Abstract
Previously, a facilitating effect of hyperbaric oxygenation (HBO2) on aortic ring responses to angiotensin-(1-7) in healthy rats was reported, with epoxyeicosatrienoic acids (EETs) possibly playing an important role. The aim of this study was to assess whether HBO2 exerts similar effects in diabetic rats and to further explore the role of specific cytochrome P450 (CYP) enzymes in changes induced by HBO2. Aortic relaxation to angiotensin-(1-7) was significantly higher in HBO2 diabetic rats compared to control diabetic rats, while HBO2 had no effect on angiotensin II contraction. N-methylsulphonyl-6-(2-propargyloxyphenyl/hexanamide inhibited the facilitation of angiotensin-(1-7) responses in HBO2 rats, suggesting an important role of EETs in this modulation. mRNA expression of CYP2J3 and protein expression of CYP2C11 were significantly upregulated in HBO2 diabetic rats, whereas CYP4A1, CYP4A2 and CYP4A3 mRNA and CYP2J3 protein expression was similar between groups. Mean arterial pressure, ferric reducing ability of plasma and Thiobarbituric Acid Reactive Substances levels and serum angiotensin-(1-7) concentrations were not significantly changed.
Background
Hyperbaric oxygenation (HBO2) is every medical and experimental application of pure (100%) oxygen at a pressure level higher than atmospheric pressure. 1 Treatment with HBO2 was traditionally used for a number of classic indications, such as gas gangrene, carbon monoxide poisoning, decompression sickness and others, but in recent years it has been shown to be beneficial in various conditions with vascular pathology. 1 The most clinically impressive effect of HBO2 in that regard is its contribution to healing of ischaemic ulcerations in diabetic patients, but there are also other observed effects of HBO2 therapy including neurological improvement after stroke, recovery from myocardial infarction and acute peripheral extremity ischaemia or reduction of atherosclerotic plaques.1–6 Although these effects have been documented clinically and in experimental models, the exact mechanisms involved in the mediation of these HBO2 actions are largely unknown, and studies regarding the influence of HBO2 on vascular structure and function are scarce. 1 The effects of HBO2 in pathologic conditions such as wound healing cannot be simply explained with the compensation of the lack of oxygen and it has become clear that oxygen plays important roles in convoluted signalling pathways instead of being simply a nutrient.1,2 Better understanding of the actions of HBO2 on vascular function in diabetes mellitus and other disorders is the crucial requirement for more efficient clinical use, better prediction of outcome or unwanted/adverse results, more precise decisions about suitability and further development of therapeutic methods. It is, however, also a prerequisite for completely elucidating the roles of oxygen as a key factor in physiological processes and for further advances in experimental research.
In previous studies, which investigated the effects of HBO2 on aortic ring reactivity to angiotensin-(1-7) (ANG-(1-7)) and angiotensin II (ANG II) in healthy Sprague–Dawley rats, it has been found that HBO2 enhances aortic vascular reactivity to ANG-(1-7), but does not alter reactivity to ANG II. 7 Furthermore, it has been shown that the specific epoxidation inhibitor n-methylsulphonyl-6-(2-propargyloxyphenyl)hexanamide ((MS-PPOH) which inhibits specific cytochrome P450 (CYP) enzyme reactions that produce epoxyeicosatrienoic acids (EETs)) reverses the observed changes of HBO2 on vascular reactivity to ANG-(1-7). 7 This implies that EETs may have an important role in mediating the changes of vascular reactivity to ANG-(1-7) induced by HBO2. 7 This is extremely interesting, since both ANG-(1-7) (which has dilative properties that counteract ANG II contraction) and EETs have beneficial effects on vasculature in diabetes, as shown in various earlier studies.8–14 A number of works discovered disturbances of vascular reactivity in diabetes – notably, increased reactivity to physiological vasoconstrictors and decreased reactivity to vasodilators,15–21 as well as impaired cerebral blood flow autoregulation. 22 In streptozocin-induced diabetic rats, vasodilation to acetylcholine is impaired, as is arteriolar flow-dependent dilation.20,23,24 In such rat models of diabetes, contraction to noradrenalin in aorta, skeletal arteries and mesenteric arteries is increased.15,25,26 ANG-(1-7) was demonstrated to improve the damaged vascular reactivity to various constrictors and dilators in diabetes and to facilitate recovery from ischaemia-reperfusion injury in hearts of streptozocin-induced diabetic rats.8,10 The synthetic ANG-(1-7) agonist NorLeu(3)-ANG-(1-7) accelerates healing of diabetic wounds. 11
The biological roles of EETs in vascular function are extremely important – they serve as an endothelium-derived hyperpolarizing factor, with their vasodilating effect being comparable to that of acetylcholine. 27 Besides causing vasodilation, by increasing potassium influx into smooth muscle cells, they have pro-angiogenic, anti-inflammatory, anti-apoptotic and pro-fibrinolytic effects.28–31 In their role of regulating blood flow, some EETs can also exhibit vasoconstrictive properties, for example, in the kidney where they can cause constriction of the afferent arteriole.27,32 They are synthesized in epoxidation reactions (as arachidonic acid metabolites) catalysed by a number of specific CYP enzymes capable of epoxidation.27,33 EETs are capable of modulating vascular responses to other stimuli, such as hormonal and paracrine agents. For instance, vasopressin-induced increase in cytosol calcium in mesangial cells is enhanced by EETs and reduced with inhibition of EETs synthesis. 27 Responses of afferent arterioles to ANG II, endothelin-1 and noradrenaline increase with inhibition of EETs. 27 Importantly, EETs were shown to be protective against streptozocin-induced diabetic nephropathy12,34 and coronary artery disease, 13 whereas inhibition of EETs synthesis worsens stroke in rats with streptozocin-induced diabetes mellitus type 1. 14 Meanwhile, diabetes induced with streptozocin in rats causes a decrease in levels of protective EETs. 14 Some CYP enzymes were also found to act as oxygen sensors in tissues, with production of EETs and hydroxyeicosatetraeonic acids (HETEs) directly correlating with pO2.35–37 This fact brings CYP enzymes (probably also those capable of EETs synthesis) into focus as possible targets of HBO2.
A facilitation of vascular reactivity to ANG-(1-7) in diabetes, similar to the facilitation reported in healthy rats, would therefore explain some of the beneficial effects of HBO2 in diabetic wound healing and in other vascular pathologic conditions associated with diabetes. The aim of this work was to investigate the influence of HBO2 on aortic ring reactivity to ANG-(1-7) and ANG II in diabetic rats and to test whether inhibition of EETs synthesis reverses effects of HBO2. Furthermore, the effect of HBO2 on mRNA and protein expression of important rat CYP enzymes (synthesizing EETs) in aorta was assessed. The main hypothesis is that HBO2 enhances aortic relaxation after ANG-(1-7) addition, without significantly altering aortic reactivity to ANG II, and that EETs play an important role in this enhancement. Under this hypothesis, inhibition of EETs synthesis would be expected to reverse any facilitation of vascular reactivity induced by HBO2 and HBO2 would possibly cause an upregulation of CYP enzymes that synthesize EETs.
