Abstract
Objective:
The aim of this study was to determine whether high fitness attenuates the defects in left ventricular (LV) structure, function and triglyceride (TG) content in patients with type 2 diabetes mellitus (T2DM).
Materials and methods:
Patients (n = 74) with T2DM and ≥1 additional cardiac risk factor were recruited to participate in this cross-sectional study. Outcome measures of interest were LV structure and function by magnetic resonance imaging (MRI) and myocardial TG content by 1H-magnetic resonance spectroscopy (MRS). The primary exposure variable was cardiorespiratory fitness defined by peak oxygen consumption scaled to fat-free mass (FFM; VO2peak-FFM).
Results:
Mean age was 53.5 years; 42.9% were women and mean glycosylated haemoglobin (HbA1c) was 8.0% with the mean duration of T2DM 8.2 years. VO2peak-FFM was crudely associated with both LV end systolic (r = 0.35, p = 0.002) and diastolic volumes (r = 0.32, p = 0.004), but not with ejection fraction (r = −0.15, p = 0.206), myocardial TG (r = −0.04, p = 0.734) or early diastolic peak filling rate (PFR; r = −0.01, p = 0.887). In multiple linear regression analyses, among measures of LV structure/function, VO2peak-FFM was independently associated only with LV end-diastolic volume (EDV) (β = 1.037, p = 0.038).
Conclusion:
In individuals with T2DM at increased cardiovascular (CV) risk, cardiorespiratory fitness is not associated with LV morphology, function or myocardial TG content.
Introduction
Type 2 diabetes mellitus (T2DM) is an established risk factor for atherosclerotic cardiovascular (CV) disease and congestive heart failure. 1 The increased CV morbidity and mortality associated with T2DM is generally attributed to conventional risk factors including dyslipidemia, hypertension, dysglycaemia, obesity and inflammation. 2 More recently, our group and others revealed that T2DM is associated with excessive deposition of triglyceride (TG) in the myocardium or cardiac steatosis.3,4 In turn, steatosis is associated with left ventricular (LV) hypertrophy, 5 impaired diastolic filling dynamics, 6 as well as apoptosis contributing to LV systolic dysfunction. 7 The factors that contribute to ectopic deposition of lipids within the myocardium and the associated changes in LV structure and function remain poorly understood.
Low cardiorespiratory fitness is a robust determinant of CV disease morbidity and mortality,8,9 independent of chronic disease status. 10 Among individuals with T2DM, low cardiorespiratory fitness is associated with an increased inflammatory state, 11 arterial stiffening 12 and reduced life expectancy. 13 Low cardiorespiratory fitness is associated with an increased risk of ectopic lipid deposition 14 and lower LV diastolic function; 15 it is possible that fitness may contribute to features of diabetic cardiomyopathy, including abnormalities of cardiac structure and function and myocardial steatosis.
In light of these observations, the main objective of this study was to determine whether cardiorespiratory fitness is associated with abnormalities of cardiac structure, function and myocardial steatosis in individuals with T2DM. Our hypothesis was that individuals with T2DM and high cardiorespiratory fitness would have lower levels of myocardial TG and enhanced LV diastolic relaxation compared with individuals with T2DM and low cardiorespiratory fitness.
Materials and methods
Study design and participants
We relied on cross-sectional data collected at baseline in 104 individuals with T2DM enrolled in a therapeutic randomized controlled trial who were selected to undergo baseline cardiac imaging with magnetic resonance imaging (MRI) and 1 H-magnetic resonance spectroscopy (MRS). 16 Participants were recruited from outpatient clinics, research study rosters and public advertisement. Inclusion criteria were age >18 years, medically treated T2DM (previous diagnosis of T2DM or current use of hypoglycaemic medical therapy) and the presence of ≥1 other CV disease risk factors, including smoking, hypertension (≥140/90 mmHg), dyslipidemia (treated, physician diagnosed or self-reported), albuminuria, family history of coronary artery disease or high-sensitivity C-reactive protein >3 mg/L. Exclusion criteria included atherosclerotic disease (i.e. coronary artery disease determined by clinical history and confirmed with an exercise stress test to exhaustion with a 12-lead electrocardiogram (ECG)), stroke, transient ischaemic attack, carotid or peripheral atherosclerosis, determined from patient history, the use of thiazolidinedione treatment within the prior 6 months, prior thiazolidinedione intolerance, prior heart failure, aspartate transaminase or alanine transaminase >3 times the upper limit of normal or inability to perform treadmill exercise. The University of Texas Southwestern Medical Center Institutional Review Board approved the study. All participants provided written informed consent.
Outcome measures
The primary outcome measure for the present analyses was myocardial TG content, measured non-invasively with 1 H-MRS and expressed as ratio fat/water (%), as previously described.4,16 Spectroscopy data were collected at end expiration and end systole, triggered by simultaneous respiratory and ECG gating. Spectra were collected using a whole body coil and a 17-cm surface coil for signal reception. Point RESolved Spectroscopy (PRESS) was used for spatial localization and signal acquisition. The interpulse delay TR was 4 s and the spin echo time (Te) was 25 ms. Data points (1024) were collected over a 1000-Hz spectral width and averaged >64 spectra. Areas under the curve for resonances of water and methylene groups in fatty acid chains were evaluated with line-fit procedure and commercial software (NUTS; Acorn NMR Inc., Freemont, CA, USA). Signal decay due to a spin–spin relaxation was calculated from a monoexponential relaxation equation 17 using a mean T2 relaxation time of 40 ms for water and 78 ms for myocardial TG.
