Abstract
Objective:
Stroke is a severe complication of type 2 diabetes mellitus. Glucagon-like peptide-1 receptor agonists have been shown to have a neuroprotective effect in experimental diabetes. The aim of this study was to determine if their neuroprotective effect is an independent property of the drug independent of glycaemic control.
Methods:
This two-phase study used male Wistar rats. In the first phase, experimental animals were pretreated with liraglutide, while controls received only vehicle. After transient focal brain ischaemia modelling, neurological deficit and brain infarct volume were measured. In the second phase, the first and the second groups of experimental animals with type 2 diabetes mellitus received liraglutide and metformin, respectively, while control animals with diabetes received only vehicle. After transient focal brain ischaemia modelling, neurological deficit and brain infarct volume were evaluated.
Results:
Pretreatment with liraglutide in diabetic and non-diabetic animals reduced infarct size as compared to controls, while only non-diabetic liraglutide-treated rats presented neurologic deficit decreases. Despite glycaemia normalization, metformin-treated diabetic rats had no differences in stroke outcome when compared to the control group.
Conclusion:
The neuroprotective effect of liraglutide is not associated with glycaemic control amelioration in experimental type 2 diabetes mellitus.
Introduction
Type 2 diabetes mellitus (T2DM) is a major healthcare issue as its vascular complications can severely affect quality of life. 1 Recently, a new group of drugs, incretin mimetics, including dipeptidyl-peptidase-4 inhibitors and glucagon-like peptide-1 receptor (GLP-1R) agonists, have been developed and are both effective and safe in treating T2DM. To date, the neuroprotective effect of GLP-1R agonists has been demonstrated in cerebral ischaemia in non-diabetic animals and in animal models of diabetes by independent research teams.2–4 However, in most studies, the control groups consisted of diabetic animals which were not subjected to treatment, and therefore, it was impossible to elucidate the effects of glycaemic control on stroke outcome.2,3 Clinical trials have shown that GLP-1R agonists decrease the mortality from cerebrovascular accidents in T2DM, 5 but GLP-1R agonists were usually prescribed in combination with other medications, and drug interactions could not be excluded. Here, we investigate GLP-1R agonists to determine if their neuroprotective effect is an independent property of the drug independent of glycaemic control. We evaluated the neuroprotective effect of the long-acting GLP-1R agonist, liraglutide, which has high homology to endogenous GLP-1, in a rat model of transient focal brain ischaemia and T2DM.
Methods
The study was approved by Pavlov University Ethics Committee in agreement with the European Convention for the protection of vertebrate animals and is reported according to ARRIVE guidelines.
In these experiments, male Wistar rats (Rappolovo, Russia; n = 85) were used. The rats were housed in a controlled temperature and humidity environment with a 12-h-light/12-h-dark cycle and were provided with free access to water and standard food. Animals with hyperglycaemia or impaired neurological function according to the results of a baseline blood glucose level analysis (Accu-Check Performa) and neurological deficit assessment with Garcia scale 6 were excluded from the study. After allowing 2 weeks for acclimatization, the animals were randomly divided into two groups according to experimental design.
In the first part of the experiment, the rats from the first group (14–15 weeks old, 300–400 grammes) were randomly allocated to three subgroups to evaluate the neuroprotective effect of liraglutide in non-diabetic rats. A power analysis was performed to determine the number of animals in the treatment and control subgroups, and justification was based on our previous pilot studies. Cohen’s effect size (d) was calculated to identify differences in the treatment and control subgroups for a decrease in the stroke size (d = 1.1). After a two-tailed power analysis (statistical power of 80% and an α = 0.05), we calculated a sample size of 15 animals per subgroup. Experimental rats (n = 15) received liraglutide (Novo Nordisk, Denmark) 1 mg/kg per day subcutaneously for 7 days prior to stroke modelling, while control (n = 15) and sham-operated (n = 5) animals were treated with only vehicle (0.9% sodium chloride). On the seventh day, blood glucose levels were analysed, and focal cerebral ischaemia was induced by filament occlusion of the left middle cerebral artery (MCA) (Doccol Corporation, USA) for 30 min under anaesthesia with 430 mg/kg chloral hydrate as described previously. 7 Detection of the decrease in regional cerebral blood flow by 70% with ultrasound Doppler ensured successful MCA occlusion. Rectal temperature was controlled at 37.0°C during and after surgery. On the second day of reperfusion, neurological deficit was measured. After euthanasia, the brains were sliced into eight 2-mm-thick coronal sections and stained with 0.1%, 2,3,5-triphenyltetrazolium chloride (Sigma, USA). The sections were photographed and analysed with Adobe Photoshop 8.0. Infarct size of the ischaemic cortex was expressed as a percentage of total brain volume. The surgery, neurological deficit and brain infarct size evaluation were performed by three blinded investigators.
