Abstract
Purpose:
To determine the effects of a 12-week cardiac rehabilitation programme of aerobic and resistance exercise training on arterial stiffness, peak calf vasodilatory reserve, and haemostatic markers in patients with type 2 diabetes.
Methods:
Observational cohort study examining effects of 12 weeks of exercise training in 23 subjects (13 men, 10 women; mean age of 56.1 ± 10.1 years) with type 2 diabetes mellitus. Subjects performed exercise training for 12 weeks [aerobic training 5 days/week, 70%–75% peak cardiovascular fitness (VO2peak) and resistance training 2–3 days/week, 60% of one repetition maximum]. Vascular stiffness (pulse-wave velocity), augmentation index, peak calf vasodilatory reserve, and VO2peak were measured pre- and post-exercise training. Secondary outcomes included heart rate variability and haemostatic measures.
Results:
VO2peak increased by 16% (20.1 ± 5.5 vs 23.2 ± 8.8 mL/kg/min, p = 0.002) and abdominal circumference was reduced (101.9 ± 13.3 vs 97.9 ± 12.7 cm, p < 0.03). Vascular function was improved including central arterial stiffness (central pulse-wave velocity: 8.44 ± 1.75 vs 8.02 ± 1.60 m/s, p = 0.026) and the aortic augmentation index (21.7 ± 10.6% vs 18.3 ± 12.6%, p = 0.005); peak calf vasodilatory reserve increased from 30.3 ± 10.6 mL/100 mL/min to 38.0 ± 15.3 mL/100 mL/min (p = 0.04). No changes were seen in heart rate variability, blood lipids, glycated haemoglobin and C-reactive protein.
Conclusion:
A 12-week cardiac rehabilitation programme of aerobic and resistance training significantly reduces arterial stiffness and improves aerobic fitness in individuals with type 2 diabetes mellitus.
Introduction
It is widely accepted that combined aerobic and resistance training improves blood glucose control, aerobic fitness, body composition,1,2 heart rate variability (HRV) and high-sensitivity C-reactive protein (hs-CRP) levels.3,4 A programme of combined aerobic and resistance training may offer greater benefits than aerobic exercise alone for people with diabetes.5,6 However, data from the Behavioral Risk Factor Surveillance System conducted by the Centers for Disease Control and Prevention report that only 12% and 41% of US adults with diabetes meet the American Diabetes Association guidelines for resistance and aerobic exercise, respectively. 7 Moreover, participation was related to previous education in diabetes management. Despite a publicly funded health care system in Canada, only 20.6% of people with newly diagnosed non-gestational diabetes accessed a diabetes education programme within 6 months of diagnosis. 8 Notwithstanding readily available and publicly funded health care, the dearth of lifestyle programmes to support people with a diagnosis of diabetes has prompted some to advocate for cardiac rehabilitation (CR) programmes to provide lifestyle modification and risk factor management. 9 In addition to usual clinical practice of pharmacological treatment and visits with a diabetes educator, people with diabetes are likely to benefit from more intensive behavioural therapy. The aerobic and resistance training components as well as risk factor interventions and assessments offered by an interprofessional health care CR team can reduce vascular risk in this population. 10 The cardiopulmonary exercise stress test is a recommended standard of care in most CR programmes but is not routinely administered in most traditional diabetes education programmes. The stress test informs an individualized target training intensity and mitigates adverse outcomes by identifying clinically relevant abnormalities. CR programmes also include supervised exercise sessions where individuals receive guidance on the progression of resistance and aerobic exercise parameters for continued improvement and adherence to behaviour change. However, while the benefits of a comprehensive CR programme are well established in the cardiac population,11,12 there are limited prospective data describing the effects of a CR programme on cardiovascular fitness and arterial stiffness, which remain two important outcome measures for people with type 2 diabetes mellitus (DM2).
Arterial stiffness increases gradually with age; 13 however, in those with DM2 it is accelerated and is an independent risk factor for premature cardiovascular disease (CVD) and mortality.14–16 The general disturbances to vascular function in DM2 are likely multi-factorial, including increases in inflammatory activity,17,18 reduced large artery elasticity and autonomic nervous system activation. 19 Aggressive lifestyle intervention may postpone the manifestation of DM2 in high-risk individuals and the development of premature arterial stiffening. 20 In healthy individuals, our group 21 and others22–24 have demonstrated that aerobic fitness is associated with lower arterial stiffness.25,26 Improving cardiovascular fitness is also an important outcome for people with DM2, as low levels of aerobic fitness are associated with increased risk of CVD mortality in men with diabetes. 27 Therefore, given the importance of optimizing cardiovascular fitness, and the association between vascular stiffness, accelerated CVD and autonomic dysfunction with progression of DM2,28,29 further study regarding the effects of a CR exercise intervention on vascular function, arterial stiffness and cardiovascular fitness is warranted.
