Abstract
The study was designed to discover the biological function of endotheliocyte-derived microparticles in diabetes condition. A quantitative shotgun proteomics methodology was performed to study the proteome of these high-glucose-activated endothelial microparticles. A total of 1428 proteins were identified, containing 1421 and 1423 proteins in control and high-glucose groups, respectively. According to the ExoCarta database, 669 proteins have previously been identified in microparticles. The proteins associated with disease were identified in this study, and notably, 30 proteins have been reported to be associated with Alzheimer’s disease, including amyloid beta A4 protein. Besides, the peptide abundance of amyloid beta A4 protein from control group was much less than that from high-glucose group. In conclusion, this work revealed the proteome of endothelial microparticles in mimic diabetes condition and provided a new proteomic evidence for Alzheimer’s disease to be counted as the type 3 diabetes.
Introduction
Microparticles are membrane-enclosed vesicles of 30–1000 nm in diameter. Microparticles can mediate intercellular communication and transfer components within microparticles to targeted cells. 1 Their content has been reported to be regulated under multiple clinical conditions, including cardiovascular disease, ischaemic attacks, diabetes, sepsis, metabolic disease, autoimmunity disease and cancer. It is also known that microparticles can be affected by various physiologic factors, such as the cholesterol level in plasma, development of chronic or acute diseases and even certain physiologic activity, for example, pregnancy and ageing. Specifically, endotheliocyte-derived microparticles (EMPs, also named as endothelial microparticles) have been described as a new marker of endothelial cell dysfunction. 2
Elevated EMPs have been observed in metabolic diseases, such as type 2 diabetes and obesity.3,4 Studies have demonstrated that EMPs released from human coronary endothelial cells under stimulation of high glucose concentrations mimic the diabetic conditions and hold higher nicotinamide adenine dinucleotide phosphate (NADPH) oxidase activity. These high-glucose-induced EMPs promote endothelial inflammation and exacerbate endothelial dysfunction. However, compared with the relatively abundant studies of the behaviour of microparticles in multiple diseases, the proteomic analysis of protein content is still inadequate. A recent paper utilized proteomic approaches to study changes in the EMP proteome after in vitro treatment of endothelial cells by tumour necrosis factor (TNF)-α and revealed several changes due to inflammation.
In this study, a quantitative shotgun proteomic analysis was performed to compare the proteome change occurring in EMPs after high-glucose treatment and thereafter to explore the biological function of EMPs under high-glucose condition, a mimic condition of diabetes.
Methods
Sample preparation
Human umbilical vein endothelial cells (HUVECs) were extracted and cultured in Endothelial Cell Medium (ScienCell). HUVECs between passages 3 to 5 were treated by high glucoses (22.2 mM) or normal condition (5.6 mM) when reaching around 90% confluence. Culture medium was collected after 4 h of treatment for immediate centrifugation at 800g for 5 min at 4°C to deplete cell debris. The debris-free medium was ultracentrifuged at 100,000
Proteomic analysis
The resuspended microparticles were lysed with radio immunoprecipitation assay buffer. The protein of the microparticles was fractionated by a sodium dodecyl sulphate polyacrylamide gel electrophoresis, and the gel lanes were cut into 14 pieces and digested using trypsin. The tryptic peptide mixtures were analysed on a LTQ-Orbitrap Elite system equipped with a Dionex 3000 UPLC. For the Mascot searches, the results were filtered using a threshold peptide ion score of 40 and at least two matching peptides. For the sequence searches, the results were filtered based on the XCorr scores <1.5 (+1 ions), >2.0 (+2 ions), >2.5 (+3 ions). The false discovery rate (FDR) for these searches was determined to be less than 1%.
The differential analysis of proteins in these samples was performed using the label-free spectral count method. The relative abundant change of certain protein was considered significant as long as its cut ratio (CR) exceeds 2.0, which ensured the standard error was less than 20% for a moderate-to-high abundant protein.
Results
As a result, 1428 proteins were identified, containing 1421 presented in control group (CG) and 1423 in high-glucose-treated group (HG). Between two groups, 83 different proteins were detected, among which 48 were up-regulated and 36 were down-regulated by high-glucose treatment (Supplement Figure 1C).
