Abstract
Background:
Annexin A5 (anxA5) is involved in processes which are crucial in atherogenesis. However, anxA5’s relationship with atherosclerotic lesion extension and plaque composition in high-risk patients with type 2 diabetes remains unclear. Thus, we characterized the association between circulating anxA5 levels with atherosclerotic burden and coronary plaque composition in diabetes mellitus patients.
Methods:
Intima-media thickness was determined in 96 diabetes mellitus patients with stable coronary artery disease. Furthermore, intracoronary optical coherence tomography was performed in 106 lesions to determine plaque composition.
Results:
AnxA5 plasma levels of patients with intima-media thickening were higher (3.49 ± 2.19 ng/mL) compared to patients with normal intima-media thickness (2.24 ± 1.67 ng/mL, p = 0.002). Furthermore, anxA5 was associated with intima-media thickening on univariable [odds ratio = 1.445 (1.106–1.889), p = 0.007] and multivariable [odds ratio = 1.643 (1.166–2.314), p = 0.005] logistic regression analysis when adjusted for multiple cardiovascular risk factors and biomarkers. Furthermore, receiver operating characteristic analysis demonstrated that anxA5 predicted intima-media thickening with low–moderate diagnostic efficiency [area under the curve = 0.700 (0.592–0.808)]. In contrast, there was no association between anxA5 levels and coronary plaque composition as assessed by optical coherence tomography including the presence of lipid, calcified, fibrous plaque or the minimal thickness of the fibrous cap overlying the necrotic lipid core (p = ns).
Conclusion:
Circulating anxA5 levels are associated with carotid intima-media thickness but not coronary plaque composition in high-risk patients with diabetes mellitus.
Introduction
Annexin A5 (anxA5) is a single-chain protein which has initially been discovered by Reutelingsperger et al. 1 as an anticoagulant protein from arteries of the human umbilical cord. AnxA5 is part of the annexin gene superfamily and is known for its binding to phosphatidylserine (PS) with high affinity and in a calcium-dependent manner. 2 PS is expressed on stressed and dying cells, and anxA5 has been demonstrated to exert anti-inflammatory, anti-apoptotic and anticoagulant properties via binding to PS and shielding this phospholipid from participating in pro-inflammatory and procoagulant reactions.3,4 AnxA5 may also dampen inflammation by inhibiting other pro-inflammatory lipids such as oxidized cardiolipin 5 and lysophosphatidylcholine. 6 Previous studies demonstrated beneficial effects of anxA5 on restenosis and vein graft patency in murine models,7,8 and work from our own laboratory could demonstrate anti-inflammatory effects of anxA5 in a mouse model of atherosclerosis. 9
AnxA5 is also a promising and emerging cardiovascular biomarker in addition to its effects on atherogenesis. 10 Recently, we could demonstrate that anxA5 has also a role in heart failure and improves the prediction of mortality in these patients when added to N-terminal pro-B-type natriuretic peptide (NT-proBNP). 11 Furthermore, anxA5 has been associated with subclinical atherosclerosis and endothelial dysfunction in patients with systemic lupus erythematosus. 12 However, other studies found contradicting results by demonstrating a negative correlation between anxA5 levels and vascular disease, 13 and the association between anxA5 levels and atherosclerosis presently awaits confirmation.
The intima-media thickness (IMT) of carotid arteries is easily determined by carotid ultrasound and yields excellent image quality due to the superficial carotid arteries.14,15 Given that atherosclerosis is a systemic process, the carotids are the arteries of choice to non-invasively image IMT as a surrogate parameter for systemic atherosclerosis, 15 and recently, carotid IMT has even been associated with cardiovascular events. 14
Optical coherence tomography (OCT) is a novel intravascular imaging modality with a resolution 10-fold higher than intravascular ultrasound. OCT uses the reflection of light to allow the exact visualization and quantification of both intraluminal structures of coronary artery lesions as well as plaque structures with an excellent reproducibility. 16
Taken together, the preclinical data regarding anxA5 suggest this protein to be a promising biomarker both for atherosclerosis extension as well as plaque composition. Given that this relationship is particularly important in high-risk patients with type 2 diabetes, we investigated the association between circulating anxA5 levels, on one hand, and carotid IMT as well as coronary plaque composition as determined by OCT, on the other hand, in this high-risk population.