Therefore, the aim of this study was to determine the effects of a 12-week CR programme of aerobic and resistance exercise training on aerobic fitness, vascular function and selected haemostatic factors in patients with DM2 with no known CVD. We hypothesized that short-term combined aerobic and resistance training would improve aerobic fitness and measures of arterial stiffness and vascular function.
Research design and methods
Study design
This was a 12-week exercise intervention study in a cohort of subjects with DM2 participating in the Diabetes, Exercise and Healthy Lifestyle Programme at Toronto Rehab (TR). The programme is based on the traditional CR programme model that includes group-based weekly supervised sessions and support from an interprofessional team of physicians, nurses, kinesiologists, dietitians, a psychologist and a social worker. Along with individualized exercise, nutrition, and psychosocial support, 13 group education sessions were offered including the benefits of exercise, risk factor profiling, goal setting, lifestyle change and motivation, targets for diabetes control, exercise safety/exercise in hot and cold weather, stress and diabetes, the role of food and meal planning, portion distortion, understanding food labels, cholesterol and fibre, foot care, sleep apnoea and medications.
All subjects were evaluated for aerobic fitness and anthropometric measurements on a different day than for vascular assessments. Participants were asked to refrain from caffeine and alcohol 12 h before vascular assessments to minimize confounding effects. Given this was a diabetic population, we were not able to control for diet; however, all assessments were conducted at least 3 h following a meal or prior to any exercise. All tests were performed at the same time of day, before and after the chronic exercise intervention.
Participants
In total, 27 adults with DM2 (aged 33–72 years) were recruited from the TR programme, referred to the programme by their family physician. They were not engaged in any significant physical activity prior to recruitment. All subjects provided informed consent, and the study was approved by the local research ethics boards, conforming to the Helsinki Declaration involving human subjects. The inclusion criteria included a diagnosis of DM2 according to the criteria of the Canadian Diabetes Association, treatment by diet and/or oral hypoglycaemic agents, men aged >30 years and women confirmed to be post-menopausal (more than 1 year since menses). Subjects were excluded from the study if they were taking insulin, had documented coronary artery disease, chronic inflammatory disease, uncontrolled hypertension, orthostatic hypotension, ankle-brachial index <0.8, foot ulcers, neuropathy, peripheral vascular disease, the presence of implanted electrical device such as a pacemaker or defibrillator, cardiac arrhythmia and/or were unable to exercise as per the TR protocol. Pre-menopausal women or those taking hormone replacement therapy were also excluded. Medications including oral hypoglycaemic agents, antihypertensive, lipid lowering, and anticoagulant medications were maintained throughout the study.
Exercise testing
A resting 12-lead electrocardiogram (ECG), medical history and anthropometric measures were collected prior to each symptom-limited cardiopulmonary exercise stress test. Exercise testing was performed on an electrically braked cycle ergometer (Ergoline 800 EL) with a pedalling rate of 60 r/min, with resistance increased every minute by 16.7 W until voluntary fatigue or signs/symptoms appeared that necessitated termination of the test. 30 Breath-by-breath gas samples were collected and averaged over a 20 s period via calibrated metabolic cart (Vmax SensorMedics 2900, Yorba Linda, CA, USA) to determine cardiovascular fitness (VO2peak) with continuous monitoring of 12-lead ECG (Marquette Case 80, GE, UK), and blood pressure measures taken every 2 min. Indications for discontinuing the test included achieving physiological maximum or if the patient had adverse clinical signs or symptoms.
