Abstract
Introduction:
Olfactory dysfunction is suggested to be a clinical manifestation of central diabetic neuropathy. The aim of the study was to assess olfactory function in adult patients with type 1 diabetes.
Materials and methods:
A total of 106 patients with type 1 diabetes and 30 healthy subjects were included in the study. We evaluated the metabolic control of diabetes and the presence of chronic complications. Olfactory function was assessed with Sniffin’ Sticks.
Results:
We found a negative correlation between olfactory identification scores and body mass index (Rs −0.2; p = 0.04) and triglycerides (Rs = −0.2; p = 0.04). We showed lower olfactory identification scores in neuropathy group versus non-neuropathy group [8 (interquartile range, 7–9) vs 10 (interquartile range, 9–11) points; p = 0.005]. In multivariate linear regression, impaired olfaction was independently associated with neuropathy (beta, −0.3; p = 0.005). In multivariate logistic regression, diabetes duration (odds ratio, 1.06; 95% confidence interval, 1.00–1.11; p = 0.04) and olfactory identification score (odds ratio, 0.61; 95% confidence interval, 0.43–0.85; p = 0.003) were independently associated with neuropathy.
Conclusion:
Olfactory dysfunction is observed in patients with type 1 diabetes and diabetic peripheral neuropathy.
Introduction
Type 1 diabetes (T1D) is a chronic disease requiring complex and demanding treatment based on insulin. One of the treatment goals is the prevention of chronic microvascular complications of the disease such as retinopathy, nephropathy or neuropathy. 1 Prolonged hyperglycaemia can lead to the development of chronic complications of diabetes in several ways. These include activation of the protein kinase C pathway, formation of advanced glycation end-products (AGEs), accumulation of sorbitol through the aldose reductase pathway and activation of the hexosamine pathway and the polyADP-ribose polymerase (PARP) pathway, which regulates several inflammatory response gene expressions and causes neuronal dysfunction.2–4 Acute and chronic hyperglycaemia and high variability of glycaemia are associated with low-grade inflammation and oxidative stress. A generation of reactive oxygen species (ROS) has been proposed as the key element of different pathogenic pathways. 5
Diabetic neuropathy is the most common chronic complication with axonal atrophy, demyelination, nerve fibre loss and blunted regeneration of nerves identified as known pathologies. Although peripheral symmetric polyneuropathy is the most prevalent clinical manifestation, in some people with diabetes, central forms of neuropathy, affecting grey matter and cranial nerves, may also be diagnosed. Olfactory dysfunction could be one of the clinical manifestations of central neuropathy. The studies reporting the relationship between diabetes and olfactory function are focused mainly on people with type 2 diabetes. 6
The aim of this study was to assess olfactory function in adult patients with T1D.
Methods
The study was conducted in patients with T1D referred to the outpatient clinic of Department of Internal Medicine and Diabetology, Poznan University of Medical Sciences, in cooperation with the Department of Otorhinolaryngology of the City Hospital of Raszeja in Poznan. It was approved by the local ethics committee and conducted according to the guidelines of the Declaration of Helsinki on biomedical research involving human subjects. All participants provided written informed consent before enrolment in the study. Exclusion criteria were diabetes duration less than 5 years, age above 65 years, diabetic ketoacidosis at the time of enrolment and use of drugs affecting nasal mucosa (antihistamine, local steroids). All patients underwent a complete physical examination with anthropometric and blood pressure measurements. The patients underwent ear, nose and throat (ENT) examination with nasal endoscopy to exclude other factors disabling sense of smell. In all, 14 of 120 initially examined patients were excluded due to nasal pathologies potentially influencing the sense of smell (acute or chronic sinusitis, rhinitis, nasal polyposis, severely deviated nasal septum).
We finally included 106 subjects. They were asked not to drink anything but water or eat or smoke cigarettes at least 1 h prior to examination. Olfactory function was assessed with Sniffin’ Sticks. For the assessment of odour identification, 12 pens dispensing different odours were used. After removing the cap, the tip of the pen was placed 2 cm below patient’s nostrils for 2 s. Each of the 12 odours was presented with the list of four possible answers. The patient should select 1, which best described each odour for every pen. The possible scores ranged from 0 to 12, scores 11–12 were considered normal olfactory function (normosmia), while scores 0–10 signified olfactory dysfunction. We assessed the metabolic control of diabetes and the presence of diabetic retinopathy, peripheral neuropathy and diabetic kidney disease based on standard diagnosis criteria.7–9 Blood samples were obtained after overnight fast. HbA1c, serum lipids and creatinine were measured with standard techniques. The control group consisted of 30 healthy people. The clinical characteristics of the study group are presented in Table 1.