In the second part of the experiment, the rats from the second group (7–8 weeks old, 200–250 grammes) were randomly allocated into four subgroups to evaluate the neuroprotective effect of liraglutide in experimental T2DM. T2DM was induced using a high-fat diet combined with nicotinamide and low-dose streptozotocin injections as described previously. 8 Following the development of T2DM, experimental rats from the first (n = 15) and second (n = 15) subgroups received liraglutide 0.03 mg/kg per day subcutaneously, and metformin (Sigma, USA) 200 mg/kg per day orally, respectively, for 7 days prior to stroke modelling, while control (n = 15) and sham-operated (n = 5) animals received vehicle treatment. On the seventh day of treatment, the same procedures as in the first part of the experiment were conducted to analyse glycaemia and to model focal brain ischaemia. On the second day of reperfusion, neurological deficit and brain infarct size were measured as described above.
Data were expressed as a median and interquartile range and analysed with StatSoft STATISTICA 10. For comparisons between two groups, the Mann–Whitney U test was used, while the matched samples were compared with Wilcoxon signed-rank test. In all cases, p < 0.05 was the criterion for statistical significance.
Results
In the first part of the study, non-diabetic, sham-operated animals exhibited no cerebral infarct areas and no neurological deficit, while liraglutide-treated rats had significantly smaller brain infarct volume (p < 0.01) in comparison with control animals. At baseline, the rats had no neurological deficit, but after MCA occlusion, all the animals except sham-operated rats had neurological impairment. Treatment with liraglutide significantly reduced neurological deficit after MCA occlusion in non-diabetic rats (p = 0.03) compared with control treatment. No significant difference in glycaemia levels between liraglutide-treated and control rats was noted (Figure 1).
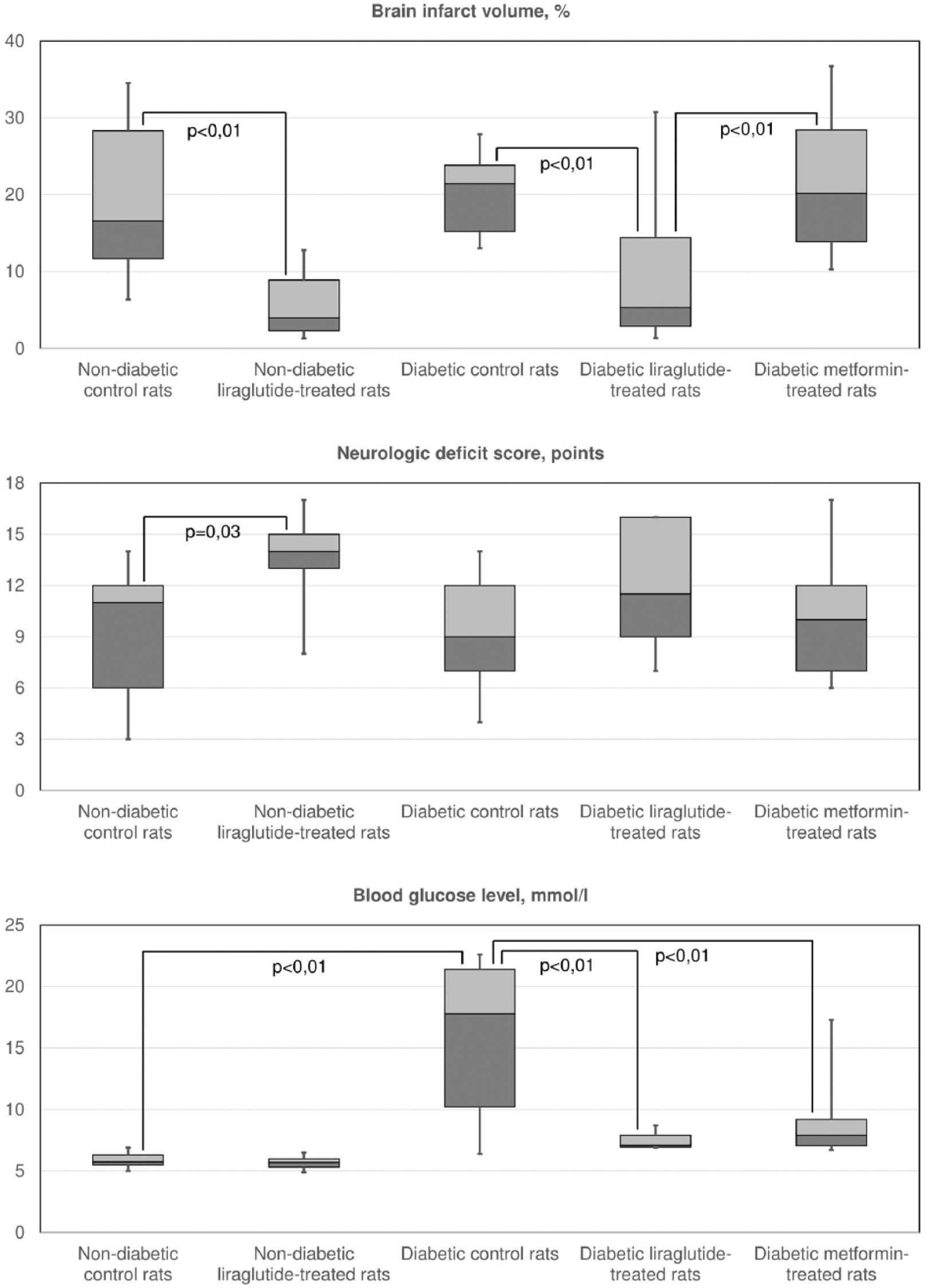
Brain infarct size (%), neurological deficit evaluation (points) and glycaemia (mmol/L).
In the second part of the study, diabetic, sham-operated animals exhibited no cerebral infarcts areas or neurological deficit, while liraglutide-treated rats had significantly smaller brain infarct volume (p < 0.01) in comparison with control animals and metformin-treated animals. The infarct volume in metformin-treated animals did not significantly differ from control animals. At baseline, the rats showed no neurological deficit, but after MCA occlusion, all the animals except sham-operated rats had neurological impairment. Treatment with liraglutide and metformin did not reduce neurological deficit after MCA occlusion in diabetic rats compared with control treatment ones, and there were no significant differences in neurologic deficit between metformin-treated and liraglutide-treated rats. Liraglutide and metformin treatment helped to achieve significantly lower glycaemia in comparison with vehicle treatment (p < 0.01 and p < 0.01, respectively); nevertheless, no significant difference in glycaemia between liraglutide-treated and metformin-treated rats was noted.