The secondary outcome measures included LV filling dynamics, ejection fraction, LV volumes and LV mass quantified using standard cardiac MRI techniques as previously described. 16 Briefly dynamic images were used to quantify LV diastolic and systolic volume using short-axis slices. LV mass was computed as the product of LV end-diastolic volume (EDV) and myocardial density (1.05 g/mL), divided by body surface area. All MRI and spectroscopy measures were obtained using a whole body 1.5 T Gyroscan Intera clinical scanner (Philips Medical Systems, Cleveland, OH, USA) equipped with a spectroscopy package and were analysed by the same technician.18,19 Systolic function was estimated by ejection fraction and as we did not perform ultrasound in this study, diastolic function was estimated by early LV diastolic peak filling rate (PFR), from MRI cine images, rather than transmitral flow velocities or tissue Doppler-based estimates of tissue relaxation rates.
Primary exposure variable
The primary exposure variable was cardiorespiratory fitness, measured as peak oxygen uptake (VO2peak-FFM) determined with a graded treadmill exercise test to exhaustion.20,21 To determine treadmill speed for each participant, a warm-up phase prior to the cardiopulmonary exercise testing was performed. Each participant walked on the treadmill at an initial workload of 1.7 mile/h and 0% grade. Treadmill speed was gradually increased until the participant attained a comfortable stride. Thereafter, further adjustments in speed were made until 60%–70% of the participant’s age-predicted maximal heart rate was achieved and/or until the participant reported a rate of perceived exertion of 11–13 using the Borg scale. 22 The participant walked for 4 min at the workload that elicited 60%–70% of the participant’s age-predicted maximal heart rate to become familiar with the pace/effort. Following 5–10 min of rest, the graded treadmill test was performed at the workload needed to elicit 60%–70% of the participant’s age-predicted maximal heart rate while the grade was increased by 2% every 2 min until volitional exhaustion. Expired gases and volumes were quantified breath-by-breath using the MedGraphicsTM Cardiopulmonary Exercise System (CPX/D; Medical Graphics Corporation, St. Paul, MN, USA), at rest and throughout the treadmill exercise test. Maximal values for each of the gas exchange variables were calculated as the average of the two highest consecutive 30-s values for that variable. VO2peak was scaled to fat-free mass (FFM; VO2peak-FFM) to control for the potential confounding effect of adiposity on the outcome measures. Additional measures collected at the end of the graded maximal exercise test included the respiratory exchange ratio (RER) = [(rate of CO2 expired)/(rate of O2 consumed)], heart rate and the ratio of minute ventilation to oxygen to determine whether the effort met criteria for a maximal exercise test.
Confounding variables
Anthropometric measures and body composition
Body weight was measured to the nearest 0.1 kg on a calibrated scale. Waist circumference was measured to the nearest 0.1 cm with a flexible tape measure parallel to floor at 1 cm above the iliac crest at end expiration. Height was obtained with a standard stadiometer. Body mass index (BMI; kg/m2) was calculated using the standard formula: weight (kg)/height (m2). Skin-fold thickness was measured with calipers prior to each exercise test from four locations in duplicate and averaged, and percent body fat was calculated by the method of Durnin and Womersley. 23
Metabolic profile
The following cardiometabolic risk factors were measured after a 10-h overnight fast using standard clinical lab methods: serum glucose, TGs, high-density lipoprotein (HDL)-cholesterol and urine analysis for albumin to creatinine ratio. Plasma and serum samples were multiply aliquoted and frozen at −70 °C for subsequent analyses, including but not limited to assessment of B-type natriuretic peptide (BNP) using a commercially available assay (Biosite, Inc., San Diego, CA, USA), while glycosylated haemoglobin (HbA1c) was measured on fresh plasma samples. Resting systolic blood pressure (BP) and diastolic BP were measured in triplicate in a sitting position using a Welch Allyn series 52,000 oscillometric automated BP machine, as recommended by the National Committee on Preventive, Detection, Evaluation and Treatment of High BP. 24 The mean of the three different measures was used in the analysis.
Race/ethnicity and smoking status
Race/ethnicity and smoking status (non-smoker, former smoker or current smoker) were self-reported by the participant during the medical interview.
Statistical analysis
Data are presented as mean ± standard deviation for continuous variables and as n (%) for categorical variables. Bivariate Spearman’s correlations were performed to investigate the associations between the exposure variable and primary and secondary outcome measures. Spearman’s correlations were used because the primary and the secondary outcome measures were not normally distributed. Log transformations were performed to normalize the outcome measures for all other group-wise regression analyses. To investigate the independent association between cardiorespiratory fitness and myocardial TG content, LV morphology and function, two strategies were used. First, multiple linear regression models were used to investigate the continuous association between cardiorespiratory fitness and myocardial TG content, LV structure and LV function independent of confounders. Second, the cohort was divided into data-derived tertiles based on VO2peak-FFM and were used in multiple-regression models using the lowest tertile as the reference group. Statistical analyses were adjusted for age, sex, race/ethnicity, smoking status, systolic BP, dyslipidemia, BMI, diabetes duration and insulin use. Multiple linear regression models forcing all the confounding variables were used based on reported influence of each on the LV structure/function metrics assessed. All analyses were performed with SAS software (SAS Version 9.1.3; Cary, NC, USA) using two-tailed tests at a significance level of 0.05, with no corrections made for multiple comparisons.
