Abstract
Aim:
To evaluate the effects of the glucagon-like peptide-1 receptor agonist, exenatide, on blood pressure and heart rate during an intraduodenal glucose infusion in type 2 diabetes.
Methods:
Nine subjects with type 2 diabetes were randomised to receive intravenous exenatide or saline control in a crossover design. Glucose (3 kcal min−1) was infused via an intraduodenal manometry catheter for 60 min. Blood pressure, heart rate, and the frequency and amplitude of duodenal pressure waves were measured at regular intervals. Gastrointestinal symptoms were monitored using 100 mm visual analogue scales.
Results:
During intraduodenal glucose infusion (0–60 min), diastolic (p(0–60) = 0.03) and mean arterial (p(0–60) = 0.03) blood pressures and heart rate (p(0–60) = 0.06; p(0–120) = 0.03)) were higher with exenatide compared to placebo. The increase in the area under the curve for diastolic blood pressure and mean arterial blood pressure was related directly to the suppression of the duodenal motility index with exenatide compared to control (p = 0.007 and 0.04, respectively).
Conclusion:
In type 2 diabetes, intravenous exenatide increases mean arterial blood pressure and heart rate during an intraduodenal glucose infusion, supporting the need for further research with exenatide for its potential use in postprandial hypotension.
Introduction
Postprandial hypotension (PPH), that is, a fall in systolic blood pressure (SBP) >20 mmHg after a meal, triggered by pooling of blood in the splanchnic vasculature, occurs frequently and may result in syncope and/or falls, especially in older people and patients with type 2 diabetes and/or autonomic dysfunction. 1 The hypotensive response to oral glucose is dependent on the rate of gastric emptying; accordingly, pharmacological interventions that slow gastric emptying could ameliorate PPH. 1 In this regard, glucagon-like peptide-1 (GLP-1) receptor stimulation has gained attention as a potential treatment for PPH.
Even though studies involving chronic, subcutaneous administration of exenatide have reported a decrease in SBP, this has not been specifically measured in the postprandial phase and is apparent mainly in those who have relatively higher blood pressure (BP) at baseline. 2 In contrast, acute intravenous administration of exogenous GLP-1 has been reported to increase BP in some human studies, albeit not consistently,3–5 and attenuate the hypotensive effect of not only oral 1 but also intraduodenal 6 glucose loads, suggesting that mechanisms other than slowing of gastric emptying could be contributing to its pressor effects.
We recently reported that in patients with type 2 diabetes, intravenous infusion of the GLP-1 receptor agonist, exenatide, suppressed small intestinal motility and flow of chyme and slowed the small intestinal transit of a glucose load, associated with an acute reduction in glucose absorption. 7 We now report the effects of intravenous exenatide on SBP, diastolic blood pressure (DBP), mean arterial blood pressure (MAP) and heart rate (HR) in that study.
Patients and methods
Subjects
BP and HR were evaluated in nine patients with type 2 diabetes, managed by diet alone (six males and three females; mean age = 60.7 ± 2.4 years; body mass index (BMI) = 29.8 ± 1.4 kg m−2; known duration of diabetes = 5 ± 1 years and HbA1c = 6.2% ± 0.2% (44.7 ± 2.5 mmol mol−1)). 7 In one other patient, these data were unavailable as a result of technical errors in recording BP. The inclusion and exclusion criteria have been reported, 7 and the protocol was approved by the Human Research Ethics Committee of the Royal Adelaide Hospital and conducted in accordance with the principles of the Declaration of Helsinki as revised in 2000. All subjects provided written informed consent prior to their inclusion in the study.
Protocol
Each patient visited the laboratory on 2 days, at least 5 days apart, when a combined manometry and impedance assembly, which included a multilumen silicone catheter (Dentsleeve International, Mississauga, ON, Canada), was advanced into the stomach through an anesthetised nostril and allowed to pass into the duodenum by peristalsis. 7 The position of six manometry side holes (perfused with degassed water) and an infusion port within the duodenum was monitored continuously by measuring the antral and duodenal transmucosal potential difference via a proximal duodenal and a distal antral side hole perfused with 0.9% saline, and the numbers and amplitudes of the duodenal pressure waves were recorded digitally for subsequent analysis. Patients were randomised in double-blind fashion to receive either intravenous exenatide (7.5 mcg administered between −30 and 120 min) or volume-matched saline control, while glucose (3 kcal min−1) was given intraduodenally from 0–60 min. Venous blood was sampled at regular intervals using a cannula inserted in a cubital fossa vein, for measurement of blood glucose and serum insulin concentrations. 7 SBP, DBP, MAP and HR were measured every 3 min from −30 to 120 min, using an automatic sphygmomanometer (DINAMAP ProCare 100, Milwaukee, WI, USA).
