Abstract
Metabolic syndrome (MetS) is a clustering of atherosclerotic coronary heart disease risk factors. This post-hoc analysis compared the effects of switching to ezetimibe/simvastatin 10/20 mg or rosuvastatin 10 mg in a cohort of 618 high-risk hypercholesterolaemic patients with (
Introduction
Metabolic syndrome (MetS) represents an important segment of the at-risk population as determined by the number of patients who display characteristics of MetS and have had a cardiovascular event. Based on an analysis of the 1988–1994 National Health and Nutrition Examination Survey III (NHANES III), an age-adjusted estimated 24% of all adults and >40% of adults over 60 years of age in the United States had the MetS. 1 An updated NHANES 1999–2000 analysis demonstrated that there was a continued increase, up to 27%, in the age-adjusted prevalence of MetS. 2
Components of the MetS include abdominal obesity, atherogenic dyslipidaemia, hypertension, insulin resistance, prothrombotic state, and proinflammatory state. Although it has not been possible thus far to determine how each of these factors individually contributes to coronary heart disease (CHD), together they accentuate the risk in the presence of reduced levels of high-density lipoprotein cholesterol (HDL-C). 3
Expert organisations, such as the National Cholesterol Education Program Adult Treatment Panel (NCEP ATP) III, have developed guidelines that recommend reducing elevated levels of low-density lipoprotein cholesterol (LDL-C) as the primary therapeutic target for reducing the risk of CHD in patients with the MetS.4,5 These guidelines also recommended management of the MetS by reversing obesity and increasing physical activity. 5
Ezetimibe, an inhibitor of intestinal cholesterol absorption, significantly lowers LDL-C, modestly decreases triglycerides (TG), and raises HDL-C levels. 6 When combined with simvastatin, the two drugs provide complementary improvements in the atherogenic lipid profile of patients with hyperlipidaemia.7,8 Rosuvastatin represents the most potent statin currently available in terms of both the lower statin dosage and LDL-C lowering.
Many studies have evaluated the effects of lipid-altering drugs in patients with the MetS.9-16 However, it is unclear whether MetS status is a better predictor of atherosclerotic CHD risk than an assessment of its individual components. Given the lack of clarity of whether the dyslipidaemia in patients with the MetS truly represents a distinctly different clinical entity than the dyslipidaemia in patients without MetS, it is clinically relevant to know how hyperlipidaemic patients respond to lipid-altering therapies, with or without meeting the criteria for the diagnosis of MetS.
This post-hoc analysis was undertaken to examine the effects of ezetimibe/simvastatin compared with rosuvastatin in dyslipidaemic patients with and without the MetS. Patients in the study had been on previous statin monotherapy, but had not achieved the target LDL-C levels prescribed in the NCEP ATP III guidelines. 5 The differences between the two treatments for their various lipid-altering effects were evaluated. Also evaluated were the odds ratio of high-risk patients achieving LDL-C levels of <2.59 mmol/L, non-HDL levels of <3.37 mmol/L and apolipoprotein (apo) B levels of <0.9 g/L and for highest-risk patients reaching levels of <1.81 mmol/L for LDL-C, <2.59 mmol/L for non-HDL-C, and <0.8 g/L for apo B, as recommended in the NCEP ATP III guidelines and the American Diabetes Association and the American College of Cardiologists.5,17
Materials and methods
Patients and study design
Full details of the study design are reported elsewhere. 18 Briefly, this multicentre, randomised, double-blind trial, active-controlled, parallel group study (ClinicalTrials.gov: NCT00479713) enrolled 618 patients with documented hypercholesterolaemia (LDL-C 2.59–4.92 mmol/L at the screening visit and 2.59–4.14 mmol/L at randomisation) and high cardiovascular risk who were taking a stable daily dose of one of several statin medications for at least 6 weeks prior to the study randomisation visit. Patients entered a 6-week open-label stabilisation/screening period during which they continued to receive their pre-study statin dose. Patients were deemed to be of high cardiovascular risk if they met one or more of the following criteria: (i) history of CHD (i.e. stable and unstable angina, revascularisation procedure, myocardial infarction, documented myocardial ischaemia) or with established vascular atherosclerotic disease (i.e. peripheral vascular disease, ischaemic stroke); (ii) type 2 diabetes without a history of vascular disease and with high cardiovascular risk (i.e. renal impairment [proteinuria >300 mg/24 h or creatinine clearance standardised for body surface area <1.002 ml/sec] and/or at least two CHD risk factors per Framingham risk calculation); (iii) CHD risk >20% over 10 years as determined by Framingham risk calculation. 4 Fasting TG levels had to be ≤350 mg/dl 1 week prior to the randomisation visit (i.e. Week 0/baseline) to allow for the calculation of LDL-C by the Friedewald equation. 19
