Abstract
We report here the efficacy and safety of GFT505, a novel liver-targeted peroxisome proliferator-activated receptor alpha/delta (PPARα/δ) agonist, in the
Introduction
Peroxisome proliferator-activated receptors (PPARs) are ligand-activated transcription factors implicated in the regulation of glucose homeostasis, lipid metabolism and vascular inflammation. Numerous synthetic PPAR ligands have been developed to treat metabolic disorders including type 2 diabetes mellitus (T2DM), dyslipidaemia and non-alcoholic steatohepatitis (NASH). 1
PPARα agonists such as fibrates lower serum triglycerides (TGs) and increase high-density lipoprotein (HDL)-cholesterol in patients with hyperlipidaemia. 2 PPARγ agonists such as rosiglitazone and pioglitazone are insulin sensitizers that improve insulin sensitivity in T2DM patients. 3 The PPARδ isotype is also a potential target for the treatment of metabolic diseases. Indeed, preclinical studies suggest that PPARδ regulates glucose metabolism and improves insulin sensitivity 4 and β-cell function. 5 In humans, the synthetic PPARδ agonist GW501516 improves dyslipidaemia and insulin resistance and reduces liver fat in moderately overweight subjects. 6
Given the potential pharmacological benefit of activating multiple PPAR isoforms, numerous synthetic dual-PPAR and pan-PPAR agonists have been developed to obtain synergistic actions on lipid and glucose homeostasis for the treatment of metabolic disorders including T2DM, dyslipidaemia and NASH. 7 However, several PPARα/γ agonists (glitazars) were discontinued because of safety concerns during clinical development.8,9 The most limiting safety issue associated with PPARγ agonist compounds relates to fluid retention, haemodilution, oedema and subsequent effects on cardiac remodelling and risk of heart failure. 10 Most recently, the development of the PPARα/γ agonist aleglitazar was terminated in Phase III due to safety concerns including an increase in heart failure. 11
GFT505 is a novel liver-targeted PPAR agonist with preferential activity on PPARα (half-maximal effective concentration 10–20 nmol/L) and additional activity on PPARδ (half-maximal effective concentration 100–150 nmol/L).
12
GFT505 has been shown to improve dyslipidaemia, hepatic and peripheral insulin sensitivity and liver enzymes in abdominally obese patients.12,13 Here, the efficacy and safety of GFT505 were assessed in the
Materials and methods
Compounds and chemical reagents
Rosiglitazone was acquired from Yick-Vic Chemicals & Pharmaceuticals Ltd (Hong Kong). Aleglitazar and GFT505 were synthesized at Genfit and verified by nuclear magnetic resonance (NMR) and liquid chromatography–mass spectrometry (LC-MS).
Diabetic db/db mice studies
Animals
Male
Treatments
In Experiment 1, mice were treated with GFT505 (10 and 30 mg/kg/day), rosiglitazone (10 mg/kg/day), aleglitazar (0.3, 1 and 3 mg/kg/day) or vehicle for 56 days. An oral glucose tolerance test (OGTT) was performed after 43 days. In Experiment 2, mice were treated with GFT505 (3, 10 and 30 mg/kg/day), rosiglitazone (10 mg/kg/day) or vehicle for 56 days. In Experiment 3, mice were treated with GFT505 (10 and 30 mg/kg/day) or vehicle for 14 days, and a pyruvate challenge test was performed. At the end of each experiment, plasma and selected organs were collected.
Glucose tolerance (OGTT) and pyruvate challenge tests
Blood chemistry
TGs and HbA1c were measured using the RX Daytona™ automatic analyzer (Randox, UK). Plasma adiponectin levels were determined using mouse enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems, UK). HDL-cholesterol levels were measured using a kit from Biomerieux (France).