Anthropometric measures
Height (m) and body mass (kg) were measured in exercise clothing prior to the exercise test and with shoes removed. Body fat was measured by bioelectrical impedance (Tanita TBF-300A, Tokyo, Japan), with bare feet. Waist circumference was measured at the narrowest part of the torso between the iliac crest and xiphoid process or at the level of the iliac crest after normal exhalation. 30
Vascular function
Pulse wave velocity and aortic augmentation index
Triplicate measures of supine arterial blood pressure were obtained 1 min apart using an automated sphygmomanometer (Dinamap 1846 SX Monitor; Critikon Inc., Tampa, FL, USA) following 5 min of supine rest. Measures of arterial stiffness were then assessed using a commercial device with proprietary software (SphygmoCor; AtCor Medical, Sydney, Australia). Arterial pressure waveforms were recorded by a tonometer probe (Millar Instruments, Houston, TX, USA) applied to the arterial site, captured by a high-fidelity transducer and recorded for subsequent analysis. 31 The shape of the incident and reflected waves are determined by the structural and functional characteristics of the vasculature,32,33 and with increasing arterial stiffness, the incident wave is augmented by reflected waves returning to the central circulation. Aortic augmentation index (AIx) was calculated by a validated transfer function32,33 determined from peripheral pressure waveforms collected from the right radial artery and calibrated against the supine blood pressure. In order to control for the influence of heart rate on AIx, 34 AIx data were normalized to a heart rate of 75 beats/min (AIx at 75 beats/min) by proprietary software. Following the assessment of AIx, pulse-wave velocity (PWV), defined as the speed of travel of the pulse between two sites along the arterial branch, 32 was calculated by the time delay between sampling sites determined by concurrent measurements of ECG-derived QRS complexes and arterial waveforms. Distance between sites was measured by anthropometric tape using specific anatomical landmarks. For central PWV, tonometry measures were taken at the right carotid and femoral arteries, and for peripheral PWV, at the right femoral and dorsal pedal arteries. The reliability and reproducibility of AIx and PWV have been demonstrated by others,31,35,36 and unpublished data from our laboratory indicate a within-observer mean difference for AIx, central PWV and peripheral PWV of 0.17% ± 4.31%, 0.02 ± 0.17 and 0.04 ± 0.21 m/s, respectively.
Peak calf vasodilatory reserve
Resting and peak calf blood flow was measured in the right calf by venous-occlusion strain-gauge plethysmography 21 using an indium-gallium strain gauge (Medasonics model SPG16). Concurrent measurements of heart rate and blood pressure (Finapres®; Ohmeda Inc., Englewood, CO, USA) were performed to calculate vascular conductance [blood flow/mean arterial pressure (MAP) = diastolic blood pressure (DBP) + 1/3 pulse pressure]. Following the assessment of resting blood flow, peak calf blood flow was recorded immediately following 5 min of local calf ischaemia produced by an occlusion cuff (200 mmHg) followed by ischemic local calf exercise to exhaustion. Digital data were acquired and analysed using custom software (LabVIEW; National Instruments, Austin, USA). Peak calf blood flow (mL/100 mL/min) was determined from the steepest slope from a series of time-calf volume curves obtained within 2 s of cuff release, and peak vascular conductance was calculated using the MAP recorded concurrently with maximal blood flow. The peak calf vasodilatory reserve (PCVR) was determined as the difference between resting and maximal blood flow.
Haemostatic markers
Venous blood samples were obtained in a rested state for determination of fasting plasma glucose, blood lipids, HbA1c and Hs-CRP. Blood lipids were obtained by an enzymatic method using commercial kits, and an immunoturbidimetric method was used to determine HbA1c samples. Fasting blood glucose was assessed by the glucose dehydrogenase method. To determine glucose control at the time of testing, a random grab-sample of random blood glucose (RBG) was measured in the supine position using a single drop of blood from a finger capillary (OneTouch Ultra; LifeScan, Milpitas, CA, USA) just prior to the start of 10 min of supine rest, prior to arterial stiffness assessment. RBG testing was repeated immediately after the acute exercise bout in the supine position. Samples were obtained within 1 week prior to, and following, the training programme.
Exercise training
Subjects were part of a usual care exercise programme provided to participants of the TR programme. Subjects attended one supervised and four independent or home-based aerobic sessions per week for 12 weeks. After 8 weeks of training, two sets of 15 repetitions of nine resistance training exercises were added to the aerobic training regimen at an intensity of 60% one-repetition maximum. These included five upper body (supine chest fly, lateral raise, dumbbell row, bicep curl and triceps extension), three lower body (half squat, leg curl and heel raise) and one trunk stabilizing exercise (abdominal curl ups). The initial walking prescription was set at a distance of approximately 1.6 km/day and an intensity equivalent to the ventilatory anaerobic threshold and/or 60%–75% of
Statistical analysis
All data were analysed using SPSS for Windows (Version 14; SPSS, Chicago, IL, USA). Paired t-tests with a Bonferroni correction were used to examine whether there were significant changes in variables before and after exercise training. Associations among variables were determined using the Pearson correlation coefficient. Data are presented as mean ± standard deviation (SD). A significance level of p < 0.05 was set for all analyses.