Patients who did not achieve their minimum recommended LDL-C levels (i.e. LDL-C <2.59–4.14 mmol/L) after taking a stable dose (≥6 weeks) of open-label statin monotherapy were stratified by study site and potency of their pre-randomisation statin medication/dose (low [Stratum 1: atorvastatin 10 mg, fluvastatin 80 mg, lovastatin 20 mg, pravastatin 40 mg, simvastatin 20 mg] or high [Stratum 2: atorvastatin 20 mg, rosuvastatin 5 mg, simvastatin 40 mg]) and subsequently randomised in equal proportions to receive double-blind ezetimibe/simvastatin 10/20 mg (
Statistical analyses
Efficacy and safety analyses were conducted in the overall analysis population as well as in patient subgroups defined by the presence and absence of MetS. The overall analysis population consisted of all randomised patients with known MetS status at baseline. Patients were included in the MetS subgroup if they met at least three of the following criteria at baseline: (i) body mass index (BMI) ≥30 kg/m2 (surrogate for increased waist circumference); (ii) TG ≥1.7 mmol/L; (iii) HDL-C <1.03 mmol/L (men) and <1.29 mmol/L (women); (iv) diagnosis of hypertension or systolic blood pressure (SBP) ≥130 mm Hg or diastolic blood pressure (DBP) ≥85 mm Hg or taking antihypertension medications; (v) diagnosis of type 2 diabetes or fasting plasma glucose (FPG) ≥5.6 mmol/L or taking medication for elevated blood glucose. The non-MetS subgroup for this analysis consisted of all patients with known MetS status at baseline, excluding those patients with MetS. Fifteen patients were excluded from this analysis because their baseline MetS status was unknown.
The primary efficacy objective was to assess the effects of treatment on the mean percentage change from baseline to study endpoint (i.e. last post-baseline measurement) in LDL-C. Other efficacy endpoints included the percentage change from baseline in total cholesterol (TC), TG, HDL-C, non-HDL-C, LDL-C/HDL-C ratio, TC/HDL-C ratio, apo B, and high-sensitivity C-reactive protein (hs-CRP). The percentage of patients achieving recommended treatment levels for LDL-C (<2.59 mmol/L and <1.81 mmol/L), non-HDL-C (<3.37 mmol/L and <2.59 mmol/L), and apo B (<0.9 g/L and <0.8 g/L) at study endpoint were assessed.
Adverse experiences (AEs) were assessed throughout the study. The investigators determined the severity and relationship to study drug. Pre-specified AEs of special interest included those that were gastrointestinal related, gallbladder related, an allergic reaction or rash, hepatobiliary related, elevations in alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST) ≥3× upper limit of normal (ULN), and creatine kinase elevations ≥10× ULN with or without muscle symptoms.
This subgroup analysis was performed on the full-analysis set (FAS) population with known MetS status, which included all patients who received at least one dose of study medication, had a baseline efficacy measurement, and at least one post-randomisation efficacy measurement.
Continuous efficacy results for percent change from baseline in normally distributed parameters (i.e. LDL-C, TC, HDL-C, non-HDL-C, LDL-C/HDL-C ratio, TC/HDL-C ratio, and apo B) were analysed using a parametric analysis of variance (ANOVA) model with terms for treatment, stratum, baseline efficacy variable (categorised based on quartiles), study centre, MetS status (with, without) and treatment-by-subgroup interaction. Least squares (LS) means and 95% confidence intervals (CIs) within each patient subgroup (i.e. MetS/non-MetS) using the above model (except the last two terms involving subgroup) were computed and used to quantify the differences between treatment groups.
Continuous efficacy results for the percentage change from baseline in non-normally distributed parameters (i.e. TG and hs-CRP) were analysed using an ANOVA model on ranks of these efficacy variables with terms for treatment, stratum, baseline efficacy variable (categorised based on quartiles), study centre, MetS status (with, without) and treatment-by-subgroup interaction. Differences between treatment groups were quantified as differences in medians and 95% CIs using Hodges–Lehmann estimates within each patient subgroup (i.e. MetS/non-MetS).
The percentages of patients achieving lipid/lipoprotein recommended levels at study endpoint were analysed using a logistic regression model with terms for treatment, stratum, baseline efficacy variable, MetS status (with, without), and treatment-by-subgroup interaction. Odds ratio estimates and 95% CIs using the above model (except the last two terms involving subgroup) were computed and used to quantify the treatment effect within each patient subgroup (i.e. MetS/non-MetS).
