Abstract
Recent studies have investigated the potential of type 1 diabetes mellitus–related autoantigens, such as heat shock protein 60, to induce immunological tolerance or to suppress the immune response. A functional 24-residue peptide derived from heat shock protein 60 (P277) has shown anti-type 1 diabetes mellitus potential in experimental animals and in clinical studies, but it also carries a potential atherogenic effect. In this study, we have modified P277 to retain an anti-type 1 diabetes mellitus effect and minimize the atherogenic potential by replacing the P277 B epitope with another diabetes-associated autoantigen, insulinoma antigen-2 (IA-2), to create the fusion peptide IA-2-P2. In streptozotocin-induced diabetic C57BL/6J mice, the IA-2-P2 peptide displayed similar anti-diabetic effects to the control P277 peptide. Also, the IA-2-P2 peptide did not show atherogenic activity in a rabbit model. Our findings indicate the potential of IA-2-P2 as a promising vaccine against type 1 diabetes mellitus.
Introduction
Type 1 diabetes mellitus (T1DM) is a chronic autoimmune disease characterized by the self-destruction of pancreatic beta-cells. 1 Antigen-based intervention has been studied,2,3 including autoantigens, such as heat shock protein 60(Hsp60).4,5 Peptide P277, a 24-residue fragment of Hsp60 (437-460), has been used to ameliorate T1DM in animal models.6,7 Moreover, P277 has been developed into the vaccine, DiaPep277,8–10 which raised C-peptide and improved the preservation of beta-cell function in phase II clinical trials.11–13 However, Hsp60/65 is also an atherosclerosis (AS)-related protein, with high frequencies of anti-human Hsp60 antibodies detected in AS patients.14–16 Also, P277 has been shown to induce endothelial damage in C57BL/6J mice. 17 In our previous studies, intranasal immunization of HSP65-6 × P277-treated New Zealand White rabbits induced AS. 18 Furthermore, a follow-up study noted that subcutaneous immunization with P277 emulsified in incomplete Freund’s adjuvant (IFA) can aggravate the AS in high cholesterol diet (HCD)-fed rabbits, 19 and this effect was related to the up-regulation of anti-P277 antibodies. 20
Elias et al. 4 reported that the key diabetes-specific T-cell epitope in P277 appears to be contained within the 12 amino acids at positions 449-460 (P2 fragment). The P2 fragment is capable of T1DM prevention, but the effect is weaker than intact P277, which may result from the absence or incomplete B epitopes. 21 Therefore, we screened several B epitopes of diabetic autoantigens as potential replacements for the original P277 B-cell epitope.
Insulinoma antigen-2(IA-2) is one of the major autoantigens against T1D. Vaccination with IA-2 was able to prevent late prediabetic NOD mice from developing diabetes. 22 Also, knowing the humoral autoreactive epitope map, 23 it is suitable replacement for the original B-cell epitope in peptide P277.
In this study, a novel peptide vaccine, IA-2-P2, was constructed by substituting the 12 amino acids in the N-terminal P277 with the B epitope of IA-2 to investigate whether this new peptide could retain the intrinsic anti-diabetic effects and eliminate the pro-atherosclerotic side effects.
Materials and methods
Materials and reagents
Streptozotocin (STZ) and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma–Aldrich, USA; peptides IA-2-P2 and P277 were synthesized by GL Biochem in Shanghai, China (high-performance liquid chromatography (HPLC) purity >90%). Lipofundin (20%) was from B. Braun, Germany. Mouse interleukin (IL)-10 and interferon (IFN)-γ enzyme-linked immunosorbent assay (ELISA) kits were purchased from Biolegend, USA. 3, 3′, 5, 5′-tetramethylbenzidine (TMB) was from Sunbio, Nanjing, China. Four-week-old male C57BL/6J mice were purchased from the Comparative Medicine Center of Yangzhou University (Yangzhou, China, No. SCXK 2012-0004) and raised under pathogen-free conditions with a 12 h light–dark schedule. All experiments were approved by the Animal Care and Use Committee of the authors’ institution.