Methods
Study population
A total of 96 patients with type 2 diabetes undergoing coronary angiography due to stable angina pectoris (SAP) and subsequent percutaneous coronary intervention of the coronary target lesion were identified at the Department of Cardiology, University Hospital of the RWTH Aachen, Germany. OCT analysis, laboratory testing and clinical history taking were performed in all patients as previously described.17 –19
Furthermore, inclusion criteria were type 2 diabetes, age >30 years and written informed consent to the study protocol. Type 2 diabetes was based on clinical history of diabetes mellitus, ongoing insulin/oral antidiabetic therapy or an HbA1C level exceeding 6.5%.
Exclusion criteria were left main coronary artery stenosis, bypass graft lesions, cardiogenic shock, hemodynamic or rhythmic instability, acute or chronic renal insufficiency (serum creatinine level > 1.5 mg/dL) and pregnancy. We furthermore excluded severely tortuous and calcified vessels, not allowing the safe advancement of the OCT catheter and the necessity to predilate a target lesion prior to OCT in order to achieve Thrombolysis in Myocardial Infarction (TIMI) flow grade III. The study was approved by the local Ethics Committee at the University Hospital in Aachen and is in accordance with the Declaration of Helsinki on ethical principles for medical research involving human subjects.
Carotid ultrasound
Carotid ultrasound was performed using a high-performance, premium ultrasound system (Vivid i; General Electric, Boston, MA, USA) and a 4- to 13-MHz transducer (8L-RS). The maximal IMT of each side was measured and computerized using a commercially available software (General Electric). The larger of the two sides was taken for further statistical analysis. A cut-off of 0.83 mm was used to define patients with normal and thickened IMT, as this value has been shown to be the mean in patients with vascular disease.20,21
AnxA5 measurements
AnxA5 levels were determined from ethylenediaminetetraacetic acid (EDTA) plasma (Sarstedt, Nümbrecht, Germany; 1.6 mg K3-EDTA/mL blood). All specimens were put on ice immediately following blood drawings and processed within 30 min, and then stored in −80°C until further analysis. AnxA5 levels were measured using a commercially available kit (Hyphen Biomed, Neuville-sur-Oise, France) following three cycles of freezing and thawing to unbind all non-calcium-bound anxA5.
OCT image acquisition and analysis
