Abstract
Aims:
We aimed to evaluate asymmetric dimethylarginine levels in young patients with Type 1 diabetes mellitus according to diabetes duration and to examine the relationship between these levels and measures of atherosclerosis and myocardial function.
Materials and methods:
In total, 83 patients (8.5–22 years) with Type 1 diabetes mellitus were stratified by diabetes duration: 12–60 months (Group 1, n = 27), >60–120 months (Group 2, n = 29) and >120 months (Group 3, n = 27). Asymmetric dimethylarginine levels were assessed. Carotid intima–media thickness was measured. Myocardial function was assessed by M-mode, conventional Doppler and tissue Doppler echocardiography.
Results:
Asymmetric dimethylarginine level was significantly higher in Group 1, while carotid intima–media thickness was significantly greater in Group 3 (
Conclusion:
In contrast to adult diabetics, asymmetric dimethylarginine concentration decreases as diabetes duration increases in young Type 1 diabetic patients and is associated with worsening measures of cardiovascular risk and poorer diastolic function.
Introduction
Cardiovascular disease (CVD) is a common cause of mortality in patients with diabetes mellitus (DM). 1 CVD is more common in patients with Type 2 diabetes than those with Type 1 diabetes mellitus (T1DM). However, deaths due to CVD in patients with T1DM occur at younger ages. 2 The risk of CVD increases in patients with T1DM and CVD may be seen as early as adolescence. 3 Increased oxidative stress due to hyperglycaemia has been implicated in the development of cardiovascular complications in diabetes. 4 Hyperglycaemia-induced overproduction of superoxide is accompanied by increased generation of nitric oxide (NO). Thus, particularly in patients with poor metabolic control, tissue damage may occur because of high concentrations of powerful oxidants. 5
NO is synthesized in the vascular endothelium by endothelial nitric oxide synthase (eNOS) using L-arginine as a substrate and plays an important role in the maintenance of vascular homeostasis. Asymmetric dimethylarginine (ADMA) is an endogenous inhibitor of eNOS by competing with L-arginine.6,7 An increase in ADMA levels causes endothelial dysfunction by reducing NO bioavailability. 8 Therefore, higher circulating ADMA levels are recognized as a biomarker of atherosclerotic CVD risk and mortality. 7 Higher ADMA levels have been found in adults with T1DM compared with healthy controls. 9 However, in recent studies of ADMA levels in children and adolescents with T1DM, contradictory data were obtained.10–12
Increased intima–media thickness (IMT) of the common carotid artery and the presence of carotid plaques are considered to be markers of subclinical atherosclerosis and are also independent predictors of future cardiovascular events. 13 Although adult investigations have shown a positive relationship between circulating ADMA levels and carotid intima–media thickness (CIMT), 14 studies in children and adolescents are inadequate, and the conclusions are inconsistent. Studies have suggested that there was no direct relationship between ADMA and CIMT in children.15,16 Furthermore, there is a paucity of data concerning ADMA concentrations and IMT in children and adolescents with T1DM.
Diabetes mellitus is associated with cardiovascular complications, one of which is cardiomyopathy. Many factors play a role in the aetiology of diabetic cardiomyopathy, including microangiopathy and endothelial dysfunction. 17 Impaired biventricular function has been documented as an early sign of diabetic heart muscle disease in children with T1DM without overt cardiac disease. 18 Previous investigation has suggested that plasma ADMA concentration may be correlated with age-associated changes in left ventricular (LV) mass in a normal, untreated, ageing population. 19 To date, there is only one report of the relationship between ADMA levels and echocardiographic parameters, and the authors only used one methodology, M-mode echocardiography. 20
In this study, we aimed to evaluate circulating ADMA levels according to diabetes duration and to determine the relationship between ADMA levels and subclinical atherosclerosis assessed by CIMT and cardiac muscle function evaluated by ultrasonographic Doppler tissue imaging in children and adolescents with T1DM.
Participants and methods
This cross-sectional study was conducted with pubertal, Type 1 diabetic children and adolescents. All cases were diagnosed and treated in a single unit, and all were receiving intensive insulin therapy. Patient inclusion criteria were as follows:
Diabetes duration greater than 1 year;
Basal-bolus regimen with a total daily insulin dose requirement greater than 0.5 U/kg/day (indicating that the so-called honeymoon period is over).
