muscle content, ‘stiffening’ of the peripheral arteries could be predominantly functional because of an increase in the vascular smooth muscle tone. It is therefore possible that peripheral and central arteries may associate differentially with endothelial function.
Regional arterial stiffness can be assessed by measuring the pulse wave velocity (PWV) across different segments of the arterial tree. The carotid to femoral PWV is indicative of the stiffness in the central-elastic artery, while the carotid to radial PWV can provide information about the peripheral-muscular arteries. Additionally, the central aortic pressure waveform can be derived from the radial artery pressure wave using a transfer function analysis. Augmentation index estimated from this aortic pressure wave reflects the arterial properties of the central-elastic as well as the peripheral-muscular arteries via changes in PWV. Ultrasound-based assessment of brachial artery diameter before and after reactive hyperaemia has been used for estimation of ‘recruitable’ endothelial function. Percentage flow-mediated dilation (FMD) measured by this method has been shown to correlate with coronary endothelial function. 12
Metabolic syndrome is a complex web of interrelated risk factors which includes dysglycaemia, raised blood pressure, elevated triglyceride levels, reduced high-density lipoprotein (HDL) and obesity (particularly central adiposity). 13
Interestingly, each component of metabolic syndrome (increased blood pressure, 14 elevated blood sugar, 15 dyslipidemia 16 and obesity 17 ) can independently cause endothelial dysfunction. Additionally, each of these components can increase arterial stiffness either directly (increased blood pressure, 18 increased blood glucose 19 and obesity20–22) or indirectly via endothelial dysfunction. 18 Metabolic syndrome, hence, is a conglomeration of structural and functional damage to the vasculature, and interrelationship between these factors is critical in understanding the pathophysiology of CVDs. Additionally, the alteration in the central and peripheral pressures as a result of arterial stiffness and endothelial dysfunction may also differentially influence the interrelationship between these parameters in the central and peripheral vessels.
This study aims to assess the association between haemodynamic parameters, endothelial function and arterial stiffness in the central and peripheral vessels in patients with metabolic syndrome.
Materials and methods
Thirty-seven patients of metabolic syndrome [63.0 (57.5–66.0) years, 26 males] were recruited for the study based on the National Cholesterol Education Program – Adult Treatment Panel-III (NCEP ATP III) criteria given in 2009 by the Joint Scientific Statement Clinical criteria. 13 Written informed consent was obtained from all patients before starting the study, which was conducted in the department of physiology at All India Institute of Medical Sciences, New Delhi, India. The institute ethics committee for research on human subjects approved the study.
Adult patients with presence of three or more of the following components: elevated blood pressure, systolic ⩾130 mmHg or diastolic ⩾85 mmHg, fasting blood glucose ⩾100 mg/dL, triglycerides ⩾150 mg/dL, HDL ⩽40 mg/dL for males and ⩽50 mg/dL for females and waist circumference (for Asian population) of ⩾90 cm for males and ⩾80 cm for females or on medication(s) for any of these components were considered as metabolic syndrome as per the NCEP ATP III criteria 13 and included in the study. Patients with any recent infection and co-existing renal disease, peripheral vascular disease, autoimmune vasculitis and collagen vascular disease were excluded from the study. Patients with history of smoking but no clinical signs or symptoms of peripheral vascular disease were not excluded from the study.
The PWV was used to assess the arterial stiffness at both the central and peripheral arteries for estimating the structural changes in arterial wall properties. Additionally, the arterial stiffness along with the changes in wave reflection at the aorta was measured using augmentation index.
Vascular endothelial function was evaluated by measuring the endothelium-dependent flow-mediated vasodilation.
Peripheral haemodynamic variables were measured by two consecutive measurements of brachial blood pressure by manual sphygmomanometry, while central haemodynamic parameters were derived from the tonometrically recorded radial pressure waveform by a validated transfer function analysis 23 (SphgmoCor ®).
The PWV was assessed at two different arterial sites – carotid to radial for peripheral arterial stiffness and carotid to femoral for measuring central arterial stiffness. An applanation tonometer (SphgmoCor® CVMS CPVH System, Atcor Medical, Australia) was used along with simultaneous recording of Lead II ECG to allow for R-wave-gated reference to time the arrival of the pressure pulse. The waveform was recorded sequentially till a reproducible signal was obtained from the carotid and the radial sites for measuring the carotid-radial pulse wave velocity (crPWV) and from carotid and femoral sites for measuring the carotid-femoral pulse wave velocity (cfPWV). The distance was measured from the sternal notch to the best palpable point for the arteries. The arm was abducted at an angle of 90° and parallel to the floor while measuring the radial artery distance. The distance to the femoral artery was measured by first measuring the distance from the suprasternal notch to the umbilicus and then from the umbilicus to the best palpable point of the femoral artery pulsation. Automated calculation of the PWV was performed by the software using the time elapsed between the onset of the wave at the proximal (carotid) site and the distal (radial/femoral) site.