Real-time polymerase chain reaction (PCR)
Nucleospin® 96 RNA kit (Macherey Nagel, France), RNeasy Fibrous Tissue Mini Kit (Qiagen, France) and guanidinium thiocyanate were used to extract total RNA from liver, skeletal muscle and adipose tissue, respectively. After synthesis of complementary DNA using Moloney Murine Leukemia Virus Reverse Transcriptase (MMLV-RT), the real-time PCR measurement of individual cDNAs was performed using iQ SYBR Green Supermix kit to measure duplex DNA formation with the MyiQ Single-Color Real-Time PCR Detection (Biorad, France). The PCR primers used are shown in Table S1. Expression levels were normalized using 36B4 (liver) or 18S (skeletal muscle and adipose tissue) genes.
Twelve-month toxicology study in cynomolgus monkeys
Animals and treatments
Male and female purpose-bred cynomolgus monkeys (
Haematology and blood chemistry
Bone marrow smears were prepared from the sternum of all animals at necropsy and stained with May Grünwald Giemsa. The bone marrow differential cell count determination was performed by microscopic examination. For haematology, samples were collected into ethylenediaminetetraacetic acid (EDTA) tubes and analyses were performed using an ADVIA 120 Hematology Analyzer (Siemens, Germany). For blood chemistry, samples were collected into lithium heparin tubes and analyses were performed using an ADVIA 1650 (Siemens).
Clinical examinations
Electrocardiography and blood pressure measurements were performed on all animals prior to treatment and at 13, 26 and 52 weeks of treatment. Echocardiography examinations were performed on four animals per sex in the vehicle and high-dose (50 mg/kg/day GFT505) groups prior to treatment and at 26 and 52 weeks of treatment. At the end of the 52-week treatment period, organs were dissected and weighed, and macroscopic and microscopic organ examinations were performed.