Exclusion criteria were as follows:
Pre-pubertal;
Symptoms suggestive of cardiovascular system affection, such as palpitation, chest pain and presence of systemic hypertension;
Medications known to modify cardiac function;
Obvious cardiac disease;
History of acute complications, such as ketoacidosis or hypoglycaemia, in the 6 months preceding the study;
The presence of microalbuminuria;
Presence of chronic complications such as neuropathy, nephropathy or retinopathy.
Patients were divided into three groups according to the duration of diabetes as follows: 12–60 months duration were designated Group 1, >61–120 months duration were Group 2 and >121 months duration formed Group 3. Clinical and laboratory parameters were compared between the three groups.
Clinical and laboratory evaluation
All children and adolescents with T1DM underwent physical examination and laboratory studies (ADMA and lipid profile) following descriptive data collection including age, gender, weight and height. Tissue Doppler echocardiography (TDE) and M-mode echocardiography were performed on the same day. Glycolated haemoglobin (HbA1c) was measured in all patients to evaluate the degree of diabetic control. The mean HbA1c values of the patients were calculated for each group.
Echocardiographic measurements
The ultrasound device Vivid 7 system (GE-Vingmed Ultrasound AS, Horten, Norway) and 3S-RS probe (with an operating frequency of 3.5 MHz) were used for echocardiographic investigation, in a supine position and left lateral decubitus. All echocardiographic Doppler measurements were performed using the averages of three consecutive cycles. The two-dimensional (2D), M-mode and Doppler echocardiographic techniques were used to take images from parasternal and apical positions. M-mode echocardiographic measurements were obtained under the standards outlined by the American Society of Echocardiography. 21 Briefly, interventricular septum thickness (IVST) and left ventricle posterior wall thickness (LVpWT) were measured through the parasternal long axis at the end of diastole.
The conventional Doppler echocardiographic measurements were obtained from the parasternal long axis image with the pulse wave Doppler sample volume traced at the junction of the LV outflow tract and the anterior mitral valve. Peak systolic velocity (S), peak early (E) and peak late (A) diastolic myocardial velocities, deceleration time of E (DT), ejection time (ET), isovolumic relaxation time (IVRT) and isovolumic contraction time (IVCT) were measured. Myocardial performance index (MPI) was calculated with the Tei index formula (IVRT + IVCT/ET). 22
Tissue Doppler imaging (TDI) was obtained from the apical four-chamber image with the pulse wave Doppler sample volume traced at the mitral-lateral annulus. Peak systolic velocity (Sm), peak early (Em) and peak late (Am) diastolic myocardial annular velocities, ET, IVRT and IVCT were measured. MPI was calculated with the Tei index formula (IVRT + IVCT/ET). 22 All measurements were performed by the same experienced cardiologist.
CIMT
We prospectively studied all subjects using carotid arterial Doppler ultrasound with 9–4 MHz transducers (Siemens Acuson Antares; Siemens Medical Solutions, Inc.,). Doppler ultrasound examinations were performed by the same experienced radiologist. Patients lay in a supine position with their necks in extension. Common, internal and external carotid artery lumens were scanned bilaterally. Flow velocity and its characteristics were evaluated. IMT was measured from the leading edge of the first bright line (lumen–intimal interface) to the leading edge of the second bright line (media–adventitia interface).
Biochemical analysis
Fasting (>8 h) blood was carefully collected from subjects to avoid haemolysis, into plain and ethylenediaminetetraacetic acid (EDTA) tubes in the morning, following 30 min of supine rest and immediately centrifuged after collection.
For ADMA measurement, blood samples were centrifuged at 2000g for 10 min at 4°C and then serum samples were stored at −70°C until use. Serum ADMA levels were measured by enzyme-linked immunosorbent method [ADMA®-enzyme-linked immunosorbent assay (ELISA); DLD Diagnostika GmbH, Hamburg, Germany] according to the manufacturer’s guidelines.
The fasting values of total cholesterol (TC), triglyceride (TG) and high-density lipoprotein cholesterol (HDL-C) were determined by commercially available kits (Beckman Coulter, USA). Low-density lipoprotein cholesterol (LDL-C) was calculated by Friedewald et al. 23 equation.