For image acquisition of the target lesion, we used the frequency-domain-OCT C7XR system and the DragonFly catheter (St. Jude Medical Systems, LightLab Imaging, Inc., Westford, MA, USA) with an automated pull back device at a rate of 20 mm/s. Blood clearance was achieved with the non-occlusion OCT-technique by injection of iso-osmolar contrast (Iodixanol; GE Healthcare, Chicago, IL, USA).
Quantitative intraluminal dimensions and plaque morphologies were assessed as previously described17 –19,22,23 and a representative OCT image is displayed in Figure 1. All measurements were performed in adaptation to the published consensus for quantitative and qualitative assessment.16,24
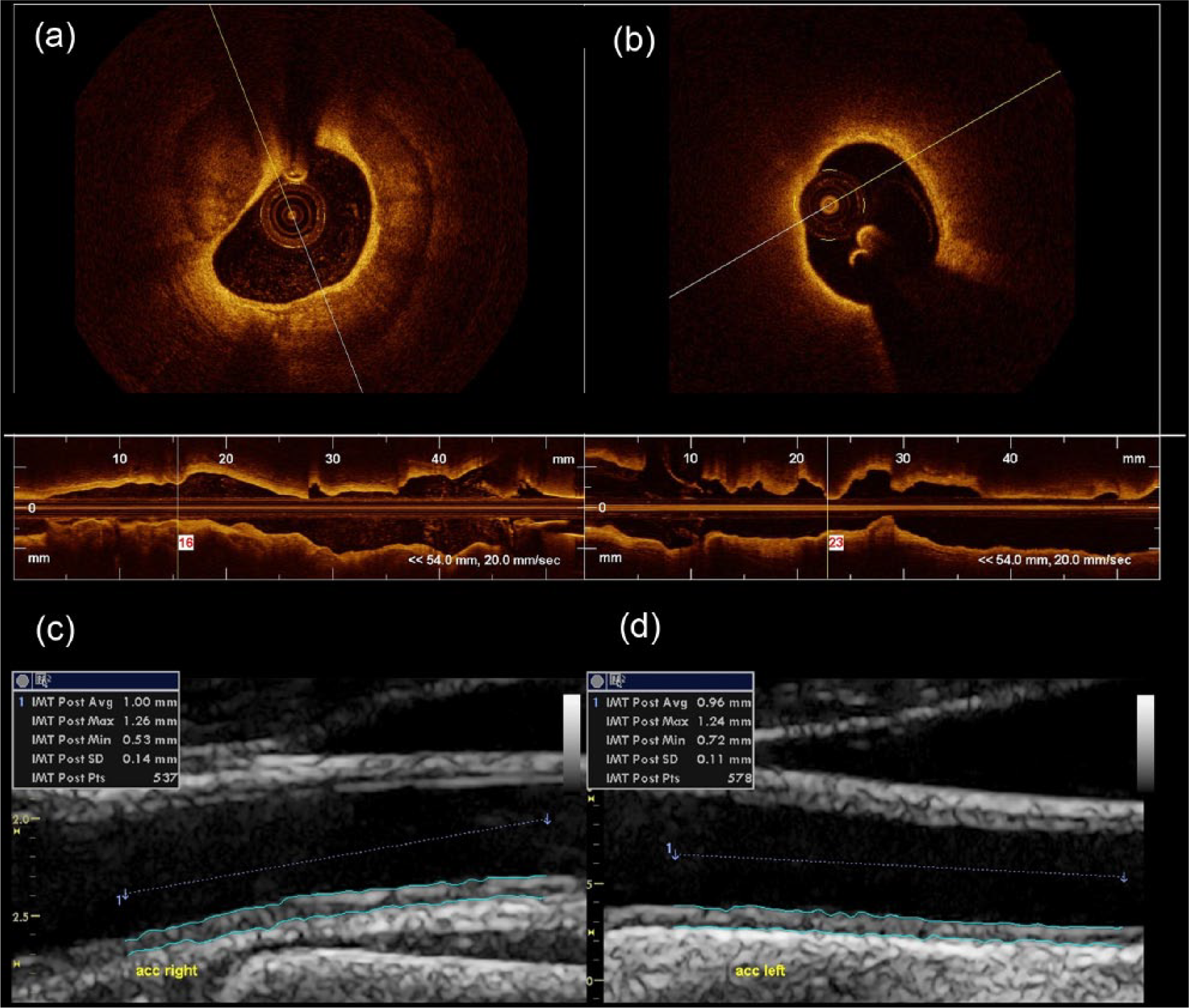
Representative intracoronary optical coherence tomography and carotid ultrasound images. (a and b) Representative intracoronary optical coherence tomography images are displayed: (a) a plaque with calcification which is signal-poor with defined borders and (b) a lipid plaque with the signal intense fibrous cap overlying the necrotic lipid core. Representative ultrasound images of the (c) right and (d) left carotid artery (ACC) are displayed which show the determination of the intima-media thickness using a computerized and commercially available software.
Statistical analysis
Statistical analyses were performed with SPSS software (IBM Corp., Armonk, NY, USA). Categorical variables were summarized as count (%), continuous variables as mean ± standard deviation. Continuous variables were compared with Student’s or Welch’s t-test where appropriate. Pearson’s χ2 test was used to compare nominal variables.
Non-linear regression analysis was performed to determine the association between anxA5 levels and IMT. Univariable and multivariable binary logistic regression analyses were performed to identify risk factors for a thickened IMT. Sensitivity, specificity, positive and negative predictive values as well as optimal cut-off-values were calculated from the receiver operating characteristic (ROC) curve to predict a thickened IMT. The value with the highest Youden index (sensitivity + specificity − 1) was identified as optimal cut-off-value. This optimal cut-off value was used to distinguish between patients with normal and elevated anxA5 levels. A classification of the diagnostic efficacy for anxA5 levels according to the values of the area under the curve (AUC) was used as described elsewhere. 25 Patient and IMT data were analysed on a per-patient basis, and the OCT data were analysed on a per-stenosis basis. A p value of <0.05 was considered to indicate statistical significance.