Statistical analyses
Experimental results are expressed as mean ± standard deviation (SD). Statistical analyses were performed using a one-way analysis of variance followed by a post hoc Bonferroni analysis and compared to the corresponding control group with
Results
GFT505 treatment improves glucose control and plasma lipids in diabetic db/db mice
Eight-week treatment of
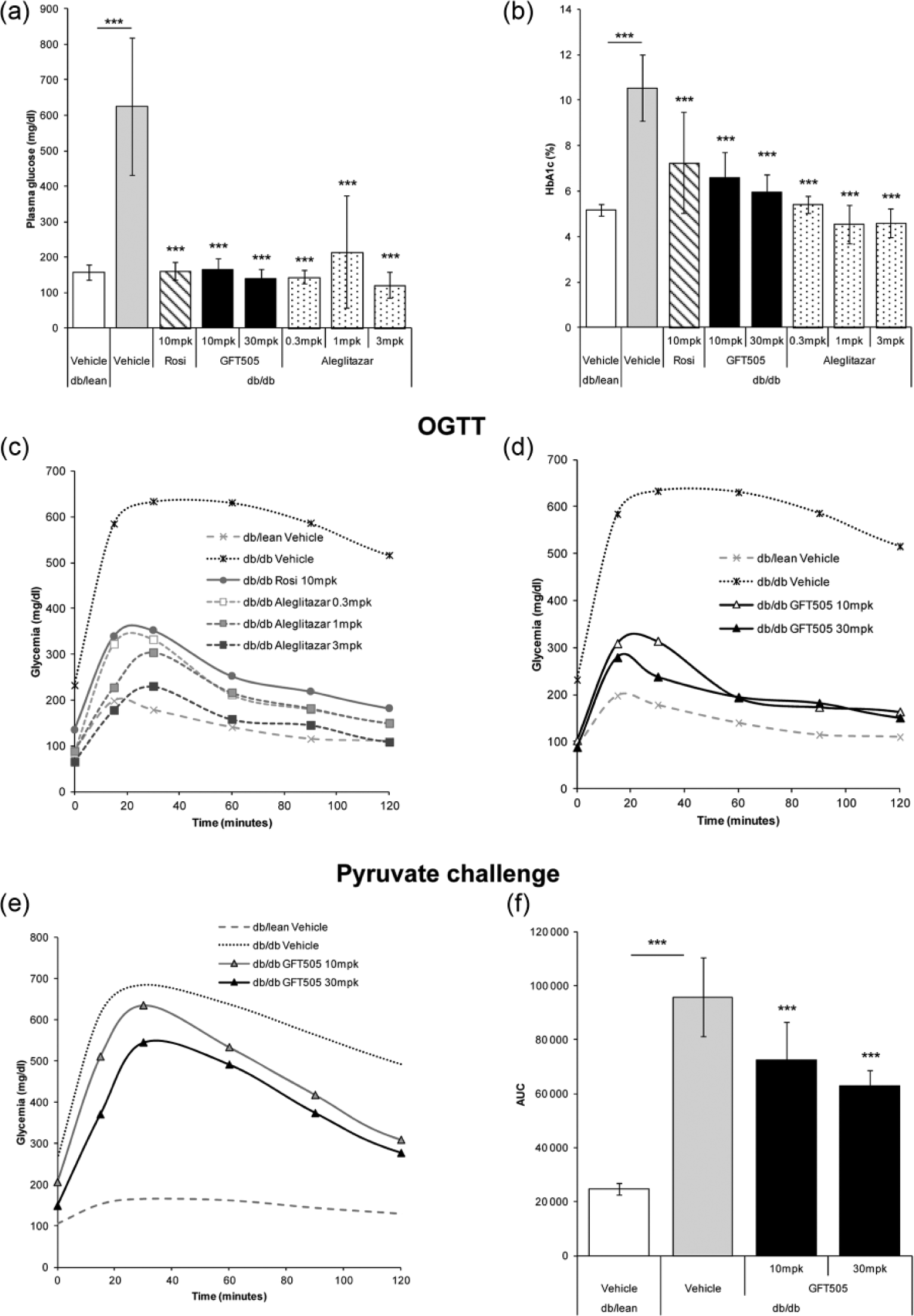
Effect of GFT505 on glycaemic control in insulin-resistant
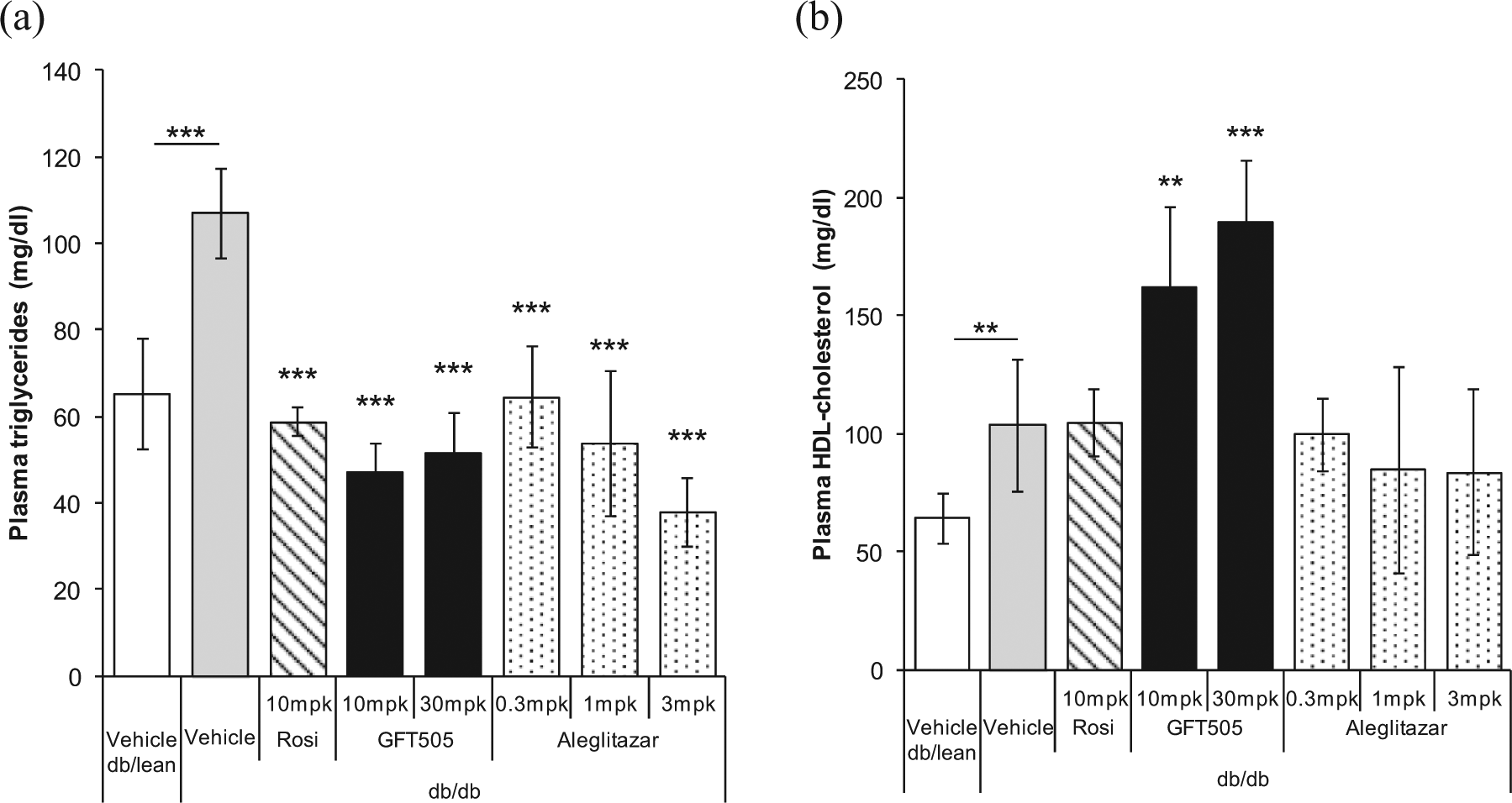
Effect of GFT505 on plasma lipids in insulin-resistant
To determine the mechanism of action, gene expression levels were analysed in
Effects of GFT505 (3, 10 and 30 mg/kg/day) and rosiglitazone (10 mg/kg/day) on gene expression in liver, skeletal muscle and adipose tissue of insulin-resistant
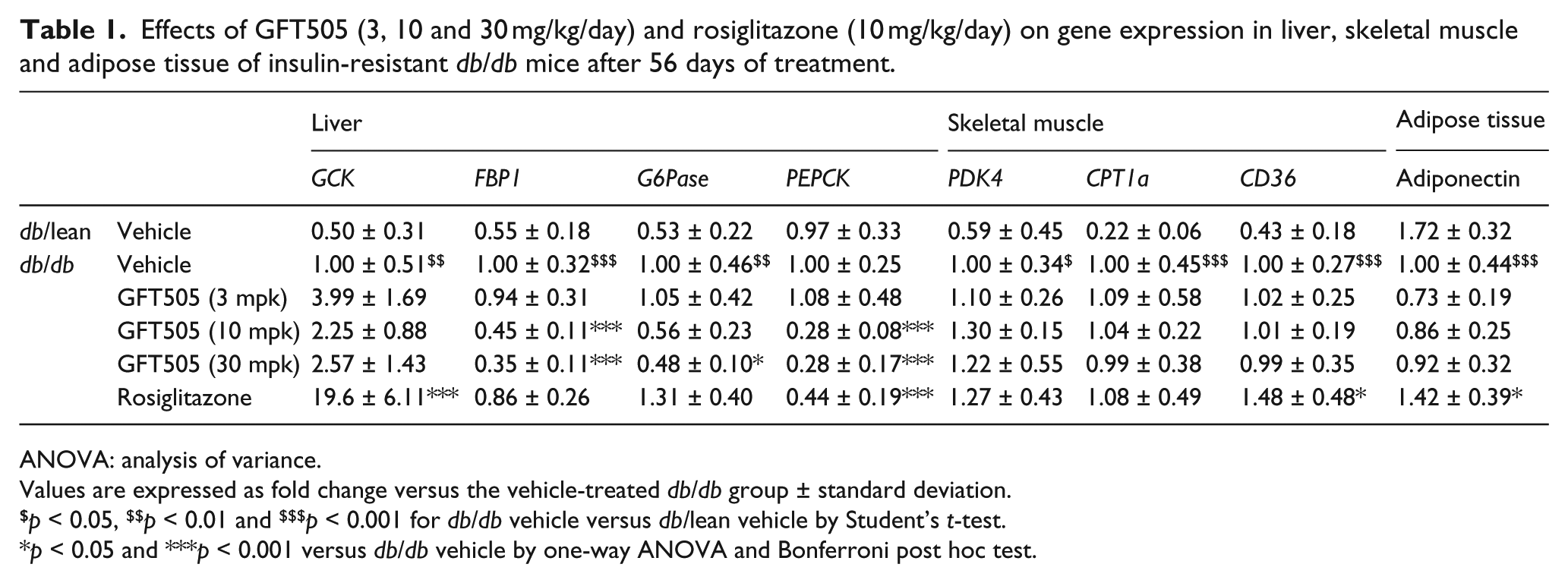
ANOVA: analysis of variance.