Results
General characteristics of the study population
Of the 104 study participants volunteering to undergo cardiac MRI/MRS imaging, 74 participants completed all phases of the imaging protocol with valid measures of LV filling and myocardial TG content. Baseline characteristics for these 74 participants are provided in Table 1 stratified by VO2peak-FFM tertile. On average, participants were 53.5 ± 9.3 years of age, mostly men (58%), obese (average BMI = 34 ± 6 kg/m2), with modest glycaemic control (HbA1c = 8.0% ± 2%) and with T2DM duration of 8 ± 6 years. No trends were observed in crude analyses between general characteristics across tertiles of VO2peak-FFM, with the exception of BMI (p = 0.03). Approximately 35% of participants (26/74) did not reach an RER of 1.1.
General characteristics overall and stratified by cardiorespiratory fitness tertiles.
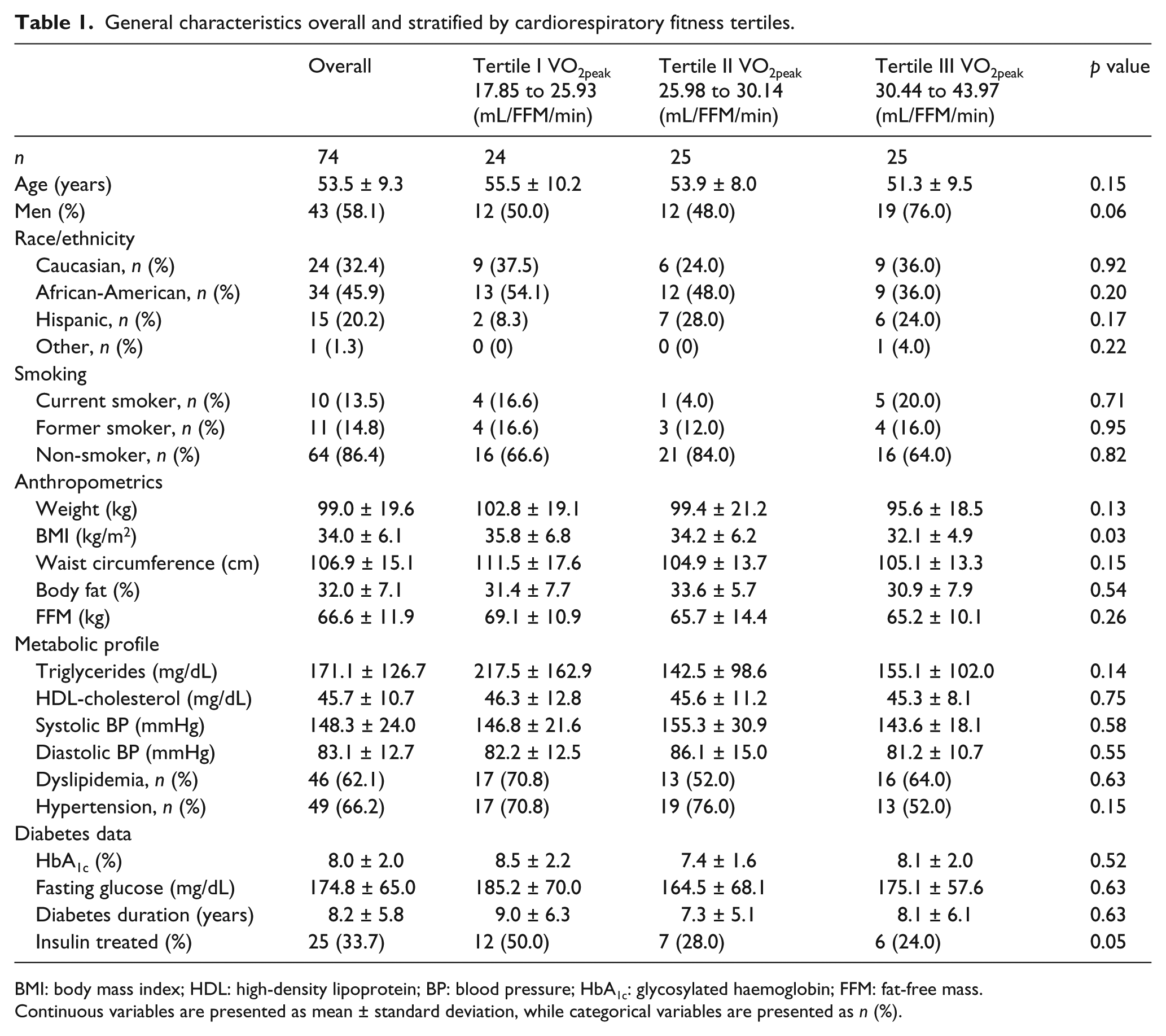
BMI: body mass index; HDL: high-density lipoprotein; BP: blood pressure; HbA1c: glycosylated haemoglobin; FFM: fat-free mass.
