Abstract
Purpose:
To investigate the expression and function of serum response factor in podocyte epithelial–mesenchymal transition of diabetic nephropathy.
Methods:
The expression of serum response factor, epithelial markers and mesenchymal markers was examined in podocytes or renal cortex tissues following high glucose. Serum response factor was upregulated by its plasmids and downregulated by CCG-1423 to investigate how it influenced podocyte epithelial–mesenchymal transition in diabetic nephropathy. Streptozotocin was used to generate diabetes mellitus in rats.
Results:
In podocytes after high glucose treatment, serum response factor and mesenchymal markers increased, while epithelial markers declined. Similar changes were observed in vivo. Serum response factor overexpression in podocytes induced expression of Snail, an important transcription factor mediating epithelial–mesenchymal transition. Blockade of serum response factor reduced Snail induction, protected podocytes from epithelial–mesenchymal transition and ameliorated proteinuria.
Conclusion:
Together, increased serum response factor activity provokes podocytes’ epithelial–mesenchymal transition and dysfunction in diabetic nephropathy. Targeting serum response factor by small-molecule inhibitor may be an attractive therapeutic strategy for diabetic nephropathy.
Keywords
Introduction
The incidence of diabetic nephropathy (DN) in diabetes mellitus (DM) patients is approximately 20%–40%, and DN is the most severe complication and chief cause of death in DM. 1 With the rapid increase in the number of DM patients, the incidence of DN rises year by year. It has become the first reason leading to end-stage renal disease (ESRD) in some countries. 2 The main pathological changes of early stage DN are glomerulus hypertrophy, the thickening of glomerular basement membrane and the progressive accumulation of extracellular matrix (ECM) in mesangial area. Late-stage changes are interstitial fibrosis of glomerulus and renal tubules, eventually leading to proteinuria and renal failure. 3
Podocytes play a fundamental role in glomerular filtration. Podocyte epithelial–mesenchymal transition (EMT) is considered as the beginning process of podocyte injury, which can enhance the motility of podocytes and lead to proteinuria and glomerular fibrosis. 4 In the process of EMT, epithelial markers are downregulated along with the upregulation of mesenchymal markers. Snail, Zeb and helix loop helix (HLH) family are important transcriptional repressors of P-cadherin and other genes about EMT. 5 However, these factors cannot fully explain the molecular mechanism of podocyte EMT. So, new transcriptional factors about EMT will need to be found.
Serum response factor (SRF) is a highly conserved DNA binding protein of MADS-box transcription factor family and a main switch for the transcription of cytoskeletal and contractile genes in almost all cells across various species. 6 SRF can be activated through two different ways according to the sensitivity of target genes to the Ras–extracellular-regulated kinase (ERK)–ternary complex factors (TCFs) or Rho–actin–myocardin-related transcription factors (MRTFs). 7 SRF has been shown to have an essential role in tumour progression, especially in the EMT and metastasis of epithelial tumour cells, such as hepatocellular carcinoma, 8 gastric cancer 9 and prostate cancer. 10 In transgenic mice, high SRF expression can cause myocyte hypertrophy, interstitial fibrosis and muscle fibre damage. 11 And SRF knockout of coronary arterial smooth muscle cells (SMCs) could reduce the migration and proliferation ability of SMCs. 12 In addition, SRF plays a key role in transforming growth factor (TGF)-β-induced lung myofibroblast differentiation, and using protein kinase A (PKA) to reduce SRF expression and activity can inhibit this process. 13 However, how SRF regulates EMT of podocytes remains largely unknown. Herein, this study seeks to examine the regulation and function of SRF in cultured mouse podocytes and a rat model of DN. Furthermore, the therapeutic potential of this signalling was also investigated.
Materials and methods
Materials
Animal protocols
Male Wistar rats, 8 weeks of age and weighing 250–280 g, were obtained from Animal Center of Shandong University and were given free access to food and water throughout the study. After 1 week of adaptation and an overnight fast, rats obtained a single intraperitoneal injection of 60 mg/kg STZ in citrate buffer (pH 4.5), while the control rats received the same volume of citrate buffer only. The diabetic status was confirmed 72 h later as random glucose level higher than 16.7 mmol/L for 3 continuous days. Animals were randomly stratified into three groups with equal average initial body weight: (1) non-diabetic normal rats (control group, n = 6), (2) STZ-induced diabetic rats (DM group, n = 18) and (3) STZ-induced diabetic rats treated with CCG-1423 that is diluted in a 1:100 solution of dimethyl sulphoxide (DMSO): phosphate-buffered saline (PBS) as vehicle (DM + CCG group, n = 12). CCG-1423 was administered by daily intraperitoneal injection at doses of 0.01 and 0.02 mg/kg body weight for 8 weeks since the day after STZ injection. As a control, the same volume of vehicle (100 µL/100 g body weight) was given to both the control group and DM group. After the induction of diabetes, rats were sacrificed at 8, 12 or 16 weeks, respectively, for various analyses. All procedures complied with the guidelines of the Animal Ethics Committee of the Shandong University, which were consistent with the Animal Management Rules of the Ministry of Health of the People’s Republic of China (document no. 55, 2001).
Sample collection and biochemical analysis
At the end of the study, rats were weighed and housed in metabolic cages to collect 24-h urine. Rats were given euthanasia through intraperitoneal injection of pentobarbital (50 mg/kg), and then blood samples were collected from the left ventricle. Systemic perfusion with PBS through the left ventricle was carried out to wash out remnant blood. Kidneys were quickly removed, decapsulated, weighed and dissected into two parts. One part was snap-frozen in liquid nitrogen and stored at −80°C for molecular biological studies, while the other was stored in 4% paraformaldehyde solution for histopathological analysis. Serum glucose, serum creatinine (Scr), blood urea nitrogen (BUN) and 24-h urinary protein (24-h UP) were measured by the Department of Pathology at the Second Hospital of Shandong University. The renal mass index that was revealed by the ratio of kidney weight to body weight was calculated as well.
Histopathological analysis
To assess the glomerular sclerotic injury, kidney tissues were embedded in paraffin, and 3 µm thick sections were prepared for periodic acid–Schiff (PAS) and Masson staining to demonstrate fibrosis in renal tissues. Glomerulosclerosis index (GSI) was used to determine the severity of glomerulosclerosis, 14 which was performed by an examiner blinded to the specimen source through a light microscope with a camera (Nikon Eclipse 90i LH-M100CB-1; Nikon, Tokyo, Japan). Scores 1, 2, 3 and 4 represented the sclerotic lesion involving <25%, 25–50%, 50–75% and >75% of the glomerular tuft area, respectively. In all, 50 glomeruli were randomly selected in each mouse, and the average score was used for analysis.