Values are expressed as fold change versus the vehicle-treated
Finally, the effects of GFT505 on hepatic gluconeogenesis were directly assessed in
GFT505 does not induce cardiac adverse effects of PPARγ-activating agonists
Eight-week treatment of
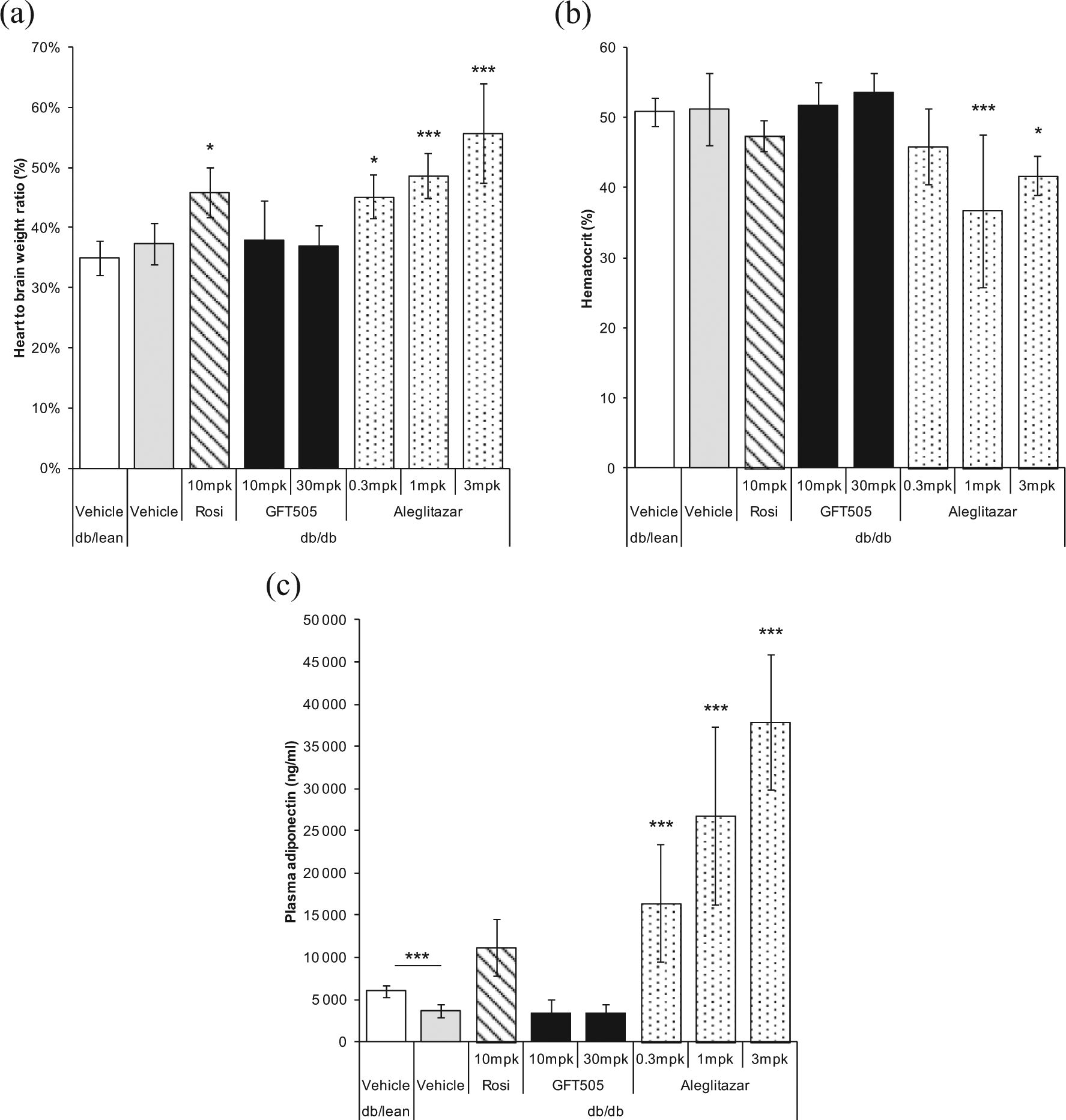
Safety profile of GFT505 in insulin-resistant
The cardiac safety of GFT505 was also studied during a 12-month toxicity study in cynomolgus monkeys. GFT505 at up to 50 mg/kg/day showed no significant effect on heart weight, and there were no treatment-related histological changes in cardiac tissues (