Aortic augmentation Index (AIx@75) was measured by placing the same tonometer probe at the radial artery, and validated transfer function generated the central aortic waveform from the acquired radial artery tonometric signal. The amount of augmentation of the aortic pressure waveform corrected for heart rate was calculated and expressed as AIx@75.
FMD was used to assess endothelial function using brachial artery ultrasonography. Longitudinal ultrasound images of the brachial artery were captured in B-mode using Vivid-e (GE Healthcare). A 12-MHz probe was used to visualize the artery with the frame rate set at 47. Simultaneous recording of Lead II ECG was done to correlate brachial artery diameter with end-diastolic phase of the cardiac cycle. A cuff was wrapped around the forearm, and baseline brachial artery images were obtained 2–3 cm above the antecubital fossa. This was followed by a 5-min period of occlusion during which the cuff was inflated to a pressure of 50 mmHg above the systolic blood pressure of the patient. The brachial artery images were captured at every 10 s after release of the occlusion for a period of 3 min. Electronic calipers were used to manually measure the diameter at three points on each image from the intima-to-intima, which was then averaged to obtain the diameter for each image. The maximum increase in the brachial artery diameter after release of occlusion was used to calculate the percentage FMD.
Statistical analysis
Normally distributed data are expressed as mean ± standard deviation, while non-Gaussian data are expressed as median with inter-quartile range. Correlation between continuous variables was tested with Pearson’s test for normally distributed data and with Spearman’s test for non-parametric data. Multiple regression analysis was done to identify the independent predictors of vascular parameters. Principal component analysis (PCA) was done to further identify unifying factors influencing the data. All analysis was done using R version 3.2.3.
Results
The demographic characteristics of the patients and descriptive statistics of the various study parameters are mentioned in Tables 1 and 2, respectively. The cfPWV could not be assessed in six patients due to presence of high central adiposity. FMD could not be assessed in four patients due to inadequate image quality.
Patient demographic and anthropometric characteristics.
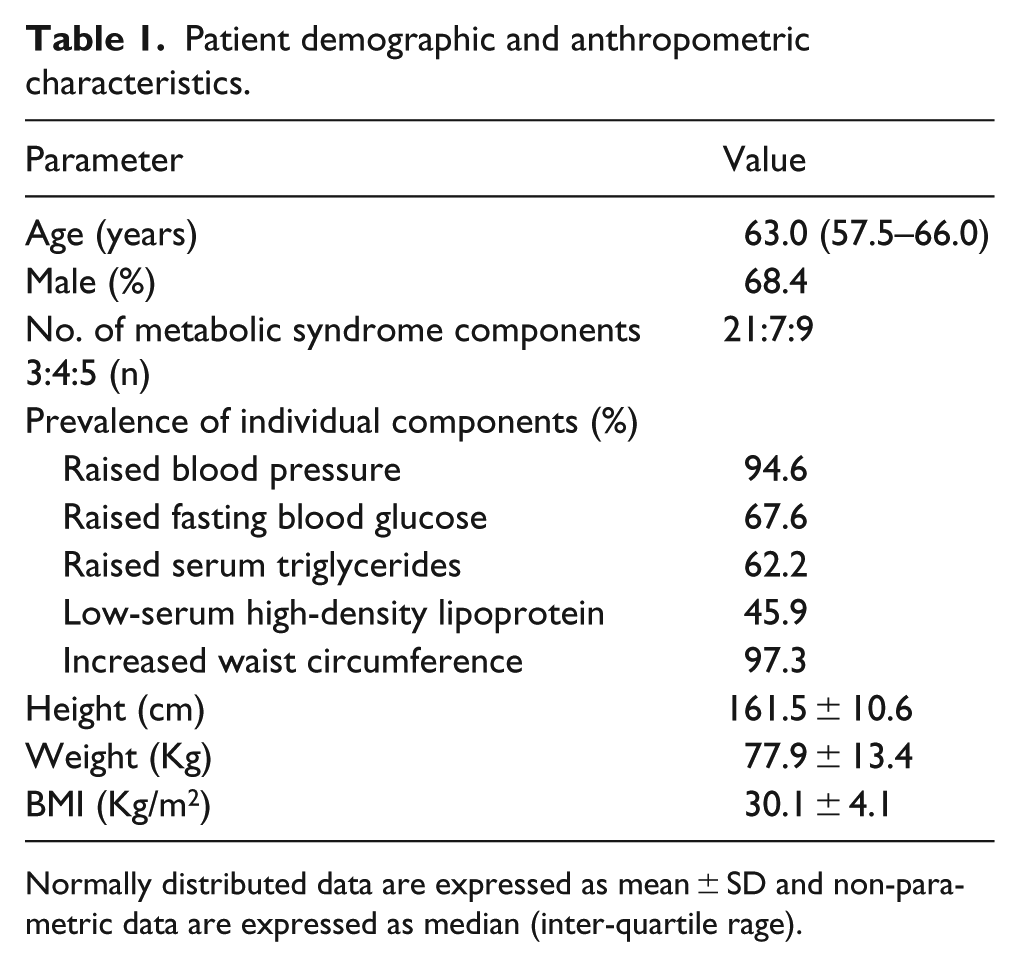
Normally distributed data are expressed as mean ± SD and non-parametric data are expressed as median (inter-quartile rage).