HbA1c was measured using EDTA blood samples. The HbA1c levels were determined by a high-performance liquid chromatography (HPLC) method (G7 HPLC analyzer; Tosoh, Japan).
Statistical analysis
Data were analysed using SPSS version 20 statistical package (SPSS, Chicago, IL). Quantitative data were expressed as mean ± standard deviation (SD). The minimum and maximum values are shown only in Table 1. All data were tested for normality using the Kolmogorov–Smirnov test. Non-parametric tests were used for all comparison between groups because our groups were below 30 patients. Kruskal–Wallis test was used for comparison between the three groups. The comparison between the two groups was made by the Mann–Whitney U test. Pearson’s and non-parametric Spearman’s correlation analyses were also used for correlations between parameters.
Comparison of mean ± SD (range) age, diabetes duration, ADMA level and CIMT between the three groups.
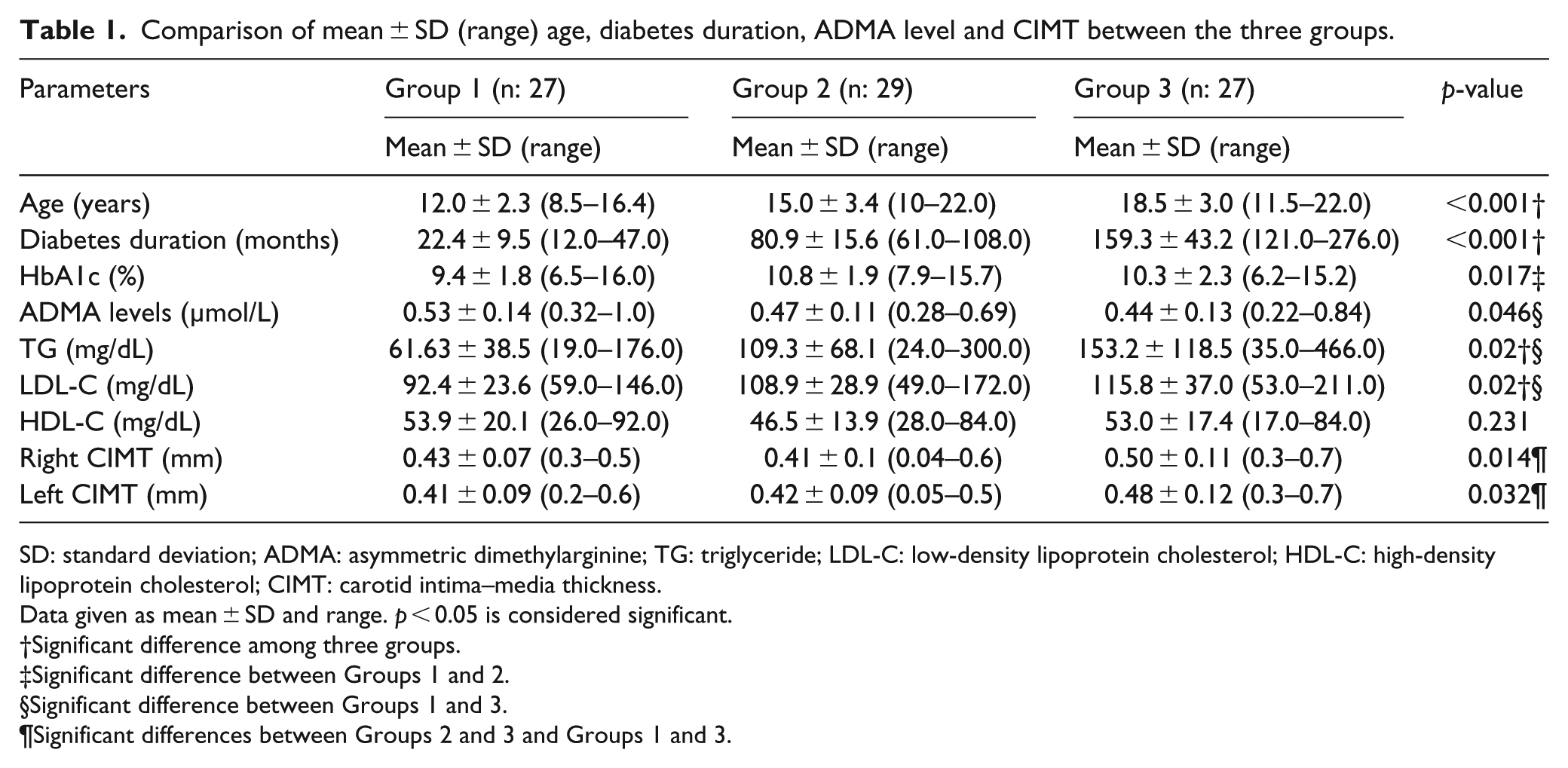
SD: standard deviation; ADMA: asymmetric dimethylarginine; TG: triglyceride; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; CIMT: carotid intima–media thickness.