Methods
Animals and model of diabetes
Male Sprague–Dawley rats were housed doubly in shoebox style cages with free access to standard rat chow and tap water, maintained on a 12:12-h light:dark cycle. A type 1 diabetes mellitus model was induced by injecting streptozocin (60 mg/kg) intraperitoneally at 6 weeks of age. Blood glucose levels were checked 1 week after the injection and again on the day of the experiment (at an age of 14 weeks). All animals that did not develop diabetes at 1 week after the injection (or without confirmed diabetes again at 14 weeks) were euthanized and not used in further experiments. From experience in our laboratory, we set the minimum blood glucose cut-off at the age of 7 weeks at 15 mmol/L (with ~80% of injected rats in our facility satisfying this requirement), because with such a high initial cut-off, most of the remaining rats indeed successfully develop diabetes. Therefore, only three rats (3.85%) planned for the aortic vascular reactivity assay and four rats (4.88%) planned for the other experiments had to be excluded (prior to entering experiments) at the 14-week blood glucose check for not developing diabetes. A OneTouch Ultra (LifeScan, Inc., Milpitas, CA, USA) glucometer and the tail cut method were used to measure blood glucose levels (with the vast majority of successfully developed diabetic animals at 14 weeks of age showing a level ‘higher than 33.3 mmol/L’ – the measurements were used only as a confirmation of successful development of the diabetes model).
At the age of 14 weeks, rats were divided into a control group and an HBO2 group which underwent the HBO2 protocol. Rats from the HBO2 group were treated in a hyperbaric chamber (containing CO2 adsorbent) with 100% O2 (using pressure of 2 bar) for 2 h a day (with addition of 15 min for the compression phase and 15 min for the decompression phase) during four consecutive days. On the fifth day, the aortic ring experiments were conducted. For that purpose, the rats were anaesthetized with a combination of ketamine (75 mg/kg) and midazolam (2.5 mg/kg) and afterwards decapitated with a guillotine. The abdomen and thorax were surgically opened and the lungs, heart, oesophagus and adjacent tissue removed. The thoracic aorta was carefully and promptly isolated, placed into an oxygenated Krebs–Henseleit solution and cleaned of adherent tissue. The procuration of animals, the husbandry and the experiments conformed to the ‘European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes’ (Council of Europe No 123, Strasbourg 1985). The experiments were approved by the Ethics committee of the Faculty of Medicine, University of Osijek.
Reagents
ANG II and ANG-(1-7), noradrenaline and acetylcholine were purchased from Sigma–Aldrich. Ketamine and midazolam were obtained from Pfizer, New York, NY, USA. Streptozocin was purchased from Sigma–Aldrich. The Krebs–Henseleit solution (composition: 113 mM NaCl, 4.7 mM KCl, 1.2 mM MgSO4 × 7H2O, 22 mM NaHCO3, 1.2 mM KH2PO4, 11 mM glucose, 2.5 mM CaCl2 × 2H2O, 0.026 mM ethylenediaminetetraacetic acid (EDTA); pH 7.4) was prepared from EDTA and purchased from Sigma–Aldrich, CaCl2 × 2H2O and NaHCO3 from Merck KGaA, Darmstadt, Germany, with the rest of the chemicals purchased from Kemika, Zagreb, Croatia. MS-PPOH, a selective inhibitor of the epoxidation reactions catalysed by specific CYP450 isozymes, was a gift from John R. Falck, Southwestern Medical Center, Dallas, TX, USA. MS-PPOH is a reaction-specific inhibitor and is not isoform-specific: it selectively inhibits epoxidation in various CYP isoforms that are capable of catalysing these reactions, 38 although it is often mentioned that it inhibits CYP4A2 and CYP4A3, which are merely some of the first isoforms on which this epoxidation inhibition was confirmed. 38 The CO2 adsorbent Drägersorb 800 Plus was produced by Dräger, Lübeck, Germany.
Aortic vascular reactivity assay
Short segments from each end of the isolated aorta were severed and discarded, whereas the rest of the vessel was cut into rings (of about 3–4 mm in length). These rings were mounted in tissue bath chambers containing Krebs–Henseleit solution (maintained at 37°C) with 95% O2/5% CO2 compressed gas mixture bubbling through and connected to pressure transducers as part of an Experimetria vessel ring preparation setup (purchased from Experimetria Ltd, Budapest, Hungary). The data were continuously recorded on a computer and later analysed. Passive tension for each ring was set at 2.0 g. The vessels were allowed to equilibrate and stabilize for 1 h, replacing the Krebs–Henseleit solution every 15 min with fresh solution and readjusting passive tension to 2.0 g as needed. Subsequently, intactness of endothelium was tested by precontracting the rings with 10−7 M (final concentration) noradrenaline, letting stabilize for 5 min and inducing relaxation with 10−5 M acetylcholine. If the vessel ring failed to relax, it was not used for further studies (in this series of experiments, one ring from the diabetic control group and one from the HBO2 group were excluded, which accounts for 1.83% of the overall aortic rings). If the vessel ring relaxed, it was washed three times with fresh solution and allowed to equilibrate for 30 min, with washing at 10-min intervals. After the rings were stabilized, maximal contraction was induced with 60 mM KCl + 10−7 noradrenaline. When plateau was reached, the rings were washed three times with fresh solution and allowed to equilibrate for 30 min, washing at 10-min intervals.
After this phase, aortic ring responses to ANG II or ANG-(1-7) were tested, by treating every ring of a certain animal with a different peptide/protocol and only once. The concentrations for the peptides were chosen on the basis of previous studies where such concentrations were effective.39–41 In one chamber, 10−6 M ANG II was applied. A second aortic ring was treated with 10−6 M ANG II + 10−6 M ANG-(1-7) – to test the diminishing effect of ANG-(1-7) on ANG II contraction. Another ring was precontracted with noradrenaline for 5 min, after which 10−6 M ANG-(1-7) was added and the tension read after 3 min. In this way, in experiments which test a certain substance response (e.g. response to ANG II or response to ANG II + ANG-(1-7)), the number ‘n’ corresponds simultaneously to the number of animals and to the number of rings (because every aortic ring tested for this substance is from a different animal). The peak contraction force of the responses to ANG II and ANG II + ANG-(1-7) was expressed as percentage of maximal contraction of the particular ring, and thereby, the contractile response for a certain substance was normalized to the maximal contraction of that ring. The responses to ANG-(1-7) were expressed as percentage of precontraction decrease (after 3 min of ANG-(1-7) presence). Aortic responses to noradrenaline were also evaluated.
To explore the role of EETs in mediating the potential facilitation of vasodilatory effects of ANG-(1-7) by the HBO2 protocol, the selective epoxidation inhibitor MS-PPOH was used. In a series of experiments with HBO2 rats, MS-PPOH (10−5 M final concentration) was added 15 min before precontraction with noradrenaline (and subsequent addition of ANG-(1-7)).
CYP mRNA expression studies
Quantitative polymerase chain reaction (Q-PCR) was performed to detect the expression levels of CYP4A2 and CYP4A3 (which are some of the important isoforms inhibited by MS-PPOH), as well as CYP2J3 and CYP4A1 in HBO2 and control samples of diabetic rats. Aorta samples were collected and stored in RNAlater (Qiagen, Hilden, Germany) on −80 °C until RNA isolation. Total RNA was extracted using TRI Reagent (Molecular Research Center, Inc., Cincinnati, OH, USA) according to the manufacturer’s instructions. RNA concentration and purity was assessed using NanoDrop (Thermo Scientific, Waltham, MA, USA). Using a Deoxyribonuclease kit (Sigma, St Louis, MO, USA), total RNA was additionally purified from gDNA. Reverse transcription was performed with High-Capacity cDNA Reverse Transcription kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer’s instruction on MyCycler Thermal Cycler (BioRad, Hercules, CA, USA). Quantitative real-time PCR was performed on an AB7500 (Applied Biosystems) platform, determining the expression of mRNA for CYP4A1, CYP4A2 and CYP4A3. For that purpose, TaqMan Gene Expression Assay products Rn04224034_s1, Rn01417068_g1 and Rn00598412_m1 were used. CYP2J3 mRNA expression was determined with the use of custom-made primers designed on Primer Express (Applied Biosystems) using ABsolute Q-PCR SYBR Green low ROX master mix (Thermo Scientific) – also on the Applied Biosystems 7500 real-time PCR System. Gene expression (CYP mRNA expression) was normalized to the expression of two housekeeping genes – hypoxanthine-guanine phosphoribosytransferase (HPRT) and 18S.