Immunohistochemical staining
After deparaffinization, rehydration and blockage of endogenous peroxidase activity, slices were submitted to Tris-EDTA (pH 9.0) in a microwave for 17 min for antigen retrieval, and then the slices were incubated overnight at 4°C with a primary antibody. The slices were thoroughly rinsed by PBS, followed by incubation with a secondary antibody labelled by horseradish peroxidase (HRP) at room temperature for 40 min. The detection process was carried out by the use of diaminobenzidine and haematoxylin (ZSGB-BIO, Beijing, China). A negative control was omission of the primary antibody to verify the antibody specificity. Imaging of all slices was performed by a Leica microscope electronic imager. The expression of SRF, P-cadherin, α-SMA and FN was quantified by Image-Pro plus version 6.0 software (Media Cybernetics, Inc., Rockville, MD, USA) to determine the integral optical density. All analyses were performed in a blinded fashion.
Cell culture and treatment
Mouse conditionally immortalised podocyte cell line (mpc5) was illustrated previously 15 and kindly provided by Dr Peter Mundel (Mount Sinai School of Medicine, New York, NY, USA). Cells were propagated at 33°C in Dulbecco’s Modified Eagle medium (DMEM) (HyClone, Logan, UT, USA) containing 10% foetal calf serum (Gibco, Langley, OK, USA) and 50 U/mL mouse recombinant interferon-γ (Invitrogen, Carlsbad, CA, USA). Podocytes were cultivated at 37°C without interferon-γ for 10–14 days to induce differentiation for the proposed experiments (Supplementary Material available online). Podocytes were treated with low glucose (5.5 mM) and high glucose (30 mM) with or without CCG-1423 (1, 2 and 5 µM). CCG-1423 was dissolved in DMSO, and a same volume of DMSO was added to controls. A treatment of 5 mM glucose + 25 mM mannitol was implemented as an osmolarity control.
Plasmids and transfection
pcDNA3.1-SRF containing full-length complementary DNA (cDNA) sequence of mice SRF and control pcDNA3.1 vectors were gifted by Dr Eric Olson (Department of Molecular Biology, UT Southwestern Medical Center, Dallas, TX, USA). Podocytes were transfected with these plasmids using lipofectamine 2000 (Invitrogen) according to the manufacturer’s protocol. Infected cells were incubated for 5 h and then restored to complete medium. After 72 h, whole-cell lysates were collected for gene and protein expression analyses. Infected podocytes were submitted to immunofluorescence staining with the SRF antibody as well.
Quantitative and semi-quantitative reverse transcription polymerase chain reaction
Total RNA was isolated using TRIzol (Invitrogen). The first-strand cDNAs were synthesised from 2 µg of total RNAs in 20 mL reaction using Prime Script RT reagent kit (TaKaRa Biotechnology, Dalian, China). Quantitative PCR was performed in Eppendorf Mastercycler ep realplex detection system (Eppendorf, Hamburg, Germany) by FastStart Universal SYBR Green Master (Roche, Penzberg, Germany) according to the manufacturer’s protocol. The relative messenger RNA (mRNA) levels were normalised to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and calculated as 2−ΔΔCT.
The cDNA amplification procedure for semi-quantitative PCR was performed using 2× Taq PCR Master Mix (Tiangen, Beijing, China) according to the manufacturer’s protocol. PCR products were visualised on 1.5% agarose gels stained with GelRed (Biotium, Hayward, CA, USA) under UV transillumination. GAPDH was used as a control to indicate equal loading of the cDNA samples. All the primer sequences are reported in Table 1.
The primers used in this article.
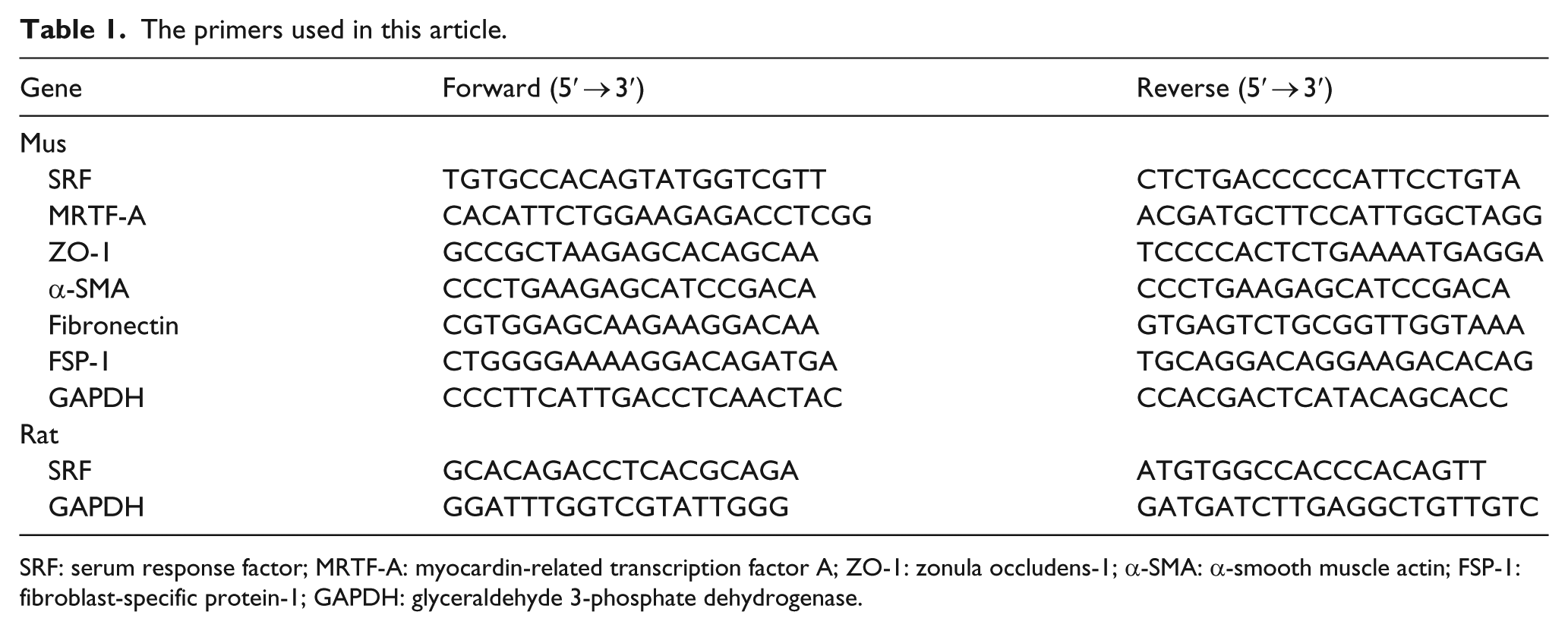
SRF: serum response factor; MRTF-A: myocardin-related transcription factor A; ZO-1: zonula occludens-1; α-SMA: α-smooth muscle actin; FSP-1: fibroblast-specific protein-1; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Immunoblotting
Kidney tissues or cell homogenates were lysed and stored at −80°C until assayed. Protein concentrations were determined through Coomassie reagent. An equal amount of whole protein (50 µg) was subjected to 12% sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA). The blots were blocked for 1 h at room temperature with 5% skimmed milk in Tris-buffered saline (pH 7.4) containing 0.1% tris-buffered saline Tween 20 (TBST), followed by incubation for 16 h at 4°C with primary antibodies. After further washing with TBST, the blots were incubated with HRP-conjugated secondary antibodies for 2 h at room temperature. Immune complexes were detected using enhanced chemiluminescence (Millipore). The immunoreactive bands were quantified by Image J analysis software (NIH, Bethesda, MD, USA).