Due to the exploratory nature of this analysis, no multiplicity adjustments were employed. Between-group differences and treatment-by-subgroup interaction tests with a
The safety analysis was based on the all-patients-as-treated population of patients with known MetS status who received at least one dose of study medication.
Results
Of the 618 patients enrolled in this study, 603 (98%) had sufficient information to determine MetS status at baseline; 306 patients were randomly allocated to the ezetimibe/simvastatin 10/20 mg group and 297 to the rosuvastatin 10 mg group. Within each patient subgroup (MetS/non-MetS), baseline demographic and anthropomorphic characteristics were generally well balanced across the treatment groups (Table 1). Patients with MetS had higher mean baseline BMI, SBP, DBP, and FPG values at baseline than did patients without MetS. In addition, a higher percentage of patients in the MetS subgroup had a medical history of hypertension and type 2 diabetes mellitus. With regard to lipid/lipoprotein/biochemical parameters, patients with MetS had higher median baseline TG and hs-CRP levels and lower mean HDL-C levels than did non-MetS patients. Baseline LDL-C values were generally similar between treatment groups for patients with MetS versus those without MetS (Table 1).
Baseline characteristics for randomised patients with and without metabolic syndrome a
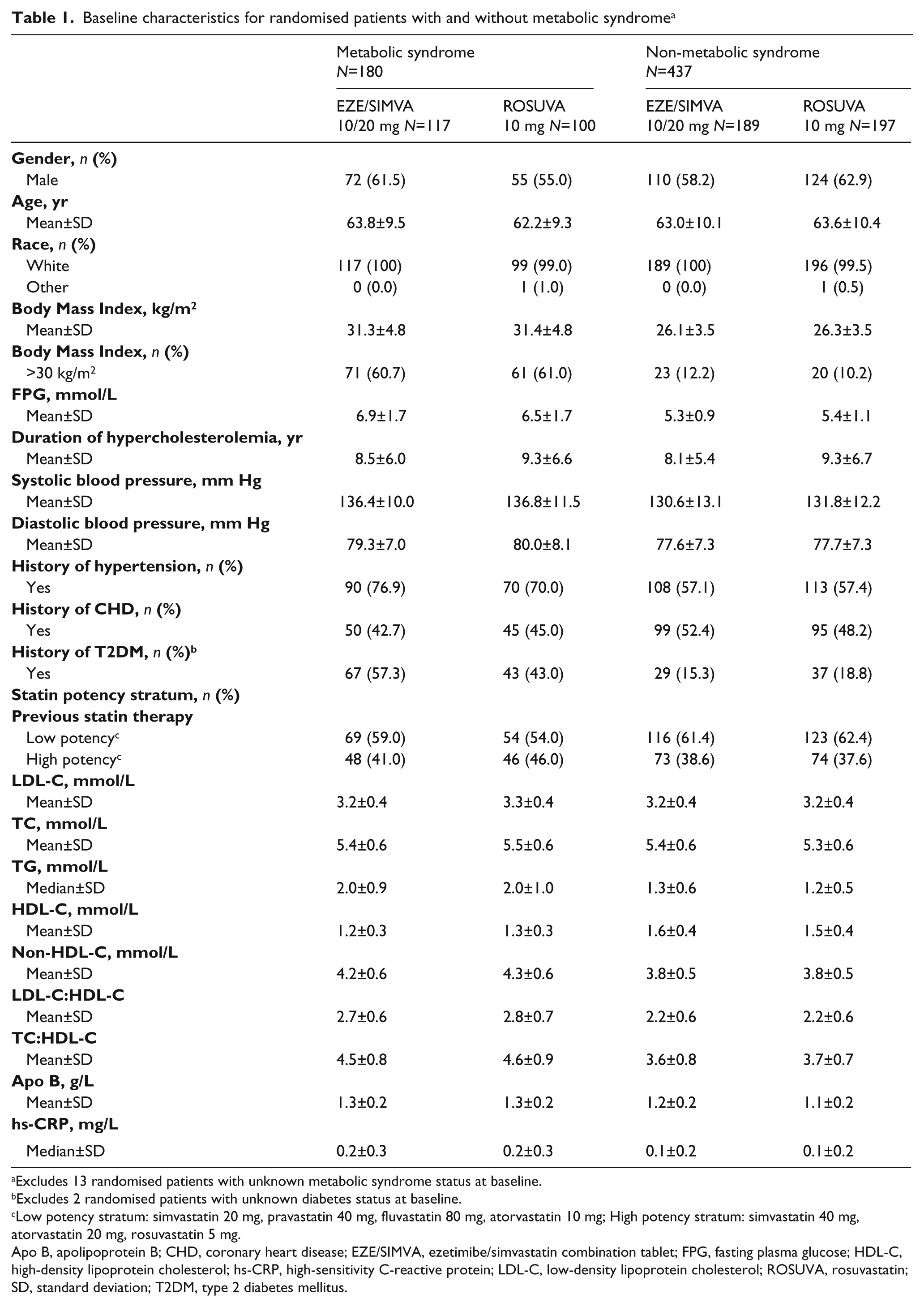
Excludes 13 randomised patients with unknown metabolic syndrome status at baseline.
