Abstract

Keywords
Authors/Task Force Members: Lars Rydén (ESC Chairperson) (Sweden), Peter J. Grant (EASD Chairperson) (UK), Stefan D. Anker (Germany), Christian Berne (Sweden), Francesco Cosentino (Italy), Nicolas Danchin (France), Christi Deaton (UK), Javier Escaned (Spain), Hans-Peter Hammes (Germany), Heikki Huikuri (Finland), Michel Marre (France), Nikolaus Marx (Germany), Linda Mellbin (Sweden), Jan Ostergren (Sweden), Carlo Patrono (Italy), Petar Seferovic (Serbia), Miguel Sousa Uva (Portugal), Marja-Riita Taskinen (Finland), Michal Tendera (Poland), Jaakko Tuomilehto (Finland), Paul Valensi (France), and Jose Luis Zamorano (Spain)
ESC Committee for Practice Guidelines (CPG): Jose Luis Zamorano (Chairperson) (Spain), Stephan Achenbach (Germany), Helmut Baumgartner (Germany), Jeroen J. Bax (Netherlands), Héctor Bueno (Spain), Veronica Dean (France), Christi Deaton (UK), Çetin Erol (Turkey), Robert Fagard (Belgium), Roberto Ferrari (Italy), David Hasdai (Israel), Arno W. Hoes (Netherlands), Paulus Kirchhof (Germany), Juhani Knuuti (Finland), Philippe Kolh (Belgium), Patrizio Lancellotti (Belgium), Ales Linhart (Czech Republic), Petros Nihoyannopoulos (UK), Massimo F. Piepoli (Italy), Piotr Ponikowski (Poland), Per Anton Sirnes (Norway), Juan Luis Tamargo (Spain), Michal Tendera (Poland), Adam Torbicki (Poland), William Wijns (Belgium), Stephan Windecker (Switzerland).
Document Reviewers: Guy De Backer (Review Coordinator) (Belgium), Per Anton Sirnes (CPG Review Coordinator) (Norway), Eduardo Alegria Ezquerra (Spain), Angelo Avogaro (Italy), Lina Badimon (Spain), Elena Baranova (Russia), Helmut Baumgartner (Germany), John Betteridge (UK), Antonio Ceriello (Spain), Robert Fagard (Belgium), Christian Funck-Brentano (France), Dietrich C. Gulba (Germany), David Hasdai (Israel), Arno W. Hoes (Netherlands), John K. Kjekshus (Norway), Juhani Knuuti (Finland), Philippe Kolh (Belgium), Eli Lev (Israel), Christian Mueller (Switzerland), Ludwig Neyses (Luxembourg), Peter M. Nilsson (Sweden), Joep Perk (Sweden), Piotr Ponikowski (Poland), Željko Reiner (Croatia), Naveed Sattar (UK), Volker Schächinger (Germany), André Scheen (Belgium), Henrik Schirmer (Norway), Anna Strömberg (Sweden), Svetlana Sudzhaeva (Belarus), Juan Luis Tamargo (Spain), Margus Viigimaa (Estonia), Charalambos Vlachopoulos (Greece), and Robert G. Xuereb, (Malta).
1. Introduction
This is a summary of the second iteration of the European Society of Cardiology’s (ESC) Guidelines on the management of diabetes mellitus (DM), pre-diabetes, and cardiovascular disease (CVD) developed in collaboration with the European Association for the Study of Diabetes (EASD). These guidelines are designed to assist clinicians and other health care workers to make evidence-based management decisions. The growing awareness of the strong relationship between DM and CVD prompted these organizations to collaborate to generate guidelines relevant to their joint interests, the first of which were published in 2007.
The processes involved in generating these guidelines can be found at: http://www.escardio.org/guidelines-surveys/esc-guidelines/about/Pages/rules-writing.aspx.
EASD and ESC appointed Chairs to direct the activities of the Task Force. Its members were chosen for their particular areas of expertise. Initial editing and review of the manuscripts took place at the Task Force meetings, with systematic review and comments provided by the ESC Committee for Practice Guidelines and the EASD Panel for Overseeing Guidelines and Statements.
To complement the Guidelines, several other documents, based on the full text version, are available. Thus, besides this summary, there are also pocket Guidelines, summary slides, booklets with essential messages and an electronic version for digital applications (Smartphones etc.). These versions are all abridged; thus, if needed, one should always refer to the full text version, which is freely available on the ESC website.
2. Abnormalities of glucose metabolism and cardiovascular disease
2.1 Definition, classification, and diagnosis
The classification of DM is based on recommendations from the World Health Organization (WHO),1,2 and the American Diabetes Association (ADA; Table 1).3 –5 Glycated haemoglobin A1c (HbA1c) has been recommended as a diagnostic test for DM,6,7 but there remain concerns regarding its sensitivity in predicting DM, 8 and values <6.5% do not exclude DM that may be detected by blood glucose measurement.6,7,9
Comparison of 2006 World Health Organization (WHO) and 2003/2011 and 2012 American Diabetes Association (ADA) diagnostic criteria.
