Abstract
Introduction:
Fibrinolytic factors, plasminogen activator inhibitor-1, tissue plasminogen activator, tissue plasminogen activator/plasminogen activator-complex and the haemostatic factor von Willebrand factor are known markers of cardiovascular disease. Their plasma levels are adversely affected in patients with dysglycaemia, and glucose normalization with insulin glargine might improve the levels of these factors.
Methods:
Prespecified Swedish substudy of the Outcome Reduction with an Initial Glargine Intervention trial (ClinicalTrials.gov number, NCT00069784). Tissue plasminogen activator activity, tissue plasminogen activator antigen, plasminogen activator inhibitor-1 antigen, tissue plasminogen activator/plasminogen activator inhibitor-1 complex and von Willebrand factor were analysed at study start, after 2 years and at the end of the study (median follow-up of 6.2 years).
Results:
Of 129 patients (mean age of 64 ± 7 years, females: 19%), 68 (53%) and 61 (47%) were randomized to the insulin glargine and standard care group, respectively. Allocation to insulin glargine did not significantly affect the studied fibrinolytic markers or von Willebrand factor compared to standard care. Likewise, there were no significant differences in plasminogen activator inhibitor-1, tissue plasminogen activator antigen and von Willebrand factor. During the whole study period, the within-group analysis revealed a curvilinear pattern and significant changes for tissue plasminogen activator/plasminogen activator inhibitor-1 complex, tissue plasminogen activator antigen and von Willebrand factor in the insulin glargine but not in the standard care group.
Conclusion:
In people with dysglycaemia and other cardiovascular risk factors, basal insulin does not improve the levels of markers of fibrinolysis or von Willebrand factor compared to standard glucose-lowering treatments.
Introduction
Endothelial-derived fibrinolytic factors such as plasminogen activator inhibitor-1 (PAI-1) antigen, tissue plasminogen activator antigen (tPAag), tissue plasminogen activator/plasminogen activator inhibitor-1 (tPA/PAI-1) complex and the haemostatic factor, von Willebrand factor (vWF), are known predictors of myocardial infarction and stroke.1–4 They are also related to dysglycaemia. Thus, PAI-1 levels are correlated with the levels of glucose,5–7 and PAI-1, tPAag and vWF all predict type 2 diabetes. 8 These relationships may reflect underlying endothelial dysfunction and/or an underlying inflammatory process. 9
Exogenous insulin improves measures of endothelial function in people.10,11 However, the effect of insulin on endothelium-derived markers of fibrinolysis in people with dysglycaemia and cardiovascular risk factors remains unclear. 12 We therefore tested the hypothesis that administering insulin (as basal insulin glargine) to people with elevated glucose levels and additional cardiovascular risk factors would improve the endothelial-derived fibrinolytic factors [tissue plasminogen activator (tPA) activity, tPAag, PAI-1 antigen and tPA/PAI-1 complex] and vWF more than the standard glucose-lowering treatment as part of a prespecified substudy of the Outcome Reduction with an Initial Glargine Intervention (ORIGIN).
Material and methods
A detailed description of the design and results of the ORIGIN trial have published elsewhere. 13 Briefly, 12,537 people (mean age of 63.5 years) with impaired fasting glucose, impaired glucose tolerance or type 2 diabetes, who all had additional cardiovascular risk factors and whose average baseline HbA1c level was 6.4%, were studied. Participants were randomly allocated to receive basal insulin glargine (IG group) titrated targeted at normal fasting plasma glucose levels ⩽5.3 mmol/L or to standard glucose-lowering therapy [standard care (SC) group]. The sample analysis was performed blinded to treatment allocation according to ORIGIN-study protocol. They were also allocated to receive omega-3 fatty acids or placebo according to a factorial design. 13 All randomized participants were followed until study end. Adherence and adverse events were ascertained 0.5, 1, 2 and 4 months after randomization and every 4 months thereafter.