Characteristics of the study participants [number or median (IQR)].
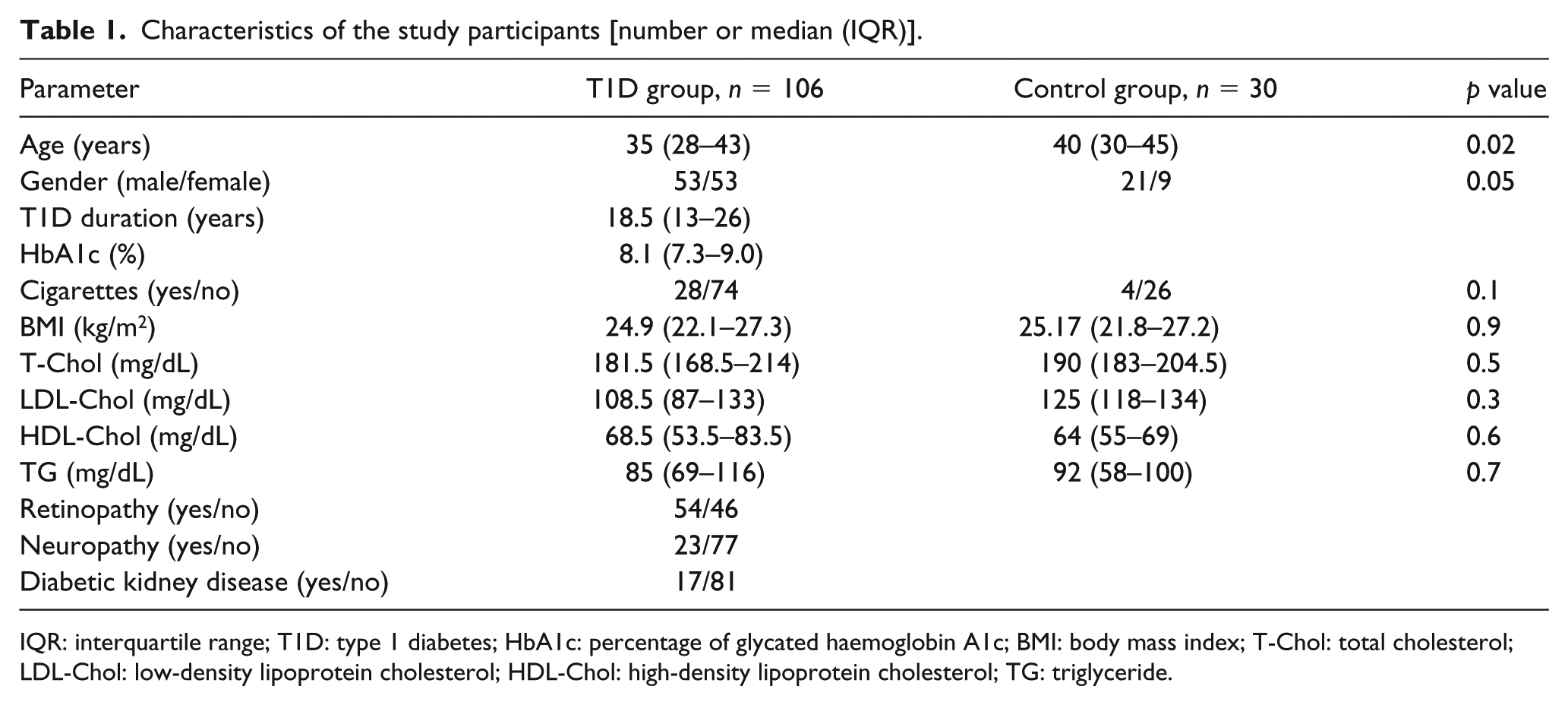
IQR: interquartile range; T1D: type 1 diabetes; HbA1c: percentage of glycated haemoglobin A1c; BMI: body mass index; T-Chol: total cholesterol; LDL-Chol: low-density lipoprotein cholesterol; HDL-Chol: high-density lipoprotein cholesterol; TG: triglyceride.