Results
Baseline characteristics
Subject characteristics including medication status are reported in Table 1. Central PWV was significantly correlated with age (r = 0.509, p = 0.013), resting blood glucose levels (r = 0.535, p = 0.008) and VO2peak (mL/kg/min) (r = −0.578, p = 0.004). Similarly, the AIx at 75 beats/min was correlated with age (r = 0.674, p < 0.001) and VO2peak (mL/kg/min) (r = −0.442, p = 0.035).
Baseline characteristics.
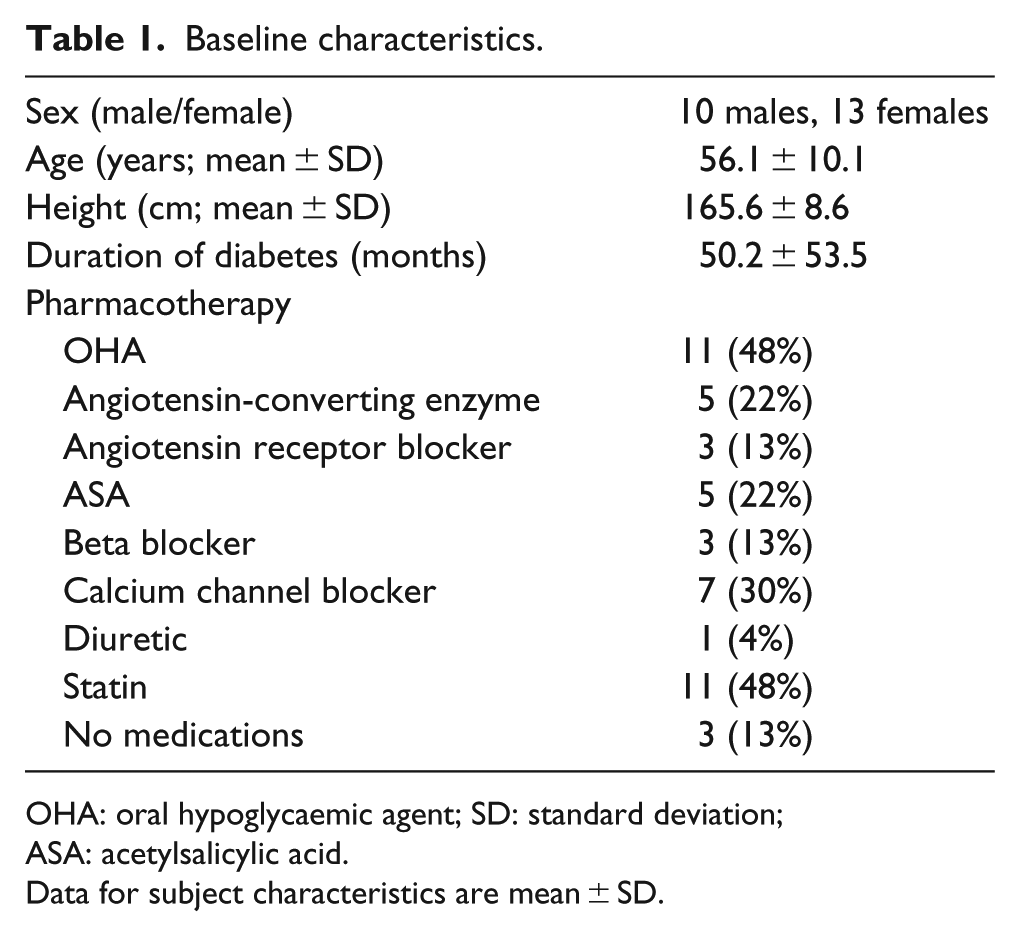
OHA: oral hypoglycaemic agent; SD: standard deviation; ASA: acetylsalicylic acid.
Data for subject characteristics are mean ± SD.
Exercise programme compliance
Of the 27 subjects recruited to the study, 2 subjects dropped out of the study because of injury unrelated to exercise (n = 1), time commitment (n = 2). Vascular assessments were not feasible in two other subjects and were removed from the analysis. In total, 23 subjects completed 12 weeks of exercise training (13 females and 10 males). Compliance to the exercise programme was determined by class attendance and records obtained from exercise diaries. The total number of expected exercise sessions was 60. Subjects attended 9.95 ± 1.66 (83%) of the 12 supervised classes and performed 33 ± 2 (69%) of 48 home exercise sessions. Exercise diaries were handed in 70.2% ± 31.9% of the time. Data compiled from the completed exercise diaries showed that subjects completed 32 ± 4.4 min of aerobic exercise 4 ± 0.4 times per week at an average rating of perceived exertion of 11 on the Borg 6–20 scale. 37 Weekly aerobic exercise from the training diaries was calculated to be 132 ± 17.8 min. Subjects also reported one resistance training session (1 set, 12–15 reps) per week. There were no exercise-related adverse events reported during training.