Data given as mean ± SD and range.
Significant difference among three groups.
Significant difference between Groups 1 and 2.
Significant difference between Groups 1 and 3.
Significant differences between Groups 2 and 3 and Groups 1 and 3.
The study was approved by the local ethics committee and conforms to the provisions of the Declaration of Helsinki (as revised in Fortaleza, Brazil, October 2013). Written informed consent was obtained from the patient’s families.
Results
Mean ± SD (range) age of all patients was 15.3 ± 4.15 years (8.5–22 years). In total, 83 patients were recruited; 41 (49.4%) were female and 42 (50.6%) were male. All patients were pubertal according to Tanner staging. The mean age of the patients between the three groups was significantly different (
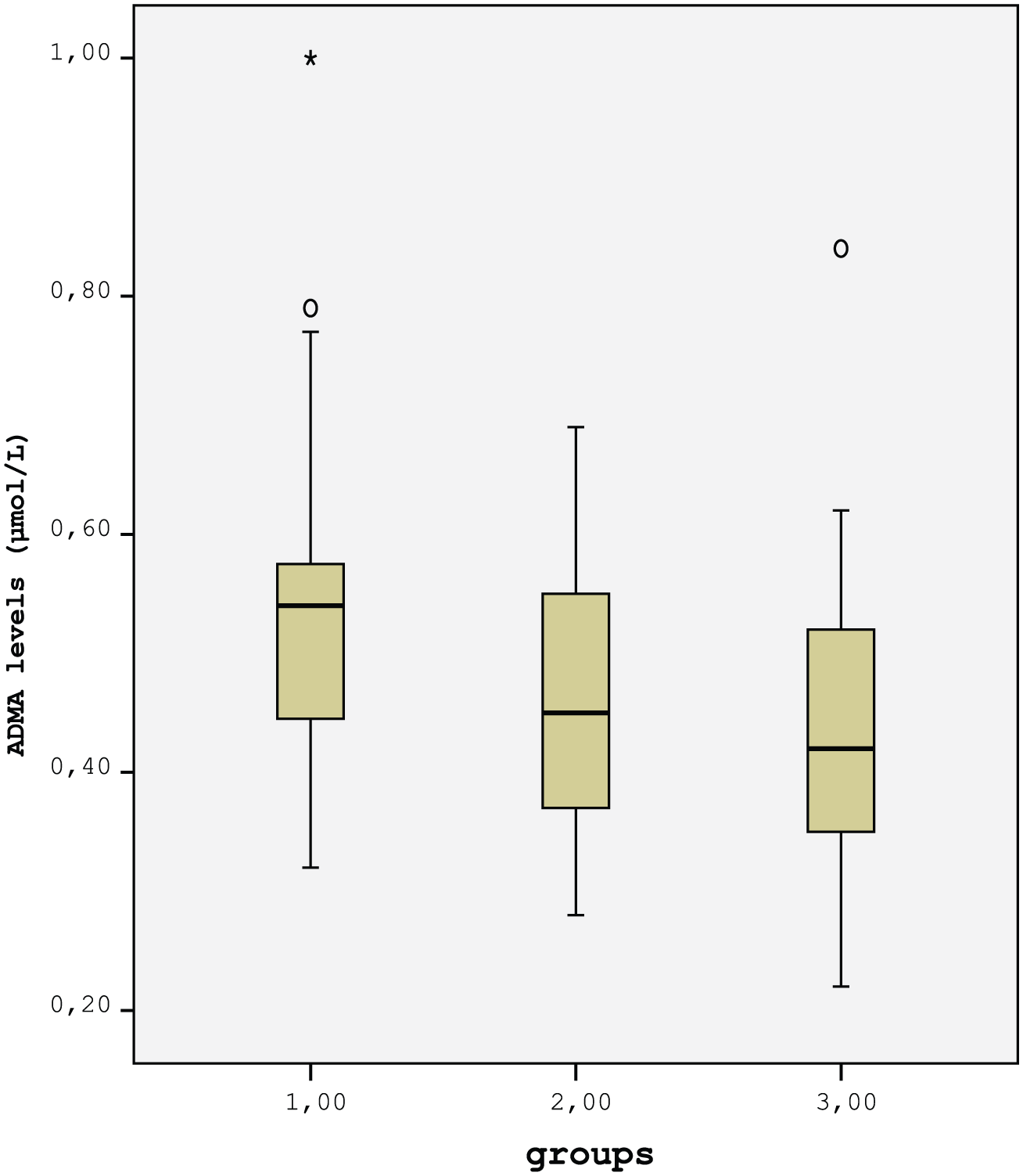
Comparison of ADMA levels between groups. ADMA levels were significantly higher in Group 1 compared to Group 3 (
Table 2 shows M-mode, pulsed Doppler and TDE indices in children and adolescents with T1DM according to diabetes duration. LV posterior wall thickness (LVpWT) and IVST were significantly lower in Group 1 compared with Group 2 and Group 3 (
Differences of echocardiographic indices among three groups.
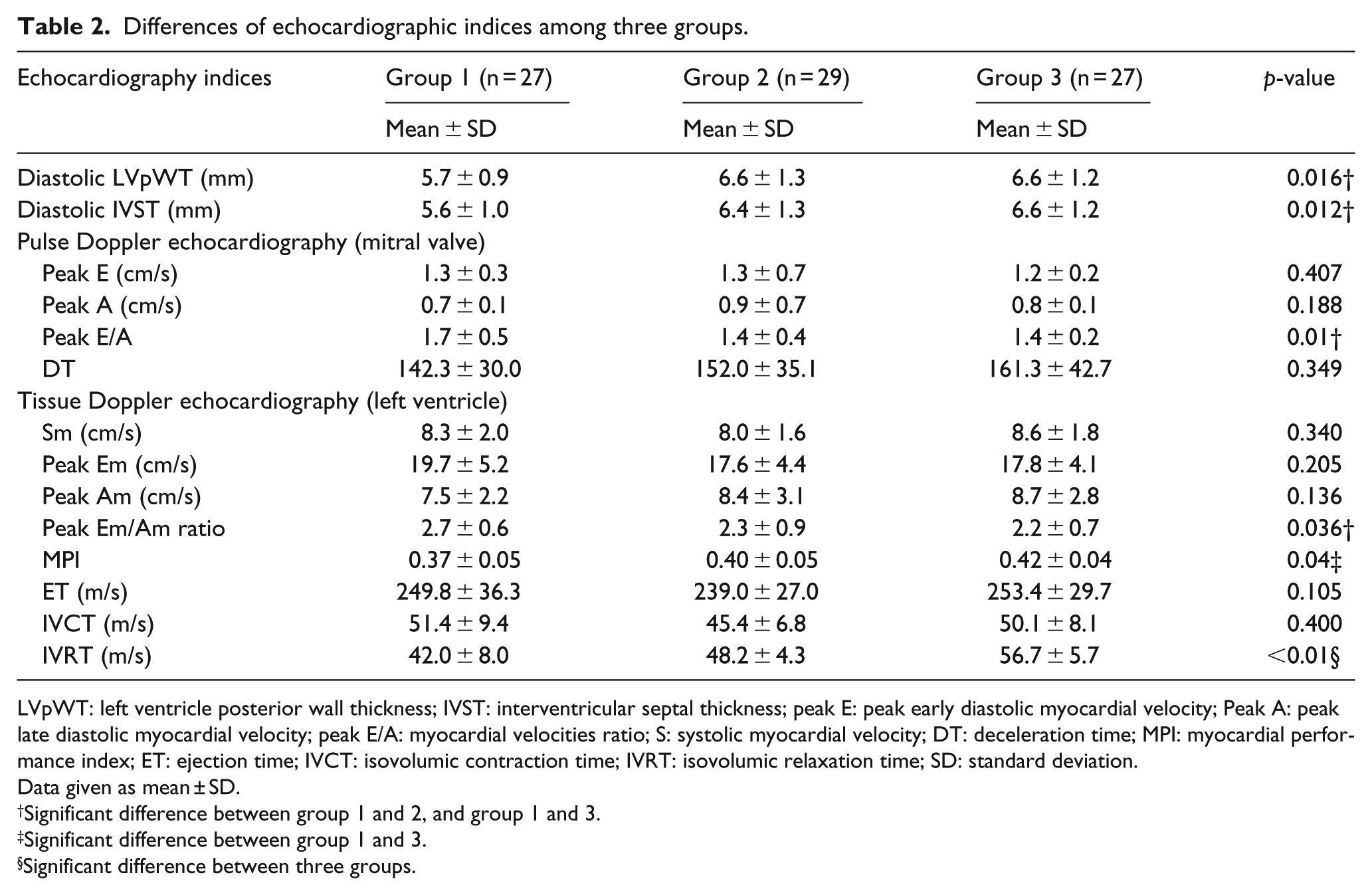
LVpWT: left ventricle posterior wall thickness; IVST: interventricular septal thickness; peak E: peak early diastolic myocardial velocity; Peak A: peak late diastolic myocardial velocity; peak E/A: myocardial velocities ratio; S: systolic myocardial velocity; DT: deceleration time; MPI: myocardial performance index; ET: ejection time; IVCT: isovolumic contraction time; IVRT: isovolumic relaxation time; SD: standard deviation.