According to ExoCarta database, 669 proteins in EMPs have been identified in microparticles previously, while the remaining 759 proteins were reported for the first time (Supplement Figure 1D). The most previously reported microparticle proteins include HSP90, HSP70, annexin A2, annexin A6, actin and elongation factor-1 alpha, all of which were detected in our study at high concentration. In order to demonstrate the protein composition of EMPs, we focused on the 1416 proteins that were identified in both groups. Gene ontology analysis results were shown in Supplement Figures 2A and B. Several pathway analyses were carried out in order to establish a full perspective of the microparticles proteome (Supplement Figures 2C to F).
The proteins associated with disease were identified in the study. Predictably, the most significant relationship was identified to be atherosclerosis (Supplement Figure 2F). However, it was interesting to note that 30 proteins were identified in this analysis, which have been reported to be associated with Alzheimer’s disease. The glycaemic index (GI) number for the associated proteins is listed as follows: 91199540, 126012571, 4503143, 16753203, 4501881, 155969707, 24797067, 24638433, 27765085, 7669492, 4502167, 169790923, 13676857, 39930351, 5901948, 156523970, 4557367, 4506141, 21361278, 38373669, 10835159, 156523968, 19923748, 33620730, 167466173, 40254422, 66529294, 4557014, 4503273, 4502027, including amyloid beta A4 protein (APP). Besides, the peptide abundance of APP from HG was much less than that from CG. A complete list of the proteins related to Alzheimer’s disease and their ratios between CG and HG are shown in Table 1.
A complete list of the proteins related to Alzheimer’s disease and their ratio between control group (CG) and high-glucose-treated group (HG).
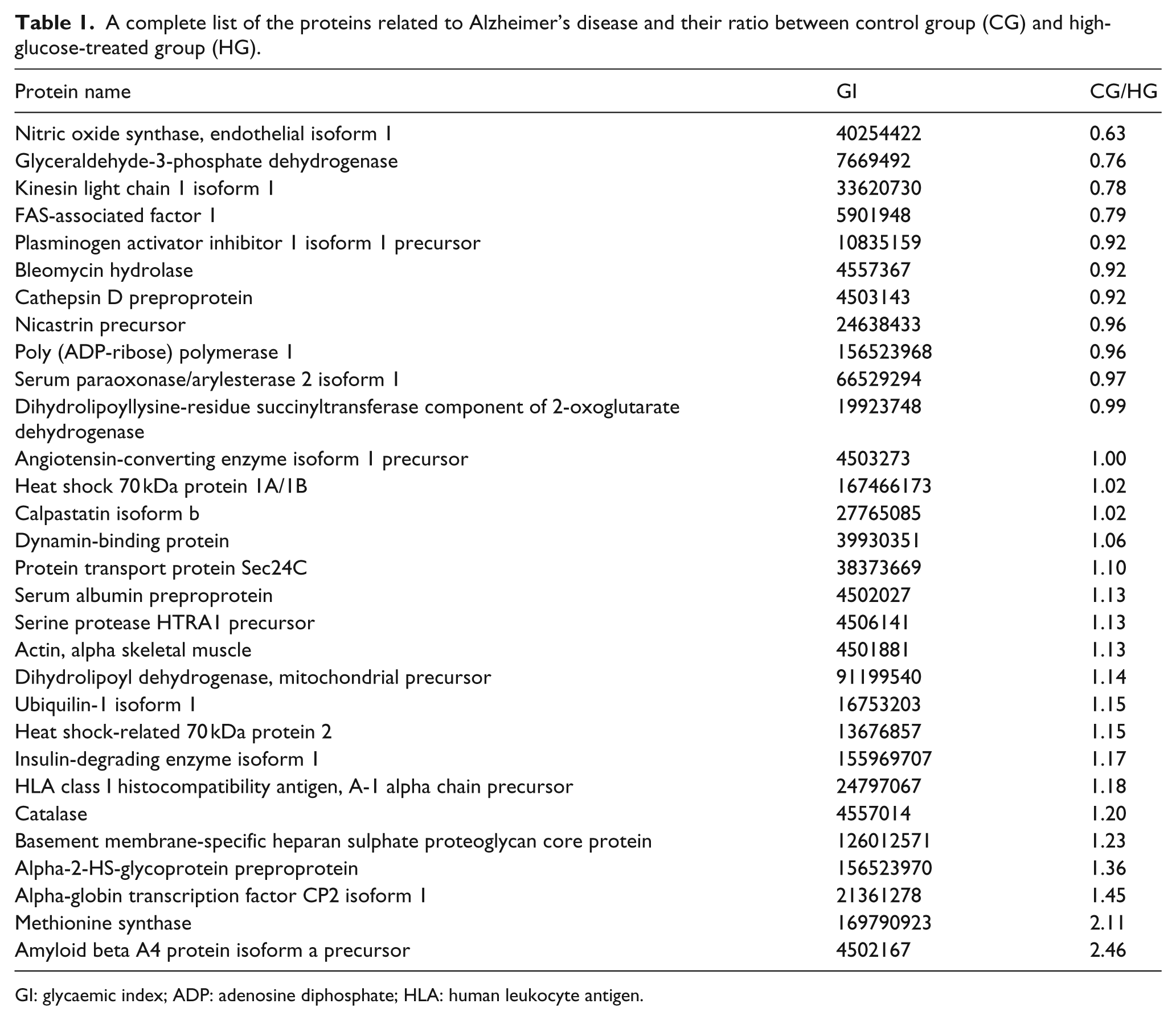
GI: glycaemic index; ADP: adenosine diphosphate; HLA: human leukocyte antigen.