Immunofluorescence staining
Podocytes were fixed for 20 min at room temperature in 4% paraformaldehyde and then permeabilized with 0.3% Triton X-100 (Sigma) in PBS for 10 min. The fixed cells were incubated with the primary antibody against SRF, followed by the appropriate fluorescein isothiocyanate (FITC)–conjugated secondary antibody (ZSGB-BIO). Non-immune IgG served as a negative control, and no fluorescence was observed. To visualise the nuclei, podocytes were incubated in a 1-µg/mL 4′,6-diamidino-2-phenylindole (DAPI) (Sigma) solution for 5 min. The slides were viewed under Nikon epifluorescence microscope, and the expression of SRF was quantified by Image J analysis software.
Transwell chamber migration assay
Podocyte migration and motility were determined using transwell chamber migration assay with tissue culture–treated polycarbonate membrane transwell filters (8 µm pore size; Corning, Cambridge, MA, USA), as previously described. 16 Podocytes after transfection with SRF cDNA or empty pcDNA3.1 vectors were seeded into the upper chamber of the filters. After 48 h of incubation at 37°C, cells were fixed with methanol for 20 min and stained with 0.1% crystal violet for 20 min. The top side of the filters was carefully scrubbed by a cotton swab. Cells that migrated to the undersurface of the filters were determined in three non-overlapping 20× fields (at a magnification of 100×) and photographed by a Nikon microscope.
Albumin filtration assay
Bovine serum albumin (BSA) filtration assay was carried out by analysing the transepithelial passage of BSA from lower to upper chamber of the collagen-coated transwell filters (0.4 mm pore; Corning, NY, USA) as previously described. 17 Plasmid-transfected podocytes were restored to complete medium for 72 h. Cells were washed twice with PBS supplemented with 1 mM CaCl2 and 1 mM MgCl2 to protect the cadherin-based junctions. After that, the upper chamber was refilled with 150 µl DMEM and the lower chamber with 1 mL DMEM supplemented with 40 mg/mL BSA, followed by incubation for 1 h at 37°C. Total protein concentration in the upper chamber was quantified by the Bradford method (Beyotime, Shanghai, China) using BSA as a standard.
Statistic analysis
All data were shown as mean ± standard error of the mean (SEM). Statistical analysis was carried out by SPSS (version 19.0; SPSS Inc., Chicago, IL, USA). Comparison between groups was performed through unpaired t-test or one-way analysis of variance. All experiments were repeated at least three times. p < 0.05 was considered statistically significant.
Results
High glucose–mediated podocyte EMT and SRF upregulation
As shown in Figure 1(a) to (d), substantial increase in protein and mRNA level of SRF was observed at 48 h after high glucose treatment, and pSRF, regarded as the active form of SRF, was also upregulated. Besides, podocytes tended to perform EMT. 18 High glucose suppressed epithelial synaptopodin and zonula occludens (ZO)-1 expression and induced collagen-1, FN, FSP-1 and α-SMA expression in a time-dependent manner (Figure 1(a), (d), (e), (g) and (h)), as shown by immunoblotting or quantitative reverse transcription polymerase chain reaction (RT-PCR) analysis. SRF is obviously transferred from podocyte cytoplasm to nucleus, and the quantity of SRF was also increased after high glucose treatment, according to immunofluorescence staining (Figure 1(i)). Of interest, MRTF-A, the upstream activator of SRF, was induced in podocytes after high glucose stimulation as well (Figure 1(j) to (l)).
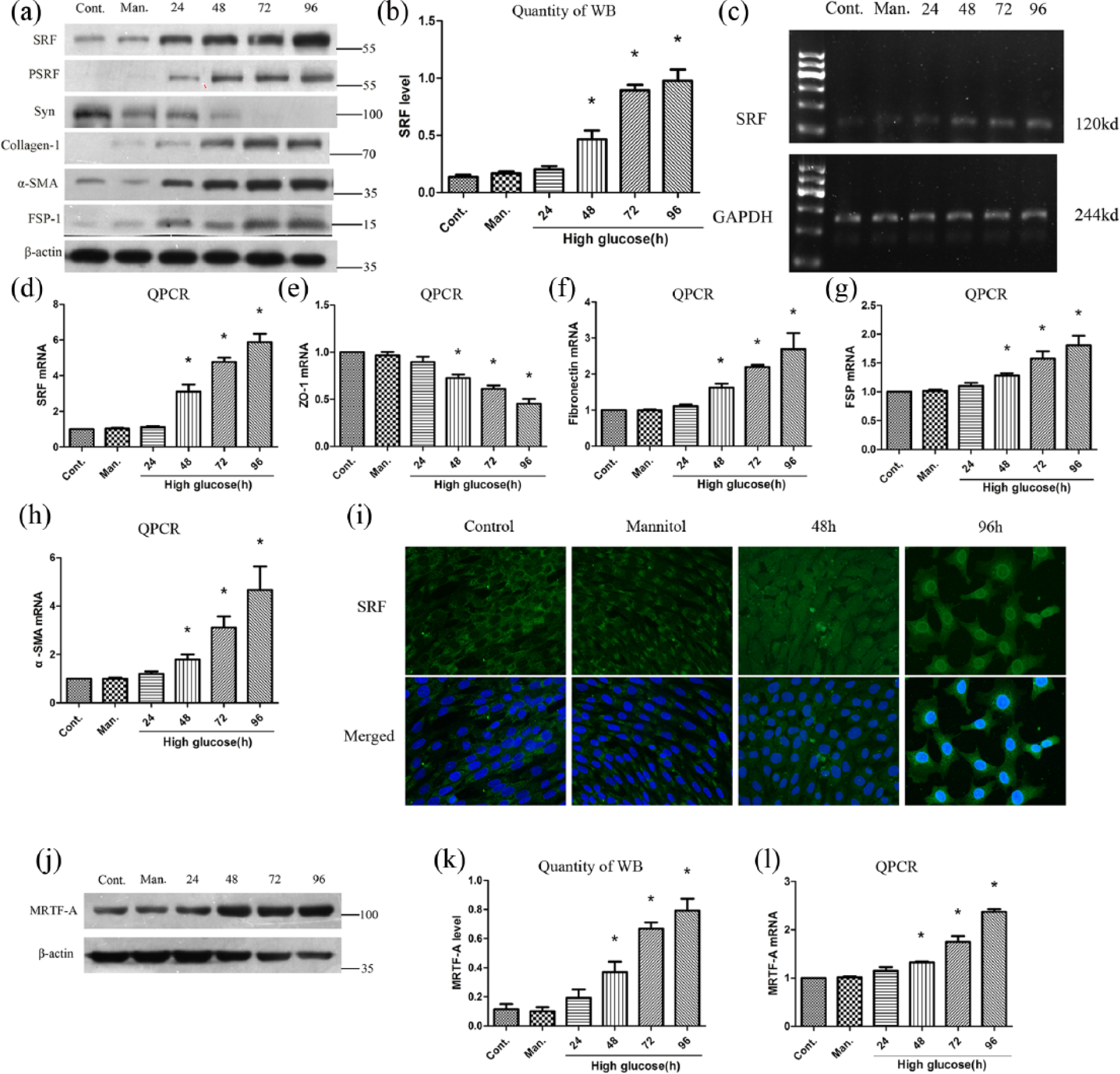
High glucose–mediated podocyte EMT and upregulation of SRF. (a) Western blot analysis showed that high glucose–mediated upregulation of SRF, pSRF, collagen-1,α-SMA, FSP-1 and downregulation of synaptopodin in a time-dependent manner. (b) Quantitative determination of relative SRF levels in different times after high glucose treatment. (c) Semi-quantitative PCR of SRF mRNA. (d) to (h) Quantitative RT-PCR of SRF, ZO-1, FN, FSP-1 and α-SMA mRNA expression. (i) Immunofluorence staining of SRF at an original magnification of 400×. The third and fourth groups were treated with high glucose for 48 and 96 h, respectively. (j) and (k) Protein expression of MRTF-A measured by western blot and quantitative analysis. (l) MRTF-A mRNA level measured by quantitative RT-PCR.