Induction of T1DM
In all, 30 C57BL/6J mice were randomly divided into control and STZ-treated groups. After fated for 8 h, mice were injected 40 mg/kg of STZ solution intraperitoneally daily for five consecutive days; the control group mice only received citrate buffer. Blood samples were obtained from the tail and glucose levels were measured using a glucose analyser (Gluco Leader GLM-77, Taiwan) at the third and seventh day after the final injection. Diabetes was defined as a blood glucose level ⩾11.1 mmol/L on two consecutive examinations. 24
Animal immunization
Following the onset of STZ-induced diabetes, mice were randomly divided into three groups (n = 10) and injected subcutaneously, with 0.1 mL emulsion containing 1 mg IA-2-P2 (HGDTTFEYQDIPALDSLTPANED) or P277 (VLGGGVALLRVIPALDSLYPANED) and 40 mg mannitol in 1 mL of 20% Lipofundin six times at 3-week intervals. Mice in the control group received six placebo injections (40 mg mannitol in Lipofundin).
A total of 30 male New Zealand White rabbits (2 ± 0.2 kg) were obtained from Jiangsu Academy of Agricultural Sciences, Nanjing, China. Animals with serum total cholesterol levels <2.6 mM (100 mg/dL) were selected and housed, individually in wire-bottomed cages at room temperature. A HCD containing 0.5% cholesterol and 5% lard was used to induce atherosclerotic lesions 20 after first immunization. Rabbits were separated into three groups (n = 8) and immunized (at six dorsal location) subcutaneously four times at 4-week intervals, with 1 mL of the protein solution (1 mg/mL) or phosphate-buffered saline (PBS) emulsified (1:1) with IFA (Sigma, USA). The animal protocol was reviewed and approved by the China Pharmaceutical University Institute Animal Care and Use Committee.
ELISA analysis
The titers of IA-2-P2- or P277-specific serum antibodies were determined by ELISA. Briefly, 96-well flat-bottomed ELISA plates were coated with 4 µg of IA-2-P2 or P277 peptide in 100 µL of coating buffer (Na2CO3-NaHCO3, pH9.6). Immunogen-coated plates were blocked with 5% bovine serum albumin (BSA) and then incubated with 100 µL/well of sera diluted 1:100. Secondary horse radish peroxide (HRP)-conjugated goat anti-mouse IgG (Sunshine, Nanjing, China) and TMB solution were used to detect antibody levels at 450 nm.
T-cell proliferation
Splenic T-cell proliferative responses were assayed to T-cell mitogen concanavalin A (ConA) (1.25 µg/mL), IA-2-P2 (5 µg/mL), and P277 (5 µg/mL) peptides in vitro. 1 × 105 T cells (in 100 µL RPMI-1640 culture medium) was added to each microplate well and detected by the MTT method at 570 nm. The stimulation index (SI) = mean absorption of cells cultured with antigens/mean absorption of cells cultured with medium alone.
Cytokine assays
Cytokines were determined by stimulation with peptide solution (terminal concentration 10 µg/mL) or medium alone for 72 h in vitro. Culture supernatant levels of mice IL-10 and IFN-γ were quantified in culture supernatants using ELISA kits (BioLegend, CA, USA) according to the manufacturer’s instructions. The lower limit of detection for the experiments was required to be 15 pg/mL.
C-peptide levels
To assess the effects of peptide immunization on pancreatic beta-cell function during the course of diabetes, changes in serum C-peptide levels in serum were measured in real time using an ELISA Kit (Jiancheng, Nanjing, China) to reflect. The ELISA process was carried out in accordance with the instruction protocols.
Analysis of atherosclerotic lesions
Rabbits were sacrificed at week 17. Intact aortas were removed from the aortic arch to the iliac bifurcation and cut longitudinally. The aortas were then fixed in 10% formalin for 4 days and stained with 0.2% (w/v) Sudan III in 75% ethanol (v/v) to visualize the areas of fat deposition. The degree of AS lesions was quantified as a staining percentage of the entire aorta area by the MapInfo Professional software (version 7.0; http://www.mapinfo.com).
Histopathological analysis of aortic arch
The aortic arches collected from rabbits were fixed in 10% formalin, dehydrated, and embedded in paraffin wax. Sections (4 µm thickness) were prepared and stained with haematoxylin and eosin. Pathological changes caused by the immunization were evaluated in a blinded fashion by a professional pathologist.
