Abstract
Aim:
To investigate, in a large cohort of 2494 individuals with diabetes mellitus, whether functional single nucleotide polymorphisms in the promoter region of tumour necrosis factor (TNF) and lymphotoxin-alpha (LTA) genes are associated with type of diabetes or presence of diabetic retinopathy.
Methods:
A total of 334 type 1 diabetes and 999 type 2 diabetes participants with sight-threatening diabetic retinopathy, and 260 type 1 diabetes and 901 type 2 diabetes participants with no diabetic retinopathy or minimal non-proliferative diabetic retinopathy, were genotyped for two single nucleotide polymorphisms (rs1800629 and rs361525).
Results:
The A allele of rs1800629 was associated with type 1 diabetes (p < 0.001; odds ratio = 0.62). After adjustment for age, sex, diabetes duration, HbA1c, hypertension and nephropathy, no significant association was found between rs1800629 or rs361525 and sight-threatening diabetic retinopathy.
Conclusion:
An association between the A allele of rs1800629 and type of diabetes was found. No association was found between two promoter variants of TNF and LTA, and diabetic retinopathy in a large cohort of Caucasian patients with type 1 diabetes and type 2 diabetes.
Diabetes mellitus (DM) is a disease of increasing prevalence, associated with significant mortality and morbidity. Microvascular complications of DM, including diabetic retinopathy (DR) and diabetic nephropathy (DN), are of complex aetiology. Pathological changes to retinal microvasculature are caused by a cascade of events involving the formation of advanced glycation end products (AGEs), in the presence of sustained hyperglycaemia. 1 The interaction of AGEs with their receptors (RAGEs) on the surface of macrophages and endothelial cells results in an increase in the synthesis and secretion of pro-inflammatory cytokines including TNF; previously known as TNF-α). 1 TNF is implicated in the pathogenesis of DR, through its contribution to blood-retinal barrier breakdown and neovascularisation. 1 Another pro-inflammatory cytokine LT-α (previously known as TNF-β) binds the same TNF receptors, with similar downstream effects. 2
TNF promoter polymorphisms have been extensively studied across a range of diseases with rs1800629 (TNF −308) and rs361525 (TNF −238) being the most widely investigated single nucleotide polymorphisms (SNPs). Individuals homozygous for the less common TNF −308 A allele have been shown to have higher circulating TNF levels than those homozygous for the G allele and have worse outcomes in response to infectious diseases. 3 The TNF −238A allele (rs361525) has also been implicated in a number of disease states with the A allele conferring protection against autoimmune diseases including rheumatoid arthritis. 3
Both the TNF and LTA genes are located on chromosome 6 in close proximity to each other, in the human leukocyte antigen (HLA) class III region. We conducted this study to investigate the regulatory regions of the TNF and LTA genes, by investigating two promoter polymorphisms (rs1800629 and rs361525) known to influence TNF and LTA expression, and their relationship to DM and DR in a large and well-characterised cohort of 2494 Caucasian patients with type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM).
Methods
Individuals with DM were recruited from ophthalmology and endocrine clinics in Australia and the United Kingdom, including Flinders Medical Centre, The Royal Adelaide Hospital, The Queen Elizabeth Hospital, The Royal Melbourne Hospital, Royal Victorian Eye and Ear Hospital, St Vincent’s Hospital, Sydney Eye Hospital, The Repatriation General Hospital and Canberra Hospital in Australia; and The National Institute for Health Research Biomedical Research Centre at Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology, London, UK. The study was approved by the Human Research Ethics Committees (HREC) in Australia (Southern Adelaide Clinical HREC, Royal Adelaide Hospital HREC, TQEH/LMH/MH HREC, Royal Melbourne Hospital HREC, Royal Victorian Eye and Ear Hospital HREC, St Vincent’s Hospital HREC, South Eastern Sydney Illawarra HREC) and The NHS Health Research Authority in London. Written informed consent was obtained from each participant and the project conformed to the tenets of the Declaration of Helsinki.
A detailed description of recruitment protocols and study participants has been reported previously. 4 In brief, patients were required to meet the following inclusion criteria: (1) at least 18 years of age and (2) on medical treatment for DM (either oral hypoglycaemic agents or insulin therapy). Individuals with T2DM were required to be on medical treatment for DM for at least 5 years prior to inclusion in the study. Social, demographic and medical history was collected via a questionnaire. Retinopathy status was established via ophthalmic examination in accordance with the modified Early Treatment Diabetic Retinopathy Study (ETDRS) criteria. Sight-threatening diabetic retinopathy (STDR) was based on the patient’s worst ever DR grading and was defined as the presence of either severe non-proliferative diabetic retinopathy (NPDR), proliferative diabetic retinopathy (PDR) or diabetic macular oedema (DME), in at least one eye. Controls were defined as those whose retinopathy grading had never been worse than minimal NPDR, with no history of DME in either eye. An additional 1445 individuals with DM included in this analysis were recruited since the previous report using identical protocols.