Continuous variables are presented as mean ± standard deviation, while categorical variables are presented as n (%).
LV structure, function and TG content across tertiles of fitness are presented in Table 2. Overall, average LV mass index (80.9 ± 20.7 g/m3), ejection fraction (65.2% ± 9.6%) and LV PFRs (68.7 ± 38.5 mL/s) were within normal ranges, and myocardial TG content was similar to previously reported values for patients with T2DM (0.95% ± 0.5%). Significant crude trends across fitness tertiles were observed for LVEDV (ptrend = 0.002), LV end-systolic volume (ESV; ptrend = 0.01) and ratio of LVPFR:LVEDV (ptrend = 0.01), while no trends were observed for myocardial TG content (p = 0.97) or ejection fraction (ptrend = 0.33). No differences in maximal RER were observed across tertiles of fitness (ptrend = 0.61).
Characteristics of LV structure, function and myocardial triglyceride content overall and stratified by cardiorespiratory fitness tertiles.
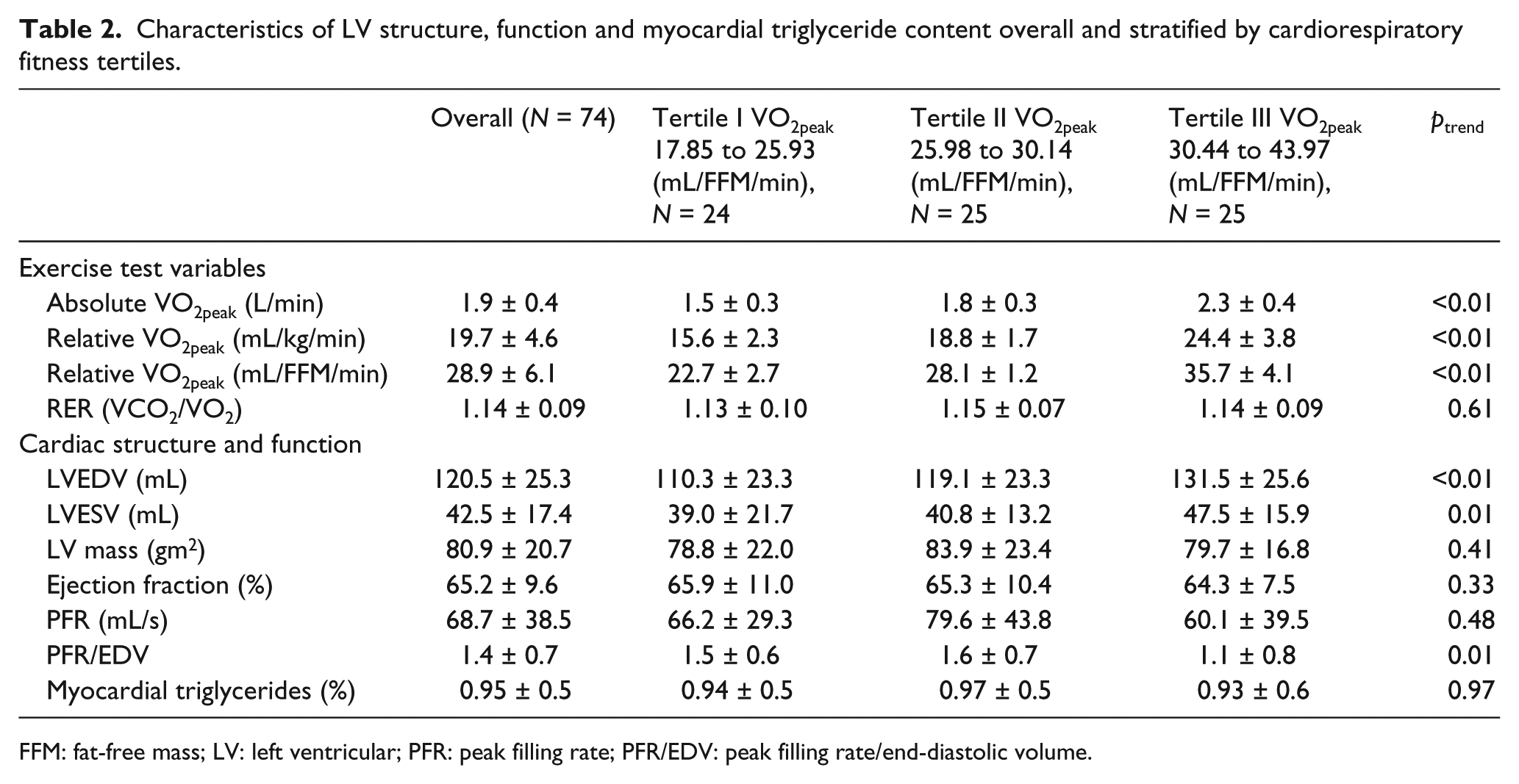
FFM: fat-free mass; LV: left ventricular; PFR: peak filling rate; PFR/EDV: peak filling rate/end-diastolic volume.