Statistical analysis
Data were expressed as means ± standard deviation (SD). All statistical analyses were performed by SPSS 11.5 software (SPSS Inc., Chicago, IL, USA). Differences between groups were analysed by analysis of variance (ANOVA). p < 0.05 was considered to indicate statistical significance marked by asterisk in figures.
Results
Effects of IA-2-P2 treatment of diabetic mice
Immunization with IA-2-P2 or P277 peptides effectively controlled hyperglycaemia of diabetic mice. Cumulative survival in the P277-, IA-P2- and placebo-treated groups were found to be 90%, 70% and 40%, respectively (Figure 1(a)). Prior to treatment, all the tested groups were hyperglycaemic with blood glucose levels >16 mmol/L. Glucose levels steadily increased to 30 mmol/L at first 12 weeks in all groups while decreased and stabilized around 16–18 mmol/L in the IA-2-P2- and P277-treated groups. At the end of observation period, numbers of normoglycemic mice in P277- and IA-2-P2-treated groups were two and one, respectively, while mice in the placebo group showed only a slight decrease in blood glucose levels (still around 30 mmol/L). Nevertheless, treatment with IA-2-P2 or P277 peptides markedly improved survival rates compared with those observed in the placebo group.
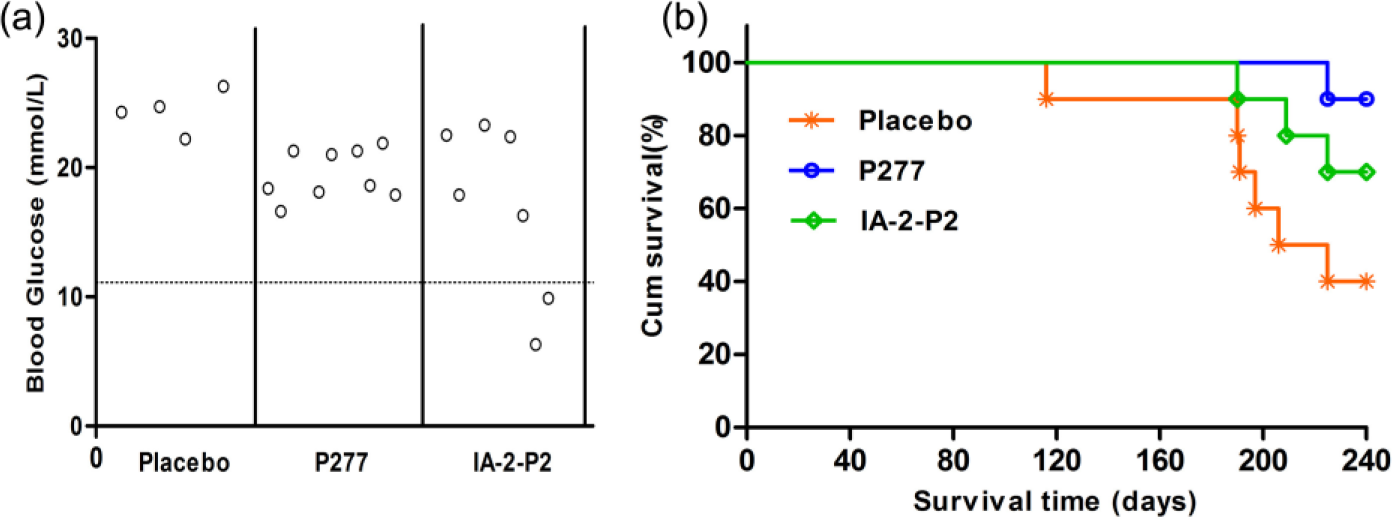
Effects of IA-2-P2 treatment on STZ-induced diabetic mice. Individual C57BL/6 male mice were treated with 100 µg IA-2-P2 or P277 peptide emulsified in Lipofundin six times after diabetes onset (n = 10). (a) The terminal blood glucose at the end of observation at day 240 and (b) survival curve of diabetic mice.
P277-specific antibodies titers in diabetic mice
Peptides were administered at 3-week intervals for total six times. Antibody titers tended to increase after each administration in the P277-treated group, but not in the IA-2-P2-treated ones. It indicates that replacement of the P277 peptide B epitope reduced the production of P277-specfic antibodies (Figure 2).