Results
Clinical characteristics
A total of 96 patients were enrolled in the study. Mean anxA5 levels were 3.00 ± 2.08 ng/mL and mean IMT was 0.88 ± 0.20 mm. All patients were divided into a group with normal (IMT < 0.83 mm, n = 38) and thickened IMT (IMT ⩾ 0.83 mm, n = 58). Patients with thickened IMT had significantly higher anxA5 levels as compared to patients with normal IMT (3.49 ± 2.19 vs 2.24 ± 1.67 ng/mL, p = 0.002). Furthermore, patients with thickened IMT tended to have a higher frequency of smoking (24.1% vs 10.5%, p = 0.095) and a higher tendency for augmented HbA1C levels (56.9 ± 13.7 vs 51.8 ± 11.8 mmol/mol, p = 0.066), whereas age, body mass index (BMI), glomerular filtration rate and C-reactive protein (CRP) were similar between groups (p = ns). For further clinical characteristics, please refer to Table 1.
Differences between thickened and non-thickened IMT.
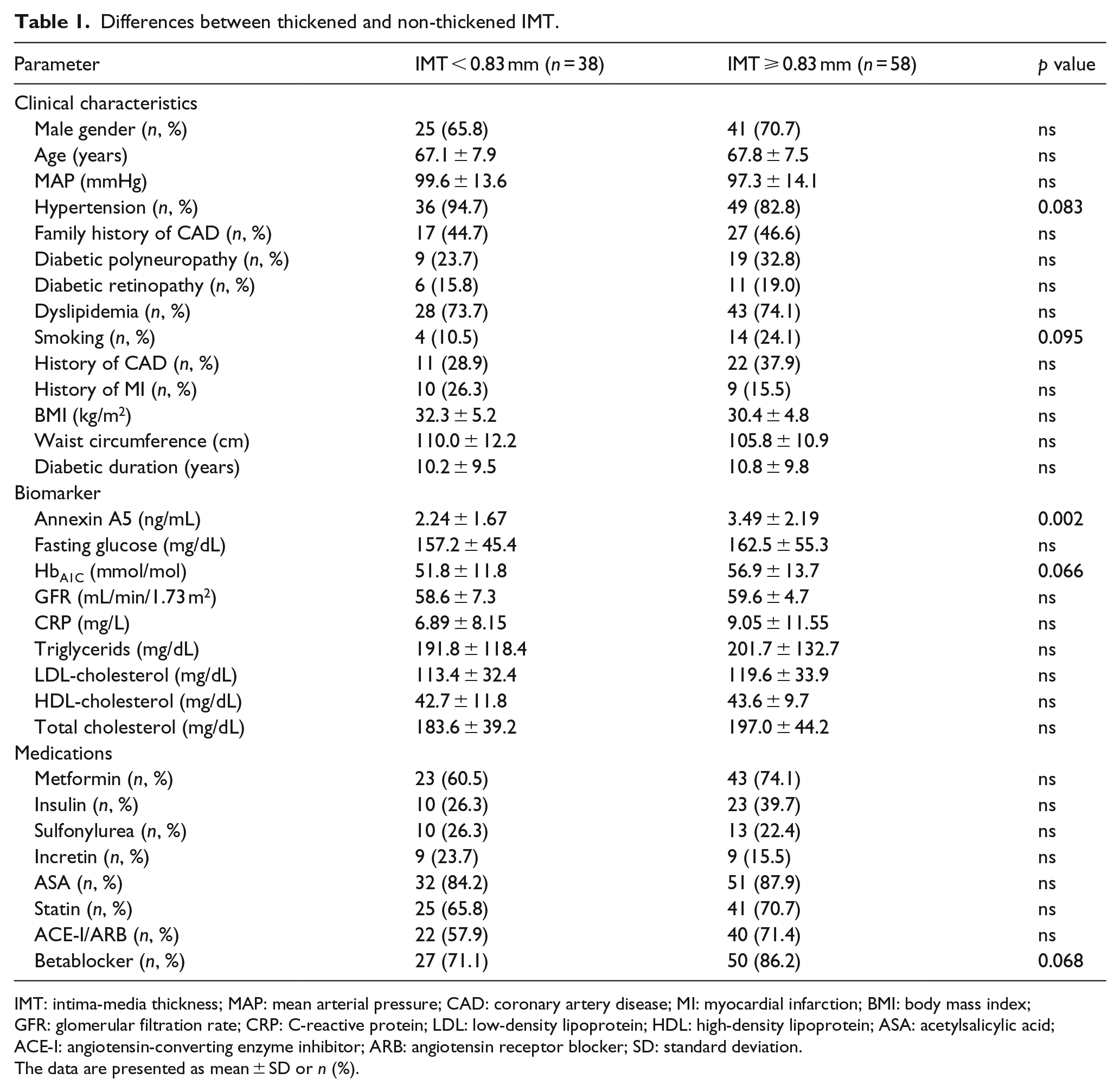
IMT: intima-media thickness; MAP: mean arterial pressure; CAD: coronary artery disease; MI: myocardial infarction; BMI: body mass index; GFR: glomerular filtration rate; CRP: C-reactive protein; LDL: low-density lipoprotein; HDL: high-density lipoprotein; ASA: acetylsalicylic acid; ACE-I: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; SD: standard deviation.