Excludes 2 randomised patients with unknown diabetes status at baseline.
Low potency stratum: simvastatin 20 mg, pravastatin 40 mg, fluvastatin 80 mg, atorvastatin 10 mg; High potency stratum: simvastatin 40 mg, atorvastatin 20 mg, rosuvastatin 5 mg.
Apo B, apolipoprotein B; CHD, coronary heart disease; EZE/SIMVA, ezetimibe/simvastatin combination tablet; FPG, fasting plasma glucose; HDL-C, high-density lipoprotein cholesterol; hs-CRP, high-sensitivity C-reactive protein; LDL-C, low-density lipoprotein cholesterol; ROSUVA, rosuvastatin; SD, standard deviation; T2DM, type 2 diabetes mellitus.
The effects of ezetimibe/simvastatin 10/20 mg and rosuvastatin 10 mg on plasma concentrations of lipids and lipoproteins in the patient subgroups are shown in Figure 1. The results in the overall analysis population, excluding the 15 patients with unknown MetS status at baseline, are provided for comparative purposes. In the overall population (i.e. all patients with and without MetS), switching from statin monotherapy to ezetimibe/simvastatin 10/20 mg resulted in significantly greater between-treatment (ezetimibe/simvastatin–rosuvastatin) reductions from baseline in LDL-C (10.6%;
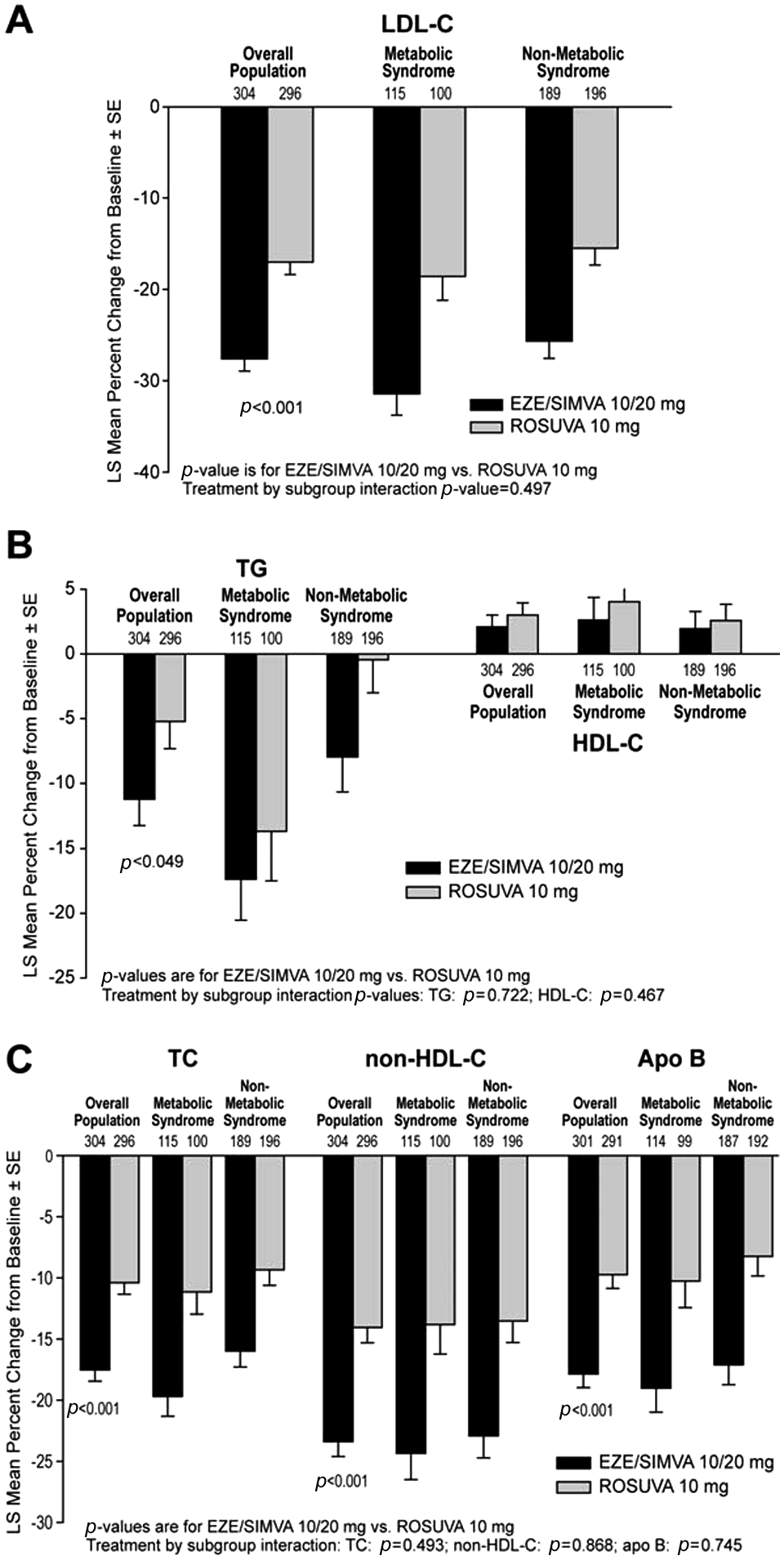
Least squares mean percent change from baseline in (A) LDL-C; (B) TG and HDL-C; and (C) TC, non-HDL-C, and apo B, for the overall analysis population and within patient subgroups defined by presence/absence of MetS.