CYP protein expression studies
Western blot was performed to assess CYP protein expression. Specific primary mouse monoclonal antibodies to CYP2C11 (Gentaur, Kampenhout, Belgium) were used in conjunction with secondary goat anti-mouse IgG antibodies labelled with horseradish peroxidase (HRP; Santa Cruz Biotechnology, Inc., Dallas, TX, USA). To detect CYP2J3 expression, a specific rabbit polyclonal antibody with attached HRP (Antibodies-Online Inc., Atlanta, GA, USA) was used. Aorta samples were promptly frozen in liquid nitrogen and stored at −80 °C until homogenization. Homogenization was performed on ice, with appropriate buffer (1 mM EDTA, 10 mM Tris (Fisher Scientific, Loughborough, UK), 0.4% sodium dodecyl sulphate (SDS; Acros Organics, USA, Geel, Belgium), protease inhibitor cocktail 0.4 µL/100 µL (Sigma–Aldrich)) and with an ULTRA-TURRAX homogenizer (IKA, Staufen, Germany). A Bradford assay (AppliChem, Darmstadt, Germany) was used to determine total sample protein concentrations. Sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) and subsequent blotting to polyvinylidene difluoride (PVDF) membranes were performed at 4 °C with BioRad protein electrophoresis and blotting systems (Mini-PROTEAN Tetra Cell and Criterion blotter) and an appropriate BioRad power supply system. After membrane blocking, primary and secondary antibody incubation phases (or in case of CYP2J3, incubation with one joint HRP-labelled antibody) and washing phases, detection was performed with a chemiluminescence method. For that purpose, Pierce enhanced chemiluminescence (ECL) Western Blotting Substrate (Thermo Scientific) was used according to the manufacturer’s instructions and the signal was recorded on X-ray films in a dark chamber. Expression of CYP2J3 and CYP2C11 for each sample was normalized to β-actin expression (primary β-actin mouse monoclonal antibodies were purchased from Sigma–Aldrich). ImageJ software was used to process and analyse the CYP2J3 and CYP2C11 protein expression (relative to β-actin expression) in concordance with the software developer’s instructions.
Arterial blood pressure and indicators of oxidative stress
We analysed potential changes in arterial blood pressure and indicators of oxidative stress in animals with regard to treatment with the used HBO2 protocol, to test whether such changes could explain HBO2 effects on vascular reactivity. Because it is known from earlier work that both arterial blood pressure changes and oxidative stress can affect vascular reactivity,42–47 it is important to observe the roles of these factors in analysing the effects of HBO2.
A separate batch of diabetic male Sprague–Dawley rats was divided into HBO2 and control animals. After the HBO2 group was subjected to the HBO2 protocol, all rats have been anaesthetized with a combination of ketamine (75 mg/kg) and midazolam (2.5 mg/kg) and a catheter made of PE-50 tubing has been surgically inserted into the left femoral artery. Body temperature was maintained constant, and the mean arterial blood pressure was measured with a Spacelabs Medical monitoring system (Spacelabs Medical, Inc., Redmond, WA, USA). After 10 min of stabilization, the blood pressure was determined as the average blood pressure during a period of 1 min.
As indicators of oxidative stress, ferric reducing ability of plasma (FRAP) 48 and Thiobarbituric Acid Reactive Substances (TBARS) 49 have been determined from blood samples in separate batches of anaesthetized HBO2 and control diabetic animals. The FRAP assay offers an index of antioxidant, or reducing, potential and uses Trolox as a standard, whereas the TBARS assay is used to detect by-products of lipid peroxidation (malondialdehyde (MDA) is used as standard).
ANG-(1-7) serum levels
Additionally, ANG-(1-7) serum levels were determined, since HBO2 was found to facilitate vascular relaxation to ANG-(1-7). This was done to assess whether a potential change in ANG-(1-7) concentration induced by HBO2 could be responsible for the mechanism of vascular reactivity changes to ANG-(1-7) – it is widely known that agonists can induce internalization and desensitization of its receptors, depending on the change of agonist quantity, leading to alterations in agonist efficacy (and signal transduction).50,51 For that purpose, enzyme-linked immunosorbent assay (ELISA) was used to measure ANG-(1-7) concentrations in serum samples of control diabetic rats and diabetic rats that underwent the HBO2 protocol. The rat ANG-(1-7) ELISA kit was purchased from USCN Life Sciences Inc. (Wuhan, China) and used by following the manufacturer’s instructions.
Statistical analysis
Statistics were performed using SigmaPlot 11.2 (Systat Software, Inc., San Jose, CA, USA). Contraction to ANG II (mean percentage of maximal contraction) was compared between the HBO2 and the control group. Similarly, in experiments measuring contraction to ANG II + ANG-(1-7), the mean percentage of maximal contraction was compared between the two groups and, likewise, experiments determining the mean percentage of precontraction decrease after ANG-(1-7) addition were analysed as well. Within the control and within the HBO2 group, the difference between contraction to ANG II and ANG II + ANG-(1-7) was tested (since every peptide was applied to a distinct aortic ring). Shapiro–Wilk test was used as normality test. If it was passed, Student’s t-test was used with significance set at p < 0.05. If the normality test was not passed, the non-parametric Mann–Whitney U test was used with significance set at p < 0.05. Results from the HBO2 group where MS-PPOH was added before application of the peptides were compared with HBO2 animals where no MS-PPOH was applied in a similar manner. Results are expressed as mean ± standard deviation (SD).
Statistical analysis of CYP expression levels, arterial blood pressure values, indicators of oxidative stress (FRAP and TBARS) and ANG-(1-7) serum levels, compared between the HBO2 and control groups, was performed with the use of Student’s t-test or Mann–Whitney U test, respective of the outcome of the Shapiro–Wilk normality test, with significance set at p < 0.05.
Results
Aortic vascular reactivity experiments
Contractile responses to ANG II were similar in both control diabetic rats and diabetic rats treated with HBO2 (Figure 1(a)). The mean percentage of maximal contraction for ANG II responses was 14.8% ± 7.1 (N = 11) in the control group and 16.4% ± 5.9 (N = 12) in the HBO2 group (p > 0.05, Student’s t-test). Contractile aortic ring responses to the combination of ANG II + ANG-(1-7), as shown in Figure 1(a), were similar between control (N = 19) and HBO2 (N = 19) rats (p > 0.05, Student’s t-test).