Data given as mean ± SD.
Significant difference between group 1 and 2, and group 1 and 3.
Significant difference between group 1 and 3.
Significant difference between three groups.
The Em/Am index showed a significant difference between the three groups using both pulse Doppler and TDE. In pulse Doppler assessment, this difference is more evident when Group 1 is compared to Groups 2 and 3 (
Other indices that differed between the groups were IVRT and MPI. As the duration of diabetes increased from Group 1 to Group 3, both indices increased. IVRT was significantly different between the three groups (
Correlation analysis
The relationship between ADMA levels and other parameters was assessed in all patients. ADMA levels showed significant inverse association with age (r = −0.455,
Correlation was evaluated separately within the groups. The negative correlation between age and ADMA levels was found only in Group 1 (r = −0.498,
Correlations of ADMA levels with other parameters.
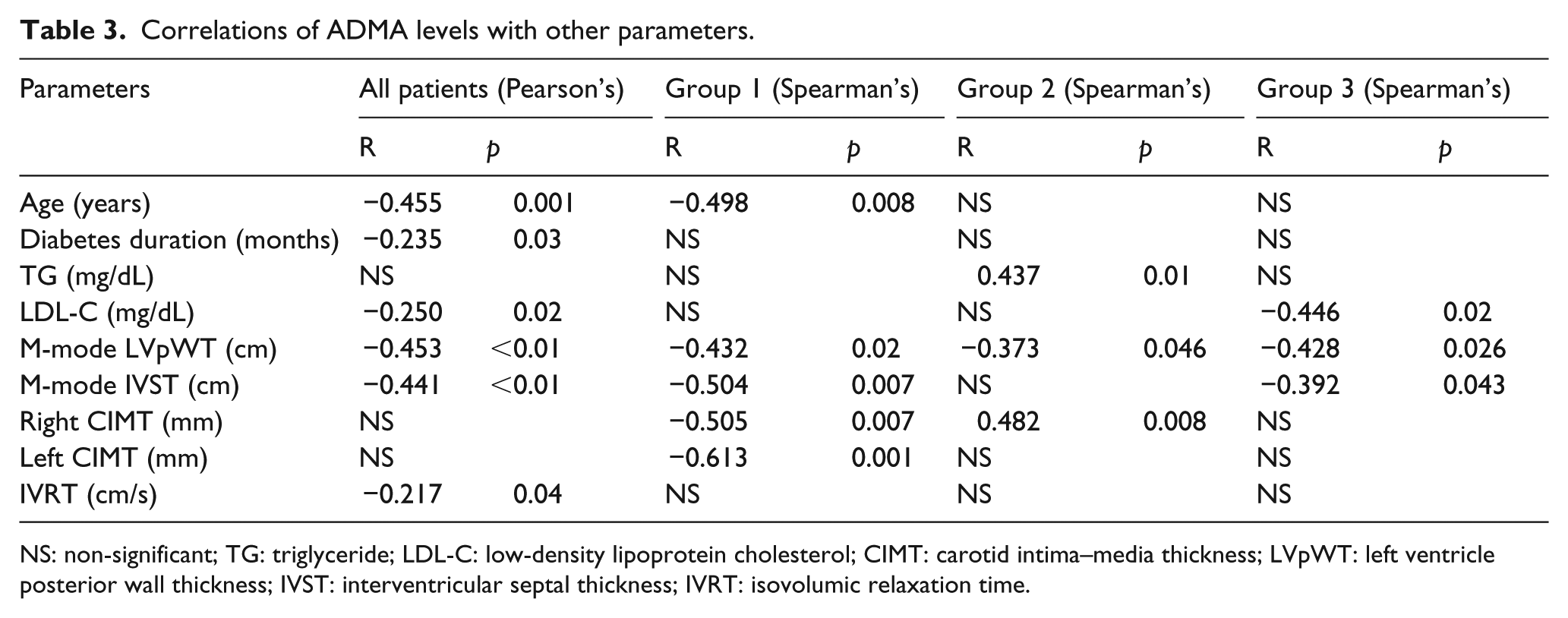
NS: non-significant; TG: triglyceride; LDL-C: low-density lipoprotein cholesterol; CIMT: carotid intima–media thickness; LVpWT: left ventricle posterior wall thickness; IVST: interventricular septal thickness; IVRT: isovolumic relaxation time.
There was no relationship between HbA1c and ADMA levels within any of the groups (
Discussion