The treatment effects within the two patient subgroups were generally consistent with the overall population (Figure 1). Ezetimibe/simvastatin 10/20 mg was more effective than rosuvastatin 10 mg at lowering LDL-C, TC, non-HDL-C, LDL-C:HDL-C, TC:HDL-C, and apo B in patients with and without MetS. Both ezetimibe/simvastatin and rosuvastatin produced similar effects on TG and HDL-C in patients with and without MetS (i.e. there were no significant between-treatment differences in HDL-C and TG irrespective of MetS status). Numerically larger between-treatment reductions from baseline were observed in MetS versus non-MetS patients for LDL-C (12.8% vs. 10.2%, respectively; Figure 1), TC (8.6% vs. -6.6%, respectively; Figure 1), non-HDL-C (10.5% vs. 9.4%, respectively) and LDL-C:HDL-C (10.1% vs. 9.4%, respectively) (Figure 1). No significant treatment-by-subgroup interactions were observed for any of the lipid, lipoprotein, or biochemical parameters, indicating there were no significant differences in the treatment effects seen between patient subgroups defined by the presence and absence of MetS (data not shown).
In the overall population, a significantly higher percentage of patients achieved LDL-C levels of <2.59 and <1.81 mmol/L (
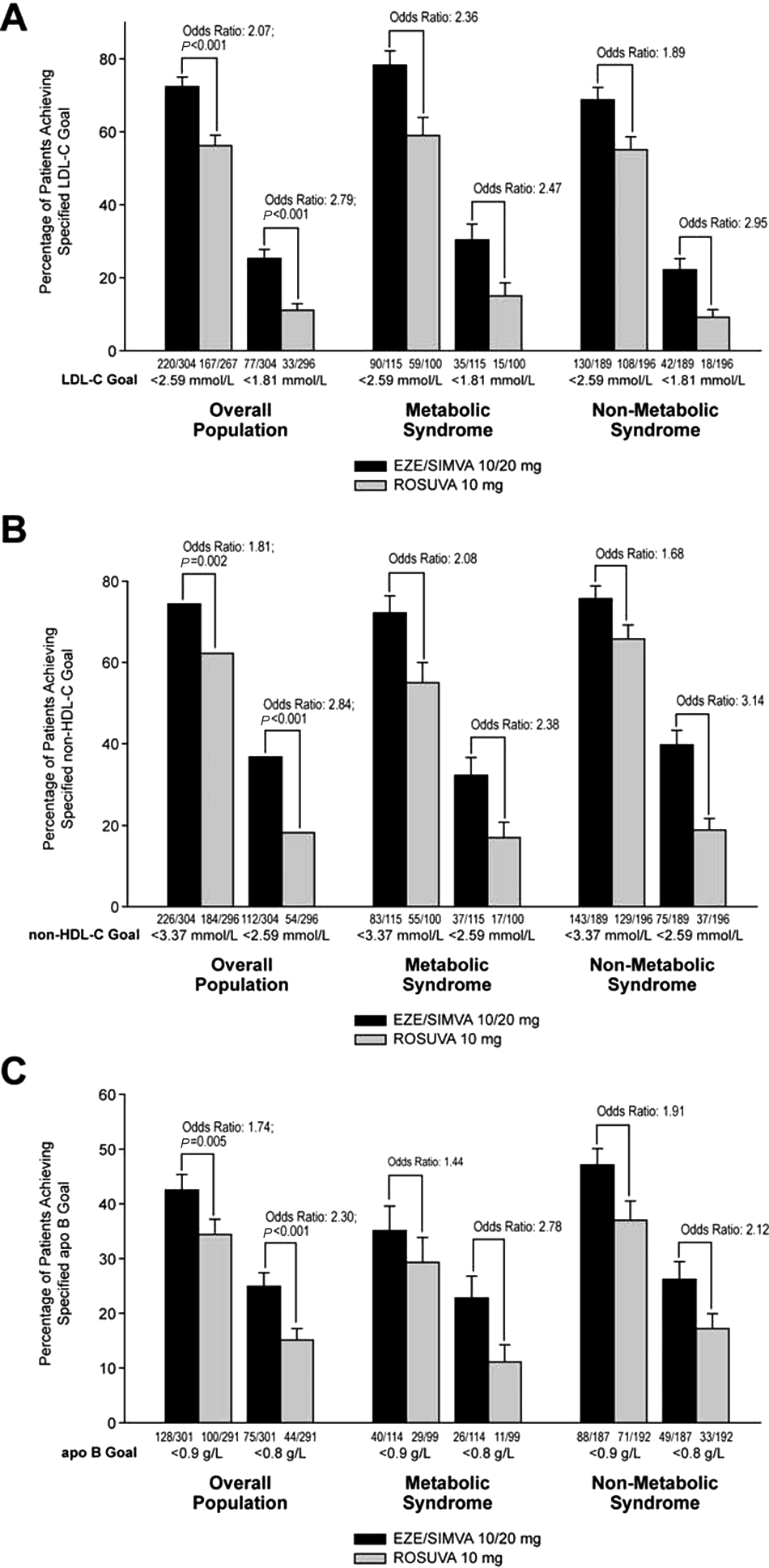
Percentages of patients who achieved (A) LDL-C levels <2.59 mmol/L and <1.81 mmol/L; (B) non-HDL-C goals <3.37 mmol/L and <2.59 mmol/L, and (C) apo B goals <0.9 g/L and <0.8 g/L at study endpoint for the overall analysis population and within patient subgroups defined by presence/absence of MetS.
Treatment with ezetimibe/simvastatin or rosuvastatin was generally well tolerated in patients in both subgroups Table 2). The incidences and types of clinical AEs were generally consistent across the patient subgroups and treatments, except for the finding of an approximate 1.5-fold increased incidence in overall clinical AEs in patients in both subgroups receiving treatment with rosuvastatin. No meaningful differences were seen between the two patient subgroups with respect to the incidences of hepatitis-related, gallbladder-related, gastrointestinal-related, or allergic reaction AEs. Presumed consecutive elevations in ALT and/or AST values ≥3×ULN were observed in one patient receiving ezetimibe/simvastatin in each of the patient subgroups; no such elevations were seen in patients taking rosuvastatin in either subgroup. There were no reports of creatine kinase elevations ≥10×ULN with any treatment in this study.