Training-induced changes in anthropometry and peak oxygen uptake
As shown in Table 2, VO2peak increased significantly (16%, p = 0.002) after the exercise intervention concomitant with reductions in weight, body mass index, abdominal circumference and body fat.
Body composition and aerobic fitness before and after exercise training.
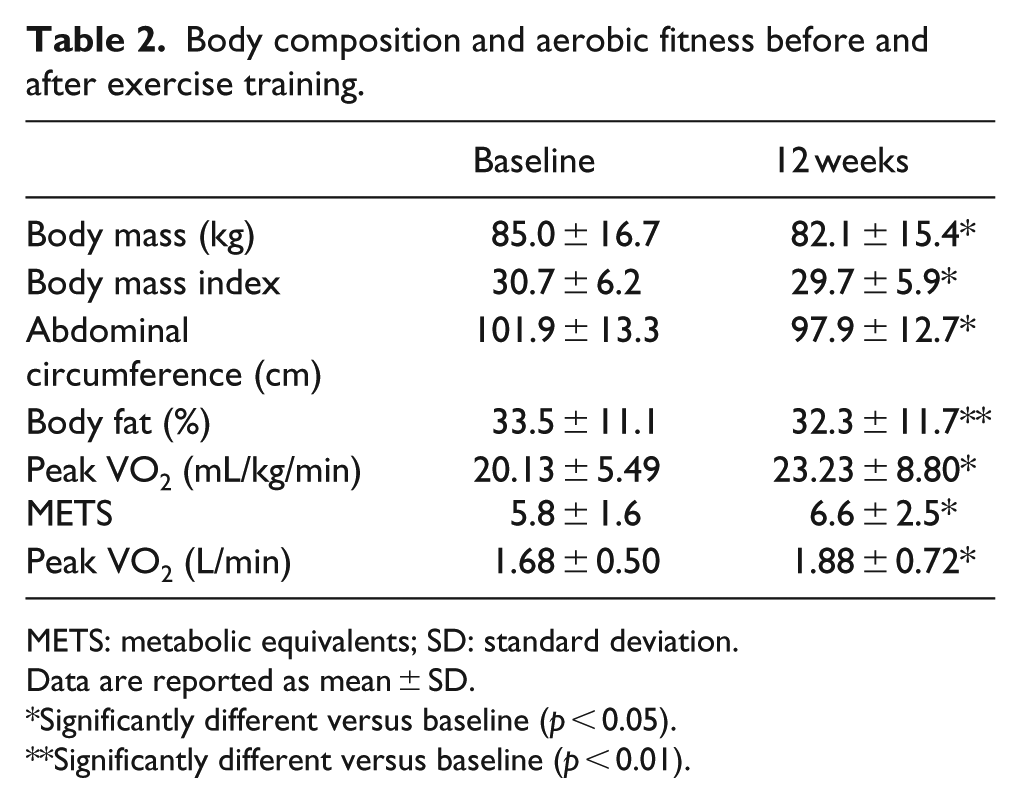
METS: metabolic equivalents; SD: standard deviation.
Data are reported as mean ± SD.
Significantly different versus baseline (p < 0.05).
Significantly different versus baseline (p < 0.01).
Vascular function
Changes in vascular function following training are reported in Table 3. A 15% increase was observed in peak calf blood flow and a 19% increase in peak vascular conductance after exercise training, but these changes failed to reach statistical significance. However, PCVR did increase significantly after training (p = 0.038). Both the AIx and central PWV decreased significantly, but peripheral PWV did not change. Changes in central PWV and AIx at 75 beats/min after chronic training were independent of changes in body mass index (BMI), aerobic fitness, hs-CRP, fasting glucose or abdominal circumference.
Cardiovascular function before and after exercise training.