In this prespecified Swedish substudy of the ORIGIN trial recruiting patients at 18 of the 22 Swedish centres, the following components of the fibrinolytic and haemostatic systems were investigated: tPA activity, tPAag, PAI-1 antigen, tPA/PAI-1 complex and vWF. Blood samples (test tube of Stabilyte) were obtained at study start (baseline; n = 129), after 2 years (Visit 2; n = 117) and by the end of the study after a median follow-up of 6.2 years (Visit 3; n = 109). To avoid diurnal variations, the blood samples were collected 08:00–10:00 am after 10 min of rest. The test tubes were centrifuged at 2000g for 15 min at 4°C and split into three aliquots that were stored at −70°C. Analyses were performed in singlets at the coordinating centre in Skellefteå after the end of the study. The samples were thawed in a 37°C water bath for 15 min and analysed immediately afterwards. Reagent kits with identical batch numbers were used for fibrinolytic analysis. ELISA kits purchased from Tcoag (Bray, Co., Wicklow, Ireland) were used for the measurement of tPAag (TriniLIZE tPA antigen®) and PAI-1 (TriniLIZE PAI-1 antigen®). The measurement of tPA/PAI-1 complex was done using ELISA kits from Technoclone (TECHNOZYM® t-PA-PAI-1, Vienna, Austria), and vWF levels were analysed with reagents purchased from DAKO (Copenhagen, Denmark). Inter-assay coefficient of variation (CV) was 4.6% for tPA, 4.0% for PAI-1, 6.6% for tPA/PAI-1 complex and 3.4% for vWF.
Statistics
Baseline characteristics are presented as percentages for categorical variables and as mean [standard deviation (SD)] or median (interquartile range) for continuous variables. Categorical and continuous variables were compared using Chi-squared or Fisher’s exact tests as appropriate or Mann–Whitney U tests, respectively. Analyses of all fibrinolytic variables were based on log-transformed data. Unadjusted and adjusted linear regression models were used to assess the effect of allocation on the difference in fibrinolytic variables and vWF from baseline to either the second or third sampling visit. Independent variables included in the linear regression models were as follows: (a) Model 1: group allocation (glargine/standard), allocation to omega-3FA/placebo, prior diabetes and prior cardiovascular disease (CVD); (b) Model 2: variables in Model 1 plus baseline metformin, baseline aspirin, baseline weight and baseline sulfonylurea or other glucose-lowering drugs and (c) Model 3: variables in Model 2 plus Visit 2 metformin, Visit 2 aspirin, Visit 2 weight and Visit 2 sulfonylurea or other glucose-lowering drugs. Unadjusted and adjusted mixed linear regression models with repeated measures were used to assess changes over all three time points (baseline, sampling Visit 2 and Visit 3). Independent variables included in these regression models were as follows: (a) Model 1: allocation to glargine/SC, allocation to omega-3FA/placebo, any diabetes and prior CVD and (b) Model 2: allocation to glargine/SC, allocation to omega-3FA/placebo any diabetes, prior CVD, metformin, aspirin, weight, sulfonylurea or other glucose drug (last four covariates were time dependent). Data were analysed with the use of SAS software (version 9.1 for Solaris). The nominal level of significance for all analyses was p < 0.05.
Sample size dimension
In an epidemiological study, a reduction of tPA mass concentration from 12.8 to 11.0 µg/L (16% reduction; SD = 4.5) was found to be clinically significant. 14 With a power of 80%, an SD of 4 µg/L and a significance level of 5%, we estimated that at least 29 people per group were required to detect a relative reduction of 20% or more in the tPA mass concentration (i.e. from 15.0 to 12.0 µg/L; SD = 4). All trial centres in Sweden were invited and 18 of 21 participated.
Ethics
This study was conducted according to the ICH E6 (Guideline for Good Clinical Practice, 1 May 1996), in agreement with the Declaration of Helsinki and in keeping with local regulations. The Regional Ethics Committee at Stockholm approved the study (Dnr: 04-041T). All participants gave their written informed consent.
Results
The total number of patients with available baseline samples was 129 (mean age of 64 ± 7 years; females: 19%). Clinical characteristics at baseline and during follow-up are displayed in Table 1. A history of CVD was present in 97% of the participants and 60% had previously known diabetes. In total, 68 (53%) of the 129 patients were randomized to the IG and 61 (47%) to the SC group. There were no significant differences in baseline characteristics between these two groups (Table 1). In total, 89% of participants allocated to insulin and 93% allocated to SC provided blood samples at the 2-year visit. The corresponding proportions at the end of the study were 85% (glargine treatment) and 84% (SC).
Baseline characteristics of the patients by allocated group (Visit 1–Visit 3).