Summary of adverse events
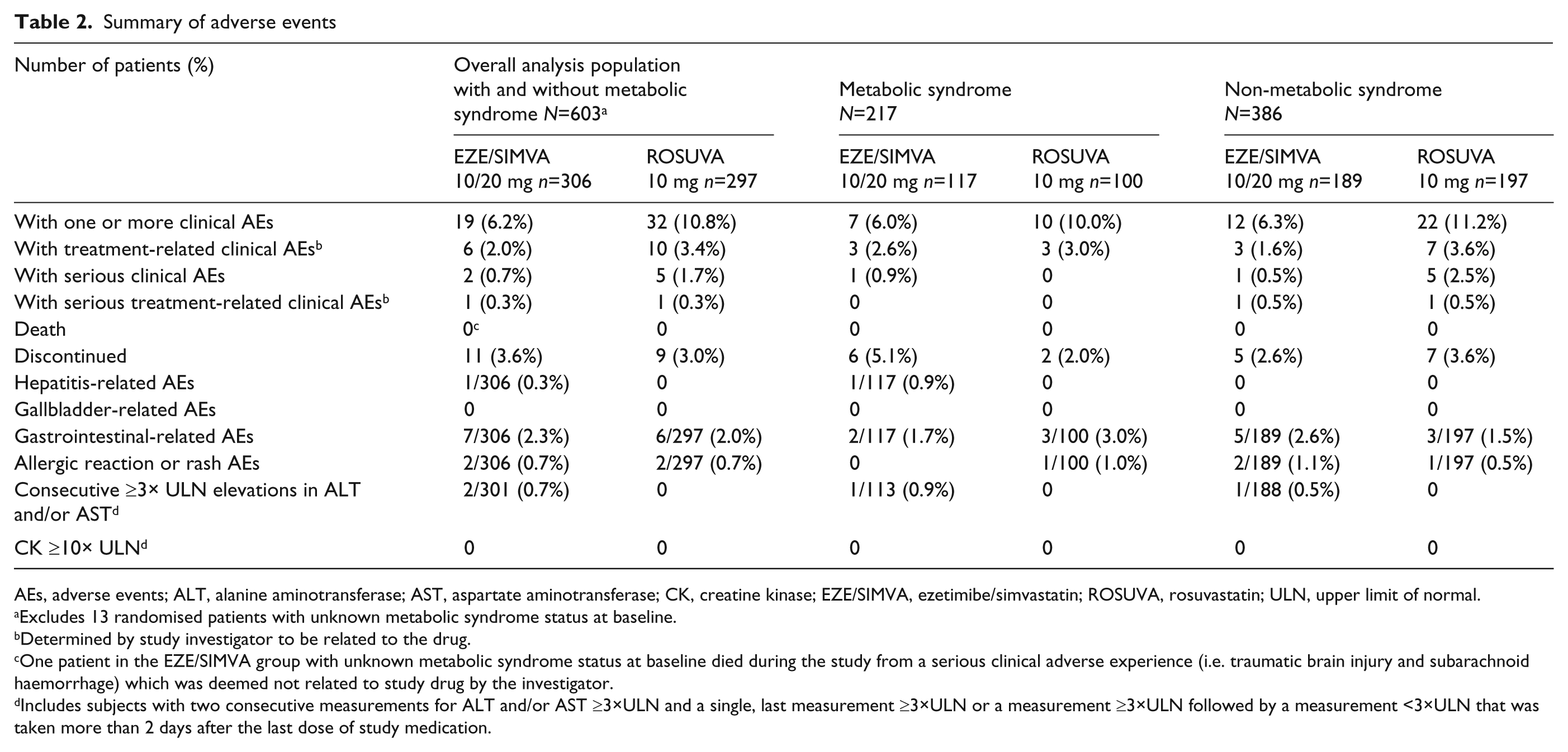
AEs, adverse events; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CK, creatine kinase; EZE/SIMVA, ezetimibe/simvastatin; ROSUVA, rosuvastatin; ULN, upper limit of normal.
Excludes 13 randomised patients with unknown metabolic syndrome status at baseline.
Determined by study investigator to be related to the drug.
One patient in the EZE/SIMVA group with unknown metabolic syndrome status at baseline died during the study from a serious clinical adverse experience (i.e. traumatic brain injury and subarachnoid haemorrhage) which was deemed not related to study drug by the investigator.
Includes subjects with two consecutive measurements for ALT and/or AST ≥3×ULN and a single, last measurement ≥3×ULN or a measurement ≥3×ULN followed by a measurement <3×ULN that was taken more than 2 days after the last dose of study medication.
Discussion
In this population of hypercholesterolaemic patients at high cardiovascular risk who failed to reach their minimum recommended LDL-C level of <4.14 mmol/L while taking statin monotherapy, switching to ezetimibe/simvastatin 10/20 mg resulted in greater improvements in most lipids and lipid ratios compared with rosuvastatin 10 mg among patient subgroups defined by the presence or absence of MetS. No interactions by subgroup were observed in this analysis, suggesting that the magnitude and direction of the treatment effects were similar both in patients with and without MetS. In addition, similar magnitude changes from baseline in results for HDL-C were observed for patients in both subgroups treated with either ezetimibe/simvastatin 10/20 mg or with rosuvastatin 10 mg. No significant within- or between-group changes from baseline in hs-CRP were seen with either treatment within the two patient subgroups. The results of this analysis in patients with and without MetS are consistent with previous reports showing the consistency in the lipid- and lipoprotein-lowering effects of ezetimibe/simvastatin therapy in diabetic and MetS patients.7-10,13-15