Statistical analysis
Results are presented as median [interquartile range (IQR), 25%–75%] for continuous variables or number (proportion) of patients for categorical variables. The relation between olfactory identification scores and laboratory parameters was assessed using Spearman’s rank correlation analysis, and Mann–Whitney U test was used to analyse the differences in olfactory scores between groups according to diagnosed chronic complications. Chi-square test was used to compare frequencies. A p value of less than 0.05 was considered statistically significant. Stepwise multivariate linear regression was used to assess factors related to olfactory function. In the logistic regression analysis, we searched for predictors of neuropathy and retinopathy. We fitted the univariate regression models to identify significant predictors of neuropathy and retinopathy and subsequently used them to select a multivariate model through stepwise selection using a p value of <0.05 as a threshold for adding a variable. Statistical analysis was performed using STATISTICA 10.
Results
Hyposmia was found in 67.9% (n = 72) of patients with T1D compared to 53.3% (n = 16) in the control group (p = 0.9). Olfactory identification score correlated negatively with body mass index (BMI) and triglyceride (TG) (Table 2). Comparison of odour identification scores between groups with and without diagnosed chronic microvascular complications showed lower scores in neuropathy group versus non-neuropathy group [8 points (IQR, 7–9) vs 10 points (IQR, 9–11); p = 0.005] and lower scores in retinopathy versus non-retinopathy group [9 points (IQR, 8–11) vs 10 points (IQR, 9–11); p = 0.03]. Stepwise multivariate linear