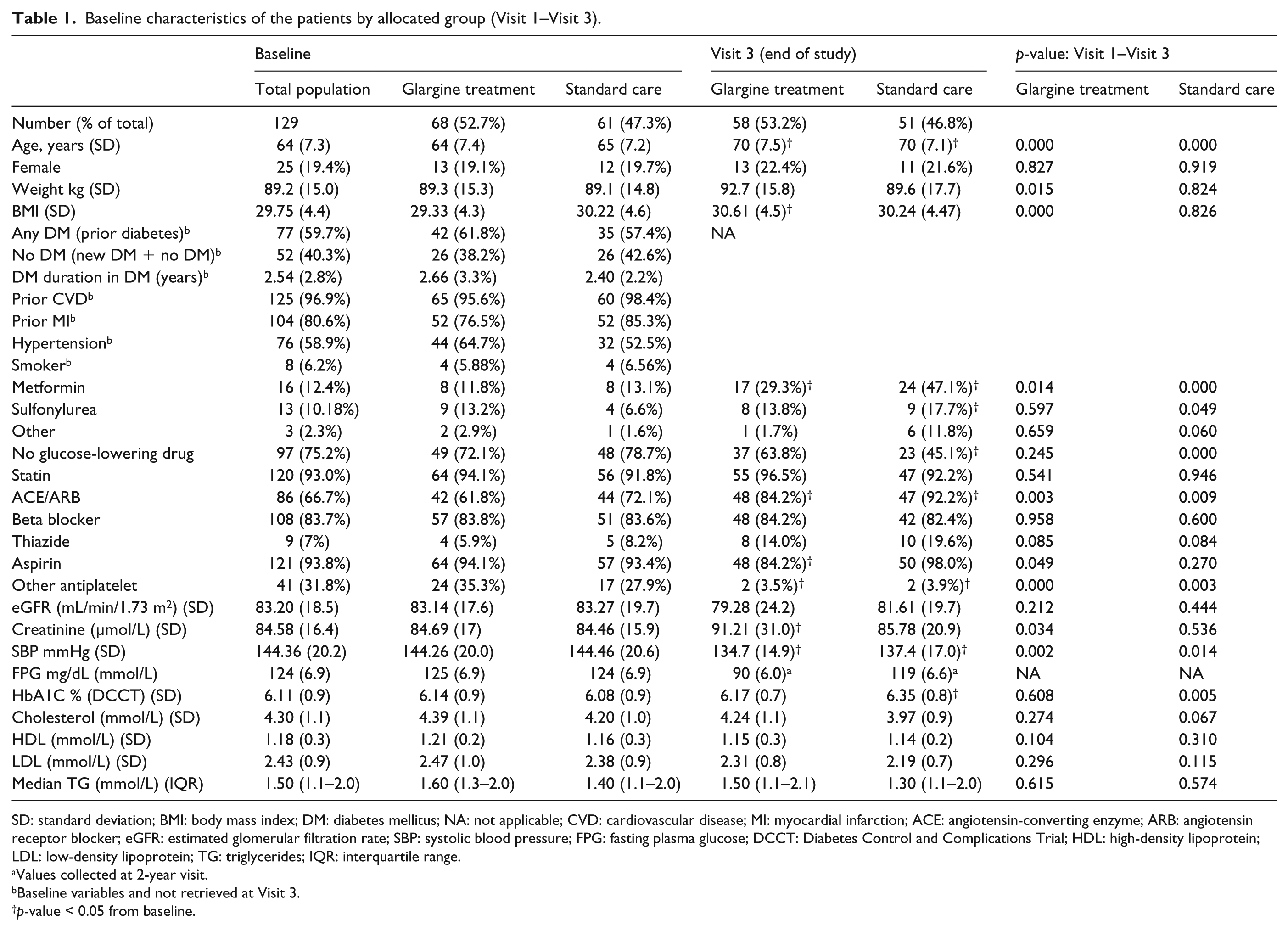
SD: standard deviation; BMI: body mass index; DM: diabetes mellitus; NA: not applicable; CVD: cardiovascular disease; MI: myocardial infarction; ACE: angiotensin-converting enzyme; ARB: angiotensin receptor blocker; eGFR: estimated glomerular filtration rate; SBP: systolic blood pressure; FPG: fasting plasma glucose; DCCT: Diabetes Control and Complications Trial; HDL: high-density lipoprotein; LDL: low-density lipoprotein; TG: triglycerides; IQR: interquartile range.
Values collected at 2-year visit.
Baseline variables and not retrieved at Visit 3.
p-value < 0.05 from baseline.
Time trends of clinical characteristics, fibrinolytic variables
In the IG group, body mass index (BMI) increased, systolic blood pressure (SBP) decreased, the use of metformin and angiotensin-converting enzyme inhibitors (ACEIs) or angiotensin receptor blockers (ARBs) increased whereas HbA1c levels did not change. In the SC group, SBP decreased, the use of metformin and sulfonylurea increased and the HbA1c level increased, while treatment with metformin and sulfonylurea increased and the proportion of patients without glucose-lowering drugs decreased. HbA1c was lower in the IG than in the SC group at Visit 2 (5.7% and 6.1%; p = 0.001), but not at the end of the study (6.2% and 6.4%; p = 0.22). There was no significant correlation between HbA1c levels and fibrinolytic markers or vWF. HbA1c levels over time are illustrated in Figure 1.