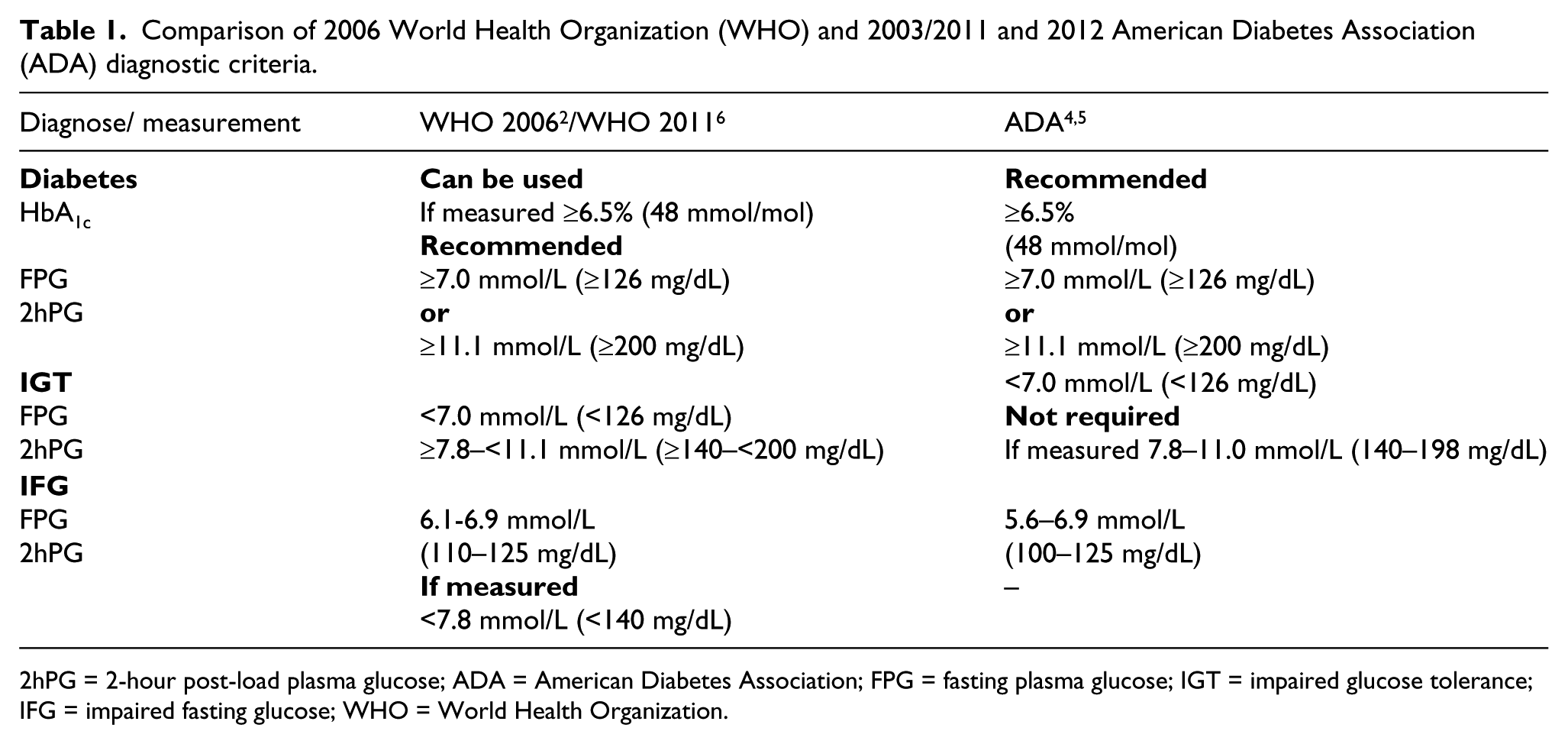
2hPG = 2-hour post-load plasma glucose; ADA = American Diabetes Association; FPG = fasting plasma glucose; IGT = impaired glucose tolerance; IFG = impaired fasting glucose; WHO = World Health Organization.
2.2 Epidemiology
The International Diabetes Federation (IDF) global estimates for 2011 suggest that 52 million Europeans aged 20–79 years have DM, and that this will increase to over 64 million by 2030. 10 A total of 281 thousand men and 317 thousand women worldwide died with DM in 2011, most from CVD. The healthcare expenditure for DM in Europe was about 75 billion Euros in 2011 and is projected to increase to 90 billion by 2030.