Bivariate associations between continuous VO2peak-FFM and various measures of LV structure and function are presented in Table 3. LVEDV (r = 0.35, p = 0.002), LVESV (r = 0.32, p = 0.004) and the ratio of LVPFR:LVEDV (r = −0.24, p = 0.037) were significantly associated with VO2peak-FFM, while ejection fraction (r = −0.14, p = 0.206), absolute LV PFR (r = −0.01, p = 0.887) and myocardial TG (r = −0.04, p = 0.734) were not associated with VO2peak-FFM. In addition, RER was positively correlated with myocardial TG (r = 035, p = 0.006), while no such association was observed with any other outcome variables.
Association between body composition, metabolic variables, myocardial steatosis, left ventricular structure and function.
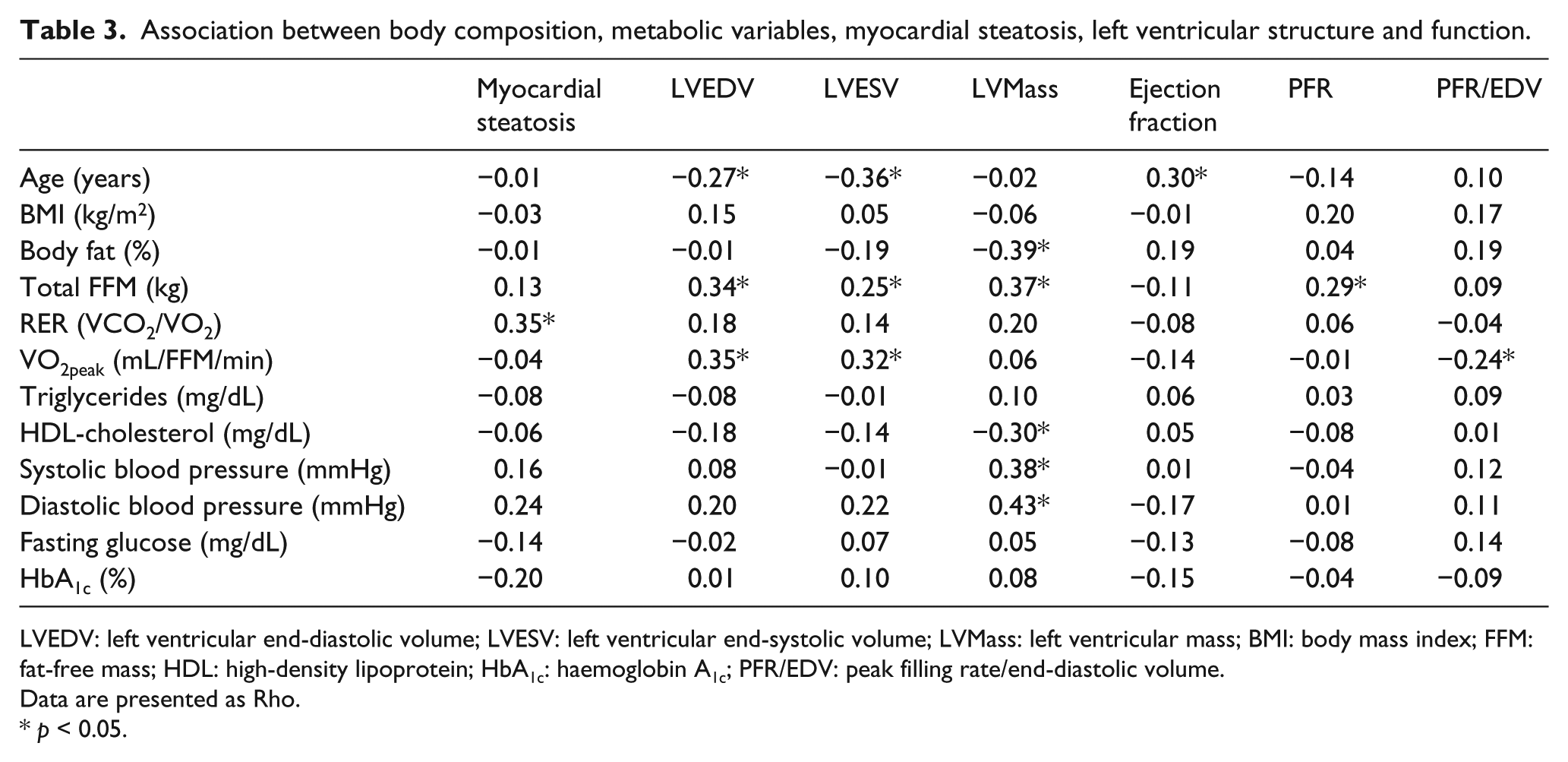
LVEDV: left ventricular end-diastolic volume; LVESV: left ventricular end-systolic volume; LVMass: left ventricular mass; BMI: body mass index; FFM: fat-free mass; HDL: high-density lipoprotein; HbA1c: haemoglobin A1c; PFR/EDV: peak filling rate/end-diastolic volume.
Data are presented as Rho.
p < 0.05.