Patient haemodynamic and vascular parameters.
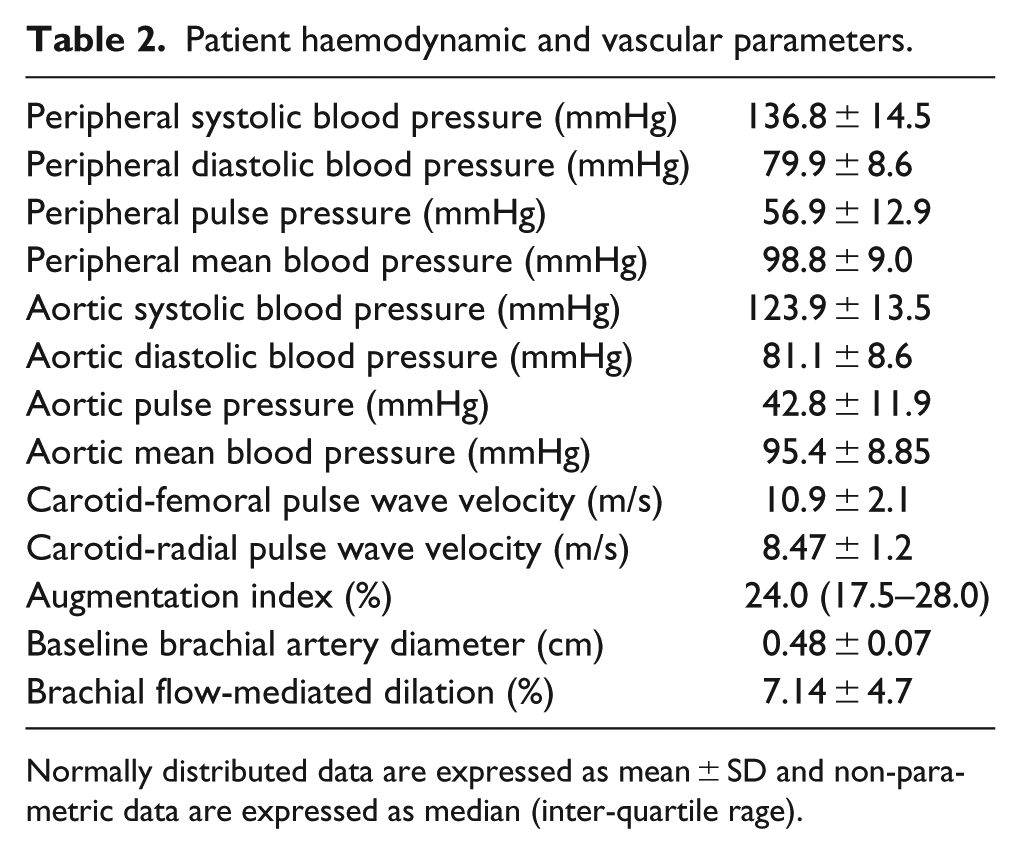
Normally distributed data are expressed as mean ± SD and non-parametric data are expressed as median (inter-quartile rage).
Correlations between vascular and demographic and anthropometric parameters
A significant positive correlation was observed between crPWV and weight (r = 0.32, p = 0.04). Both AIx@75 and cfPWV showed a significant negative correlation with height (r = −0.38, p = 0.01 and r = –0.39, p = 0.01, respectively). AIx@75 also showed a significant negative correlation with weight and waist circumference (r = −0.54, p = 0.005 and r = –0.48, p = 0.002, respectively). Additionally, AIx@75 was significantly correlated with age (r = 0.45, p = 0.005), while cfPWV showed a non-significant trend towards correlation with age (r = 0.3, p = 0.07). FMD also showed a significant negative correlation with weight and height (r = –0.35, p = 0.03 and r = –0.49, p = 0.002). None of the vascular parameters correlated with body mass index (BMI).
Correlations among vascular parameters
A strong significant negative correlation was observed between crPWV and FMD (r = –0.61, p = 0.0001). However, cfPWV did not show any correlation with FMD (Figure 1). AIx@75 correlated significantly with cfPWV (r = 0.35, p = 0.03) but not with crPWV or FMD.