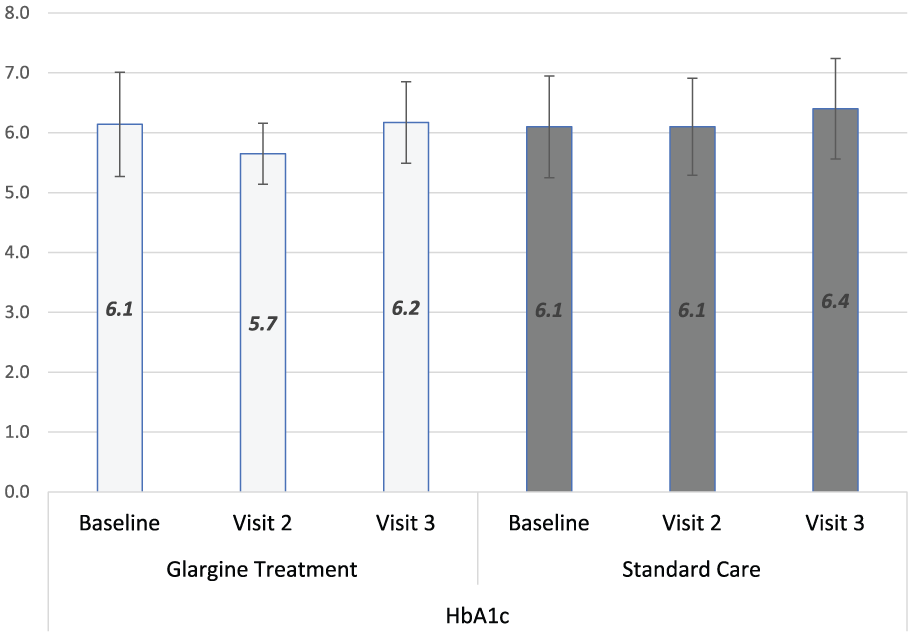
HbA1c levels Visit 1–Visit 3, glargine treatment versus standard care.
At 2 years, fasting plasma glucose was 90 mg/dL (6.0 mmol/L) in the glargine group and 119 mg/dL (6.6 mmol/L) in the standard group. Thereafter, fasting plasma glucose was not recorded in the standard treatment group. The glargine treatment group maintained the fasting plasma glucose levels below 94 mg/dL throughout the study. Although BMI increased in patients in the IG group, there was no significant difference in body weight between the two groups by the end of the study (92.7 and 89.6 kg; p = 0.337). Patients in the SC group were more frequently prescribed aspirin (84% and 98%; p = 0.013) and metformin (29% and 47%; p = 0.056) at Visit 3 than those in the IG group. Levels of fibrinolytic variables and vWF and their changeover times are shown in Table 2 and in Figure 2(a)–(e).
Levels of fibrinolytic variables and vWF and their delta values during the study (Visit 1–Visit 2–Visit 3).
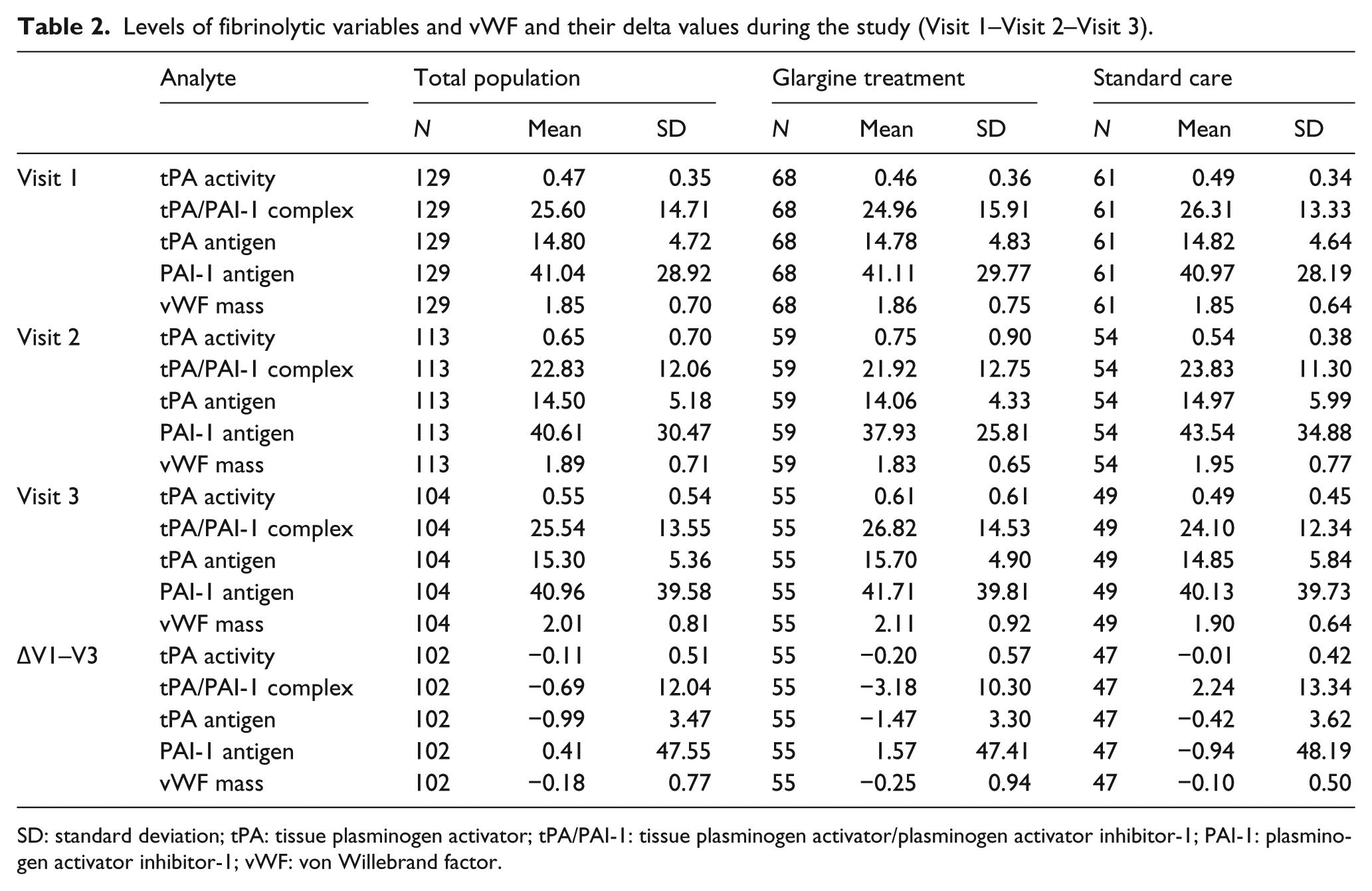