SRF overexpression–mediated podocyte EMT, migration, Snail upregulation and barrier dysfunction
To evaluate the potential effects of SRF overexpression on podocyte phenotypes and functions, cultured podocytes were transfected with SRF cDNA and empty pcDNA3.1 vectors, respectively. SRF induction after plasmid transfection was determined in both protein and mRNA levels (Figure 2(a) to (c) and (i)). Overexpression of exogenous SRF reduced epithelial nephrin and synaptopodin expression and increased collagen-1, FN, FSP-1 and α-SMA expression in podocytes, as shown by immunoblotting (Figure 2(a), (j) and (k)).
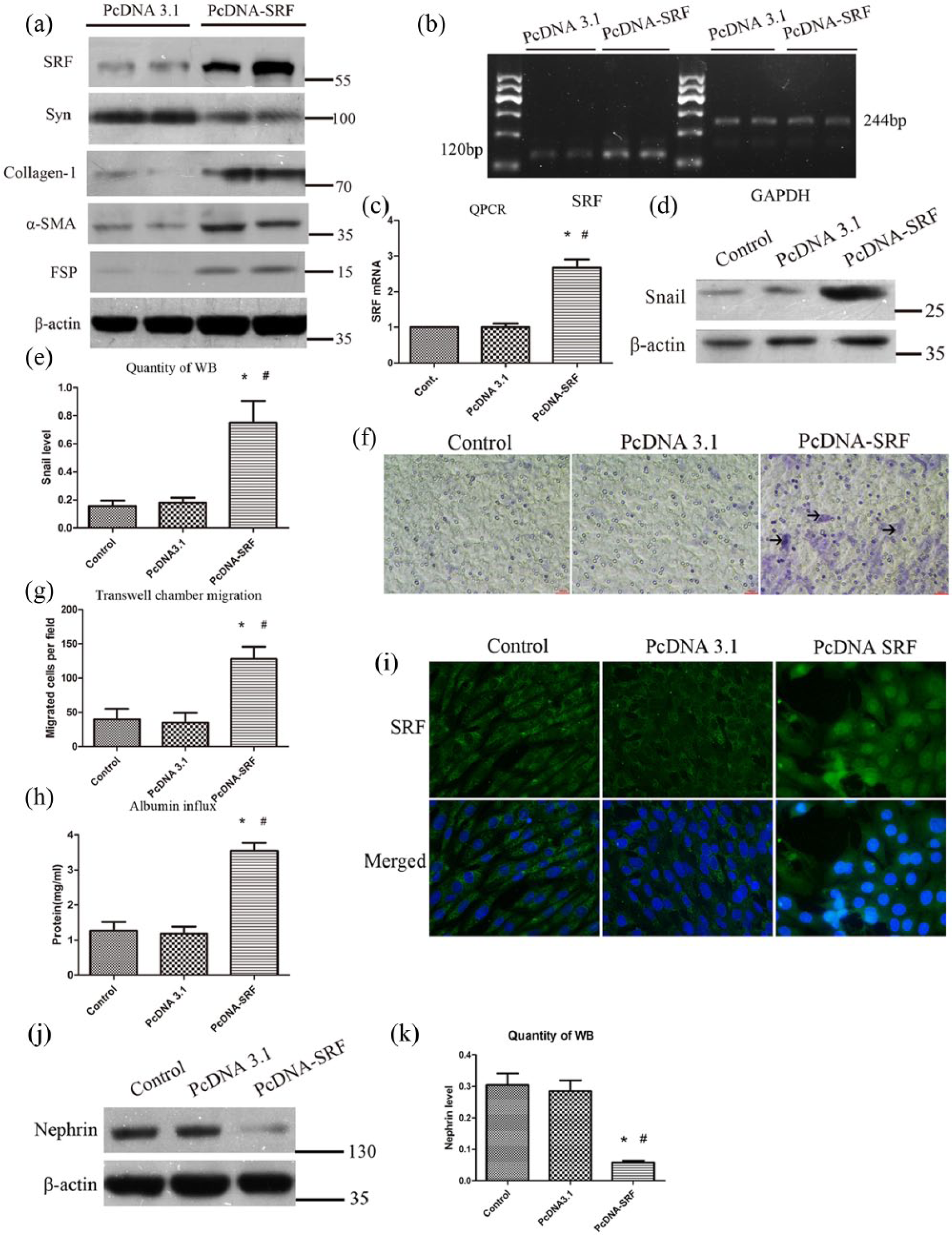
Overexpression of SRF-mediated podocyte EMT and Snail upregulation. (a) Western blot analysis showed the induction of SRF, collagen-1,α-SMA, FSP-1 and reduction of synaptopodin. (b) Semi-quantitative PCR of SRF mRNA levels. (c) Quantitative PCR of SRF mRNA expression. (d) and (e) Snail expression after SRF cDNA transfection in podocytes measured by western blot and quantitative analysis. (f) Representative micrographs of transwell chamber migration assay at an original magnification of 200×. Arrowheads indicated some of the migrated cells. Bar = 50 µm. (g) Quantitative analysis of migrated podocytes per field in three groups. (h) Albumin influx across transwell filters with podocyte monolayer in BSA filtration assay. (i) Immunofluorence staining of SRF at an original magnification of 400×. (j and k) Protein expression of nephrin measured by western blot and quantitative analysis.