Discussion
In this study, we showed that liraglutide has an independent neuroprotective effect in transient focal brain ischaemia that is not related to glycaemic control in experimental T2DM. The acquired data are consistent with the work of Briyal et al., 2 who showed that liraglutide improved stroke outcome in rats with streptozotocin-induced diabetes in comparison with insulin-treated diabetic rats; however, this rat model of diabetes refers to type 1 diabetes mellitus and no glucose blood levels were measured throughout treatment.
In our study, treatment with liraglutide in non-diabetic rats did not cause hypoglycaemia and significantly reduced brain infarct size. Although both liraglutide and metformin helped to achieve euglycaemia in experimental T2DM, only liraglutide treatment reduced ischaemic brain damage in comparison with metformin and vehicle treatment. Despite glycaemia normalization, metformin-treated rats showed no difference in brain infarct size in comparison with hyperglycaemic diabetic rats.
Therefore, liraglutide treatment has more advantages than the widely-prescribed metformin in terms of glycaemic control improvement and neuroprotection. The further demonstration of pleiotropic neuroprotective effect of GLP-1R agonists in large clinical studies with adjusted patient selection may promote their prescription in average therapeutic doses for effective reduction of stroke severity, both in patients with T2DM and in groups at risk for ischaemic stroke.
The mechanisms of GLP-1R-associated neuroprotection have been described in many experimental studies and involve the activation of the PI3K/AKT, cAMP/PKA/CREB, MAPK, ERK cascades that lead to the realization of anti-apoptotic, anti-inflammatory and regenerative strategies. 9 However, there are still unanswered questions for future investigations. It remains unknown whether the protective mechanisms could be altered by pathophysiological changes in T2DM, for example, impaired PI3K 10 and cAMP-dependent PKA signalling. 11 A recent study highlighted that the combination of metformin and liraglutide helped to achieve a more pronounced beneficial effect on endothelial dysfunction normalization. 12 Therefore, the ability to diminish stroke severity through the management of endothelial dysfunction could be an important issue for subsequent research.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