Calculations and statistical analysis
Duodenal motility indices were calculated as ln[(sum of amplitudes × number of duodenal waves) + 1] 7 and MAP was calculated as [SBP + (2 × DBP)]/3. 8 Areas under the curves (AUC) were measured using the trapezoidal rule. Mean values and AUCs were compared using Student’s paired t-test. Repeated measures analysis of variance (ANOVA; adjusted by Bonferroni’s correction) was used to analyse the outcome measures, with treatment and time as factors. The AUCs for nausea scores did not conform to normality and were compared using Wilcoxon signed rank test. Pearson’s correlation coefficient was used to assess correlations. All analyses were performed using SPSS 21 (IBM Corporation, Chicago, IL, USA). Data are mean values ± standard error; p < 0.05 was considered statistically significant.
Results
SBP, DBP, MAP and HR
Prior to (−30 to 0 min), and after the intraduodenal glucose infusion (60–120 min), the SBP, DBP and MAP, as well as the HR, did not differ between exenatide and saline control, but during the intraduodenal glucose infusion (0–60 min), SBP, DBP and MAP increased with exenatide (p < 0.01 for each), but fell with saline control (p ⩽ 0.01 for each) before returning towards baseline. The AUC(0–60) for DBP (4479 ± 195 vs 4157 ± 177 mmHg min; p = 0.03) and MAP (5692 ± 217 vs 5309 ± 186 mmHg min; p = 0.03), but not SBP (8118 ± 330 vs 7615 ± 271 mmHg min; p = 0.06), was higher with exenatide than control (Figure 1(b) and (c)).
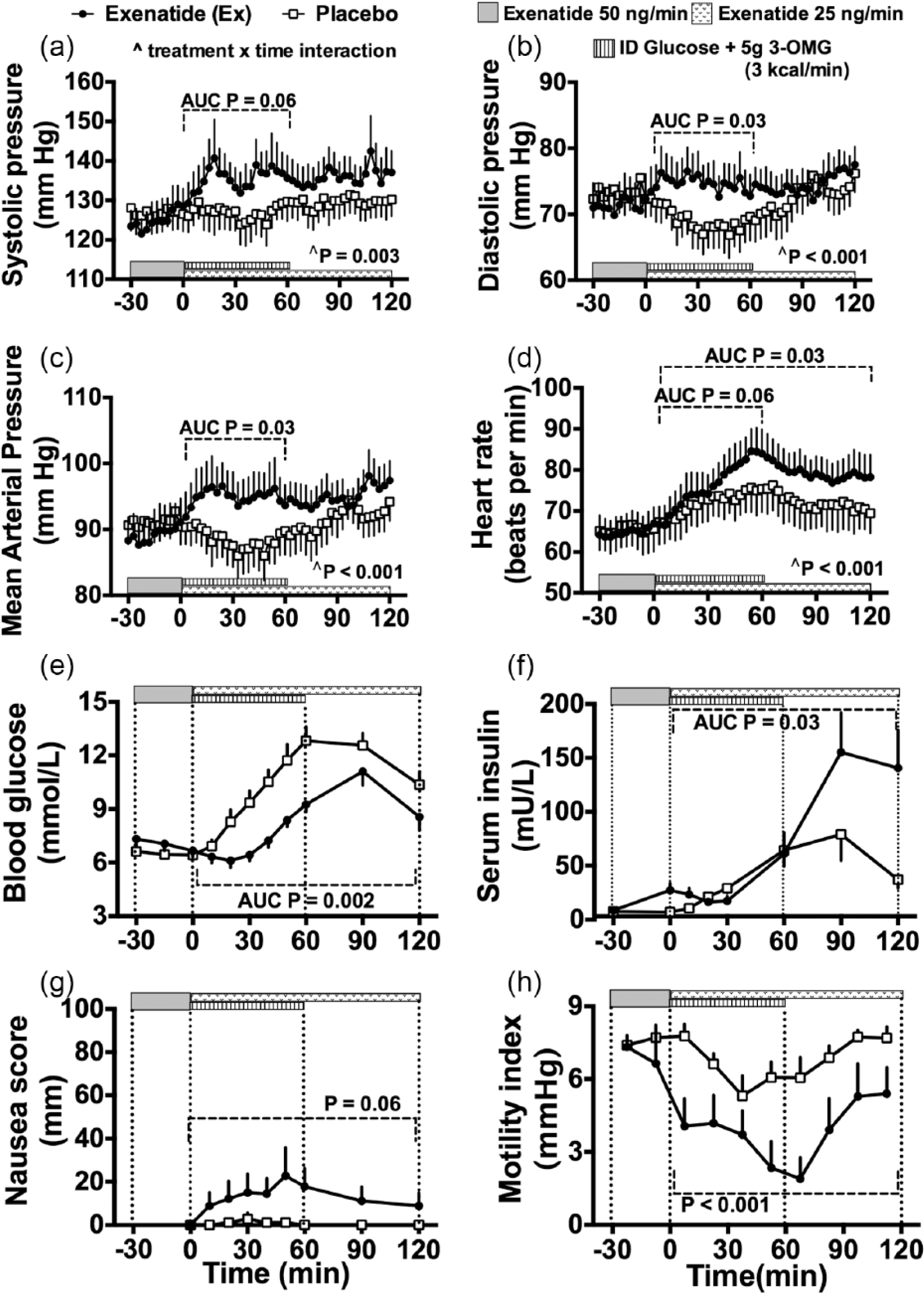
Effects of intravenous exenatide compared to saline control on (a) systolic blood pressure, (b) diastolic blood pressure, (c) mean arterial pressure and (d) heart rate, as well as (e) blood glucose, (f) serum insulin concentrations, (g) nausea scores and (h) motility index during fasting and in response to intraduodenal glucose infusion, in patients with type 2 diabetes (n = 9). Two-factor repeated measures ANOVA, with treatment and time as factors, adjusted by Bonferroni correction, was used to compare these variables. Areas under the curves were compared using Student’s t-test, other than for nausea scores, which were compared using the Wilcoxon signed rank test.