The results of our study are in striking contrast with the results of previous adult studies. 9 In this study, ADMA concentrations decreased with age and duration of diabetes. ADMA levels were found to be high in both Type 1 and Type 2 diabetic patients in previous adult studies.9,24,25 In contrast to these studies, ADMA levels in children with Type 1 diabetes were found to be low when compared with healthy children.10,26 We did not compare the ADMA levels of subjects with T1DM to a control group in this study. However, when we compared the ADMA levels of our patients with those of healthy children from the study of Huemer et al., 10 the ADMA levels of our patients with duration of diabetes less than 10 years were significantly higher than those of healthy children. Patients with diabetes duration of shorter than 5 years have significantly higher ADMA concentration. After 5 years of diabetes duration, ADMA levels had reached a nadir and did not significantly reduce further with longer diabetes duration. ADMA levels were lower in subjects with T1DM who had longer diabetes duration and older age in our study. In consistent with our study, ADMA levels also decreased with increasing diabetes duration and age in the study of Marcovecchio et al. 11
In T1DM children, endothelial function is impaired within the first decade of disease, and this is manifest in an increase in CIMT, an indicator of subclinical atherosclerosis.13,27 We found that CIMT was significantly elevated in children with duration of diabetes more than 10 years. Similar to the result of our study in diabetic children, two of the most important factors affecting the increase in CIMT were reported to be diabetes duration and age.28,29 In our study, the increase in CIMT may be related to both the duration of diabetes and age. In adults with Type 2 diabetes mellitus (T2DM), elevated ADMA level is associated with increased CIMT and a high prevalence of macrovascular disease. 23 In our study, there were different associations between ADMA and CIMT between the groups. In children with diabetes duration 1–5 years, there was a negative correlation between ADMA levels and CIMT, whereas a positive correlation was found in those with diabetes duration of 5–10 years. There are no other studies addressing this finding in children with T1DM. Our finding may suggest that the association between ADMA levels and the development of atherosclerosis in T1DM begins after the fifth year of disease in children with T1DM, but this relationship requires further investigation to confirm.
Chronic hyperglycaemia in DM damages the heart and causes myocardial dysfunction.18,30,31 In this study, we found a significant increase in LVpWT and IVST on conventional echocardiography after 5 years of diabetes duration. In previous studies, no change in IVST was detected in children and adolescents with T1DM when compared to healthy controls.18,29 As in our study, Khattab and Soliman 18 found higher diastolic LVpWT in children with T1DM diabetes compared to healthy children. The positive correlation between LVpWT and duration of diabetes has been reported by several studies.18,30,32 Our findings indicate that diastolic filling abnormalities appear to begin after 5 years of diabetes duration in children and adolescents. This result was supported by the TDE data reported here.