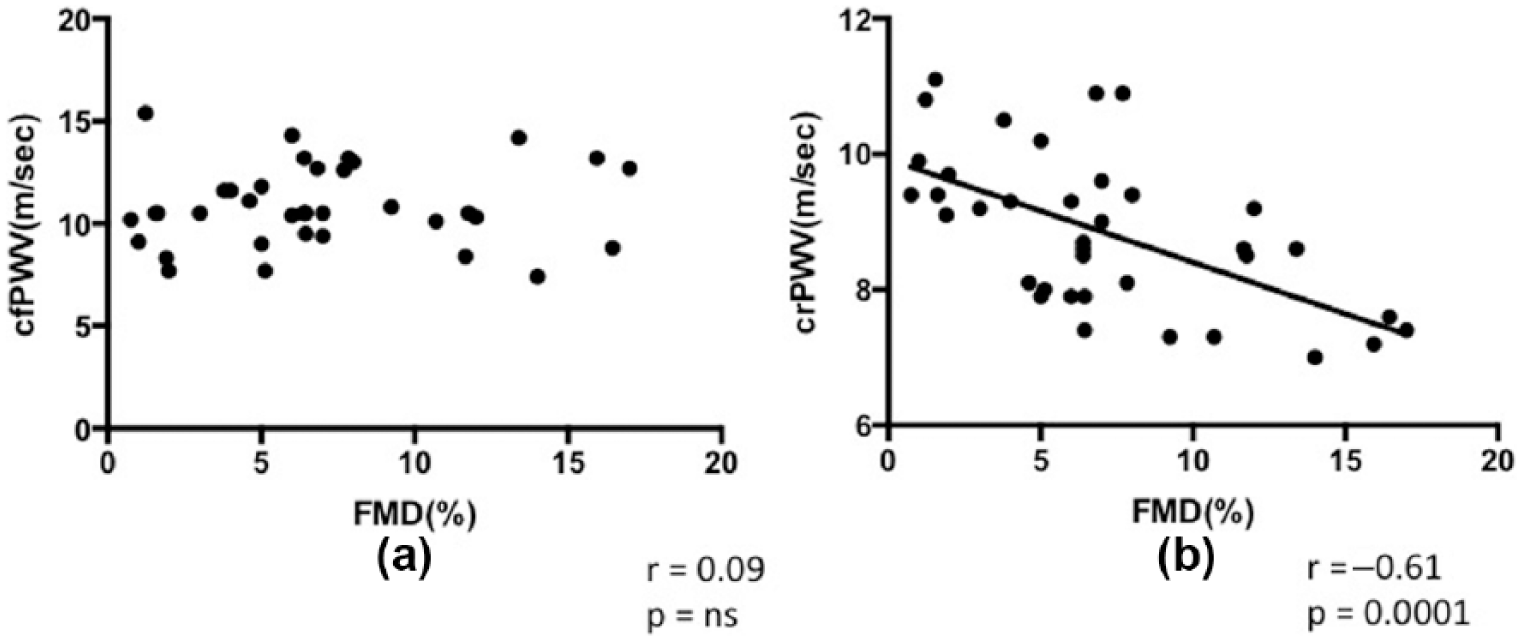
(a) Lack of correlation between carotid-femoral pulse wave velocity (cfPWV) and flow mediated-dilation (FMD). (b) Significant correlation between carotid-radial pulse wave velocity (crPWV) and FMD.
Correlations between vascular and haemodynamic parameters
crPWV correlated significantly with peripheral diastolic blood pressure (DBP; r = 0.33, p = 0.04; Figure 2). Additionally, there was a weak non-significant correlation with aortic DBP (r = 0.31, p = 0.06). No correlations were seen between the cfPWV and central or peripheral blood pressure. AIx@75 correlated significantly with aortic PP (r = 0.43, p = 0.01) but not with any other blood pressure parameters. FMD did not correlate with any central or peripheral blood pressure parameter.