The data are presented as mean ± SD or n (%).
Relationship between circulating anxA5 levels and IMT
To evaluate whether there is an association between circulating anxA5 levels and IMT, non-linear (exponential) regression analysis was performed. AnxA5 levels were significantly associated with IMT (r = 0.207, p = 0.043). When using 0.83 mm as a cut-off to distinguish between thickened and non-thickened IMT, anxA5 was associated with thickened IMT [odds ratio (OR) = 1.445 (1.106–1.889), p = 0.007, Table 2].
Univariable and multivariable logistic regression analysis for the presence of thickened IMT.
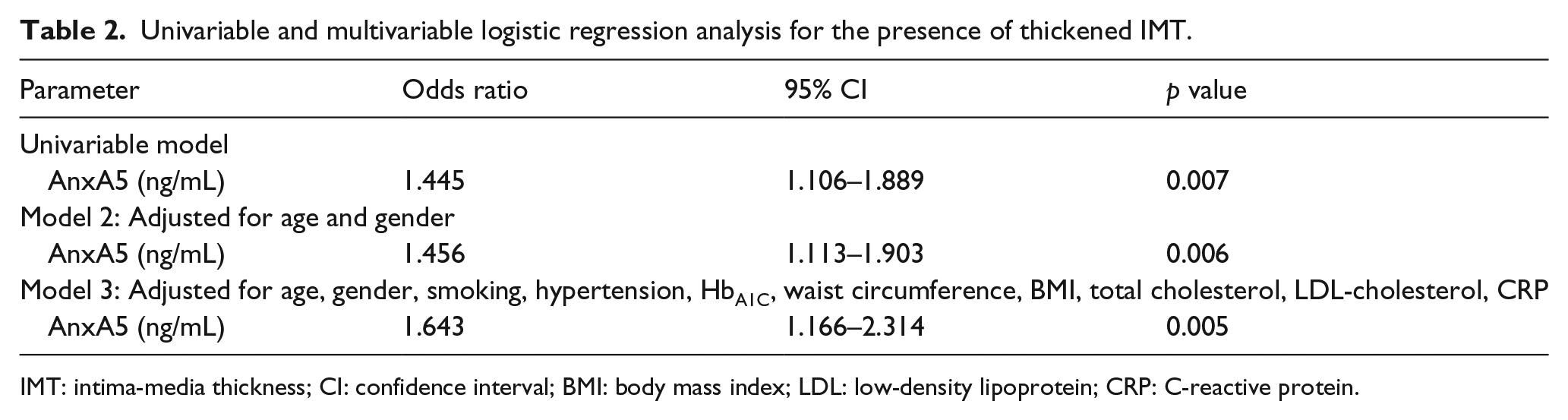
IMT: intima-media thickness; CI: confidence interval; BMI: body mass index; LDL: low-density lipoprotein; CRP: C-reactive protein.
To test if anxA5 is also an independent risk factor for the presence of thickened IMT, multivariable binary logistic regression analysis was performed. Following adjustment for age and gender, anxA5 was still associated with IMT thickening [OR = 1.456 (1.113–1.903), p = 0.006, model 2, Table 2]. Even after adjustment for multiple cardiovascular risk factors including male gender, age, smoking, hypertension, HbA1C levels, waist circumference, BMI, total cholesterol, low-density lipoprotein (LDL) cholesterol and CRP levels, anxA5 was still associated with a thickened IMT [OR = 1.643 (1.166–2.314), model 3, p = 0.005]. These data suggest that anxA5 is an independent risk factor for IMT thickening.
Diagnostic efficiency of circulating anxA5 levels to predict IMT thickening
In order to evaluate the diagnostic efficiency of circulating anxA5 levels to predict a thickened carotid IMT, ROC analysis was performed. As demonstrated in Figure 2, anxA5 levels significantly predicted the presence of IMT thickening with low to moderate diagnostic efficiency [AUC = 0.700 (0.592–0.808), optimal cut-off for anxA5 1.907 ng/mL, sensitivity at optimal cut-off 72.4%, specificity at optimal cut-off 65.8%].