Discussion
Previous epidemiologic studies also showed a strong relationship between Alzheimer’s disease and diabetes,6,7 and patients with diabetes are more inclined to dementia, 8 which may be partly contributed by the combinative damage to endothelial system. On the other hand, Alzheimer’s disease patients showed significantly higher susceptibility to insulin resistance. 6 Due to the wide overlapping pathophysiology characteristics between Alzheimer’s disease and type 2 diabetes mellitus, it has been hypothesized that Alzheimer’s disease might be ‘type 3 diabetes’. 7 Elevation of plasma levels of glucose and insulin leads to a decrease in plasma amyloid precursor protein concentration, which may be indicative of clinical significance of high blood sugar in Alzheimer’s disease pathogenesis. Both animal models and clinical studies with type 2 diabetic patients have shown that altered glucose regulation impairs cognitive capacity in general and produces classic pathological processes such as amyloid deposition, which is precipitated by APP identified found in this study. 8
Previous studies have shown that APP was contained in microparticles or exosomes in in vitro model of Alzheimer’s disease and in plaques of Alzheimer’s disease patients in vivo, and platelet microparticles is the major source of plasma APP. 9 It should be pointed out that according to some previous reports, the APP was released by exosomes, which are also membrane-secreted vesicles with diameter even smaller than microparticles. However, as shown in most previous research about EMPs, the microparticles and exosomes were not distinguished on purpose in our study, and the term ‘microparticles’ in this study refers to the combined meaning of microparticles and exosomes, that is to say, the membrane vesicles derived from samples after being ultracentrifuged at 100,000g speed.
Cerebral deposition of the amyloid β-peptide (Aβ), approximately 40 residue fragments of the integral membrane protein, APP, have been implicated as a probable cause of Alzheimer’s disease. 10 The cleavage of APP generates accumulation of Aβ-rich senile plaques and intracellular neurofibrillary tangles, triggering hyper-phosphorylated Tau and synapse loss accumulation. The molecular changes further lead to increased generation of reactive oxygen species and reactive nitrogen species, which have been confirmed to play a detrimental role in Alzheimer’s disease. 11 It is reported that APP degradation was inhibited under high-glucose treatment. 12 Whereas in our study, the high-glucose treated endothelial cell did show the upgraded expression of APP, with a down-regulation in microparticles resulting from high-glucose treatment, which may indicate that high-glucose treatment can induce the accumulation of APP in endothelium. Whether there exists an interaction between the APP released from endothelial cells and activated APP remains to be further studied.
In conclusion, this study revealed the proteome of the high-glucose-activated and normal endothelial microparticles and detected 83 different proteins between them. Notably, 30 Alzheimer’s disease–related proteins were found in comparative proteomics study, and APP from high-glucose-treated EMPs was much less, which provided an interesting evidence for Alzheimer’s disease to be counted as the type 3 diabetes.
Footnotes
Author’s contributions
Lingyun Zu and Chenguang Niu have contributed equally to this work.
Declaration of conflicting interests
The authors declare no conflicts of interest relevant to this article.
Funding
This work was supported by Grants 2011CB503900 from ‘973’ National S&T Major Project. This work was also supported by Grants 81170101, 81370235 and 81300076 from the National Natural Science Foundation of China; and Grants 7122106 and 7132195 from the Natural Science Foundation of Beijing, China. The Orbitrap Elite instrument used in this study was purchased with an NIH S10 grant 1S10RR031537-01 from the United States.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