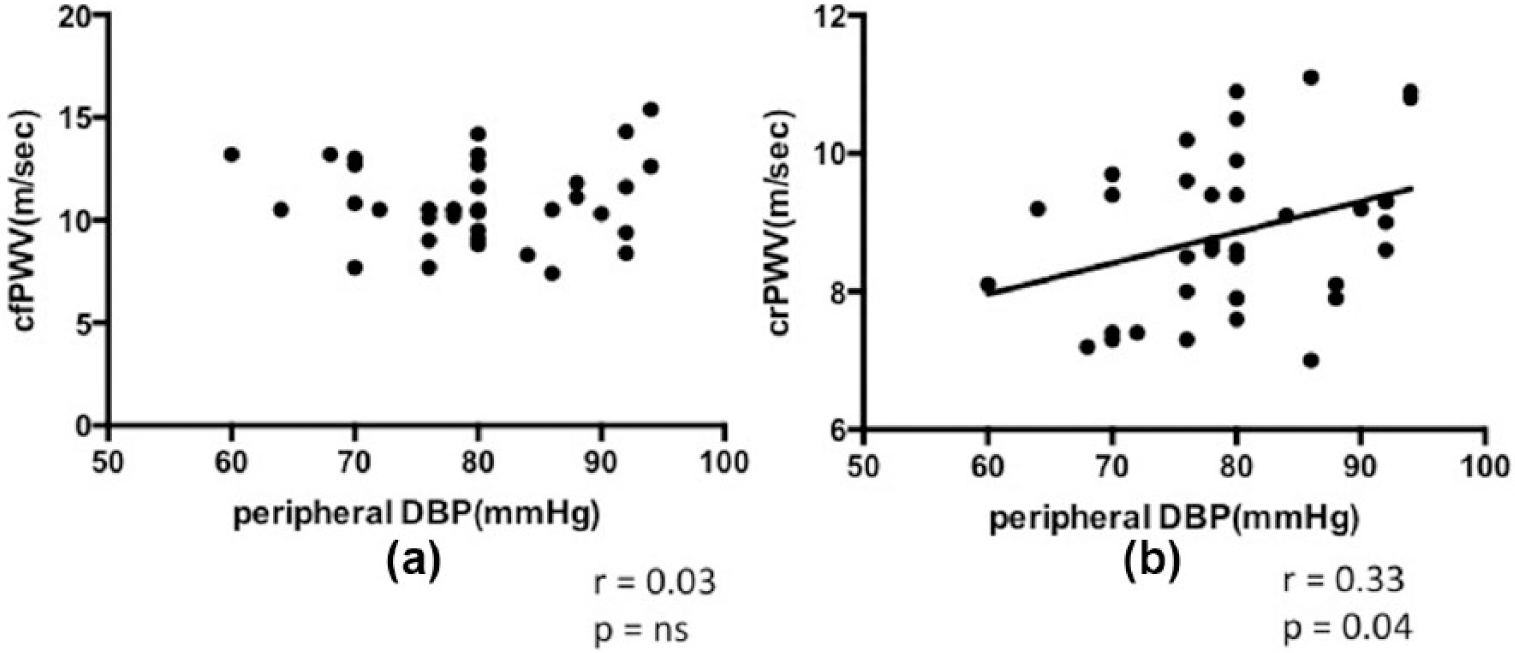
(a) Lack of correlation between carotid-femoral pulse wave velocity (cfPWV) and diastolic blood pressure (DBP). (b) Significant correlation between carotid-radial pulse wave velocity (crPWV) and DBP.
Based on the correlations obtained, multiple regression analysis was performed for crPWV to identify the variables that have an independent influence on the peripheral arterial stiffness. Peripheral DBP, FMD and weight were used in the model. Brachial artery FMD emerged as the only significant predictor of crPWV (p = 0.0005). Additionally, the model accounted for 41.4% of the variation in the dependent variable.
In addition, since a large number of correlations were observed between demographic variables, vascular and haemodynamic parameters (Figure 3), a PCA was performed to identify the unifying factors influencing the data. The demographic and anthropometric characteristics (age, height, weight and waist circumference), vascular parameters (crPWV, cfPWV, AIx@75 and FMD) and haemodynamic variables (central and peripheral systolic, diastolic, mean and PPs) were used for the PCA. The maximum contribution in the first principal component (PC1) was by haemodynamic variables, while that in the second principal component (PC2) was by vascular and demographic parameters (AIx@75, FMD, weight and waist circumference). Together, the two PCs accounted for 61.5% variance in the data. The negative associations of FMD, AIx@75, cfPWV with height and weight were also apparent in the variables plot of the PCA, which showed diagonally opposite clustering of these variables. Similarly, AIx@75 is in line with age substantiating their positive correlation in univariate analysis. In PCA, crPWV is closely clustered with both central and peripheral diastolic pressures suggesting a positive correlation, while cfPWV and AIx@75 are clustered with the central and peripheral PPs. However, no relationship emerged between cfPWV and PPs in univariate analysis. Additionally, cfPWV and AIx@75 are closely clustered in the PCA plot, while FMD and crPWV are placed diagonally opposite to each other, which corroborates the correlations observed in the univariate analysis (Figure 4).