DNA was extracted from whole blood using QIAamp Blood DNA Maxi Kits (Qiagen). SNPs rs1800629 and rs361525 were genotyped using iPLEX Gold chemistry on an autoflex Mass Spectrometer (Sequenom, San Diego, CA). SNPs were in Hardy–Weinberg equilibrium. Baseline characteristics of cases and controls were compared using the Mann–Whitney U test for non-parametric, continuous variables, and χ2 test for dichotomous variables, using SPSS version 20.0 for Mac OS X (IBM SPSS Statistics 20.0, SPSS Inc., Armonk, NY). Testing for association of each SNP with DR was undertaken with the χ2 test for univariate analysis and binary logistic regression for multivariate analysis in PLINK (version 1.06).
Findings and discussion
A total of 2494 individuals with DM were recruited and genotyped for this study. Of these, 1161 had No DR or minimal NPDR (260 T1DM and 901 T2DM), 189 had severe NPDR (25 T1DM and 164 T2DM), 734 had PDR (252 T1DM and 482 T2DM) and 909 had DME (140 T1DM and 769 T2DM). Of those, 499 with DME also had co-existing PDR or severe NPDR.
Neither rs1800629 nor rs361525 are associated with STDR, PDR or DME
This study found no association between SNPs rs1800629 and rs361525 and STDR, PDR or DME in either T1DM or T2DM patients in a multivariate logistic regression analysis controlling for age, sex, duration of DM, HbA1c, hypertension and nephropathy (Table 1).
Allelic association of rs1800629 (−308G > A) and rs361525 (−238G > A) with STDR, PDR and DME for T1DM and T2DM.
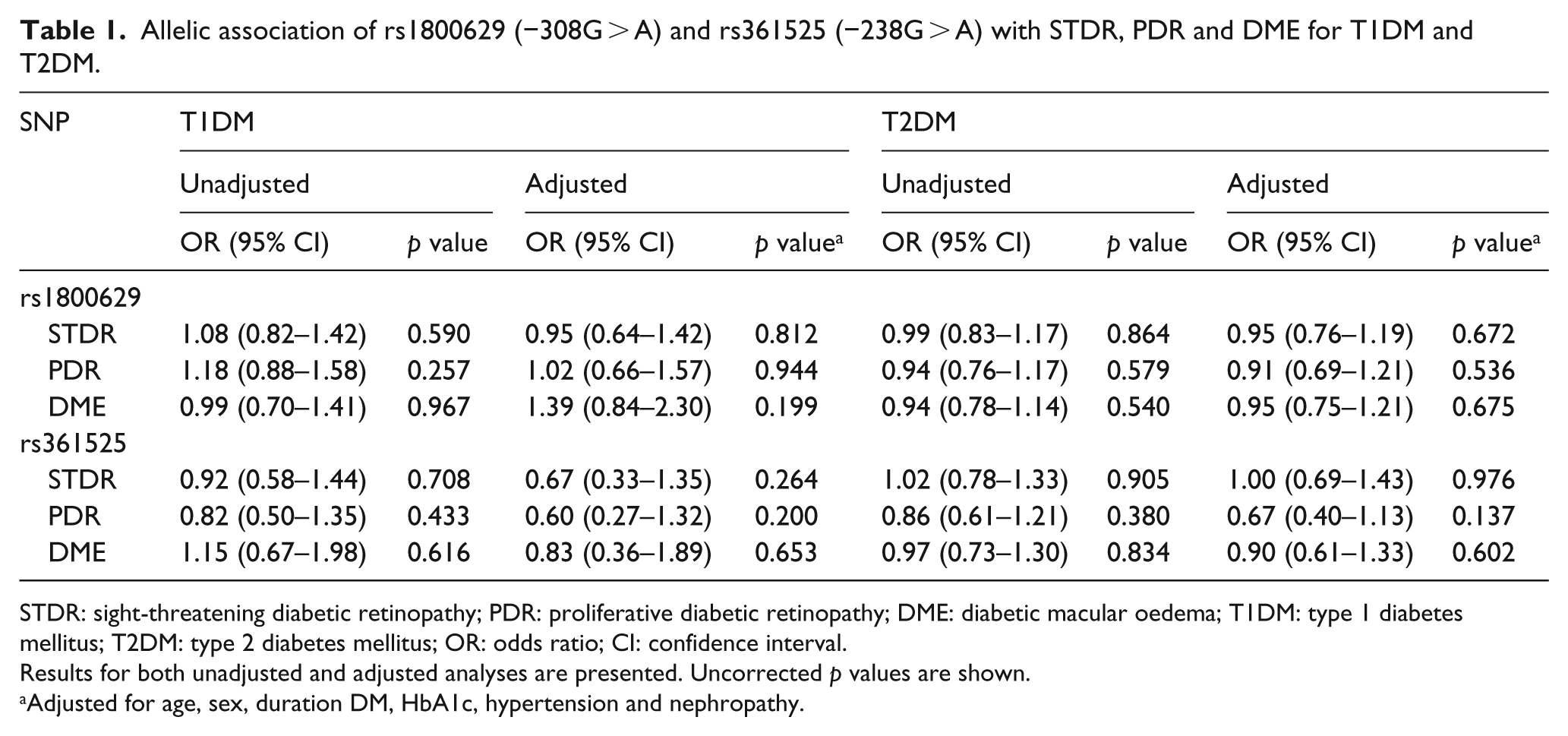
STDR: sight-threatening diabetic retinopathy; PDR: proliferative diabetic retinopathy; DME: diabetic macular oedema; T1DM: type 1 diabetes mellitus; T2DM: type 2 diabetes mellitus; OR: odds ratio; CI: confidence interval.