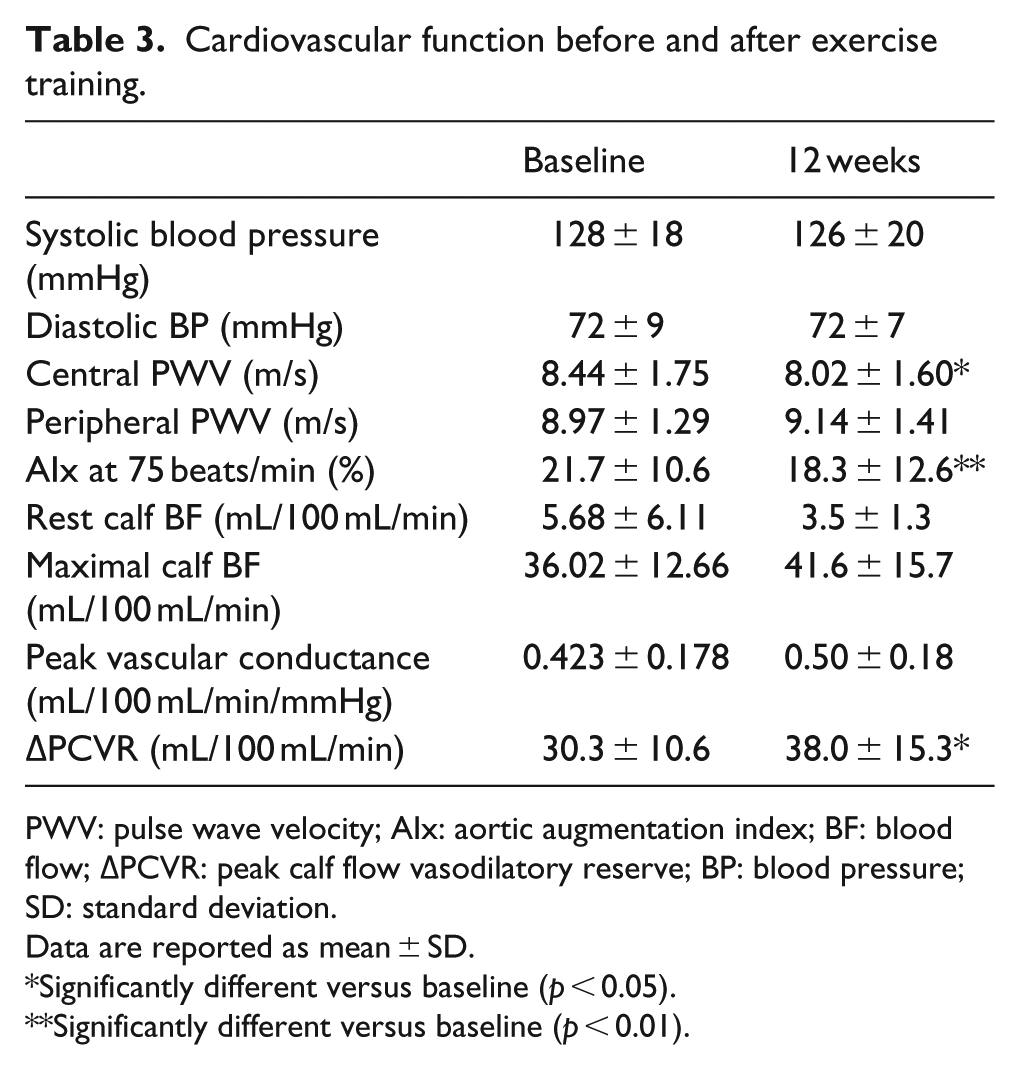
PWV: pulse wave velocity; AIx: aortic augmentation index; BF: blood flow; ΔPCVR: peak calf flow vasodilatory reserve; BP: blood pressure; SD: standard deviation.
Data are reported as mean ± SD.
Significantly different versus baseline (p < 0.05).
Significantly different versus baseline (p < 0.01).
Glucose control and lipid profile
A significant decrease in fasting glucose was observed following training (7.78 ± 1.87 vs 6.77 ± 1.24 mmol/L, p < 0.05), with no significant change in HbA1c (6.5% ± 0.7% vs 6.6% ± 0.6%) and modest but insignificant changes in the lipid profile (Table 4). The change from resting to post-exercise (20 min at 70% VO2peak) non-fasting blood glucose levels was similar before and after training.
Haemostatic markers before and after 12 weeks of exercise training.