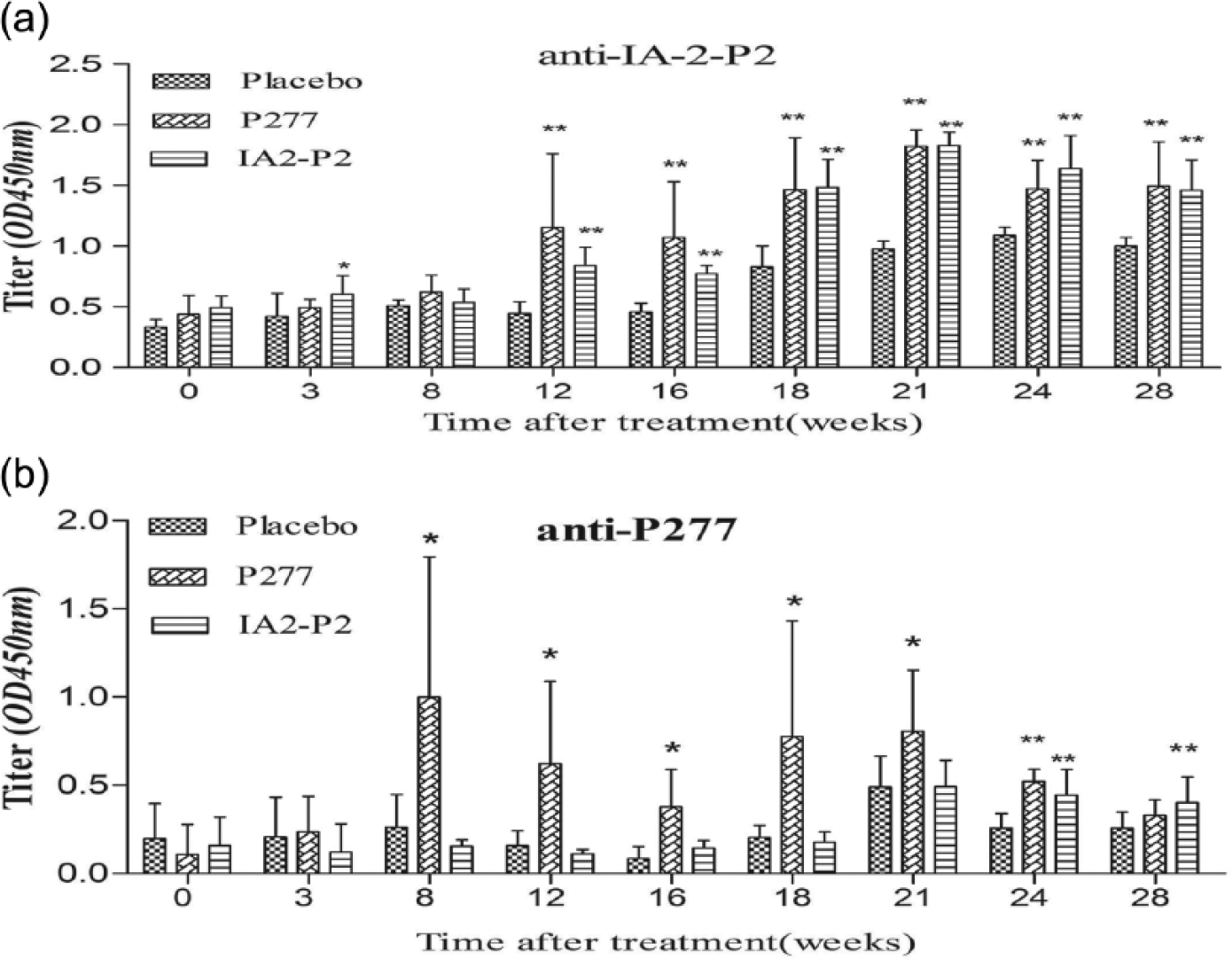
Antibodies against IA-2-P2 (a) or P277 (b) with following peptide therapy. Mice were treated with IA-2-P2 (n = 10), P277 (n = 10) or placebo (n = 10) as described in the ‘Materials and methods’ section and the titer of serum antibodies to either peptide was measured each month.