Results from a series of multivariable regression models are presented in Table 4. After adjusting for all selected confounding variables, VO2peak-FFM was positively and independently associated only with LVEDV (β = 1.037, p = 0.038). LVESV (β = 0.306, p = 0.410), LV mass (β = −0.648, p = 0.080), ejection fraction (β = −0.075, p = 0.723), LV PFR (β = 0.477, p = 0.562), the ratio of LV PFR (β = −0.011, p = 0.482) and myocardial TG (β = −0.027, p = 0.128) were not independently associated with VO2peak-FFM.
Independent association between VO2peak-FFM and measures of cardiac structure and function.
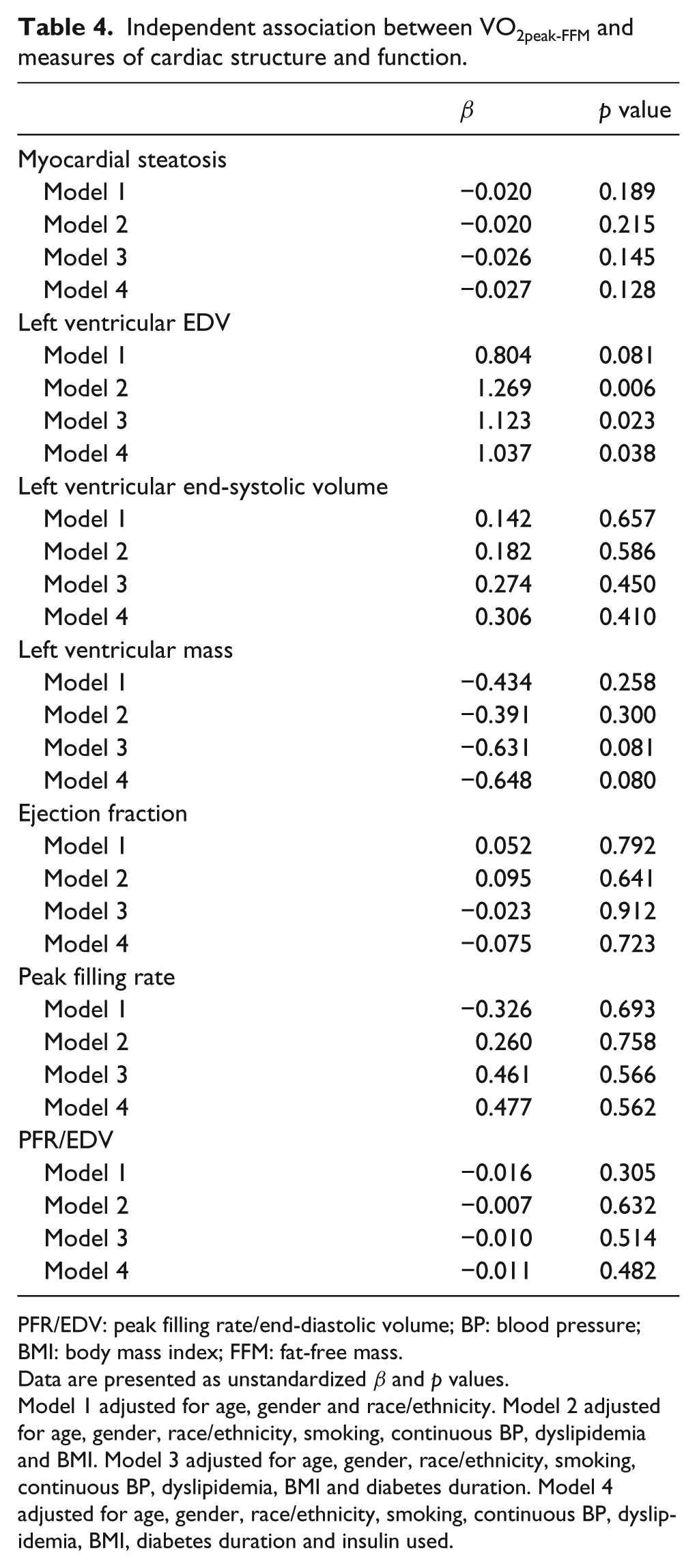
PFR/EDV: peak filling rate/end-diastolic volume; BP: blood pressure; BMI: body mass index; FFM: fat-free mass.
Data are presented as unstandardized β and p values.
Model 1 adjusted for age, gender and race/ethnicity. Model 2 adjusted for age, gender, race/ethnicity, smoking, continuous BP, dyslipidemia and BMI. Model 3 adjusted for age, gender, race/ethnicity, smoking, continuous BP, dyslipidemia, BMI and diabetes duration. Model 4 adjusted for age, gender, race/ethnicity, smoking, continuous BP, dyslipidemia, BMI, diabetes duration and insulin used.
Multiple linear regression models comparing measures of LV structure, function and TG content across tertiles of fitness after adjusting for confounding are presented in Table 5. Compared with patients in the highest tertile of VO2peak-FFM, LVEDV (β = −14.85, p = 0.032) was significantly lower in patients within the lowest tertile of VO2peak-FFM, after adjusting for confounders. Tertiles of VO2peak-FFM were not independently associated with any other measure of cardiac structure, function or myocardial TG content (p > 0.05).
Independent association between myocardial triglyceride content, hearth structure and function with tertiles of cardiorespiratory fitness.