SD: standard deviation; tPA: tissue plasminogen activator; tPA/PAI-1: tissue plasminogen activator/plasminogen activator inhibitor-1; PAI-1: plasminogen activator inhibitor-1; vWF: von Willebrand factor.
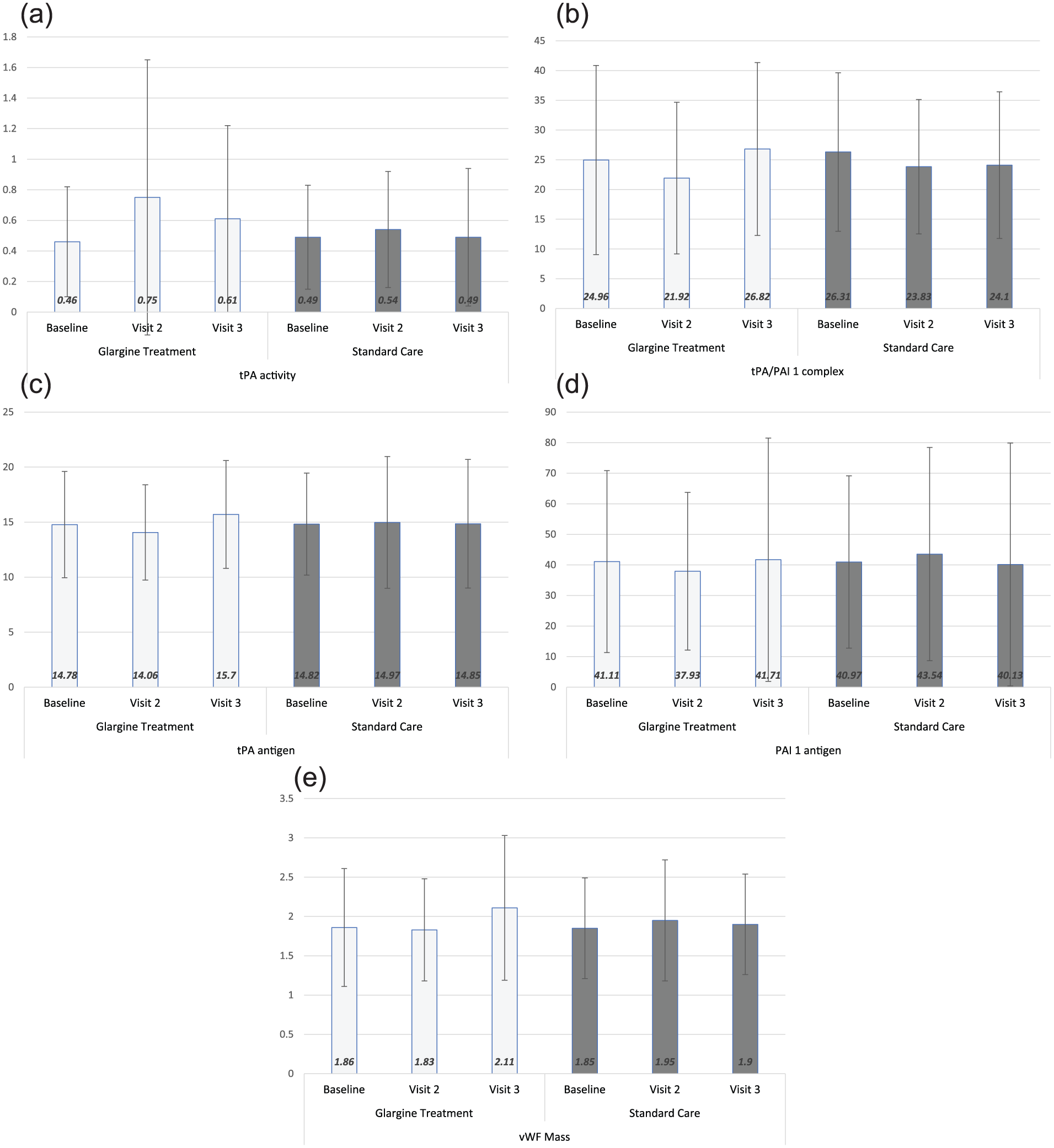
Levels of markers of fibrinolysis and vWF, Visit 1–Visit 3, glargine treatment versus standard care: (a) tPA activity, (b) tPA/PAI-1 complex, (c) tPA antigen, (d) PAI-1 antigen and (e) vWF mass.
Allocation to IG group and effects on fibrinolysis
Between-group analysis
There were no significant differences between the log-mean delta levels (Visit 1–Visit 2) of the fibrinolytic variables or vWF between the two treatment groups, as revealed by Table 3. The log-mean values (Visit 1–Visit 3) of tPAact and tPA/PAI-1 complex differed significantly between the treatment groups while PAI-1, tPAag and vWF did not (Table 3).
Difference in delta values. Visit 1–Visit 2 and Visit 1–Visit 3 in glargine versus standard care.
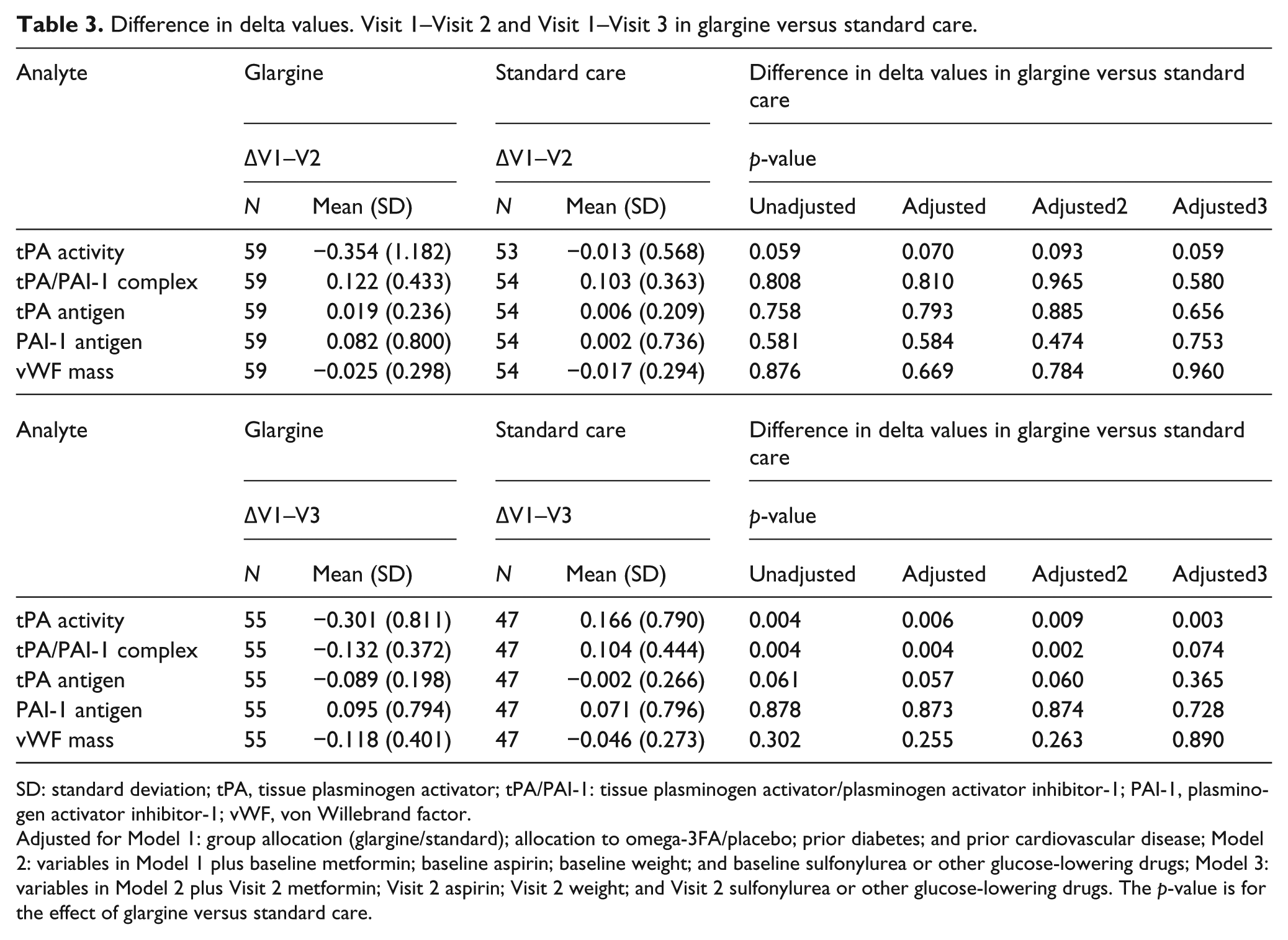
SD: standard deviation; tPA, tissue plasminogen activator; tPA/PAI-1: tissue plasminogen activator/plasminogen activator inhibitor-1; PAI-1, plasminogen activator inhibitor-1; vWF, von Willebrand factor.