During and after intraduodenal glucose infusion (0–120 min), HR increased progressively with both exenatide and control (p < 0.001 for each), before declining. The AUC(0–60) for mean HR tended to be higher with exenatide than control (4546 ± 287 vs 4307 ± 315; p = 0.06), and the AUC(0–120) was significantly higher with exenatide than control (9305 ± 562 vs 8634 ± 654 beats; p = 0.03) (Figure 1(d)).
Blood glucose and serum insulin
As reported, 7 blood glucose and serum insulin concentrations increased in response to intraduodenal glucose infusion (0–60 min), before returning to baseline. The AUC(0–120) for blood glucose was lower (p = 0.002), and the AUC(0–120) for serum insulin was higher (p = 0.03), with exenatide than control (Figure 1(e) and (f)).
Nausea
During and after the intraduodenal glucose infusion (0–120 min), nausea scores increased with exenatide, but not with control, and the difference between them (AUC) tended towards significance (p < 0.06) (Figure 1(g)).
Duodenal motility index
During and after intraduodenal glucose infusion (0–120 min), the duodenal motility index was suppressed markedly more with exenatide than control (p < 0.001) (Figure 1(h)).
Relationships between outcome measures
During intraduodenal glucose infusion (0–60 min), the increments in the AUC for DBP and MAP with exenatide compared to control were related significantly to the reduction in duodenal motility index (r = 0.82, p = 0.01 and r = 0.67, p = 0.05, respectively), but not to the changes in AUC for blood glucose or serum insulin. Neither of SBP, DBP, MAP and HR was related to nausea scores during or after the intraduodenal glucose infusion (0–120 min).
Discussion
In patients with well-controlled type 2 diabetes receiving an intraduodenal glucose infusion at a rate within the physiological range for gastric emptying (3 kcal min−1), we observed that both MAP and HR increased during intravenous administration of exenatide, and that these changes were in proportion to the suppression of duodenal motility by exenatide.