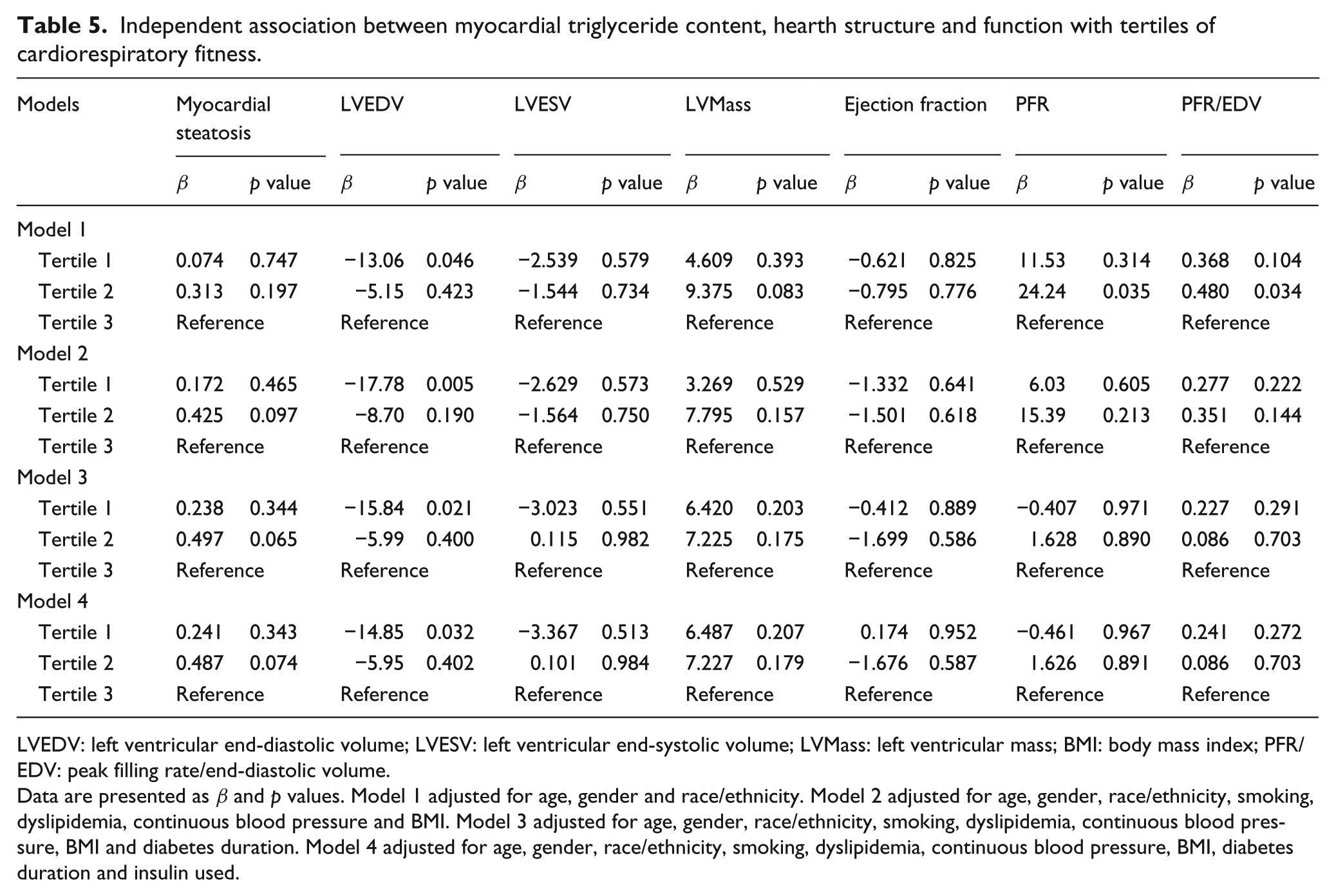
LVEDV: left ventricular end-diastolic volume; LVESV: left ventricular end-systolic volume; LVMass: left ventricular mass; BMI: body mass index; PFR/EDV: peak filling rate/end-diastolic volume.
Data are presented as β and p values. Model 1 adjusted for age, gender and race/ethnicity. Model 2 adjusted for age, gender, race/ethnicity, smoking, dyslipidemia, continuous blood pressure and BMI. Model 3 adjusted for age, gender, race/ethnicity, smoking, dyslipidemia, continuous blood pressure, BMI and diabetes duration. Model 4 adjusted for age, gender, race/ethnicity, smoking, dyslipidemia, continuous blood pressure, BMI, diabetes duration and insulin used.
Conclusion
The results from this cross-sectional study reveal that among individuals with T2DM with prevalent or increased risk of atherosclerotic CV disease, cardiorespiratory fitness is not a determinant of myocardial structure, function or TG content. In contrast with other studies,25,26 we did not find an association between LV diastolic performance and fitness determined by VO2peak-FFM. Furthermore, we did not observe an association between fitness and myocardial TG content or LV mass.