Adjusted for Model 1: group allocation (glargine/standard); allocation to omega-3FA/placebo; prior diabetes; and prior cardiovascular disease; Model 2: variables in Model 1 plus baseline metformin; baseline aspirin; baseline weight; and baseline sulfonylurea or other glucose-lowering drugs; Model 3: variables in Model 2 plus Visit 2 metformin; Visit 2 aspirin; Visit 2 weight; and Visit 2 sulfonylurea or other glucose-lowering drugs. The p-value is for the effect of glargine versus standard care.
Allocation to insulin treatment did not significantly (tPAact, p = 0.999; tPA/PAI-1 complex, p = 0.364; tPAag, p = 0.947; PAI-1ag, p = 0.602 and vWF, p = 0.620) influence any of the values of the fibrinolytic variables or vWF neither unadjusted nor after adjustments for (a) allocation to glargine/SC, (b) allocation to omega-3FA/placebo, (c) any diabetes, (d) prior CVD, (e) metformin, (f) aspirin, (g) weight and (h) sulfonylurea or other glucose drug [(e)–(h): time dependent].
Within-group analysis
Within-group analysis during the whole study period (V1–V2–V3) suggested a significant increase in tPA/PAI-1 complex (p < 0.001), tPA antigen (p = 0.006) and vWF (p = 0.032) in the IG group, with no significant effect on tPAact and PAI-1. In the SC group, there were no significant changes in any of the fibrinolytic variables or vWF.
The within-group development over time for log-transformed values of the different expressions for fibrinolysis and vWF are displayed in Supplementary Table 1 for the IG group and in Supplementary Table 2 for the SC group.
Discussion
The main finding of this substudy to ORIGIN was that the main hypothesis that IG treatment would reduce tPA antigen by at least 20% over time compared to standard glucose-lowering therapy was not confirmed. Moreover, allocation to insulin did not induce any significant changes in the markers of fibrinolysis or vWF. There were higher levels of tPAag and tPA/PAI-1 complex in the IG group in the within-group analysis. As both tPAag and tPA/PAI complex increased and there were no significant changes of PAI-1 and tPAact, no conclusion can be made of an improved fibrinolysis in the IG group. In the within-group analysis, vWF increased significantly in the IG group, a result that should be interpreted cautiously and be regarded as a hypothesis-generating finding.