Echocardiographic parameters measured in male and female cynomolgus monkeys after 12 months of treatment.
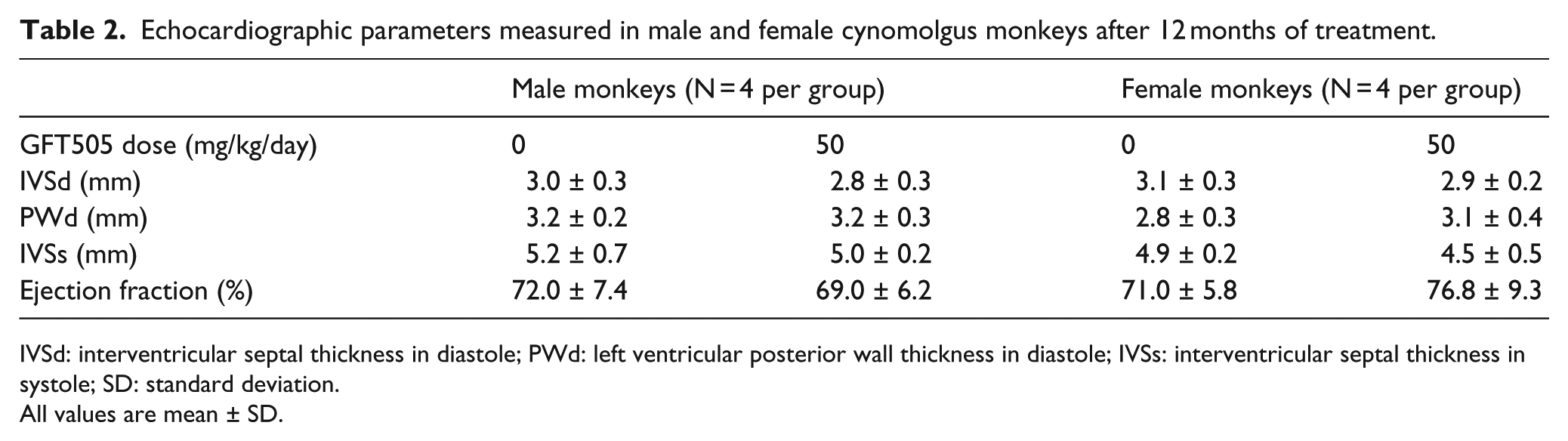
IVSd: interventricular septal thickness in diastole; PWd: left ventricular posterior wall thickness in diastole; IVSs: interventricular septal thickness in systole; SD: standard deviation.
All values are mean ± SD.
Haematology and blood biochemistry parameters measured in male and female cynomolgus monkeys after 12 months of treatment.