To explore the potential mechanism of how SRF regulated podocyte EMT, the expression of Snail was measured, which was a vital transcription factor regulating EMT in various conditions. As revealed in Figure 2(d) and (e), forced expression of SRF promoted Snail expression in podocytes. These results indicated that ectopic expression of SRF in podocytes leads to a phenotypic change that is reminiscent of EMT. Transwell chamber migration assay was used to study the impact of SRF induction on podocyte motility. As shown in Figure 2(f) and (g), overexpression of SRF significantly upregulated the migration of podocytes across the pores of transwell filters, indicating that podocyte EMT mediated by SRF is alone with an increased cell migratory capacity.
To investigate the impact of SRF on podocyte barrier function, a simple and paracellular albumin filtration assay was performed, which measured the BSA diffusion across transwell filters that contain an equal number of differentiated podocytes. As shown in Figure 2(h), albumin easily diffused across the monolayer of podocytes transfected with SRF cDNA, in contrast to that transfected with empty pcDNA3.1 vectors. These results suggested that ectopic expression of SRF led to a deficient filtration barrier and podocyte dysfunction.
Inhibition of SRF preserved podocyte phenotypes and suppressed Snail upregulation after high glucose stimulation in vitro
To evaluate whether inhibition of SRF protected podocytes from high glucose injury, a specific small-molecule SRF inhibitor, CCG-1423, was used before high glucose treatment. The potency, mechanism and specificity of CCG-1423 to inhibit Rho/MRTF/SRF pathway were verified in many types of cells.19–21 As shown in Figure 3(a) to (d), CCG-1423 selectively suppressed the expression of pSRF and SRF in a dose-dependent manner in podocytes after high glucose stimulation for 72 h. Immunofluorescence staining suggested that CCG-1423 not only inhibited SRF expression but also reduced the transfer of SRF from cytoplasm to nucleus in podocytes treated with high glucose. High glucose treatment also resulted in p38 mitogen–activated protein kinase (p38 MAPK) and Akt activation in podocytes, but CCG-1423 did not effectively reduce the phosphorylation of them (Figure 3(a)). Besides, simultaneous treatment of podocytes with CCG-1423 also significantly abolished the reduction of synaptopodin and nephrin expression and the induction of collagen-1, FN, FSP-1 and α-SMA expression, hence largely preventing podocytes from EMT (Figure 3(f), (i) and (j)). Figure 3(g) and (h) showed that CCG-1423 significantly reduced Snail expression mediated by SRF in podocytes, indicating that inhibition of SRF by CCG-1423 could block an important EMT initiator gene.
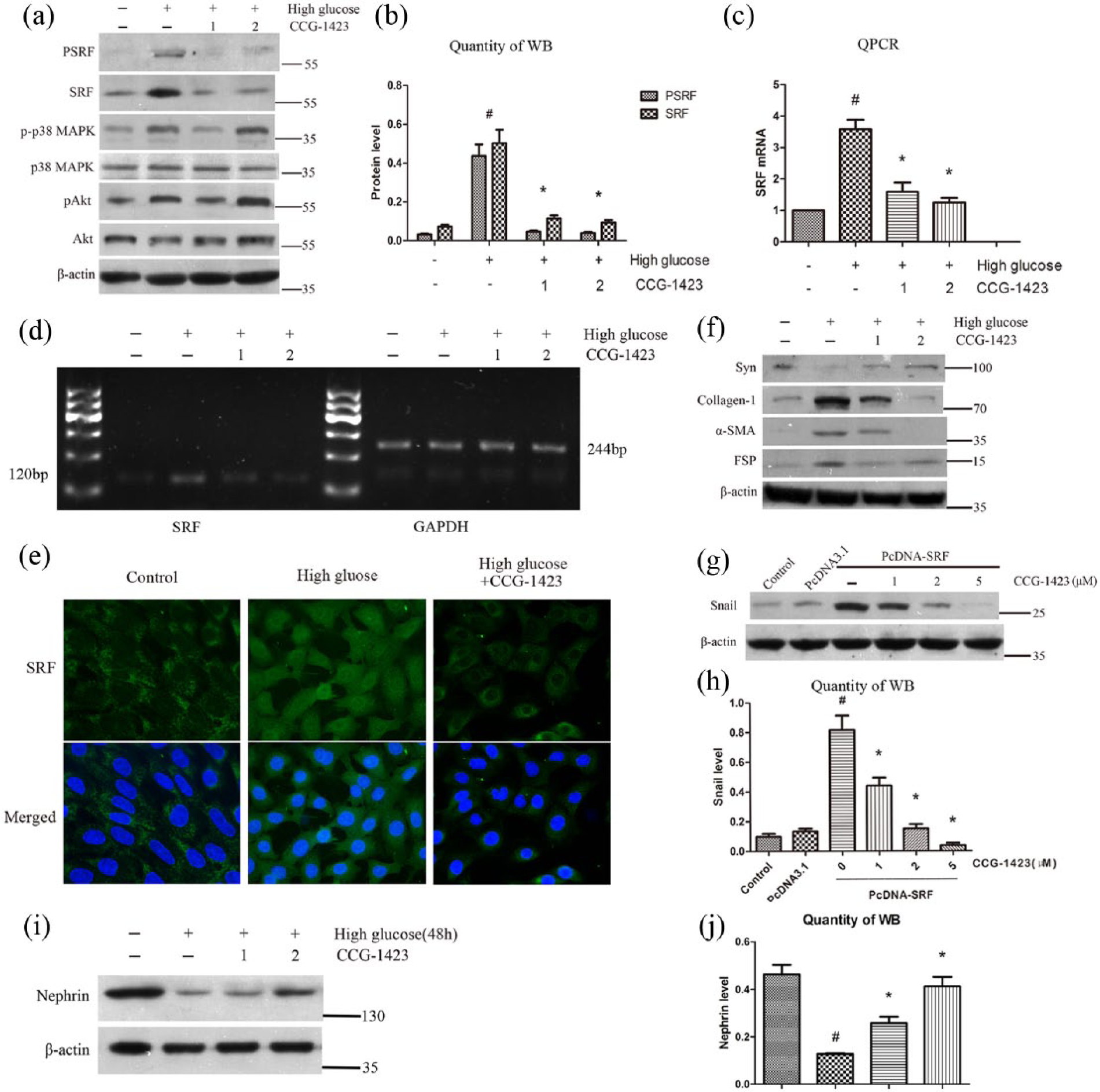
Inhibition of SRF preserved podocyte phenotypes and suppressed Snail upregulation after high glucose stimulation. Podocytes were pretreated with CCG-1423 (1 or 2 µM) or DMSO for 1 h, followed by treatment with low glucose (5.5 mM) or high glucose (30 mM) for 72 h. (a) Protein expressions of pSRF, SRF, p-p38 MAPK, p38 MAPK, pAkt and Akt measured by western blot analysis. (b) Quantitative determination of pSRF and SRF protein levels. (c) SRF mRNA expression measured by quantitative RT-PCR. (d) SRF mRNA levels measured by semi-quantitative RT-PCR. (e) Immunofluorence staining of SRF at an original magnification of 400×. CCG-1423 was used at the concentration of 2 µM. (f) Protein expressions of synaptopodin, collagen-1,α-SMA and FSP-1 measured by western blot analysis. (g) and (h) Mouse podocytes were transfected with SRF cDNA or empty pcDNA3.1 vectors. After 24 h, cells were incubated with different doses of CCG-1423 for another 24 h. Protein expressions of Snail and β-actin were measured by western blot analysis. (i) and (j) Protein expression of nephrin measured by western blot and quantitative analysis.