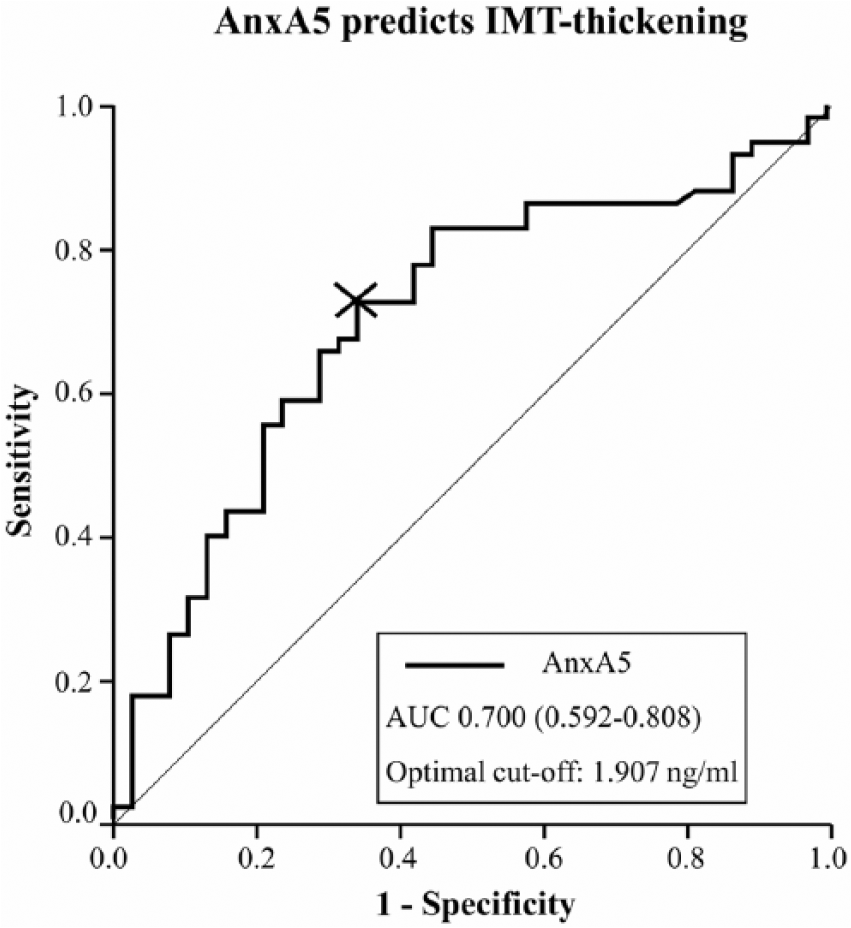
Receiver operating curve (ROC) statistics.
AnxA5 levels are not related to coronary plaque composition
After we observed a relationship between circulating anxA5 levels and carotid IMT, we investigated whether coronary plaque composition is different between patients with normal versus elevated anxA5 levels.
However, as demonstrated in Table 3, there was no difference in coronary plaque composition of patients with normal or elevated anxA5 levels including the frequency of calcified, lipid or fibrous plaque nor the incidence of a thin-capped fibroatheroma or the minimal or mean fibrous cap thickness overlying the necrotic lipid core, suggesting that circulating anxA5 levels are not related to coronary plaque composition.
OCT-derived coronary plaque characteristics: differences between lesions of patients with versus without increased anxA5 levels.