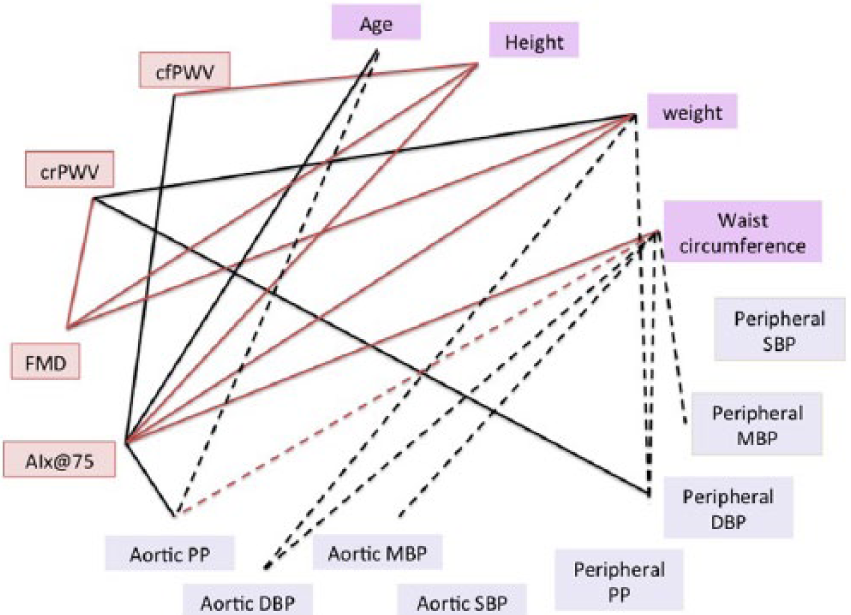
Correlation web showing correlations among vascular, haemodynamic, demographic and anthropometric parameters. Black lines indicate positive correlation, and red lines indicate a negative correlation. Solid lines indicate correlations between vascular and haemodynamic variables, and dashed lines indicate correlations between demographic or anthropometric variables and haemodynamic parameters.
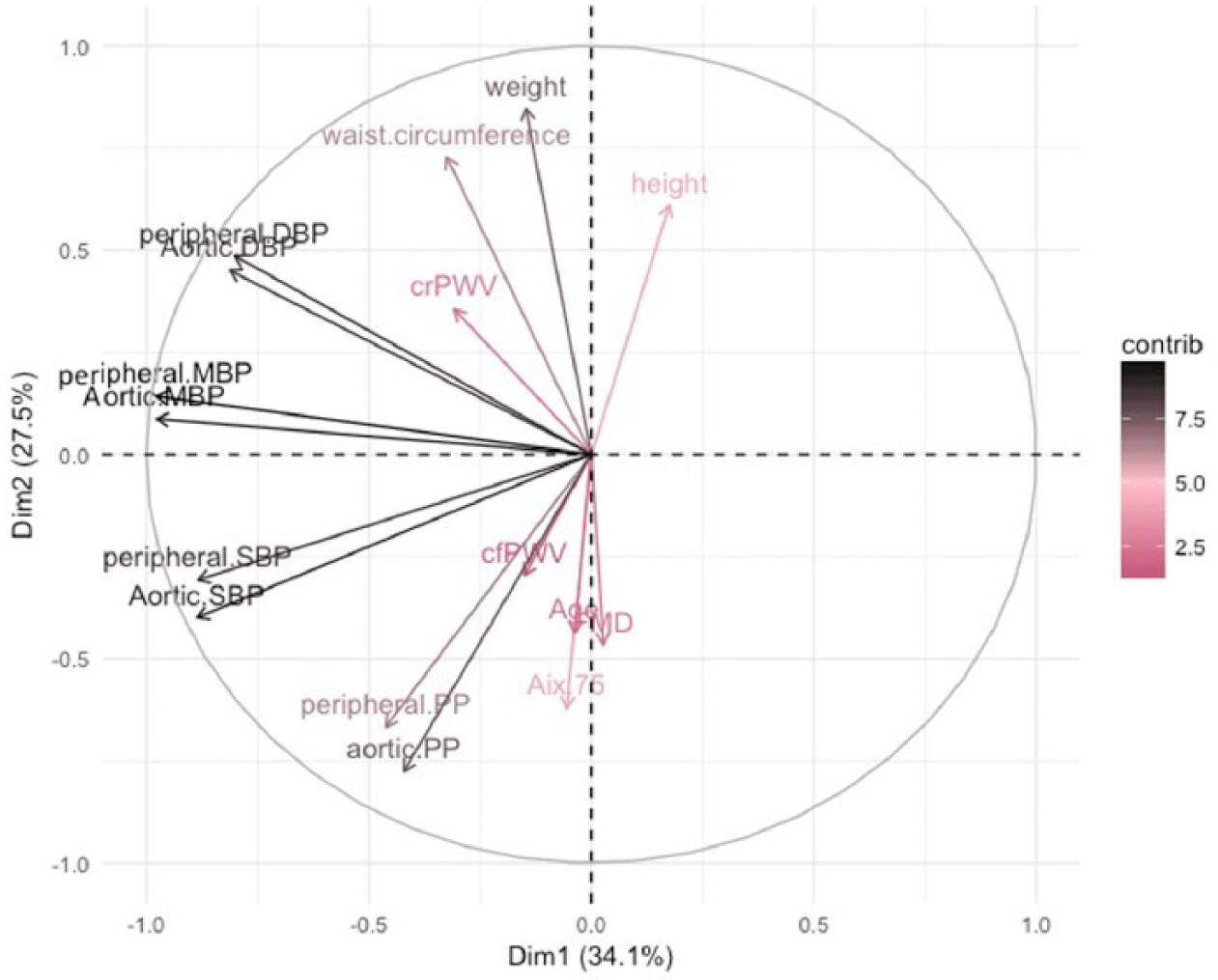
Variable factor map of first two components from principal component analysis. Variables in same direction are directly related, while those in opposite direction are inversely related and those perpendicular to each other are unrelated. Scale shows net contribution of each variable to the first two principal components.