Effect of IA-2-P2 peptide on T-cell proliferation
Splenocytes proliferative responses to IA-2-P2, P277 and ConA were assayed. As shown in Figure 3, T cells from P277- and IA-2-P2-treated mice both showed decreased response to P277 and IA-2-P2 peptides (P < 0.05). However, T cells from three groups showed similar proliferative responses to ConA. Results indicated that IA-2-P2 vaccination did not induce general inhibition of T cells reactivity but the down-regulation of specific population response to IA-2-P2, associated with prevention of diabetes.
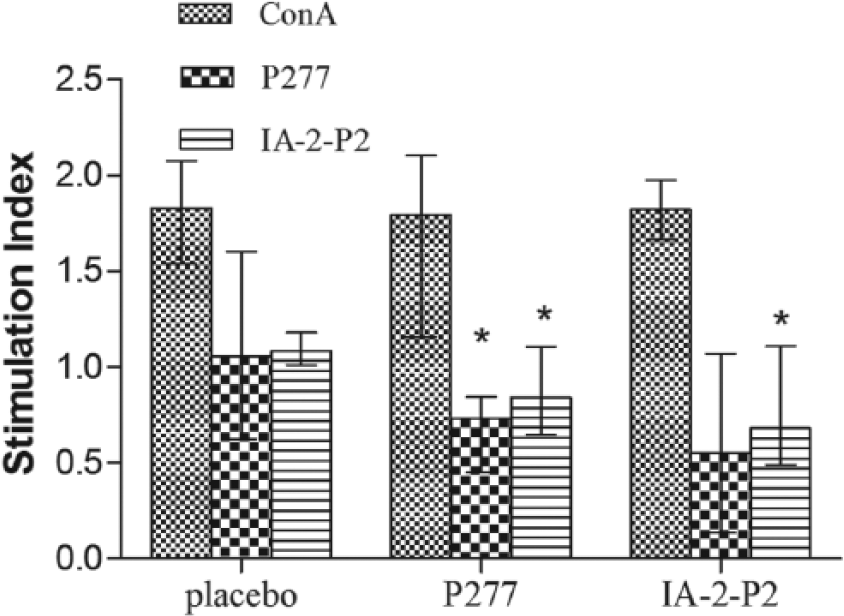
Effects of treatment on T-cell proliferation. At the end of the observation period, spleens were removed from mice (n = 3) in every group to evaluate T cells proliferative activity in vitro with 5 µg/mL of IA-2-P2, P277 or 1.25 µg/mL of T cell mitogen ConA. Data were expressed as median with interquartile range,*p < 0.05 compared with the placebo-treated group.
Effect of IA-2-P2 peptide on cytokines
IL-10 and IFN-γ production was determined after 72 h of in vitro stimulation with IA-2-P2 and P277 peptides. Immunization of mice with IA-2-P2 and P277 peptides elicited more IL-10 and less IFN-γ (p < 0.05, p < 0.01 vs the placebo-treated group), indicating an enhanced Th2 cytokine phenotype and the induction of an anti-inflammatory effect in T1DM (Figure 4).