The patients with MetS included in this analysis presented with lipid/lipoprotein and biochemical abnormalities consistent with those reported in previous studies of patients with MetS. The patients with MetS in this analysis had the expected characteristics of atherogenic dyslipidaemia at baseline, namely elevated TG levels and low HDL-C levels. In addition to dyslipidaemia, the cohort of patients with MetS had elevated hs-CRP levels relative to non-MetS patients, consistent with the known proinflammatory state of MetS. Of note, the baseline LDL-C and apo B values seen in this population of patients with MetS were similar to those observed for non-MetS patients. This study selectively enrolled patients with hypercholesterolaemia (i.e. having LDL-C values from 2.59–4.14 mmol/L) despite prior treatment with a stable statin dose for at least 6 weeks. In general, patients with MetS often have relatively normal LDL-C levels in the face of high LDL particle concentrations (i.e. high apo B levels) due to the enrichment of small, dense, cholesterol-poor LDL particles.16,20,21
Lowering LDL-C is the primary aim of lipid-lowering therapy in patients with and without MetS. Irrespective of the presence/absence of MetS, the reduction in LDL-C seen in the present study following treatment with ezetimibe/simvastatin 10/20 mg significantly exceeded that seen for rosuvastatin 10 mg at study end. The enhanced LDL-C-lowering efficacy of ezetimibe/simvastatin resulted in clinically significant increases in the percentages of patients with and without MetS achieving recommended LDL-C levels of <2.59 and <1.81 mmol/L. Recent literature suggests that non-HDL-C and apo B are more accurate measures of circulating LDL particle concentrations and hence are more reliable indicators of CHD risk, especially among patients with diabetes and/or MetS.21-24 In the current study, treatment with ezetimibe/simvastatin 10/20mg produced significantly greater reductions than did rosuvastatin 10 mg in non-HDL-C and apo B among patients in both subgroups. Furthermore, a significantly higher percentage of patients in both subgroups achieved non-HDL-C (<3.37 and <2.59 mmol/L) and apo B (<0.9 and <0.8 g/L) recommended levels with ezetimibe/simvastatin versus rosuvastatin.