SRF was upregulated and activated in diabetic rats
To evaluate the role of SRF in podocyte injury in vivo, a rat model of DN was used. The DN model was proved by biochemical test results in Table 2, which showed that serum glucose, scr, BUN, 24-h UP and kidney weight–to–body weight ratio (KW/BW) were increased in the DM group, while serum albumin was decreased. Western blot analysis, quantitative RT-PCR and immunohistochemical staining were carried out using renal cortex tissues from control group or DM group. As shown in Figure 4(a) to (e), SRF was significantly upregulated in DM group compared to control group in a time-dependent manner, suggesting that SRF also participated in the podocyte injury induced by hyperglycaemia in vivo. Synaptopodin was also detected to certify that these renal cortex tissues contained high proportion of podocytes (Figure 4(a)).
Biochemical analysis of rats.
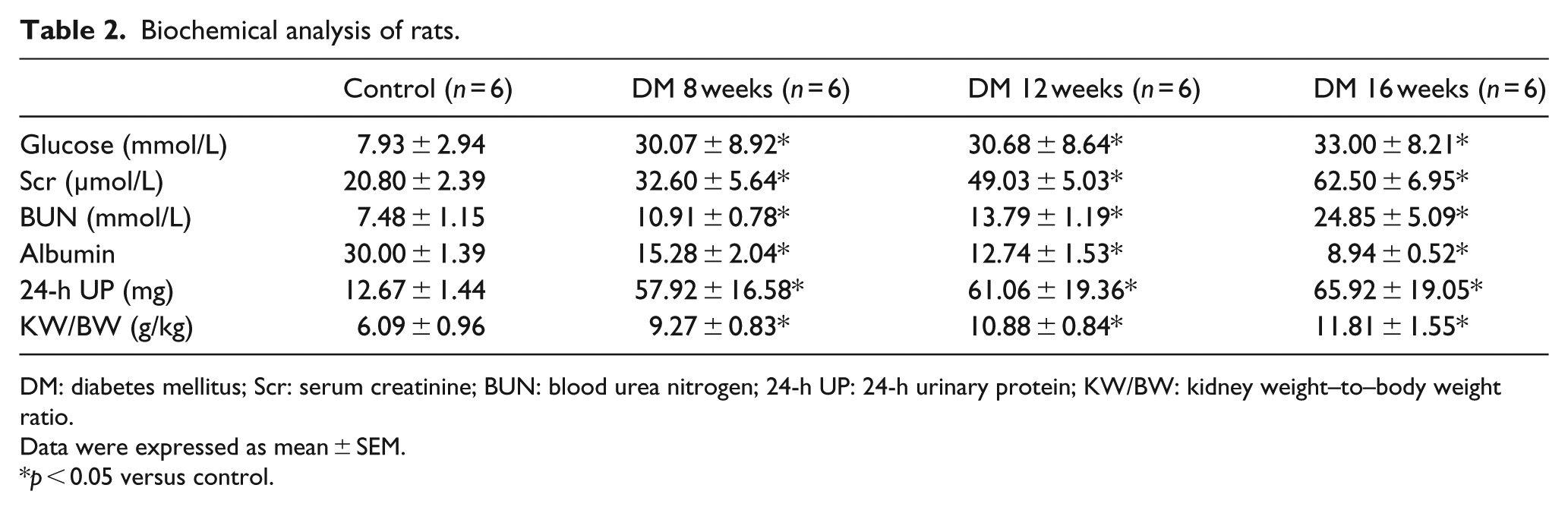
DM: diabetes mellitus; Scr: serum creatinine; BUN: blood urea nitrogen; 24-h UP: 24-h urinary protein; KW/BW: kidney weight–to–body weight ratio.
Data were expressed as mean ± SEM.
p < 0.05 versus control.
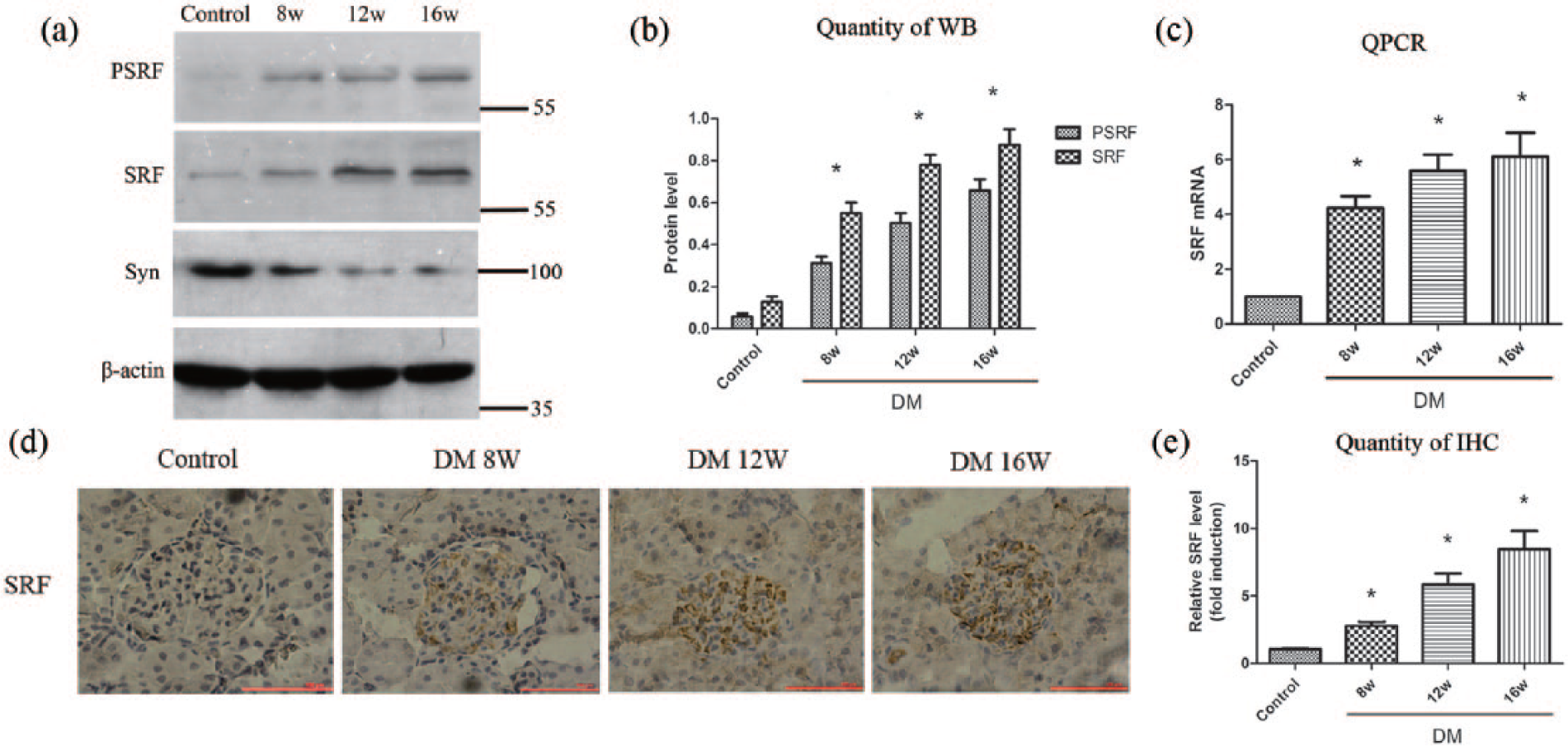
pSRF and SRF were upregulated in diabetic rats. (a) and (b) Western blot and quantitative analysis showed that pSRF and SRF were upregulated while synaptopodin was reduced in diabetic rats in a time-dependent manner. (c) Quantitative RT-PCR of SRF expression in rats. (d) and (e) SRF expression in different groups of rats measured by immunohistochemical staining and the quantitative determination.