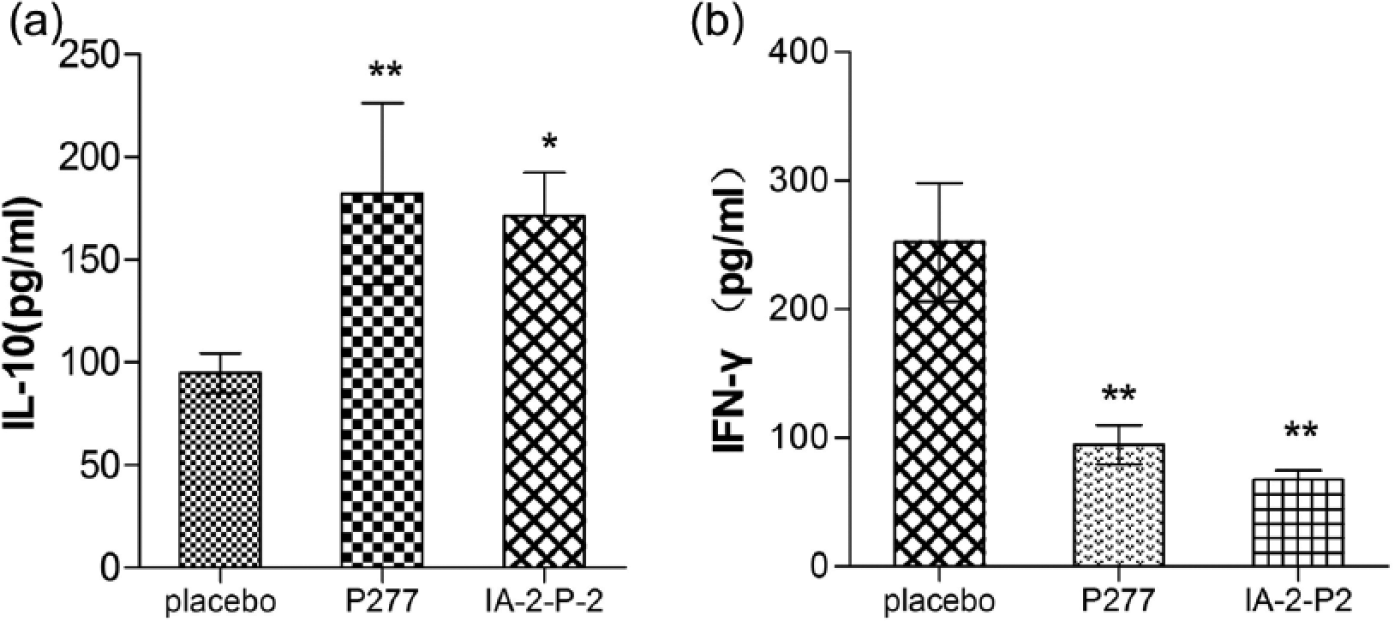
Effects of IA-2-P2 treatment on cytokines. Splenocytes supernatants were collected after 72 h of stimulation with peptides IA-2-P2 or P277 (n = 3). Mice IL-10(a) and IFN- γ (b) were quantitated by ELISA kits,*p < 0.05 and **p < 0.01 compared with the placebo group.
Effect of IA-2-P2 on preservation of C-peptide secretion
C-peptide levels, which reflects pancreatic β-cells function directly, are usually used as primary endpoints in clinical studies of T1DM. Mice treated with IA-2-P2 or P277 showed superior maintenance of C-peptide secretion than those in the placebo-treated group (Figure 5).
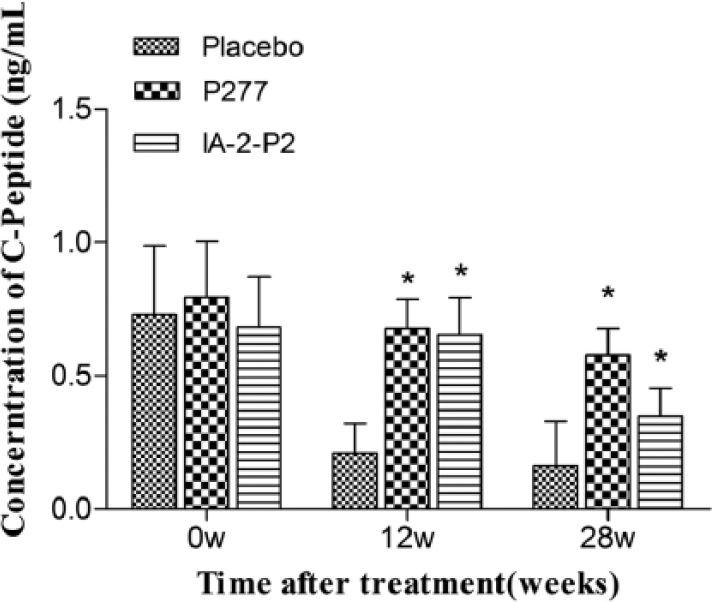
Effects of IA-2-P2 on preservation of C-peptide secretion at different periods (n = 10). *p < 0.05 compared with placebo-treated group.