In contrast to previous observations where exogenous GLP-1 infusions attenuated, but did not abolish, the hypotensive response to an intraduodenal glucose load,1,6 we found that the intravenous administration of the GLP-1 receptor agonist, exenatide, was associated with an increase in SBP, DBP, MAP and HR compared to baseline, though the increase in the SBP did not quite achieve statistical significance, possibly as a result of a type 2 error. This pressor response is likely to be due, at least in part, to an increase in cardiac output, as exogenous GLP-1 is known to increase stroke volume and HR. 9 Stimulation of atrial GLP-1 receptors 10 may have contributed to the increase in HR, which persisted even after the completion of the intraduodenal glucose infusion. In the fasting state (−30 to 0 min), exenatide was not associated with any chronotropic or pressor response. Whether the decrease in gut motility induced by exenatide could have triggered some undefined neurohumoral mechanism in the proximal gut (‘gut–heart axis’), which favoured sympathetic stimulation is open to speculation.
Nausea scores tended to be higher with exenatide than placebo during and after the intraduodenal glucose infusion, but did not show any relationship with BP or HR. Therefore, it appears most unlikely that nausea accounts for the cardiovascular effects.
Our study has several limitations. It was not primarily designed to assess cardiovascular outcomes, and the sample size was small; nonetheless, the differences in cardiovascular endpoints between treatments were quite marked. Second, as our subjects did not have PPH and had reasonably good glycaemic control, it remains to be determined whether our findings apply to patients with PPH and/or less well-controlled type 2 diabetes. Third, this study has assessed only the acute effects of intravenous exenatide after bypassing its effects on gastric emptying; in view of our observations, further studies are indicated to evaluate the effects of subcutaneous exenatide, including the potential for tachyphylaxis of cardiovascular effects after repeated dosing, as is evident for the slowing effects of gastric emptying by intravenous GLP-1. 11
In summary, intravenous infusion of exenatide is associated with increases in the MAP and HR during an intraduodenal glucose infusion, in patients with well-controlled type 2 diabetes. Our observations suggest that further studies are warranted to examine the potential use of exenatide in the management of PPH.
Footnotes
Acknowledgements
S.S.T. and C.S.M. were involved in the subject recruitment, coordination, data collection and interpretation, statistical analysis and drafting of the manuscript; T.W. was involved in the data interpretation and drafting of the manuscript; P.K. was involved in the conception of the study and study design; J.C. and J.K. were involved in the subject recruitment, coordination and data collection; R.S.R., H.L.C. and M.J.B. assisted in recruitment and data collection; K.L.J. was involved in the conception and design of the study, data analysis and interpretation; MH was involved in conception and design of the study and data interpretation; C.K.R. was involved in conception and design of the study, data analysis and interpretation, and had overall responsibility for the study. All authors critically reviewed the manuscript and have approved the publication of this final version of the manuscript. The authors wish to acknowledge the contribution of their late colleague, Antonietta Russo, in data collection and interpretation in this study. Clinical Trial Registration Number: Australia New Zealand Clinical Trials Registry number: ACTRN12608000428369.
Declaration of conflicting interests
Professor Christopher K Rayner has received funding from AstraZeneca, Merck Sharp & Dohme and Novartis. Professor Michael Horowitz has participated in advisory boards and/or symposia for AstraZeneca, Boehringer Ingelheim, Eli Lilly, Merck Sharp & Dohme, Novartis, Novo Nordisk and Sanofi and has received honoraria for this activity. Professor Karen L Jones has received research funding from Merck Sharp & Dohme and Sanofi and is supported by a NHMRC Senior Career Development Award (NHMRC ID: 627011). None of the other authors has any competing interest to declare. Dr Sony S Thazhath received scholarship funding from the University of Adelaide and the Rebecca L Cooper Foundation. Dr Chinmay S Marathe was the recipient of a scholarship from the University of Adelaide. Dr Tongzhi Wu has been supported by the Royal Adelaide Hospital Research Committee Early Career Fellowship.
Funding
This study was supported by funding from AstraZeneca and the National Health and Medical Research Council (NHMRC) of Australia.