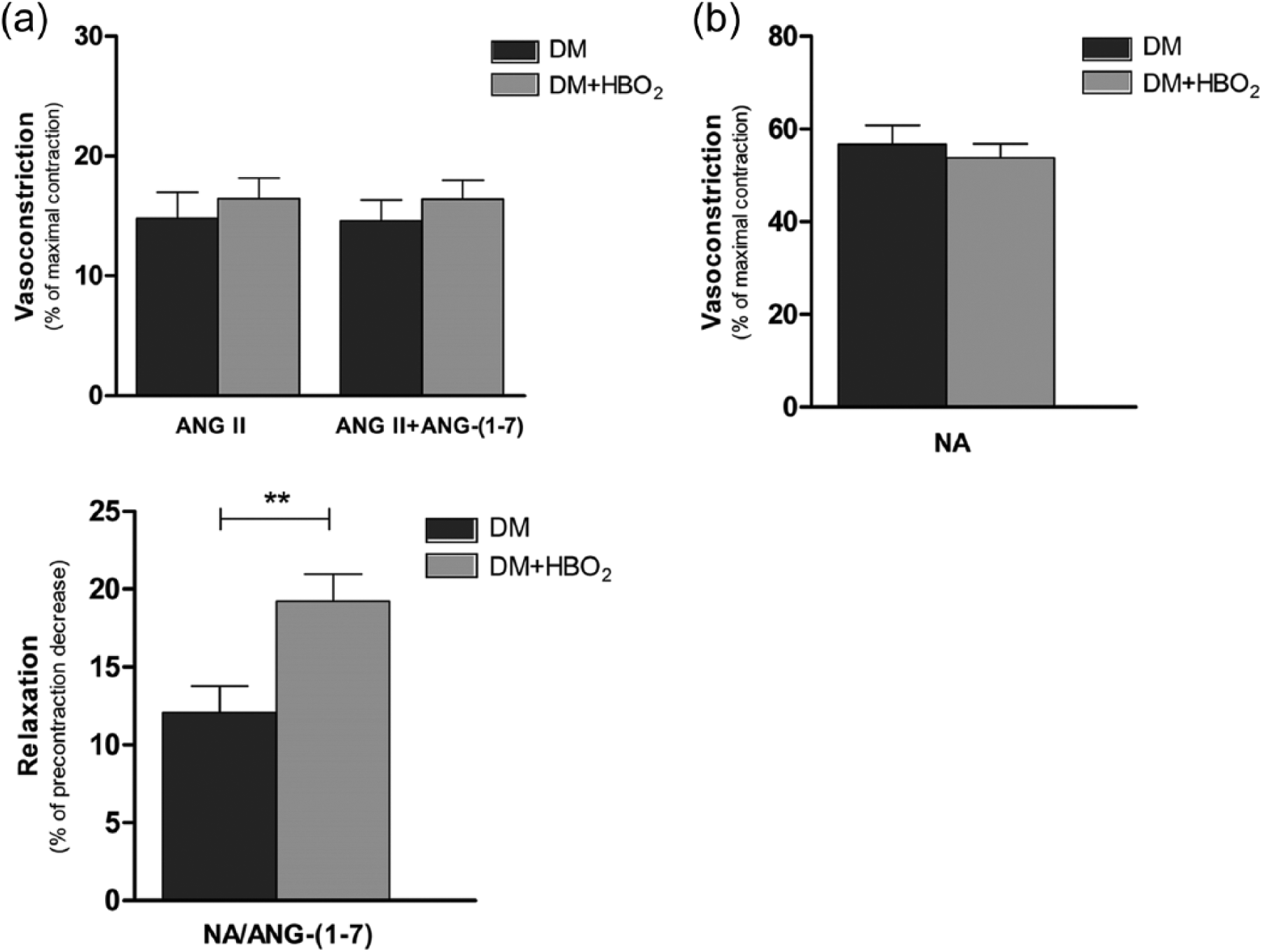
(a) Aortic vascular responses to ANG II, to the combination ANGII + ANG-(1-7) (top) and to ANG-(1-7) (bottom). Contraction responses are expressed as mean percentage of maximal contraction after peptide addition to isolated thoracic aortic rings of diabetic control rats (DM) or diabetic rats treated with HBO2 (DM + HBO2) (top). Relaxation of isolated thoracic aortic rings is expressed as mean percentage of precontraction decrease 3 min after addition of ANG-(1-7) to rings precontracted with NA (bottom). The asterisks (**) mark statistically significant difference. (b) Aortic contraction responses to noradrenaline alone. Contraction responses are expressed as mean percentage of maximal contraction after noradrenaline addition to isolated thoracic aortic rings of diabetic control rats (DM) or diabetic rats treated with HBO2 (DM + HBO2).
Relaxation to ANG-(1-7) (mean percentage of noradrenaline precontraction decrease 3 min after ANG-(1-7) addition) was 12.1% ± 6.4 (N = 14) in diabetic control rats compared to 19.2% ± 7.3 (N = 18) in diabetic HBO2 rats. This difference was statistically significant (p = 0.007, Student’s t-test) (Figure 1(a)). HBO2 had no effect on the responses to noradrenaline alone, since the contraction to noradrenaline (expressed as mean percentage of maximal contraction) was similar in both control and HBO2 animals (Figure 1(b)).
When the selective epoxidation inhibitor MS-PPOH was added before precontraction with noradrenaline and ANG-(1-7) addition to aortic rings of HBO2 diabetic rats, mean relaxation to ANG-(1-7) was 5.7% ± 5.1 (N [DM + HBO2] = 18, N [DM + HBO2/MS-PPOH] = 16). This was a statistically significantly lower relaxation response compared to responses of HBO2 rings when no MS-PPOH was used (p < 0.001, Student’s t-test) (Figure 2). Figure 5 shows contraction to noradrenaline alone of HBO2 rings treated with MS-PPOH compared to untreated HBO2 rings. There was no significant effect of MS-PPOH on contraction responses to noradrenaline (p > 0.05, Student’s t-test).
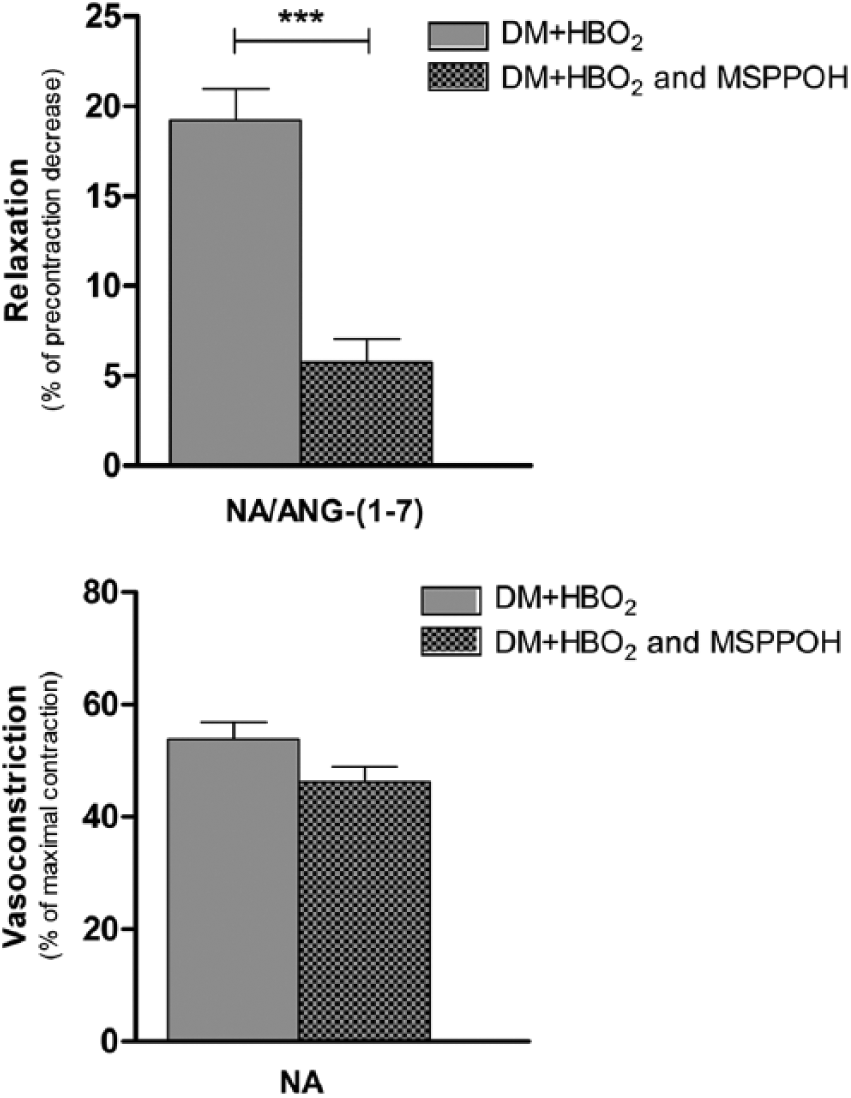
Effect of MS-PPOH on vascular responses of HBO2 aortic rings to ANG-(1-7) (top) or to noradrenaline alone (bottom). (Top) Relaxation of isolated thoracic aortic rings after addition of ANG-(1-7) to rings precontracted with NA. (Bottom) Contraction responses of isolated thoracic aortic rings after NA addition. Effect of the selective epoxidation inhibitor MS-PPOH on responses of HBO2 rings (DM + HBO2 and MS-PPOH) compared to responses of HBO2 rings when no inhibitor was used (DM + HBO2). The asterisks (***) mark statistically significant difference.