In this study, the E/A ratio was significantly decreased in patients with diabetes duration longer than 5 years compared to those with diabetes duration of between 1 and 5 years, both by conventional Doppler and TDE. This ratio continued to decline in patients who have diabetes duration of longer than 10 years, but this further decline was not significant. In addition, a negative correlation between the duration of diabetes and the E/A ratio confirms that this ratio decreases as the duration of disease increases. This finding supports the impairment of cardiac function in long-term diabetes. In contrast with our study, other recent studies have reported that there was no significant association between E/A and duration of diabetes. However, it was reported that the E/A ratio was lower in T1DM children than in healthy controls.18,30,31
We found that IVRT was prolonged, and MPI was impaired in long-term diabetes. While IVRT showed significant differences between all groups, MPI showed a significant impairment only after the fifth year. These indices were found to be significantly related to the duration of diabetes, in contrast to previous reports. 30 All findings indicate that diastolic filling defects occur when the duration of diabetes is prolonged. Similar to our findings, From et al. 33 showed a direct correlation between the duration of DM and LV diastolic dysfunction and reported that significant LV diastolic dysfunction occurs 4 years after the onset of DM, independent of coronary disease or hypertension.
We confirm the negative correlation between HbA1c levels, a well-recognized indicator of glycaemic control, and the Em/Am ratio suggesting that this ratio is the first parameter to show impairment as glycaemic control worsens. A similar result was reported by Kim and Kim 34 and Salem et al. 30 However, this has not been a consistent finding with Khattab and Soliman 18 reporting no association. However, we calculated mean HbA1c levels in patients, whereas in other studies, only the level of HbA1c at the time of study was considered.18,30,34
To date, there is only one other study showing the relationship between ADMA levels and echocardiographic parameters in childhood T1DM. Abd El Dayem et al. 20 not only found a negative relationship between NO levels and echocardiographic parameters but also showed that the most important indicator of NO levels is ADMA concentration. In this study, we found that ADMA levels were negatively associated with LVpWT and IVST. In addition, we found that IVRT was prolonged in TDE when ADMA levels decline. All of these findings indicate that an increase in ADMA levels is protective both in terms of development of subclinical atherosclerosis and myocardial function. In addition, in adult studies of patients without diabetes, higher ADMA levels have been shown to be associated with poor diastolic dysfunction and cardiovascular risk profile.35,36
Huemer et al. have suggested that lower ADMA levels in children and adolescents with T1DM are indicator of impaired protection against oxidative stress. They argued that this assumption was supported by the relationship between high HbA1c and low ADMA levels. 10 We did not find a similar relationship. But the negative association between ADMA and CIMT and IVRT, reported here, supports this assumption. Additionally, the negative relationship between ADMA and LDL-C, which is also claimed to play an important role in the atherogenic process, 37 supports the protective role of ADMA in oxidative stress.
The limitations of our study are as follows: First, there was no control group consisting of healthy children. Second, our study design is cross-sectional. Longitudinal monitoring of patients is required to make more reliable observations concerning ADMA levels, TDE and CIMT. Finally, the age range of children and adolescents included in the study was very wide because diabetes duration was used to define our three study groups.
In conclusion, significant changes were observed in CIMT, and echocardiographic parameters despite reduced ADMA levels in children and adolescents with long-term diabetes. Subclinical atherosclerosis seems to become apparent after the 10th year of the disease. Echocardiographic changes lead to diastolic dysfunction and appear to develop as early as the fifth year of disease, even in patients with no other complications. Furthermore, some changes in parameters of cardiac function may be related to poor metabolic control. Lower ADMA levels may be associated with increased cardiovascular risk and poorer diastolic function inferring a protective role for higher levels of ADMA in early paediatric T1DM, although this suggestion requires further study.
Footnotes
Acknowledgements
We would like to thank all the patients and their families for participating in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
Our study was supported by the Faculty of Medicine at Celal Bayar University, Manisa, Turkey, within the scope of our normal scientific research projects.