To the best of our knowledge, this is the first study to compare the long-term impact of insulin treatment compared to standard glucose-lowering therapy on endothelial-derived components of the haemostatic and fibrinolytic systems in people with dysglycaemia and high risk for cardiovascular events. The lack of a difference between the two treatment groups is in line with the absence of an impact of IG on cardiovascular outcomes as seen in the ORIGIN trial. 15 Insulin therapy does at least partly restore insulin-stimulated endothelial function in patients with type 2 diabetes and ischaemic heart disease. 10 We hypothesized that this might have had a favourable impact on the fibrinolytic balance through an improved endothelial function, but this assumption was not supported in this study. One possible mechanism might be that insulin stimulates both endothelin-1 and nitric oxide activity, 16 although it is unclear how an imbalance in between them would affect the endothelial-derived haemostatic factors.
The levels of tPAact were lower and tPAag and PAI-1 were higher in the present patient population than among healthy participants in the Northern Sweden MONICA survey.17,18 Our finding is consistent with a hypofibrinolytic state, characterized by low tPAact, increased tPAag and PAI-1, and this pattern was observed both at baseline and, with limited changes, throughout the period of follow-up. However, there were no significant deteriorations of the fibrinolytic components over time despite an expected increase in fibrinolytic variables and vWF as result of ageing during the study. In the northern Swedish MONICA project, for example, PAI-1 increased over 9 years by 75% and 90% in males and females, respectively. 19 This maintenance of fibrinolytic activity may have been due to the increased use of ACEIs or metformin in both treatment groups. Thus, in survivors of myocardial infarction enalapril compared to placebo is associated with significantly lower levels of tPAag. 20 Among blood pressure–lowering drugs, ACEIs have indeed been found to have the most beneficial effect on the fibrinolytic components. 21
Strengths and limitations
The size of the study and the fact that a representative number of the components of the fibrinolytic systems and vWF were followed over a long time period in a randomized trial is a considerable strength. Still, there are limitations whereof the limited number of patients is one, especially in consideration of the large the number of potential confounders that may affect both the exposition and outcome and which, by natural reasons could not be kept constant over time. Most important for the fibrinolytic system are changes in BMI and treatment with metformin.22–24 The increase in BMI in the IG group may have impacted the fibrinolytic system. 23 Weight gain may also increase inflammation, 25 which is closely linked to a deterioration of the fibrinolytic balance. 26 These confounders may have had a greater impact on changes of the fibrinolytic variables and vWF than IG treatment per se. Patients with more than one episode of severe hypoglycaemia was 1.00 per 100 person years in the IG group and 0.31 per 100 person years in the SC group (p < 0.001) and the corresponding incidence of no severe, symptomatic hypoglycaemia confirmed by a self-measured glucose level of 54 mg per dL (3.0 mmol/L) or less was 9.83 and 2.68 per 100 person years in the IG and SC groups, respectively (p < 0.001). 15 Information of hypoglycaemic effects on the fibrinolytic systemic is scarce in type 2 diabetes. In other situations, where the sympathomimetic system is activated, such as acute exercise, the effect on fibrinolytic activity is short with normalization within 24 h.27,28 Due to the relatively low rate of hypoglycaemia, it is unlikely that hypoglycaemic events have significant impact on the level markers of fibrinolysis.
Conclusion
In people with dysglycaemia and other cardiovascular risk factors, basal IG does not improve the levels of markers of fibrinolysis or vWF compared to standard glucose-lowering treatments.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.R. reports lecture honoraria from Lilly, Novo Nordisk and Roche. K.B. reports research grant reimbursement for the study, Sanofi-Aventis. H.C.G. reports consulting honoraria from Sanofi-Aventis, Lilly, Roche, Novo Nordisk, Bayer, Glaxo-Smith-Kline, Novartis, Bristol-Myers-Squibb and AstraZeneca; lecture honoraria from Sanofi-Aventis and Bayer; grants from Takeda and funds given to his institution for research or educational initiatives from Sanofi-Aventis, Lilly, Novo Nordisk, Boehringer Ingelheim, Bristol-Myers-Squibb and AstraZeneca. J.H-B., S.F.L. and M.O. reported no conflict of interest. L.G.M. reports research grants from The Swedish Heart-Lung Foundation, The Swedish Diabetes Foundation, MSD, Bayer AG, Sanofi-Aventis and lecture honoraria from MSD.
Funding
The ORIGIN trial was funded by Sanofi-Aventis. This study received funding from the Swedish Diabetes Foundation and the Swedish Heart-Lung Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