CYP mRNA expression studies
Figure 3 shows relative aortic mRNA expression levels (determined with Q-PCR) for CYP2J3, CYP4A1, CYP4A2 and CYP4A3. The CYP2J3 aortic mRNA expression (N [DM] = 9, N [DM + HBO2] = 9) is statistically significantly higher in the HBO2 group of diabetic animals compared to diabetic animals which did not undergo HBO2 protocol exposure (p < 0.05, Mann–Whitney U test). This is evident when analysed with either of the two measured housekeeping genes (HPRT and 18S). Relative aortic mRNA expression of CYP4A1 (N [DM] = 9, N [DM + HBO2] = 9), CYP4A2 (N [DM] = 8, N [DM + HBO2] = 9) and CYP4A3 (N [DM] = 9, N [DM + HBO2] = 9) is not significantly different between groups (with both housekeeping genes used for analysis) (p > 0.05, Mann–Whitney U test).
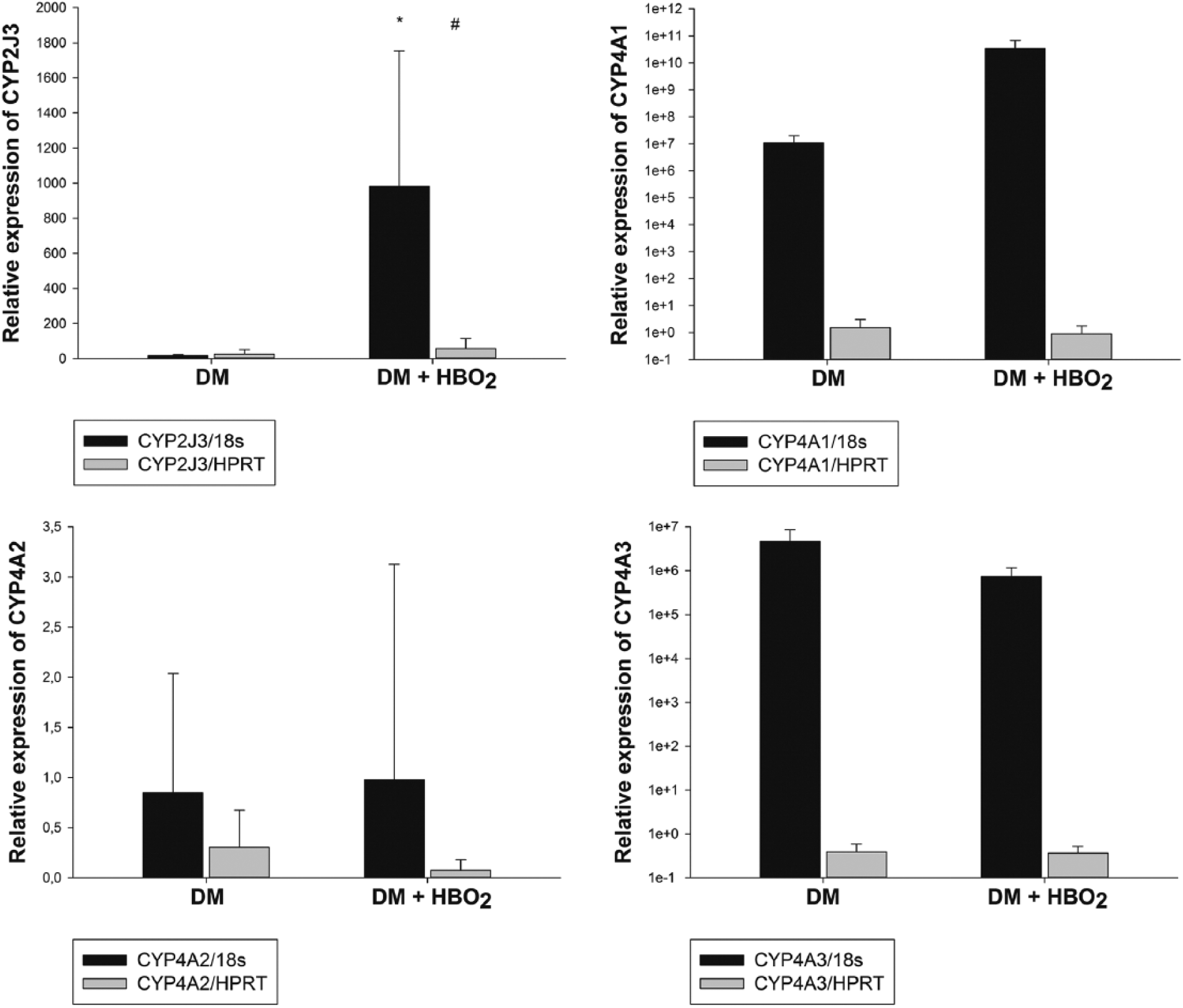
Relative aortic mRNA expression. Relative aortic mRNA expression levels of CYP2J3 (top left), CYP4A1 (top right), CYP4A2 (bottom left) and CYP4A3 (bottom right). The expression is normalized to expression of housekeeping genes (HPRT or 18S). The asterisk (*) and the hash (#) mark statistically significant difference between the diabetic control group (DM) and the diabetic HBO2 group (DM + HBO2).
CYP protein expression studies
Aortic protein expression (as assessed with Western blot) of CYP2J3 and CYP2C11 is shown in Figure 4. The relative CYP2J3 protein expression (N [DM] = 11, N [DM + HBO2] = 9) was not determined to be statistically significantly different in HBO2 diabetic animals compared to diabetic animals which were not treated with HBO2 (although HBO2 expression is non-significantly higher) (p > 0.05, Student’s t-test). A statistically significant upregulation of CYP2C11 aortic protein expression (N [DM] = 7, N [DM + HBO2] = 7) was measured in HBO2 diabetic rats compared to untreated diabetic rats (p = 0.042, Student’s t-test).