Assessment of atherosclerotic lesions
Macroscopic images of representative aortas from all immunized groups are shown in Figure 6. IA-2-P2 immunization exhibited no influence on the formation of atherosclerotic lesions compared with those observed in the PBS-immunized group. However, a significant promotion on lesion formation was observed in the P277-immunized group fed by HCD. The area of the lesion relative to that of the entire area of the aorta in the P277-immunized group was significantly greater in the IA-2-P2-immunized groups (35.48% ± 9.78% vs 17.68% ± 9.98%; p < 0.05). Result suggests that IA-2-P2 peptide has a lower potential to induce AS compared with the P277 peptide.
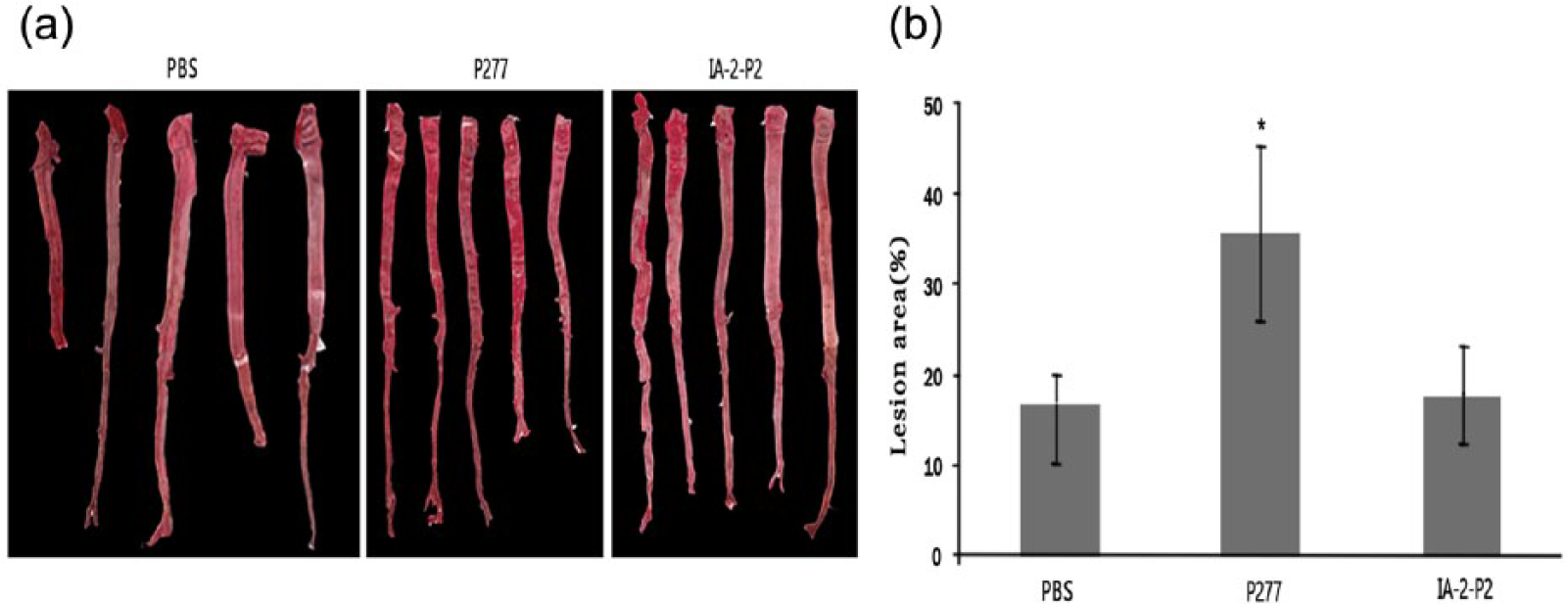
Analysis of atherosclerotic lesions in aortas. (a) Aortas (n = 5) were stained with Sudan III to visualize the area of fat deposition. (b) The percentage of Sudan III staining area relative to entire aorta area was quantified by the MapInfo Professional software (version 7.0). *p < 0.05 as compared with the placebo group.
Histopathological analysis of aortic arch
The thickness of hyperplastic aortic arches from rabbits vaccinated with PBS or IA-2P-2 was less than that observed in the P277-immunized group (Figure 7(a) and (c)). In contrast, aortic sections of rabbits showed marked intimal thickening with focal endothelial injury in the P277-immunized rabbits (Figure 7(b)).