Discussion
This study evaluated the correlations between haemodynamic parameters, endothelial function and arterial stiffness in the central and peripheral vessels in patients with metabolic syndrome.
The data show a significant correlation between augmentation index and age, and a weak non-significant correlation of central arterial stiffness with age while the same was not observed for peripheral arterial stiffness. This is expected and is in line with previous reports, which suggest that ageing influences aortic PWV to a greater extent as compared to muscular arterial PWV.2,24–26
A significant negative correlation was observed between AIx@75 and weight. While these results may seem counterintuitive, similar findings have been reported for PWV in patients of chronic kidney disease 27 and augmentation index in patients with and without diabetes. 28 Another study reported an inverse correlation between body fat and brachial arterial stiffness but not with carotid and femoral arterial stiffness. 29
Two key associations emerged in relation with the peripheral-muscular arterial stiffness: (1) a strong negative correlation with endothelial dysfunction and (2) a significant positive correlation with peripheral DBP. Both these associations hint towards a functional increase in smooth muscle tone resulting in an increased peripheral arterial stiffness in patients with metabolic syndrome. Endothelial dysfunction is characterized by an imbalance between vasodilators such as NO, EDHF, Prostaglandin I2 and vasoconstrictors like ET-1 and thromboxane A2, which modulate the contractile state of the vascular smooth muscle. This imbalance can alter the arterial distensibility by increasing the smooth muscle tone. Both NO and ET-1 have been shown to alter arterial distensibility in animal models30,31 and in humans, 32 which can result in an increase in muscular arterial PWV. In accordance with the Bramwell–Hill theory, PWV is inversely related to the square root of compliance. Endothelial dysfunction-mediated decrease in NO availability would result in a decrease in arterial compliance, thereby increasing the PWV. Additionally, the results from a recent study suggest that basal NO release is greater in the elastic arteries as compared to the muscular arteries. 33 A high muscular content, further augmented by a low basal NO production, could precipitate the effect of endothelial dysfunction-induced vasomediator imbalance in the peripheral arteries. The positive correlation between crPWV and peripheral DBP further corroborates the role of a functional increase in arterial stiffness in the peripheral-muscular arteries. It is also interesting to note that both peripheral and aortic DBP correlate with waist circumference. Obesity induced increase in DBP, and endothelial dysfunction could result in an increased muscular arterial stiffness. Additionally, endothelial dysfunction at the arteriolar level could result in an increase in the arteriolar resistance thereby increasing the DBP. Brachial FMD predominantly evaluates the macrovascular endothelial function, which could explain why we did not observe a correlation between brachial FMD and DBP (Figure 5).
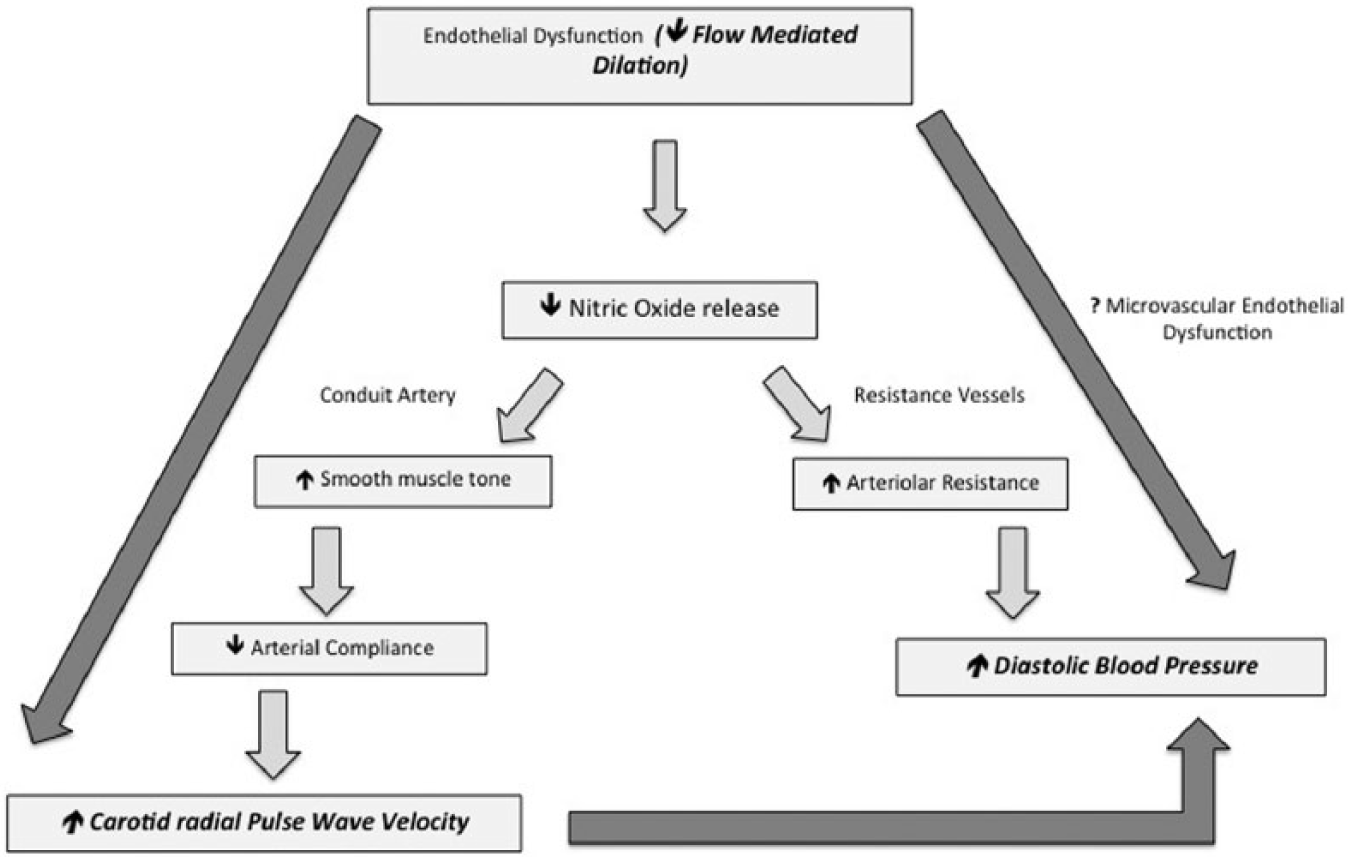