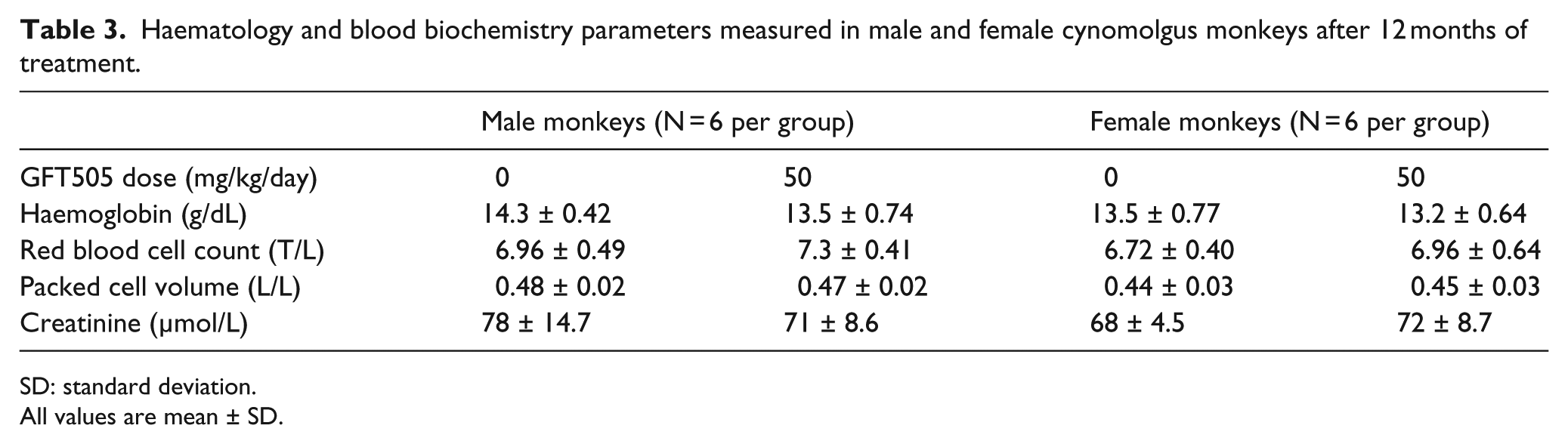
SD: standard deviation.
All values are mean ± SD.
Discussion
A mixed PPARα/δ agonist that activates both nuclear receptors has the potential to address multiple metabolic and cardiovascular risk factors of T2DM. Moreover, such an agonist should not be associated with the safety concerns encountered with PPARγ and PPARα/γ agonists, including the risk of cardiac insufficiency associated with glitazones and glitazars.9 –11 In this study, treatment with GFT505, a liver-targeted dual-PPARα/δ agonist, is shown to regulate glucose homeostasis and plasma lipids in a diabetic model, without showing PPARγ-associated adverse cardiac effects.
GFT505 shows efficacy in animal models of NASH and liver fibrosis
16
and is currently being assessed in a 1-year Phase IIb trial (ClinicalTrials.gov: NCT01694849) in NASH. In this study, treatment with GFT505 improved glycaemic control in
This study also showed that at pharmacological doses in
In summary, this study demonstrates that the dual-PPARα/δ agonist GFT505 has beneficial actions on glucose homeostasis via inhibition of hepatic gluconeogenesis. The distribution profile of GFT505, which accumulates predominantly in liver, 16 as well as its lack of PPARγ activity, likely plays an important role in its favourable efficacy/risk profile. In conclusion, GFT505 demonstrates pharmacological and safety profiles consistent with its potential use for the chronic treatment of metabolic disorders including T2DM and NASH.
Footnotes
Acknowledgements
The authors thank Raphaël Boutry and Linda Cambula for their outstanding technical assistance and Anne Rubenstrunk for valuable discussions.
Declaration of conflicting interest
BS and BC are advisors to Genfit and have been involved in the conception, analysis of the experiments and drafting of the manuscript in close interaction with the Genfit employees. All the experiments have been performed according to state-of-the-art methodology and have been analysed in an objective manner according to ethical rules of scientific research.
Funding
This work was supported by Genfit SA and an FP7/2007–2013 no. 305707 (RESOLVE) grant to B.S. B.S. also received funding from Inserm and Université Lille 2. B.S. is a member of the Institut Universitaire de France.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