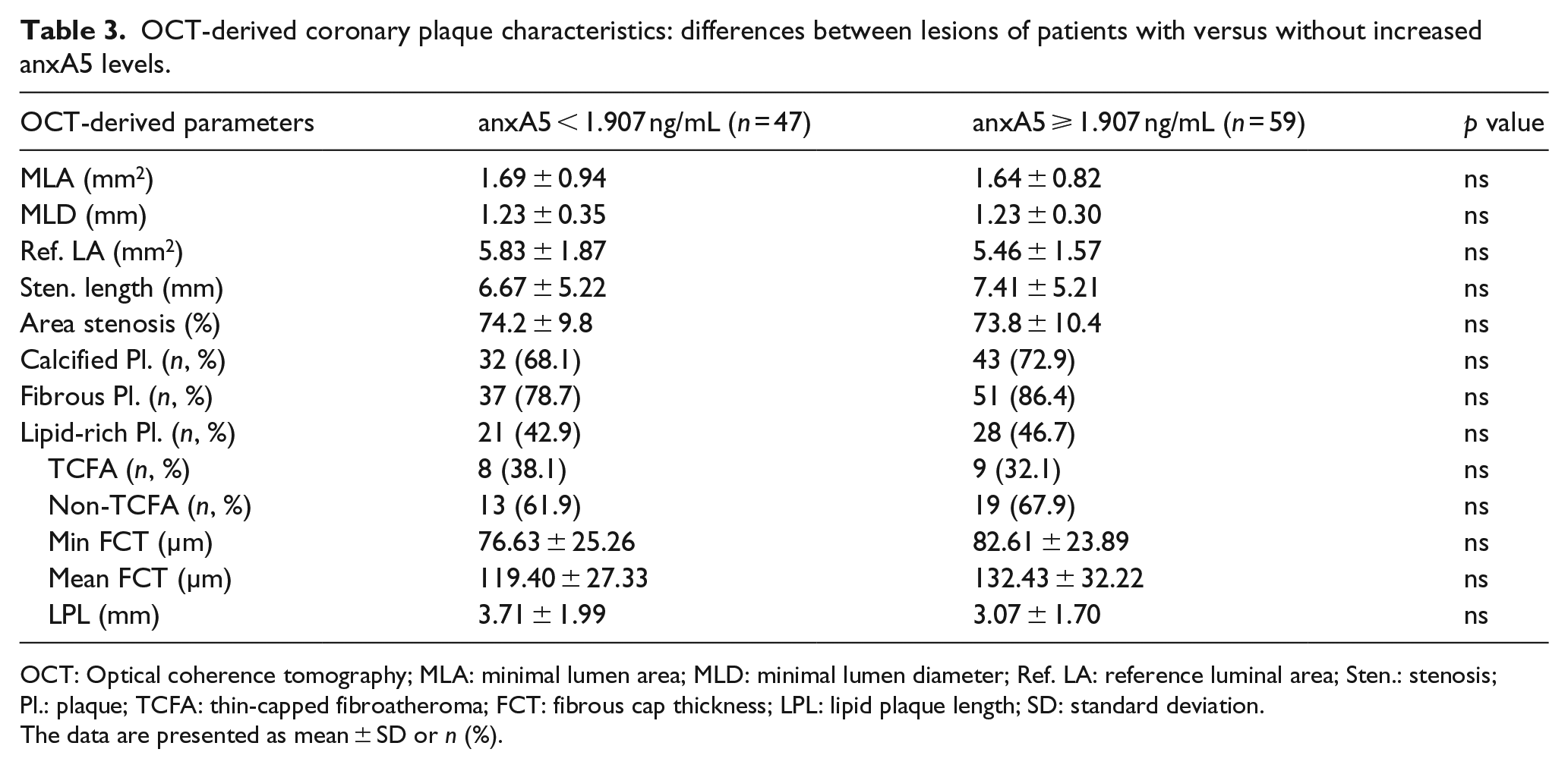
OCT: Optical coherence tomography; MLA: minimal lumen area; MLD: minimal lumen diameter; Ref. LA: reference luminal area; Sten.: stenosis; Pl.: plaque; TCFA: thin-capped fibroatheroma; FCT: fibrous cap thickness; LPL: lipid plaque length; SD: standard deviation.
The data are presented as mean ± SD or n (%).
Discussion
The main findings of this study in patients with type 2 diabetes are as follows:
AnxA5 is associated with carotid IMT. AnxA5 levels predict IMT thickening with low to moderate diagnostic efficiency.
There is no association between circulating anxA5 levels and coronary plaque morphology as determined by OCT.