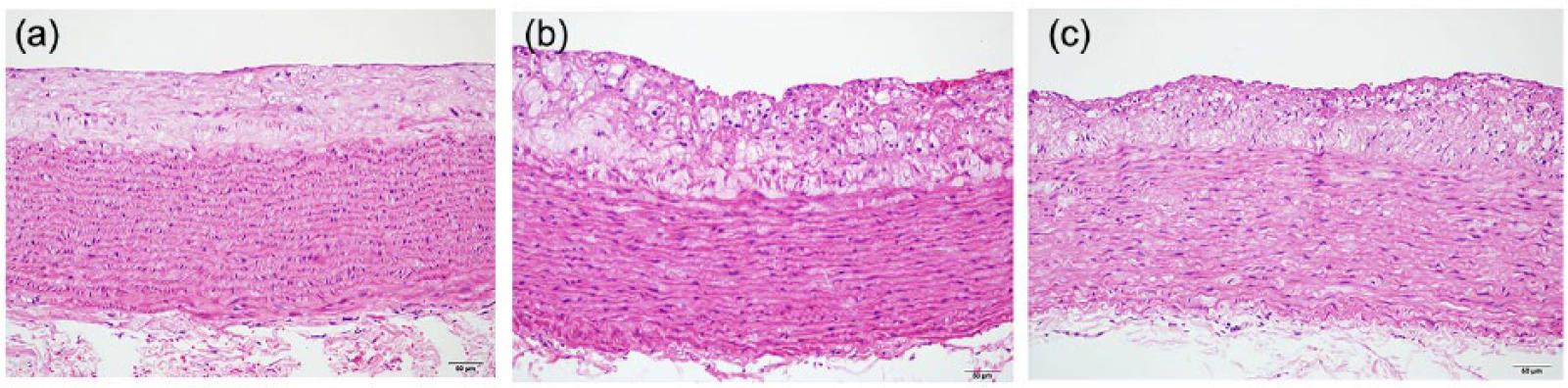
Analysis of hyperplasic aortas: (a) aortas from the PBS-immunized rabbits, (b) aortas from the P277-immunized rabbits and (c) aortas from the IA-2-P2-immunized rabbits (n = 6). Original magnification was ×100.
Discussion
It is well known that CD4+ T cells, divided into Th1 and Th2 population, play a central role 25 due to its responses to T1DM-associated antigens. In this study, cytokines analysis with the IA-2-P2 peptide revealed higher IL-10 and lower IFN-γ levels, compared with those of placebo-treated group, indicating a shift from a Th1-like pro-inflammatory response towards a Th2-like anti-inflammatory response. Moreover, T-cell proliferation analysis demonstrated a loss of proliferative reactivity to stimulated peptide, which reflects less activation-induced apoptosis or energy exhaustion, possibly due to the operational immune tolerance induced by repeated stimulation of specific peptides.
P277 antibodies have been reported to cause vascular endothelial injury 19 and monitored in entire observation period. P277-specific antibody titers increased after each peptide immunization in the P277-treated group (p < 0.05) while none were detected in the placebo-treated mice. Interestingly, the lowest level of P277-specific antibody remained in IA-2-P2-treated group, suggesting that atherogenic P277 antibody production was significantly reduced by replacement of the B epitope of P277 with IA-2. Moreover, IA-2-P2 treatment showed no influence on the formation of atherosclerotic lesions in New Zealand White rabbits compared with the PBS-immunized group, while lesion formation was significantly promoted in the P277-immunized group fed a HCD. Histological aortic analysis showed marked initial thickening with focal endothelial injury in P277-treated rabbits, revealing that IA-2-P2 reduced the atherogenic side effects relative to P277 B epitopes in animal models.
In summary, IA-2-P2 is a promising vaccine against T1DM compared with P277 for its few undesired side effects. Furthermore, this study provides novel methods to overcome important issues in the design of autoantigen-based therapeutic in autoreactive diseases.
Footnotes
Acknowledgements
L.S. and S.L. equally contributed to this paper as first authors. The authors would like to thank Jun Zou and Ruoyang Zhang for their assistance in performing the animal experiment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos 81172973 and 31270985) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