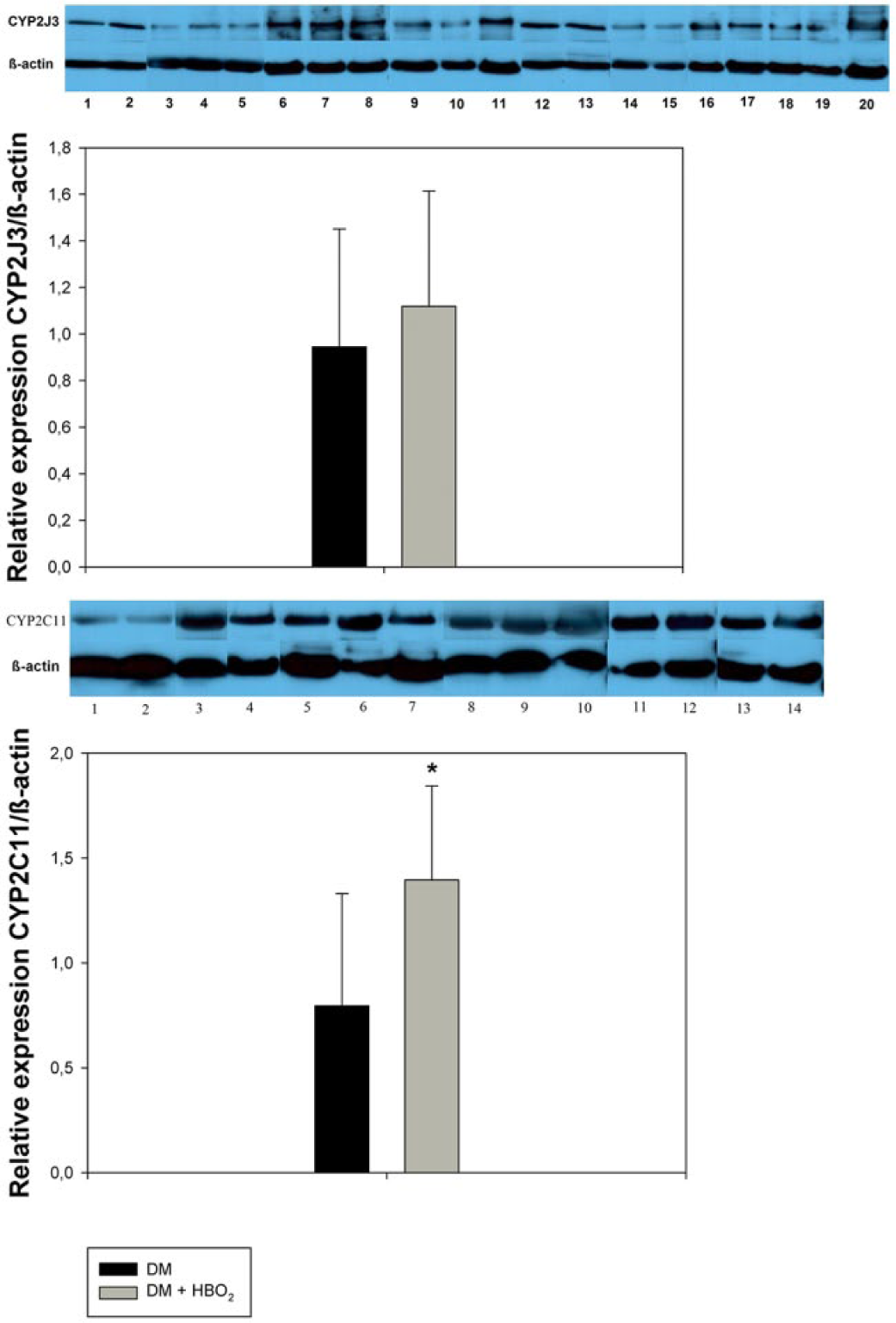
Relative aortic protein expression of CYP2J3 (top) and CYP2C11 (bottom). The expression is normalized to expression of β-actin in aorta of diabetic control rats (DM) and diabetic rats that underwent HBO2 (DM + HBO2). The asterisk (*) marks statistically significant difference.
Arterial blood pressure and indicators of oxidative stress
Results of measurements of arterial blood pressure and serum FRAP and TBARS levels of diabetic rats (N = 6) and diabetic rats treated with HBO2 (N = 6) are listed in Table 1. The results are expressed as mean ± SD. There was no significant effect of HBO2 or diabetes on either arterial pressure, or indicators of antioxidant capacity (FRAP) or lipid peroxidation (TBARS); the values between groups are similar (p > 0.05, Student’s t-test or Mann–Whitney U test).
Measurements of mean arterial pressure, FRAP and TBARS.
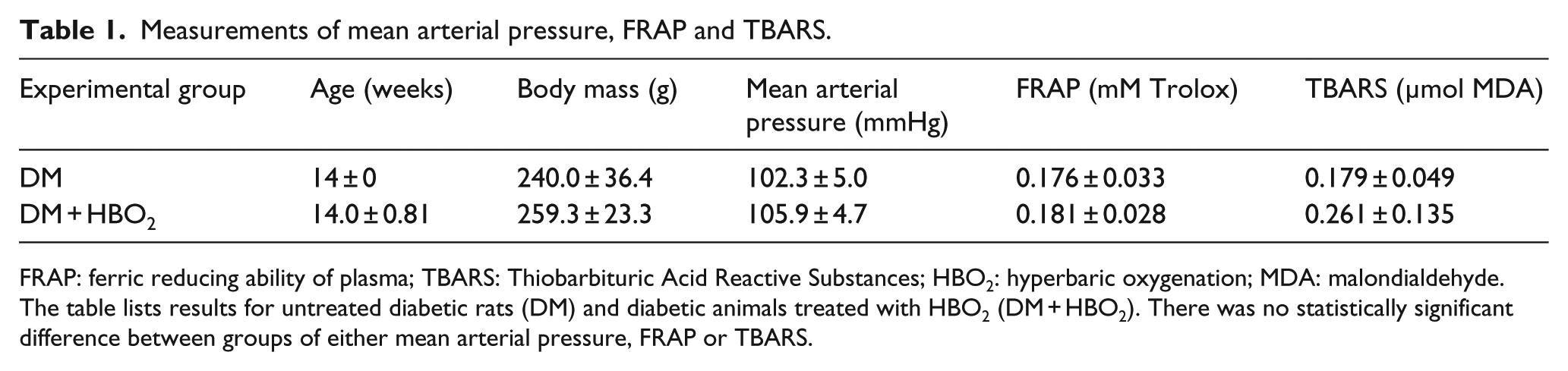
FRAP: ferric reducing ability of plasma; TBARS: Thiobarbituric Acid Reactive Substances; HBO2: hyperbaric oxygenation; MDA: malondialdehyde.
The table lists results for untreated diabetic rats (DM) and diabetic animals treated with HBO2 (DM + HBO2). There was no statistically significant difference between groups of either mean arterial pressure, FRAP or TBARS.
Serum ANG-(1-7) concentration measurements
Figure 5 shows mean serum concentrations of ANG-(1-7), as determined with ELISA, in diabetic rats (N = 8) and diabetic rats treated with HBO2 (N = 8). The serum levels are similar in both groups, with no apparent effect of HBO2 (p > 0.05, Mann–Whitney U test).
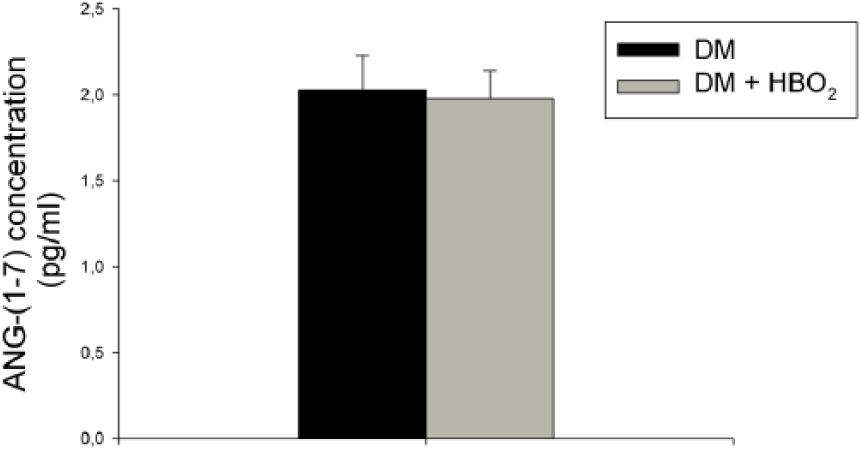
Serum levels of ANG-(1-7). Results of ELISA measurements of serum ANG-(1-7) concentrations are compared between diabetic control rats (DM) and diabetic rats treated with HBO2 (DM + HBO2).