Fitness, cardiac structure and function in T2DM
Cardiorespiratory fitness is an important modifiable prognostic indicator for CV disease-related and all-cause mortality.8–10,13 The association between fitness and mortality is evident across several chronic diseases, including T2DM.13,27 Individuals with high fitness levels for their age and sex have a significantly reduced risk of premature mortality from all causes, including T2DM.28,29 The mechanisms to explain the protective effect of high fitness against CV disease, especially in patients with T2DM, remain poorly understood. There is some evidence that this protection may be mediated through enhanced cardiac performance and vascular function.12,28,30 For example, chronic exercise training interventions that lead to 9%–13% improvements in peak oxygen uptake can improve both LV systolic and diastolic functions,30–33 and in some cases normalize diastolic filling in individuals with well-controlled T2DM. 30 The results of this study do not support observations from previous observational studies or experimental trials, 34 as cardiorespiratory fitness was not associated with LV filling parameters or ejection fraction in this study. The discrepancy between this and previous studies may be attributable to the very narrow range of fitness observed in this study cohort, at the lower end of the continuum (16.60–22.40 mL/kg/min), which was significantly lower than fitness levels reported in other studies (33.9–43.8 mL/kg/min).28,35 It is possible that fitness levels may be a determinant of cardiac structure, function and TG content at higher ranges of fitness, or perhaps fitness may have less influence on such measures among older patients with a longer duration of T2DM. Studies in larger samples of patients with broader ranges of fitness are needed to determine the relationship between fitness and alterations in cardiac structure and function in patients with long-standing diabetes.
Cardiac steatosis and T2DM
Studies by our group and others have demonstrated that myocardial steatosis is increased in states of impaired glucose tolerance4,36 and T2DM.3,20 The importance of this excessive myocardial accumulation of lipid has been a significant topic of debate.37–39 Some clinical studies of younger patients with newly diagnosed T2DM suggest that higher TG content in the myocardium is associated with impaired LV diastolic filling. 40 However, this is not a universal finding, as one experimental trial reported a significant improvement in cardiorespiratory fitness and cardiac function without any detectable change in cardiac steatosis in patients with T2DM. 41 To the best of our knowledge, only one other study has examined the association between fitness and myocardial TG content in humans. 36 Among older, obese, normoglycaemic women, cardiorespiratory fitness was weakly and negatively associated with myocardial TG content, independent of BMI, age, insulin sensitivity and serum TG (β = −0.27, p = 0.04). 36 This supports findings from experimental trials demonstrating that exercise training at doses sufficient to enhance fitness and cardiac function have little effect on myocardial TG content in patients with T2DM. 41 The data presented here also support the concept that a higher level of cardiorespiratory fitness is not a protective factor against cardiac steatosis in patients with long-standing T2DM. Specifically, cardiac TG content was approximately two-fold higher in the patients studied here relative to previously published values in otherwise healthy, normal weight adults. This difference was also evidence in the myocardial TG content in patients within the highest tertile of fitness and published norms from previous studies by our group and others (0.4%–0.6% vs 0.93% ± 0.60%).4,36,42 The results may again reflect the narrow range of fitness levels within this cohort or the heterogeneity of lipid deposition within the myocardium of these patients. Larger experimental exercise intervention trials restricted to patients with established myocardial steatosis are needed to determine the influence of increasing fitness on myocardial TG content.
Strengths and limitations
To the best of our knowledge, this is the first study evaluating the associations between cardiorespiratory fitness and myocardial structure, function and steatosis in patients with T2DM. The study is strengthened by the use of gold standard measures of cardiac structure, function and steatosis coupled with a direct measurement of cardiorespiratory fitness. Despite these strengths, the study suffers from several limitations that should be acknowledged. First, the small sample size limits the power to detect small but possibly clinically relevant differences in the cardiac measures assessed, challenging the external validity of the study findings. Second, the methods used to assess LV diastolic function and LV relaxation relative to preload are not as sensitive as more sophisticated methods (i.e. ultrasound-derived strain rates or cardiac MRI tagging methods), therefore we may not have been able to detect subtle abnormalities in LV diastolic performance associated with low fitness levels. Third, the cross-sectional nature of the study design limits any causal interpretations of the associations noted here. Despite these limitations, the data provide novel insight into the associations between fitness and cardiac structure, function and steatosis in an understudied segment of the population of patients with T2DM.
In conclusion, in adults with long-standing T2DM and prevalent CV disease or risk factors, cardiorespiratory fitness is not associated with LV structure, function or myocardial TG content. These data suggest that factors other than LV function and TG accumulation contribute to the cardioprotective effects of high aerobic fitness in individuals with T2DM.
Footnotes
Acknowledgements
We thank all the participants of the study as well as all staff member who collected the data.
Declaration of conflicting interests
No potential conflicts of interest relevant to this article were reported. The other authors have nothing to disclose.
Funding
We would like to thank the Manitoba Health Research Council, The Canadian Institute of Health Research and the Integrated and Mentored Pulmonary and Cardiovascular Training that supports the salary and the training of Dr Sénéchal. Dr McGavock is supported by a New Investigator Salary Award from the Canadian Institutes of Health Research. Dr McGuire has received research support from Daiichi Sankyo, Eli Lilly, Merck, Bristol-Myers Squibb, Boehringer Ingelheim, Genentech, F. Hoffmann-La Roche, Eisai, Omthera, Takeda, GlaxoSmithKline and Novo Nordisk and is a consultant for Janssen Pharmaceuticals, Boehringer Ingelheim, Regeneron, Genentech, Merck, Sanofi-Aventis and Takeda.