Proposed mechanism of functionally mediated increase in pulse wave velocity. Bold and italicized font indicates actual measured parameters, and dark-grey arrows indicate correlations or proposed correlation (in case of microvascular endothelial dysfunction and diastolic blood pressure).
However, though it has been established that endothelial dysfunction assessed by FMD is representative of coronary endothelial function, the technique has been criticized recently as it measures only ‘recruitable’ endothelial function by providing a supra-physiological shear stimulus and therefore is not an accurate measure of resting endothelial function. 34 Additionally, the relationship observed between FMD and crPWV could be due to assessment of the same ‘regional’ arterial segment.
No correlation was observed between cfPWV and FMD in this study. A previous study reported correlation of central arterial stiffness and FMD with correlation coefficients ranging between −0.4 and −0.8 in hypertensive patients and healthy individuals, 35 while one study did not find any correlation between cfPWV and FMD. 36 We infer that this study might be underpowered to detect this correlation. Additionally, the results of this study also suggest that in patients with metabolic syndrome, augmentation index is more closely related to stiffness in the central-elastic vessels as is evident by its correlations with cfPWV and aortic PP. While cfPWV did not correlate with aortic or peripheral PP in univariate analysis, they are closely related in the PCA variable map suggesting some association between the two. This implies that an increase in central arterial stiffness is most likely due to structural alterations in the vessel wall mediated by an increase in circumferential wall stress due to increased PP.
It is possible that in patients with metabolic syndrome, the peripheral arterial stiffness is majorly altered due to functionally mediated increase in vascular tone, while the central arterial stiffness is increased due to structural alterations in the vessel wall. Future studies in animal models of endothelial dysfunction are required to delineate the role of vasomediators and structural alteration of vessels at the elastic and muscular arterial segments and to identify which component of arterial stiffness (structural vs functional) is reflected at each of these sites.
The strength of this study is in underlining the ability of regional arterial stiffness to provide additional information over and above that afforded by the assessment of central arterial stiffness alone. However, the study is underpowered to assess the correlation of carotid-femoral PWV with the current sample size. Another limitation of our study is that we did not correct the FMD values for shear rate and did not assess endothelium-independent nitroglycerine-mediated dilation.
To conclude, the findings of this study imply that regional arterial stiffness assessed by PWV in central-elastic and peripheral-muscular arteries differentially relates to endothelial dysfunction quantified by brachial FMD, where the central-elastic arteries might be predominantly influenced by endothelial dysfunction-induced structural changes, while the peripheral-muscular arteries are majorly affected by functional alterations due to endothelial dysfunction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