Results for both unadjusted and adjusted analyses are presented. Uncorrected p values are shown.
Adjusted for age, sex, duration DM, HbA1c, hypertension and nephropathy.
The rs1800629 promoter SNP has previously been investigated in relation to DR in a large study including 742 T1DM and 2957 T2DM Caucasian patients. 5 Cases with STDR were compared with controls with no DR or NPDR, and no significant association between this promoter polymorphism and STDR risk was found. This finding is comparable to the results from our study, which evaluated differences between phenotypic extremes by comparing controls with no DR or minimal NPDR to cases with STDR. Furthermore, rs1800629 has been studied in smaller Chinese and Japanese cohorts with T2DM, again with no significant association found in either ethnic group.6,7 Conversely, Sesti et al. 8 recently reported a positive association between the A allele of rs1800629 and an increased risk of PDR in a cohort of Brazilian Caucasian patients. To date, no association has been found between rs361525 and DR, consistent with findings from the current study.
Functional studies have suggested a role for TNF as a biomarker for DR. Circulating levels of TNF are increased in the serum of patients with T1DM and PDR compared to patients with DM but without retinopathy. 9 mRNA expression of TNF as well as the level of soluble TNF receptors is elevated in the vitreous of patients with PDR. 10 Furthermore, inhibition of TNF with angiopoietin-1 has shown promising outcomes in preventing early DR in a diabetic rat model. 11
There is increasing evidence to suggest that a complex interaction exists between TNF, LTA and other genes in the HLA class III region, and this may explain the discrepancy between genetic and functional work to date. Alleles within the major histocompatibility complexb (MHC) region are in strong linkage disequilibrium making a direct association between SNPs in this region and TNF phenotypes less likely. It has been postulated that alternative pathways may be responsible for TNF expression including regulation by linked genes, interaction between the 3′untranslated region outside the TNF promoter and the −308 element and epigenetic control via TNF promoter methylation. 3 Further investigation of the relationship between these genes and factors modulating their expression is required to gain a better understanding of the role of these pro-inflammatory cytokines in the pathogenesis of DR.
rs1800629 was significantly associated with DM type
In this study, the frequency of the rs1800629 A allele was significantly higher in patients with T1DM compared to patients with T2DM (minor allele frequency (MAF) 23.5% in T1DM compared to 15.9% in T2DM) [odds ratio (OR) = 0.62; 95% confidence interval (CI) = 0.53–0.71; p < 0.001], consistent with previously reported findings. Our study also showed that the MAF of rs1800629 was significantly higher in those without hypertension (OR = 0.848; 95% CI = 0.73–0.98; p = 0.025), subjects of younger age (t = −4.0; p < 0.001), and those with longer duration of diabetes (t = 2.44; p = 0.01) when all diabetics were analysed together. These differences reflect the risk factor profile of the T1DM cohort and are driven by the T1DM association. Each of the above-mentioned significant associations survived Bonferroni correction (p < 0.025). rs361525 was not found to be associated with any covariates tested.
Recent research has shown that the TNF rs1800629 allele is in linkage disequilibrium with the MHC haplotype HLA-A1-B8-DR3, 12 which may explain the functional connection between the A allele, high TNF production and insulin-dependent DM.3,13 There is no clear evidence to suggest a direct role of SNPs in the TNF/LTA locus increasing T1DM susceptibility, independent of HLA DR- and DQ-haplotypes. 13
Conclusion
In conclusion, the risk allele of rs1800629 (TNF −308A) is associated with T1DM. We found no association between either of the two polymorphisms in the promoter region of TNF and LTA and STDR, DME or PDR, in patients with either T1DM or T2DM. It is clear that increased levels of TNF, both locally and systemically, are associated with DR risk; however, further investigation of the complex interplay between TNF, and LTA genes with regard to the HLA haplotypes, and epigenetic modifications is required to identify the specific effect of these variants on microvascular complications of DM, including DR, and determine the underlying biological drivers of these elevated levels.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by the National Health and Medical Research Council (NHMRC) of Australia project grant (no. 595918), the National Institute for Health Research (NIHR) Biomedical Research Centre at Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology. Georgia Kaidonis is funded by an NHMRC Clinical Research Postgraduate Scholarship and an Avant Doctor in Training Research Scholarship. Jamie E Craig was supported in part by an NHMRC Practitioner Fellowship and Kathryn P Burdon by an NHMRC Senior Research Fellowship.