Inhibition of SRF blocked podocyte EMT and ameliorated proteinuria in vivo
To evaluate whether inhibition of SRF protected podocytes from hyperglycaemia injury, daily intraperitoneal administration of CCG-1423 was performed in DM + CCG group. As shown in Figure 5(a), CCG-1423 ameliorated proteinuria in a dose-dependent manner. CCG-1423 at 0.02 mg/kg body weight significantly reduced 24-h UP by about 50%, compared with the vehicle controls. Besides, inhibition of SRF with CCG-1423 also significantly abrogated the reduction of synaptopodin expression and the induction of SRF, collagen-1,α-SMA, FSP-1 expression in renal cortex tissues, according to western blot analysis (Figure 5(b) to (d)). Masson and PAS staining demonstrated that renal fibrosis was present in DM group, and after 8 weeks of treatment with CCG-1423, renal fibrosis and GSI were dramatically ameliorated compared with vehicle controls (Figure 5(e), (f) and (k)). In addition, CCG-1423 significantly preserved glomerular P-cadherin and nephrin expression (Figure 5(g), (l), (j) and (o)) and suppressed α-SMA and FN expression in DN rats (Figure 5(h), (i), (m) and (n)), according to immunohistochemical staining. These results indicated that targeting SRF by small-molecule inhibitor could improve podocyte EMT and ameliorate proteinuria in vivo.
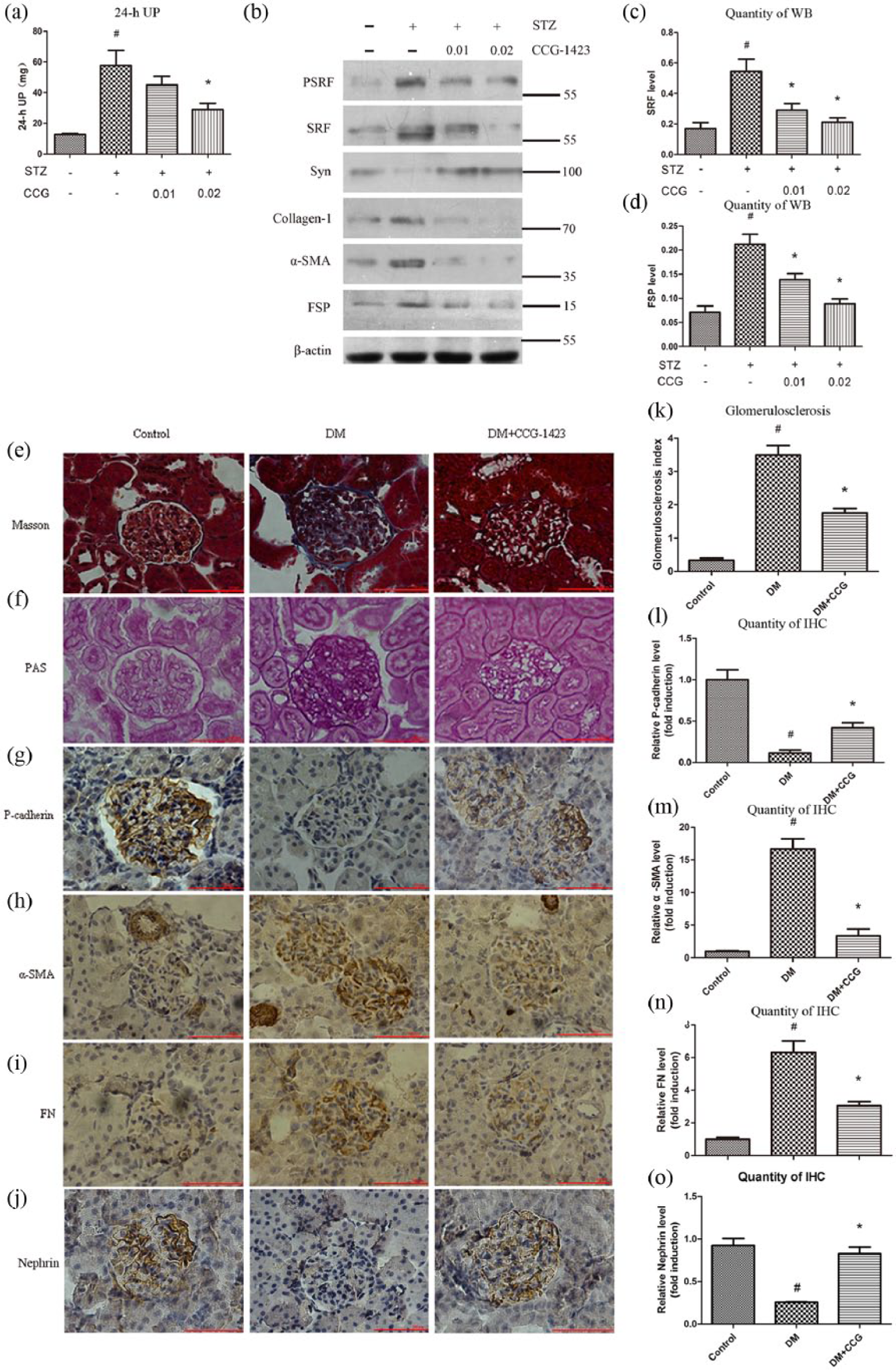
Inhibition of SRF ameliorated proteinuria and blocked podocyte EMT in vivo. (a) CCG-1423 reduced 24-h UP in a dose-dependent manner in DN rats. (b) to (d) Protein expressions of pSRF, SRF, synaptopodin, collagen-1,α-SMA and FSP-1 measured by western blot and quantitative analysis. (e) Renal fibrosis of the three groups was measured by Masson staining, and blue indicates collagen. Bar = 100 µm. (f) and (k) Pas staining and glomerulosclerosis index of three groups. Bar = 100 µm. (g) to (j) Immunohistochemical staining of P-cadherin, α-SMA, FN and nephrin of three groups. Bar = 100 µm. (l) to (o) Quantitative analysis of P-cadherin, α-SMA, FN and nephrin expression as determined by Immunohistochemical staining.