Discussion
The obtained data from the aortic vascular reactivity experiments suggest that HBO2 significantly facilitates relaxation in response to ANG-(1-7), while not affecting ANG II contraction in the animal model of diabetes. This is the first time to demonstrate these findings in diabetic rats and the findings are in concordance with previously reported effects of HBO2 on vascular reactivity of healthy rats to ANG-(1-7). 7 A similar precontraction response to noradrenaline in both HBO2 and control diabetic rats, as displayed in Figure 1(b), suggests that there was no non-specific effect of HBO2 on precontraction, which could have falsely affected results of the ANG-(1-7) responses, and this affirms the technical accuracy of the experiments. The order of magnitude of the measured ANG-(1-7) relaxations of aortic rings corresponds roughly to extents of relaxations to ANG-(1-7) in other aortic ring experiments, for similar concentrations of ANG-(1-7).52,53
Previous studies found that ANG-(1-7) improves damaged vascular function and myocardial damage in diabetes, in addition to having other positive vascular effects in diabetes.8–10,54,55 An exogenously applied agonist of ANG-(1-7) was even connected with accelerated healing of chronic diabetic wounds. 11 In light of these discoveries, the observed facilitation of vascular ANG-(1-7) actions by HBO2 seems especially important. It may partly explain how HBO2 exerts its positive therapeutic effects in conditions with vascular pathology in diabetes.
In our aim to investigate potential mechanisms of the modulation of ANG-(1-7) vascular responses induced by HBO2, we evaluated a possible role of EETs. These arachidonic acid metabolites (with predominantly vasodilatory function) were previously found to modulate vascular reactivity to various stimuli,27,32,33 to have cardioprotective and antihypertensive properties, 56 and to exert protective effects in stroke. 14 Notably, EETs show positive effects in disorders with vascular pathology in diabetes, including myocardial ischaemia-reperfusion injury, stroke, atherosclerosis, diabetic nephropathy9,12–14,34 and others. The animal model of type 1 diabetes mellitus induced by streptozocin, as used in this work, leads to a reduction of protective EETs concentrations (through increase of EETs degradation). 14 Changes in expression of certain CYP enzymes that catalyse EETs formation are connected with beneficial effects. For example, overexpression of the CYP2J group of enzymes in mice alleviates streptozocin-induced diabetic nephropathy 34 and in apoE knockout mice (an atherosclerosis model) increases EETs production and protects against ANG II-induced abdominal aortic aneurysm. 57 Upregulation of CYP2J3 inhibits apoptosis in neonatal rat cardiomyocytes after heart ischaemia, while MS-PPOH reduces this cardioprotective effect. 58 In salt-sensitive stroke-prone spontaneously hypertensive rats, salt loading down-regulated CYP2C11 expression, whereas upregulation of cerebral CYP2C11 expression using clofibrate was protective against stroke and led to increase in blood vessel diameters and cerebral blood flow. 59 Experiments in our work demonstrated that the highly selective epoxidation inhibitor MS-PPOH reversed the facilitated aortic relaxation to ANG-(1-7) in HBO2 diabetic rats. The inhibitor is highly selective and is reaction specific for epoxidation and not isoform specific, 38 which means that it inhibits EETs formation in a wide range of CYP isoforms that are capable of catalysing epoxidation reactions. The results, therefore, strongly indicate that EETs play an important role in the mechanism of HBO2-induced modulation of vascular responses to ANG-(1-7). Separate analysis of the effect of MS-PPOH on contraction to noradrenaline alone (as displayed in Figure 2) shows that there is no non-specific alteration of the precontraction to noradrenaline by MS-PPOH, indicating technical accuracy. A crucial role of EETs in the mechanism of beneficial vascular effects of HBO2 would fit well into the framework of knowledge about EETs generated in previous research. Since they can modulate vascular reactivity in diabetes27,32,33 and exert protective effects in conditions with vascular pathology in diabetes,9,12–14,34 a facilitation of EETs synthesis induced by HBO2 could further explain its beneficial clinical effects as well as the facilitation of vascular reactivity to ANG-(1-7). Furthermore, it has been revealed that EETs synthesis (as well as synthesis of 20-HETE) becomes decreased with a decrease in tissue pO2,35,36 identifying specific CYP enzymes as biological vascular oxygen sensors with their activity depending on oxygen tension.35–37 An intense increase in tissue pO2, as induced by HBO2, would therefore theoretically increase EETs formation and stimulate the CYP oxygen sensors.
To further elucidate the role of specific CYP epoxygenases as potential targets or effectors of HBO2, we tested the influence of HBO2 on aortic expression of the important rat CYP isoforms CYP2J3, CYP4A1, CYP4A2, CYP4A3 and CYP2C11. A significant upregulation of aortic CYP2J3 mRNA and CYP2C11 protein expression was detected in HBO2 diabetic rats compared to untreated diabetic rats. Such an upregulation is interesting and might partially explain the mechanisms of HBO2, but a causal relation between this upregulation and facilitation of ANG-(1-7) vascular reactivity or clinical beneficial effects cannot be determined for certain at this point. Other important epoxygenase isoforms, such as CYP4A2 and CYP4A3, were not found to be upregulated (mRNA), and an upregulation of CYP2J3 at the mRNA level could not be verified by Western blot at the protein level (the higher protein expression in the HBO2 diabetic group was not significant). It is also possible that HBO2 increases vascular sensitivity to EETs, instead of significantly increasing EETs synthesis. Namely, if MS-PPOH diminishes the effects of HBO2 (on ANG-(1-7)-induced relaxation) by inhibiting EETs synthesis, and EETs synthesis was not significantly increased by HBO2, then this suggests that HBO2 increases the vascular sensitivity to EETs. Finally, both pathways are possible in the HBO2 mechanism, based on our data, and further studies of this problem are necessary. A definitive verdict might come with future research on resistance vessels (such as the gracilis or mesenteric arteries) and in microcirculation, where the effects of HBO2 may be much more pronounced than in the aorta. The extremely low EET concentrations are difficult to measure and future high-performance liquid chromatography (HPLC) studies might investigate direct changes of EET levels induced by HBO2. Other possible pathways in the HBO2 mechanism additionally complicate this framework, including partially known influences of HBO2 on endothelial nitric oxide synthase (eNOS), other signalling cascades and inflammation 1 or conducted vasomotor responses.60,61 Considering the interaction of arachidonic acid pathways with nitric oxide pathways in oxygen sensitivity 36 and regional differences of arachidonic acid metabolite roles, 36 it is evident that the role of CYP enzymes in oxygen homeostasis is very complex. Figure 6 summarizes the discussed findings and mechanisms.