regression analysis indicated neuropathy as independent predictor of olfactory function (Table 3). In the univariate logistic regression, we found that age, diabetes duration and olfactory identification score were significant predictors of neuropathy. In the multivariate logistic regression analysis, the best model included diabetes duration [odds ratio (OR), 1.06; 95% confidence interval (CI), 1.00–1.11; p = 0.04] and olfactory identification score (OR, 0.61; 95% CI, 0.43–0.85; p = 0.003) independently associated with the presence of neuropathy, after adjusting for gender, HbA1c, BMI, cigarette smoking and high-density lipoprotein (HDL). In addition, we performed a multivariate logistic regression analysis for retinopathy. In this analysis, diabetes duration (OR, 1.45; 95% CI, 1.22–1.73; p = 0.00003), BMI (OR, 0.76; 95% CI, 0.61–0.95; p = 0.02), HbA1c (OR, 2.36; 95% CI, 1.12–4.97; p = 0.01) and olfactory identification score (OR, 0.48; 95% CI, 0.28–0.83; p = 0.005) were identified as independent predictors of retinopathy after adjusting for gender, cigarette smoking and HDL.
Correlation between various laboratory parameters and olfactory function (Spearman’s rank correlation analysis).
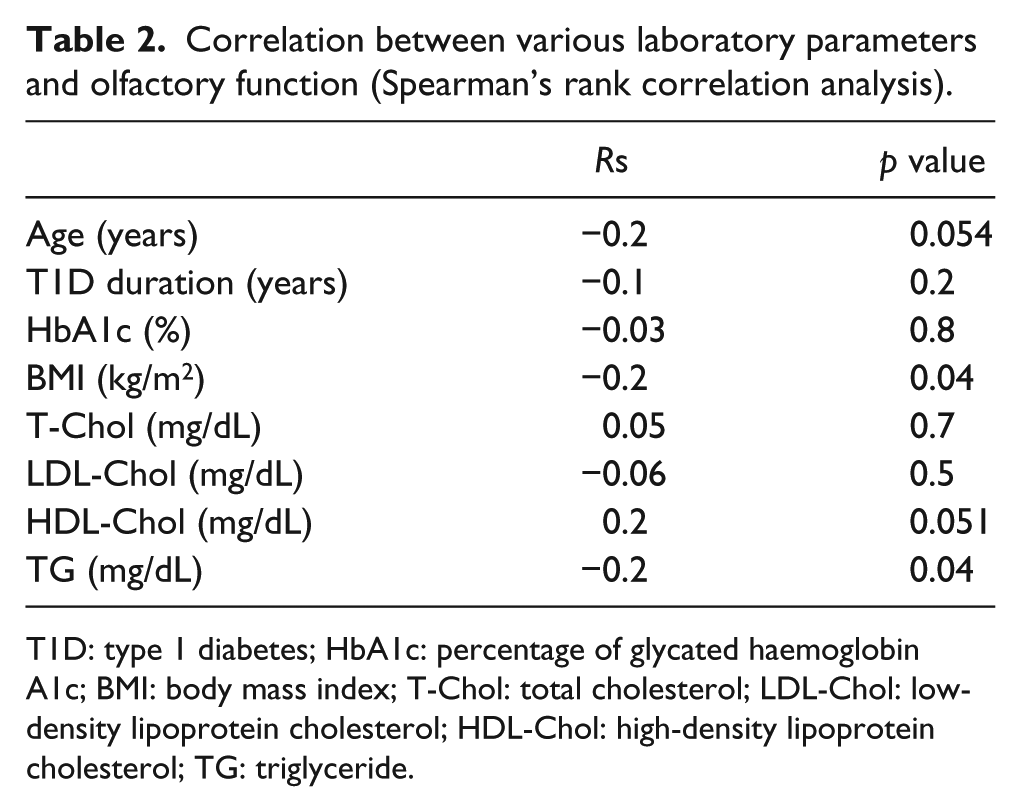
T1D: type 1 diabetes; HbA1c: percentage of glycated haemoglobin A1c; BMI: body mass index; T-Chol: total cholesterol; LDL-Chol: low-density lipoprotein cholesterol; HDL-Chol: high-density lipoprotein cholesterol; TG: triglyceride.
Stepwise multivariate linear regression analysis for olfactory identification score.
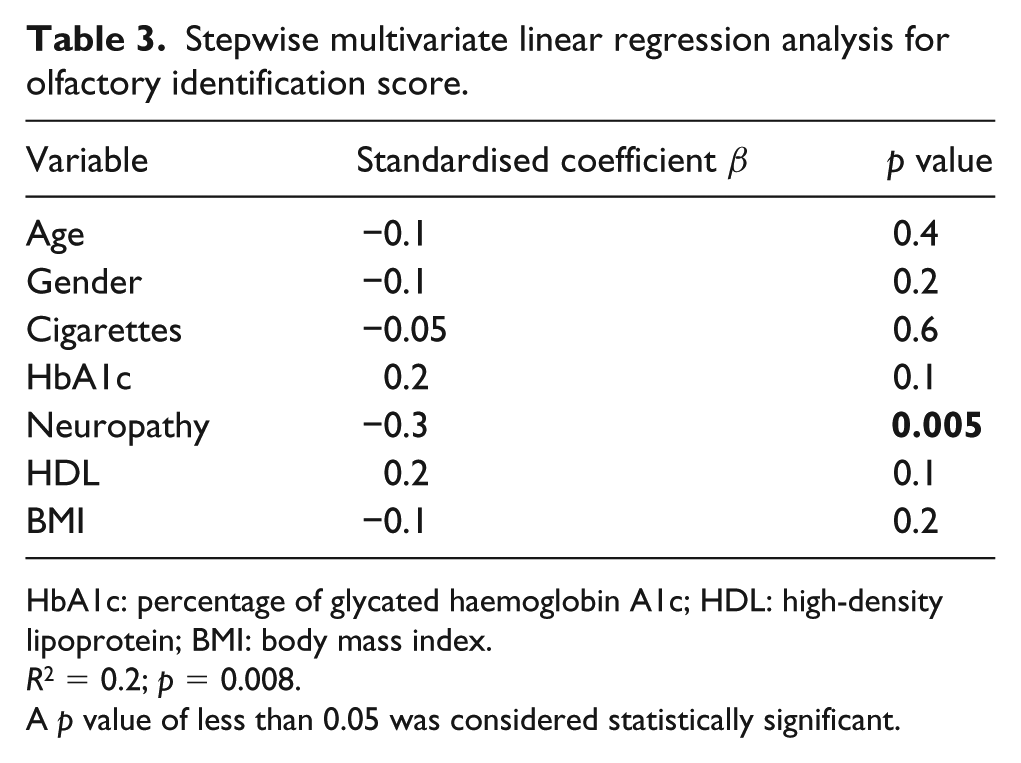
HbA1c: percentage of glycated haemoglobin A1c; HDL: high-density lipoprotein; BMI: body mass index.