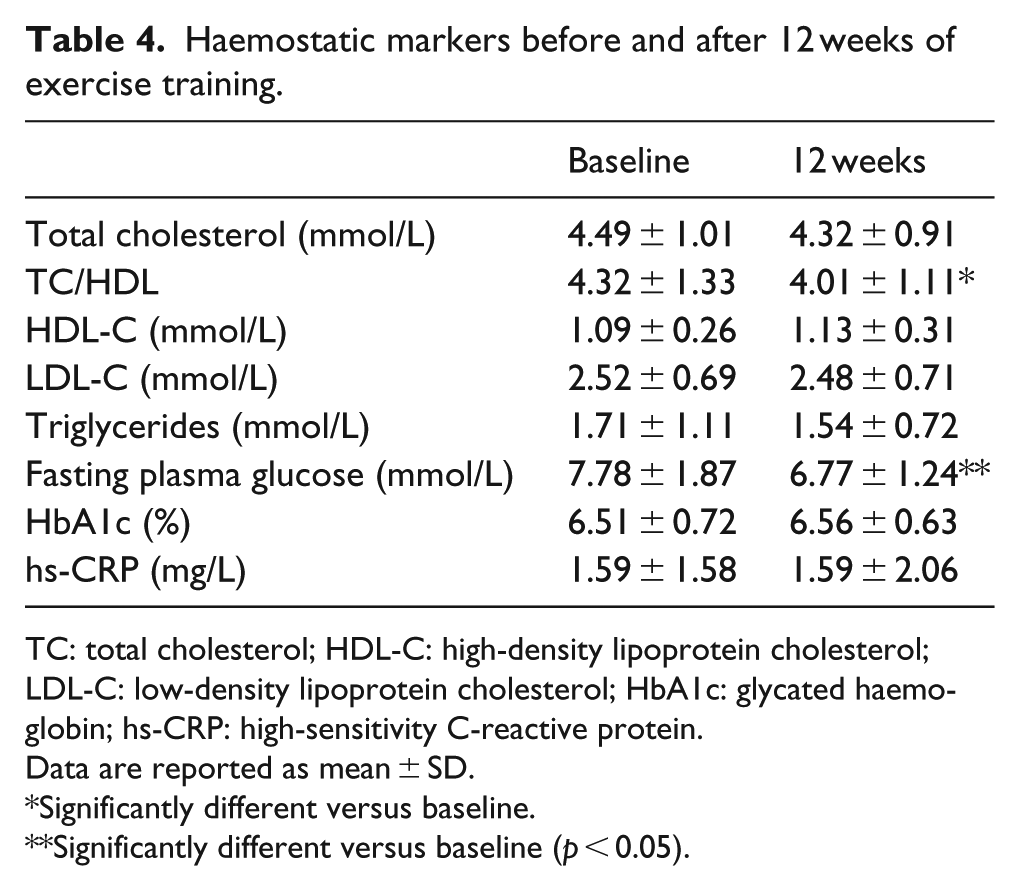
TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; HbA1c: glycated haemoglobin; hs-CRP: high-sensitivity C-reactive protein.
Data are reported as mean ± SD.
Significantly different versus baseline.
Significantly different versus baseline (p < 0.05).
Discussion
Our data indicate that a 12-week combined supervised and home-based exercise programme of aerobic and resistance training in subjects with DM2 improves aerobic power, lowers central arterial stiffness and increases PCVR. These changes were accompanied by a reduction in fasting blood glucose in the absence of changes in hs-CRP or changes in blood lipid profile.
Aerobic fitness (VO2peak) and increased habitual exercise are associated with lower arterial stiffness and increased insulin sensitivity.26,38–41 Our subjects had a significant improvement in cardiovascular fitness of close to 1 metabolic equivalent (MET) following only 12 weeks of exercise training. This is clinically relevant, as a 1 MET improvement in fitness is associated with 15%–20% lower CVD mortality risk.27,42 However, improved fitness was not correlated with changes in arterial compliance after exercise training. Madden et al. 41 demonstrated that 3 months of aerobic exercise resulted in significant improvements in arterial stiffness in people with DM2 without a change in cardiovascular fitness. Collectively, these data suggest that vascular adaptations may occur through mechanisms that are independent to changes in oxygen consumption or highly specific to vessel structure.