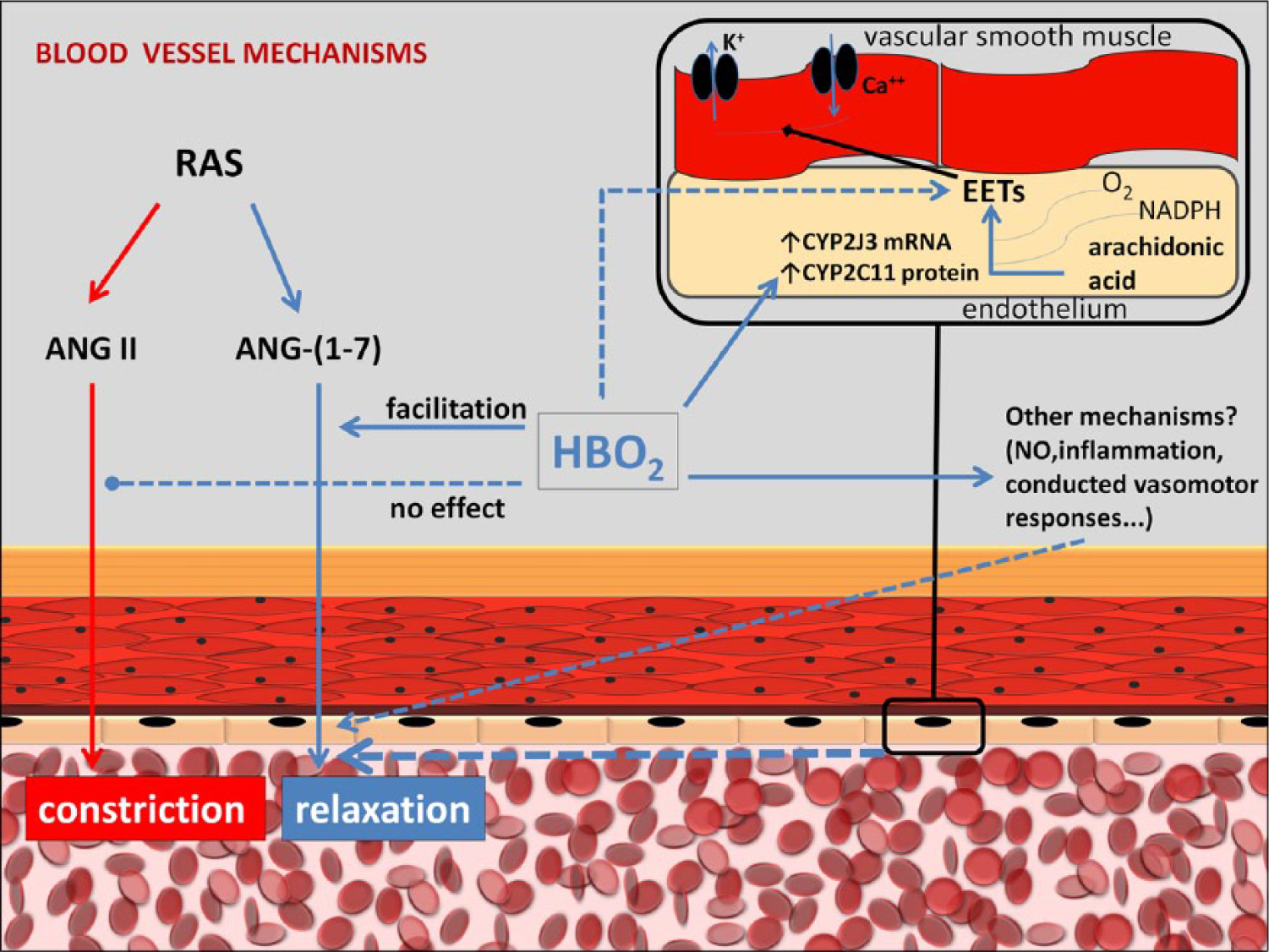
Schematic depiction of analysed HBO2 effects and the discussed potential mechanisms. The observed facilitation of the dilatory effects of ANG-(1-7) by HBO2 may partly be a consequence of epoxygenase upregulation (confirmed for CYP2J3, CYP2C11) or possibly a result of an increase of vascular sensitivity to EETs. Also, other possible factors (not involving EETs or other arachidonic acid metabolites) cannot be excluded as part of the mechanism.
The results of this work do not demonstrate any significant changes in arterial blood pressure levels or indicators of oxidative stress induced by the HBO2 protocol in diabetic rats. It was important to test this possibility, since both arterial pressure and oxidative stress affect vascular reactivity42–47 and there are also interconnections between ANG-(1-7) and oxidative stress. 62 Data from previous publications on HBO2 influence on arterial pressure are not univocal: HBO2 can by various accounts and under different conditions increase,63–65 decrease 66 or leave blood pressure unchanged.65,67,68 However, the data cannot be readily compared because of vast differences in HBO2 protocols (acute or chronic exposure, differences in exposure duration and pressure levels), underlying conditions of the treated groups (diabetic, spontaneously hypertensive, healthy) or experimental models (humans, animals). It is necessary to establish standardized reference values for specific conditions and protocols. We therefore measured mean arterial pressure for this specific protocol used in diabetic rats and found it to be unchanged in the HBO2 group. The lack of changes of FRAP and TBARS levels in our results is not surprising, since with the use of this protocol of intermittent HBO2, the experiments are performed on the fifth day of the protocol – 24 h after the last HBO2 exposure. The results are consistent with previously published investigations 69 of a single HBO2 exposure (in healthy rats) of the same duration and pressure level as used in this study, where elevated TBARS levels (and decreased FRAP) returned to normal within 24 h after the single HBO2 exposure. There is also the question of adaptation – a possible induction of antioxidant systems1,7,69 after repetitive HBO2 exposures might be responsible for normalization of oxidative stress indicators. Either way, our results suggest that changes in mean arterial pressure or oxidative stress levels do not constitute part of the mechanism of observed HBO2 effects on vascular reactivity to ANG-(1-7), although there is no way to exclude a hidden signalling effect 1 of intermittent oxidative stress elevations or antioxidant system induction.
Earlier studies showed that HBO2 could influence levels of certain components of the renin–angiotensin system, such as renin levels.70–72 The effect of HBO2 on ANG-(1-7) serum concentrations was not investigated until now. This is of particular importance here because HBO2 was found to modulate vascular responses to ANG-(1-7) and it is known that changes in agonist levels can, in general, change agonist efficacy and signal transduction – because of internalization and desensitization of its receptors.50,51 The data show that there was no change of serum ANG-(1-7) levels in diabetic rats treated with HBO2, excluding the possibility that such a change would form part of the mechanism of HBO2 influence on vascular reactivity to ANG-(1-7). This corroborates the discussed role of EETs and CYP enzymes as the primary focus of HBO2 vascular mechanisms. These results may also have broader significance, because it is useful to know that HBO2 does not seem to decrease the beneficial ANG-(1-7) in the serum of diabetic subjects.
A more complete knowledge about the exact mechanisms that mediate HBO2 effects in vasculature is an essential prerequisite for efficient clinical use and optimization of HBO2 as a therapeutic and investigative tool. HBO2 is being used for treatment and experiments for many years, without fully comprehending the changes it induces in the circulation and at the molecular and cellular level. This study represents a further step in elucidating the underlying mechanisms of HBO2 influence and is a foundation for additional investigations (that should also include human subjects). Understanding the actions of HBO2 is not only important if we want to advance protocols, evaluate indications and contraindications and improve therapeutic results, but is also the basis for predicting adverse and unwanted effects and interactions with medications. This is especially so in a time of an immense global burden of diabetes 73 and linked cardiovascular disease, 74 and a wide use of multiple medications such as ones that act on the renin–angiotensin system.
Conclusion
HBO2 significantly increases vascular responses to ANG-(1-7) in diabetic rats, what could potentially explain some of the observed positive effects of HBO2 in pathologic conditions (such as chronic diabetic ulcers). EETs seem to play an important role in the mechanism of this modulation. HBO2 increases the expression of specific CYP isoforms, but it is also possible that it increases the vascular sensitivity to EETs. The changes in vascular reactivity were not a consequence of a possible change in arterial blood pressure, oxidative stress or ANG-(1-7) blood levels. HBO2 does not significantly alter reactivity to ANG II in diabetic rats. Better knowledge of mechanisms of HBO2 effects in the vasculature of diabetic subjects is necessary for more efficient clinical use and represents a foundation for future research.
Footnotes
Acknowledgements
The authors wish to thank Dr Ana Cavka for her help with aortic ring statistical analysis.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work has been funded by a grant of the Croatian Ministry of Science, Education and Sports 219-2160133-2034.