The overall safety and tolerability profile of ezetimibe/simvastatin was similar to that seen with rosuvastatin in patients with and without MetS. There was no evidence of a clinically meaningful difference in the incidences of AE, including those related to muscle or liver toxicity in patients with or without MetS who were taking ezetimibe/simvastatin or rosuvastatin.
These collective findings suggest that combination therapy with the minimum recommended starting dose of ezetimibe/simvastatin (i.e. 10/20 mg) may allow more MetS and non-MetS patients to achieve their recommended levels of LDL-C, non-HDL-C, and apo B compared with the minimum recommended starting dose of rosuvastatin monotherapy (i.e. 10 mg) without increased safety/tolerability concerns. Further study is needed to determine whether the enhanced LDL-C-, non-HDL-C- and apo B-lowering efficacy of ezetimibe/simvastatin 10/20 mg versus rosuvastatin 10 mg therapy seen in this study of high-risk CHD patients will lead to improved coronary outcomes in patients with and without MetS.
The present analysis has several limitations that should be considered when interpreting the findings. First, this was an exploratory post-hoc analysis; therefore, the results should be viewed with caution. Due to the exploratory nature of the study, no multiplicity adjustments were made. Finally, this study was conducted in a group of high-risk hypercholesterolaemic patients with CHD who failed to achieve their minimum recommended LDL-C level (≤4.14 mmol/L) with lower-potency statin monotherapy. As a result, it is not clear whether the findings presented in this paper are transferable to drug-naïve CHD patients with MetS.
Conclusions
In this population of high-risk hypercholesterolaemic CHD patients with and without MetS, switching from statin monotherapy to ezetimibe/simvastatin 10/20 mg was more effective at reducing LDL-C, TC, non-HDL-C, and apo B than was rosuvastatin 10 mg. The enhanced efficacy of ezetimibe/simvastatin resulted in clinically significant increases in the proportions of patients with and without MetS achieving recommended LDL-C, non-HDL-C and apo B levels. The safety and tolerability profile of ezetimibe/simvastatin was similar to that seen with rosuvastatin in patients with and without MetS. Further study is needed to determine whether the enhanced LDL-C-, non-HDL-C- and apo B-lowering efficacy of ezetimibe/simvastatin 10/20 mg versus rosuvastatin 10 mg therapy seen in this study of high-risk CHD patients will lead to improved coronary outcomes in patients with and without MetS.
Footnotes
Acknowledgements
The authors thank Kathleen Newcomb of Merck, Whitehouse Station, New Jersey for assistance with preparation of this manuscript.
This work was supported by Merck/Schering-Plough Pharmaceuticals, North Wales, Pennsylvania.
Qian Dong, Arvind Shah, Amy O Johnson-Levonas, William Taggart, and Philippe Brudi are current or former employees of Merck & Co., Inc. and may own stock or hold stock options in the company. Luc Missault, Margus Viigimaa, and Maurizio Averna participated in the conduct of this study. Michel Farnier has received research grant support, honoraria, and served as consultant and/or advisory board member for several companies, including Merck and Merck/Schering-Plough Pharmaceuticals. Helena Vaverkova has received research grant support, honoraria, served as consultant, and is a Speakers’ Bureau or advisory board member for several companies, including Merck & Co., Inc. and Merck/Schering-Plough Pharmaceuticals. The authors were involved in the study design, data collection, data analysis, manuscript preparation, and publication decisions.