We have recently proposed anxA5 to be a novel cardiovascular biomarker. 10 We extend the current knowledge by demonstrating a positive relationship between anxA5 plasma levels and carotid IMT, a surrogate marker of systemic atherosclerosis, in high-risk patients with type 2 diabetes and stable coronary artery disease. 15 We could also show that anxA5 is associated with IMT thickening even when adjusted for multiple other cardiovascular risk factors including male gender, age, smoking, hypertension, HbA1C levels, waist circumference, BMI, total cholesterol, LDL-cholesterol and CRP levels. These data suggest that anxA5 is a robust predictor for IMT thickening and independent from classical cardiovascular risk factors and biomarkers. Our results are in line with Valer et al. 12 who demonstrated in patients with systemic lupus erythematosus that anxA5 is an independent predictive biomarker for both subclinical atherosclerosis as determined by carotid IMT as well as endothelial dysfunction as measured by flow-mediated vasodilatation. Whereas we demonstrate a positive relationship between anxA5 levels and IMT in diabetic patients, Van Tits et al. 13 reported a negative relationship between circulating anxA5 levels and coronary artery disease in the overall population. However, this study used population-control patients from the Rotterdam study, a population-based cohort study on chronic and disabling diseases, which had much higher levels of anxA5 (mean = 11.2 ng/mL compared to 3.00 ng/mL in our study). At this point, it is unclear whether the differential results of this study originate from a different study population or a different study design and protocol. Thus, the relationship between anxA5 levels and IMT in non-diabetic patients awaits further clarification.
Consistent with the regression analyses in our study, anxA5 is associated with IMT thickening significantly on ROC analysis but with only low to moderate diagnostic efficiency. Thus, anxA5 is not suitable to predict atherosclerosis by itself. Future studies are necessary to determine whether anxA5 levels may be useful to predict vascular disease when used in combination with other cardiovascular biomarkers and risk factors. Given the high cardiovascular risk of type 2 diabetic patients, this is particularly important in this group of patients. Future studies should also investigate if anxA5 levels are associated with cardiovascular complications in patients with type 2 diabetes.
Recently, treatment of anxA5 has been shown to modify vascular inflammation and plaque phenotype,7,8 and this was confirmed by work from our own laboratory. 9 Specifically, we have shown that anxA5 reduced plaque macrophage content in an animal model of advanced atherosclerosis. 9 Given that vascular inflammation has a tremendous impact on both the progression of atherosclerosis and features of plaque vulnerability such as the thickness of the fibrous cup overlying the necrotic lipid core,26,27 we investigated in this study the association of circulating anxA5 plasma levels with coronary plaque phenotype as determined by OCT. However, plaque characteristics were not different between patients with normal and elevated anxA5 plasma levels, suggesting that circulating anxA5 levels are not associated with coronary plaque morphology as determined by OCT. Given that anxA5 decreases plaque inflammation in an animal model of atherosclerosis, 9 these results in patients with type 2 diabetes are intriguing. Our observations on plaque composition are in line with a previous study, in which exogenous anxA5 did not change plaque collagen content, acellular mass and smooth muscle cell content in advanced atherosclerosis, 9 suggesting that the anti-inflammatory effects of anxA5 may be balanced by its inhibitory effects on efferocytosis.28,29 PS on the surface of apoptotic cells skewed macrophages towards an anti-inflammatory phenotype. 30 AnxA5 may inhibit this pathway by shielding PS on the surface of apoptotic cells. Future studies are necessary to investigate effects of anxA5 on polarization of macrophages and effects on plaque morphology in the presence and absence of type 2 diabetes. Still, the fact that anxA5 levels are associated with IMT thickening but not plaque composition is intriguing. Due to the study design, we cannot elucidate whether circulating endogenous anxA5 levels are causally involved in the atherosclerotic process or whether augmented circulating anxA5 levels merely reflect the extent of atherosclerosis which may be independent of lesion vulnerability. Work from our own laboratory recently demonstrated that anxA5 improves risk stratification in patients with heart failure potentially by assessing damage to peripheral organs. 11 Thus, it is tempting to speculate in the light of current data that anxA5 levels in this study may at least in part reflect the extent of atherosclerosis; however, this needs to be determined in future studies.
Limitations
Overall, the study population is small, and the findings need confirmation in larger trials. As this is an investigation in an exclusively diabetic cohort, we cannot draw any conclusion about the relationship between circulating anxA5 plasma levels and atherosclerosis for patients without diabetes. Furthermore, all patients had stable coronary artery disease; thus, we cannot exclude any selection bias in this work.
Conclusion
We conclude from this study that in patients with diabetes, there is a relationship between circulating anxA5 plasma levels and carotid IMT, a surrogate marker of atherosclerosis. However, no relationship between circulating anxA5 plasma levels and plaque characteristics of coronary lesions as determined by OCT could be observed. Thus, further studies are warranted to determine whether anxA5 might be suitable for risk assessment of patients with type 2 diabetes.
Footnotes
Declaration of conflicting interests
C.R. is the (co-)inventor of patents describing anxA5. The other authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