Discussion
DN is classified as a kind of podocytopathy. An early and significant event in the development of DN is podocyte EMT, which causes the loss of podocytes from glomeruli and consequently impairs the integrity of glomerular filtration barrier. 22 In this study, we find that transcription factor SRF is activated and translocated into nuclei during podocyte EMT stimulated by high glucose. Overexpression of SRF in podocytes activates key EMT regulatory gene Snail and facilitates dedifferentiation, migration and dysfunction of podocytes. To the contrary, specific inhibition of SRF by a small-molecule SRF inhibitor reverses EMT of podocytes and ameliorates proteinuria. These findings shed new light on the mechanism of podocyte EMT and indicate that inhibition of hyperactive SRF may be a new therapeutic target of proteinuria and DN.
SRF widely regulates not only transcription of cell proliferation factors but also the differentiation of contractile cells, SMCs and myocardial cells. SRF also plays an important role in maturity and differentiation of skeletal muscle cells and the expression of muscle-specific genes. It is reported that SRF is closely related to tumour invasion and metastasis and plays a key role in the process of EMT of epithelial tumour cells. 23 A recent study illustrates that SRF accelerates the high glucose–induced EMT in human peritoneal mesothelial cells, suggesting a close connection between upregulation of SRF and high glucose injury. 24 Notably, SRF expression and activation in podocytes are upregulated after high glucose treatment both in vitro (Figure 1) and in vivo (Figure 4(d)). SRF is also induced in the renal cortex tissues of a rat model of DN (Figure 4), a common kidney disease characterised by dramatical proteinuria. Interestingly, MRTF-A is upregulated in podocytes after incubation with high glucose as well (Figure 1(k) and (l)). Given that MRTF-A is a main upstream regulator of SRF, the simultaneous upregulation of both MRTF-A and SRF may lead to an amplification of SRF signalling during DN.
Growing evidence indicates that EMT, which was first discovered in epithelial tumours, appears in other diseases including kidney fibrosis. 4 Furthermore, podocytes are able to undergo EMT in DN as recently shown. 25 SRF is well documented as an important transcription factor that promotes EMT in various cell types including tubular epithelial cells. 26 So, overexpression of exogenous SRF in podocytes not only inhibits synaptopodin and nephrin expression but also upregulates the expression of many mesenchymal markers such as collagen-1, α-SMA and FSP-1 (Figure 2). These changes in cell–cell adhesion (synaptopodin) and cytoskeleton proteins (α-SMA) will result in destruction of the glomerular filtration membrane integrity. Of interest, overexpression of SRF makes podocytes motile, along with enhanced migration ability (Figure 2). This increased motility after ectopic SRF expression might be a part of or a result of EMT, which probably leads to a damaged slit diaphragm. Besides, a paracellular albumin filtration assay shows that ectopic expression of SRF also disrupts the filtration barrier function of podocyte monolayer (Figure 2). In summary, these results suggest that upregulation of SRF is sufficient to make podocyte phenotype and function alteration, which is associated with migration and a damaged filtration barrier function.
Although the molecular mechanisms by which SRF mediates podocyte EMT remain to be illuminated, it appears to be in relation to the capacity to increase Snail expression. Snail is a core EMT regulatory transcription factor that plays an essential role in fibrosis. 27 The primary mechanism by which Snail mediates podocyte EMT is by reducing P-cadherin 28 and nephrin expression.29,30 A recent research shows that SRF transferred from cytoplasm to nucleus to upregulate Snail expression and the subcellular localisation of Snail and SRF influences cellular phenotype and function. 24 Therefore, overexpression of SRF significantly increases Snail expression (Figure 2), and suppression of SRF by CCG-1423 inhibits Snail induction and abolishes high glucose–mediated podocyte EMT (Figure 3). Snail is also induced in mesenchymal-converted podocytes in the urinary sediments of DN patients. 25 Together, these data point to an important role of SRF/Snail signalling in inducing EMT and dysfunction of podocytes.
CCG-1423 is a Rho/MRTF/SRF pathway inhibitor and specifically suppresses invasion, proliferation and DNA synthesis of Rho-overexpressing cell lines. CCG-1423 is proposed to be a promising pharmacological compound in preventing the progression of prostate cancer. 31 This study also manifests that SRF may be a potential therapeutic target of DN. In the DN rat model, intraperitoneal injection of CCG-1423 ameliorates proteinuria in a dose-dependent manner (Figure 5). This beneficial impact of CCG-1423 is accompanied by a dose-dependent reduction of mesenchymal markers (collagen-1, α-SMA, FSP-1 and FN) and a preservation of podocyte-specific synaptopodin, nephrin and P-cadherin both in vitro and in vivo (Figures 3 and 5), consistent with the function of SRF in inducing podocyte phenotypic alteration.
To sum up, this research has shown that SRF is upregulated in podocytes after high glucose injury, which might play an important role in inducing EMT, migration and dysfunction of podocytes. It should be emphasised that this research still has some pitfalls and limitations since it merely used an established transformed podocyte cell line and only a type 1 DM rat model mediated by STZ. Whether these data could extend to primarily cultured podocytes and type 2 DM remains to be illuminated. What is more, it is not clear whether SRF is acting directly on the genes of the markers used in this study (nephrin, synaptopodin, snail, ZO-1, collagen-1, α-SMA, P-cadherin, etc) through binding to serum response elements (SREs) or indirectly and whether SRF plays an important role in human DN. However, our results provide a proof of principle that small-molecule inhibitor that targets SRF/Snail pathway can be an attractive therapeutic strategy for DN.
Footnotes
Acknowledgements
The authors thank Central Research Laboratory of the Second Hospital of Shandong University for providing the excellent experimental equipment and also thank the Laboratory Animal Center of the Second Hospital of Shandong University for the excellent rat care.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National Key Technology Research and Development Program of the Ministry of Science and Technology of China, 2011BA110B05.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