R2 = 0.2; p = 0.008.
A p value of less than 0.05 was considered statistically significant.
Discussion
Smell disorders are common in general population. Prevalence increases with age, affecting 1%–2% of younger subjects to above 50% in population over 65 years of age. 10 As age is considered to have a negative impact on the olfactory function, we did not achieve statistical significance of this relation in our study. 11 The loss of smell may develop due to other disorders, like upper respiratory tract infections, head trauma, multiple sclerosis, cancer, neurodegenerative disorders or diabetes.5,12–16 This study provides information that enables the assessment of olfactory function in relation with chronic microvascular complications of T1D. First, we observed that patients with T1D in general did not show any significant difference in olfactory function compared to the control group, which is congruent with other studies. 10 We must state that our control group was older than T1D group, and this is a limitation in the study. However, the prevalence of olfactory dysfunction in our study in both groups was higher than commonly reported in other studies. 12 During the last decades, a number of standardised olfactory tests have been developed. They differ in terms of methodology of odour identification and preset threshold levels. Thus, the comparison of obtained results may not be straight. 17
When analysing the T1D group, we demonstrated a negative correlation between olfactory identification scores and BMI, which previously published data do not confirm.15–18 One may postulate that BMI, commonly elevated due to excess of body fat, might be particularly detrimental. Excessive visceral adiposity in particular causes increased concentrations of free fatty acids in the plasma and induces a proinflammatory state by secreting adipokines, thus sustaining the activation of some pathogenic pathways contributing to the development of chronic complications of diabetes. 19 Yet, the proinflammatory effect of adipose tissue is characteristic for type 2 diabetes rather than type 1, where in type 2 diabetes overweight and obesity constitute a key phenotype of the disease. Furthermore, subjects included in our study were not obese, with the median BMI at the verge of overweight (24.8 kg/m2). We showed the negative correlation of TG concentration to olfactory function in people with T1D. As lipid disorders contribute to development of atherosclerosis and macrovascular diseases, TG levels may particularly reflect metabolic control of diabetes and are linked to hyperglycaemia. 20 There was no correlation between the metabolic control of diabetes expressed by HbA1c and olfactory function, in line with previous studies. 21 However, we demonstrate the relation of olfactory function to diagnosed chronic complications of diabetes. Patients diagnosed with neuropathy or retinopathy gained significantly lower olfactory identification scores compared to patients with no chronic complications. In this context, coexisting with other forms of microvascular complications, olfactory dysfunction may be perceived as a central form of neuropathy. Mehdizadeh Seraj et al. 22 revealed olfactory impairment in 60% of patients with diabetes, which is comparable to our study, yet they did not show the correlation of olfactory function to any microvascular complication of diabetes. We demonstrated that neuropathy was independently associated with olfactory status in people with T1D. Olfactory identification score and diabetes duration remained the independent predictors of neuropathy. Interestingly, olfactory identification score was identified as an independent predictor of retinopathy, together with diabetes duration, HbA1c and BMI. Retinopathy is generally considered as a microvascular complication which develops as a result of chronic hyperglycaemia. However, the neuronal apoptosis and reactive gliosis are recently postulated as early changes in diabetic retinopathy. This phenomenon is described as a neurodegeneration and suggests that diabetic retinopathy should be recognised as a neurovascular complication of diabetes. 23 In T1D, olfactory impairment is assumed to develop due to oxidative stress induced by hyperglycaemia 24 and may constitute an indicator of the function of central nervous system in general. Therefore, the assessment of olfaction may be useful in terms of diagnosing central neuropathies in T1D.
The advantage of our study is that it focuses on selected type of diabetes, exclusively on adult people with T1D.
Conclusion
In this study, we define the association of olfactory function with diabetes complications. Higher degree of olfactory dysfunction is observed in patients with T1D and diagnosed neuropathy or retinopathy. As the mechanisms of this phenomenon and a relation of olfactory function to chronic microvascular complications of T1D are still uncertain, the olfactory impairment may be an indicator of the central neuropathy in T1D.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