A number of mechanisms may be attributed to improved central vascular compliance including arterial remodelling, enhanced endothelial function and alterations in sympathetic tone. Increased arterial pressure and shear stress during acute exercise stimulates local release of growth factors that enhance central vasculature elasticity. 43 In rodent models, increased aortic distensiblity has been reported after 16 weeks of endurance training, secondary to increased aortic elastin content and reduced calcium depositions. 44 Favourable arterial remodelling occurs from chronic increases in blood flow, leading to a reduction in the thickness of the intimal and medial layers. 45 An increased vessel lumen diameter may decrease arterial stiffness, and endurance exercise training has been shown to increase both aortic 46 and femoral46,47 arterial lumen area. Our findings are in contrast to a study which failed to observe improvements in PWV after long-term (24 months) aerobic and resistance training. 48 However, our weekly aerobic training sessions were more frequent (five vs two) and compliance rates were stable throughout our training programme. Therefore, it is possible that a greater volume of exercise is required to elicit changes in central arterial stiffness. Moreover, changes in PWV following training have not been universally observed in some clinical populations. Higher baseline levels of PWV and exercise intensity may be linked to greater gains in PWV, 49 and other general or local derangements impacting peripheral vascular function and/or structure, may limit gains in certain populations such as chronic heart failure. 50
A surprising finding of this study was the reduction in central PWV but not peripheral PWV despite improvements in PCVR. Similar results have been reported elsewhere, 47 and the lack of improvement in peripheral PWV in our subjects may indicate that a different, more intensive or prolonged exercise stimulus may be required in order to increase compliance in conduit vessels of the leg. As reported in other clinical populations by our group 51 and by others, 52 we found an increase in PCVR in addition to a reduced AIx at 75 beats/min, after exercise training. Improvements in PCVR likely reflect improved endothelial function, metaboreflex response and sympathovagal modulation, 53 in addition to bio-availability or uptake of nitric oxide54,55 and other vasoactive substances. Collier et al. 56 demonstrated a training-induced reduction in central PWV (9.5%) in hypertensive subjects along with enhanced NO-mediated endothelial vasodilatation, and we have previously observed similar observations in young healthy subjects after only 6 days of intensive exercise training. 21 While our present results do not support a structural change in peripheral conduit vessels, improvements in ‘downstream’ resistance vessel function may also have contributed to lower impedance, ultimately reducing central vascular stiffness.
A reduction in fasting blood glucose levels may independently improve vascular function in DM2. Chronic hyperglycaemia in DM2 promotes the formation of advanced glycation end-products and collagen cross-links in the vascular wall, thereby accelerating arterial stiffness. We observed reductions in fasting glucose and post-exercise glucose levels, and these changes may have contributed to reduced arterial stiffness. Yamamoto et al. 39 reported a decrease in peripheral PWV (−1.20 ± 1.08 m/s) after only 2 weeks of intense hospital-based diet and exercise intervention in subjects with DM2. Although central PWV decreased to a lesser extent in this study (−0.421 ± 0.846 m/s), we also observed a reduction in AIx at 75 beats/min (−2.95%, p = 0.005).
We failed to detect changes in circulating levels of the inflammatory marker hs-CRP, which is in contrast to recent meta-analyses examining effects of exercise in people with diabetes and CVD.57,58 However, other research shows that the response of hs-CRP to exercise interventions may require an extended training period beyond the 12-week training period used in this study, as observed with other inflammatory markers not tested in this study.4,59–61 Modest reductions in body mass, abdominal fat and total body fat (%) were observed, which contrasts a recent study of older subjects with less diabetic control 41 who are more likely to realize changes in body composition and blood lipid concentrations.
Our exercise intervention reflected the recommendations of the Canadian Diabetes Association, which includes moderate- to vigorous-intensity aerobic exercise combined with resistance training. 62 Compliance to our exercise programme was lower than some studies1,63 with exercise diary data indicating a total aerobic exercise time of 132 min, less than the recommended 150 min. 62 Nevertheless, we believe our data reflect achievable and realistic compliance levels in a programme that requires most exercise sessions to be conducted independently.
Limitations
This study was not without limitations. Our population was reasonably well controlled at the onset of training. In addition, lipid and arterial blood pressure were within the treatment target for this patient population. As such, it is not surprising that we failed to detect favourable changes in hs-CRP, particularly after a relatively short time period. More significant reductions in haemostatic risk factor would be expected after an extended training period.
48
Despite the lack of a control group, there is no evidence that spontaneous recovery or changes in clinical status contributed to our findings. Our subjects had long-standing diabetes (mean duration >50 months) and were sedentary prior to initiating their exercise training. Additionally, the mean increase in
Conclusion
We conclude that a CR programme of combined aerobic and resistance exercise elicits a significant reduction in resting central arterial stiffness. These changes were accompanied by an increase in the PCVR and significant increases in aerobic capacity. Well-controlled patients with DM2 can realize training-induced vascular adaptations that are associated with lower cardiovascular risk.
Footnotes
Acknowledgements
The authors acknowledge the participation and contribution of patients and staff, including Dr Paul Oh, at Toronto Rehabilitation/University Health Network.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was supported by the Toronto Rehabilitation Institute who receives funding under the Provincial Rehabilitation Research Program from the Ministry of Health and Long-Term Care in Ontario.