The diagnosis of DM is based on the level of glucose at which retinopathy occurs but macrovascular complications such as coronary, cerebrovascular and peripheral artery disease (PAD) appear earlier and are often present when type 2 diabetes mellitus (T2DM) is diagnosed using current glycaemic criteria, and >60% of people with T2DM develop CVD.
The Diabetes Epidemiology: COllaborative analysis of Diagnostic criteria in Europe (DECODE) study (Figure 1) reported data on disorders of glucose metabolism in European populations of different ages. 11 The lifetime risk for DM is 30–40% and the prevalence of impaired glucose tolerance (IGT) increases linearly from about 15% in middle age to 35–40% in elderly Europeans.
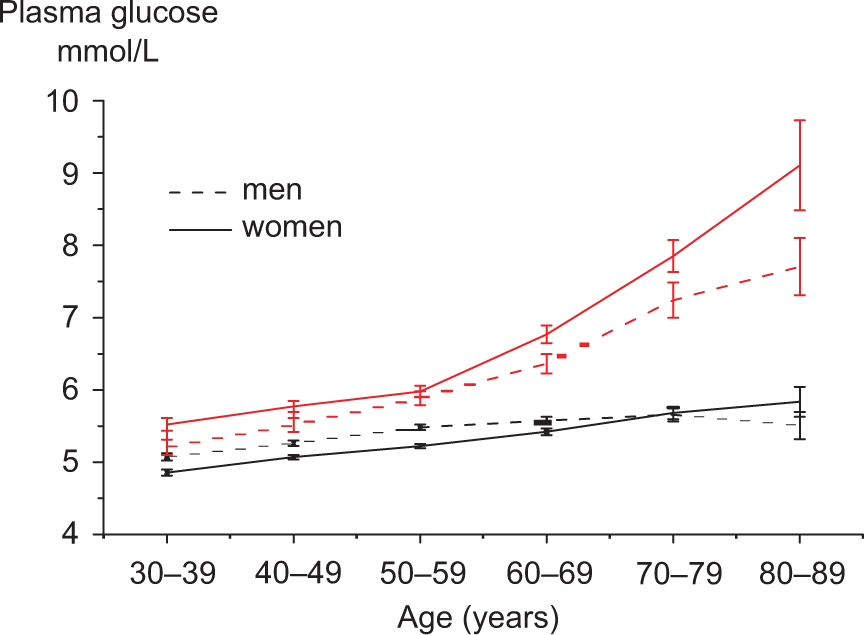
Mean FPG fasting (two lower lines) and 2hPG (two upper lines) concentrations (95% confidence intervals shown by vertical bars) in 13 European population-based cohorts included in the DECODE study. 11 Mean 2hPG increases particularly after the age of 50 years. Women have significantly higher mean 2hPG concentrations than men, a difference that becomes more pronounced above the age of 70 years. Mean FPG increases only slightly with age. FPG = fasting plasma glucose; 2hPG = 2-h post-load plasma glucose.
2.3 Screening for disorders of glucose metabolism
There is an increasing interest in identifying people with IGT, since many develop T2DM and such progress can be retarded by lifestyle interventions.12 –16 The probability of a false negative test result, compared with the oral glucose tolerance test (OGTT), is substantial when attempting to detect DM by measuring only fasting plasma glucose (FPG) and/or HbA1c. 17 Several DM risk scores have been developed, most of which perform well. 18 The FINnish Diabetes Risk SCore (FINDRISC; www.diabetes.fi/english) is the most commonly used in Europe. This tool predicts the 10-year risk of T2DM, including asymptomatic DM and IGT, with 85% accuracy.19,20 It has been validated in European populations and is available in most European languages. There are three cohorts to consider when screening: (i) the general population; (ii) people with assumed abnormalities (e.g. obese, hypertensive, or with a family history of DM) and (iii) patients with CVD. In the general population, the appropriate screening strategy is to start with a DM risk score and to investigate individuals with a high value within first-hand HbA1c and/or FPG.19,20 In CVD patients, no diabetes risk score is needed but an OGTT is indicated if HbA1c and/or FPG are inconclusive (normal), since people belonging to these groups may often have DM disclosed only by an elevated 2-hour post-load plasma glucose (2hPG). 21
2.4 Disorders of glucose metabolism and cardiovascular disease
The most convincing evidence that disorders of glucose metabolism are risk factors for CVD was provided by the European DECODE study.22 –24 Increased mortality was observed in DM and IGT but notin impaired fasting glucose (IFG). A high 2hPG predicted all-cause and CVD mortality after adjustment for other major cardiovascular risk factors, while a high FPG alone was not predictive, once 2hPG was taken into account. The highest excess CVD mortality in the population was observed in people with IGT, especially those with normal FPG. 24 The relationship between 2hPG and mortality was linear (Figure 2).
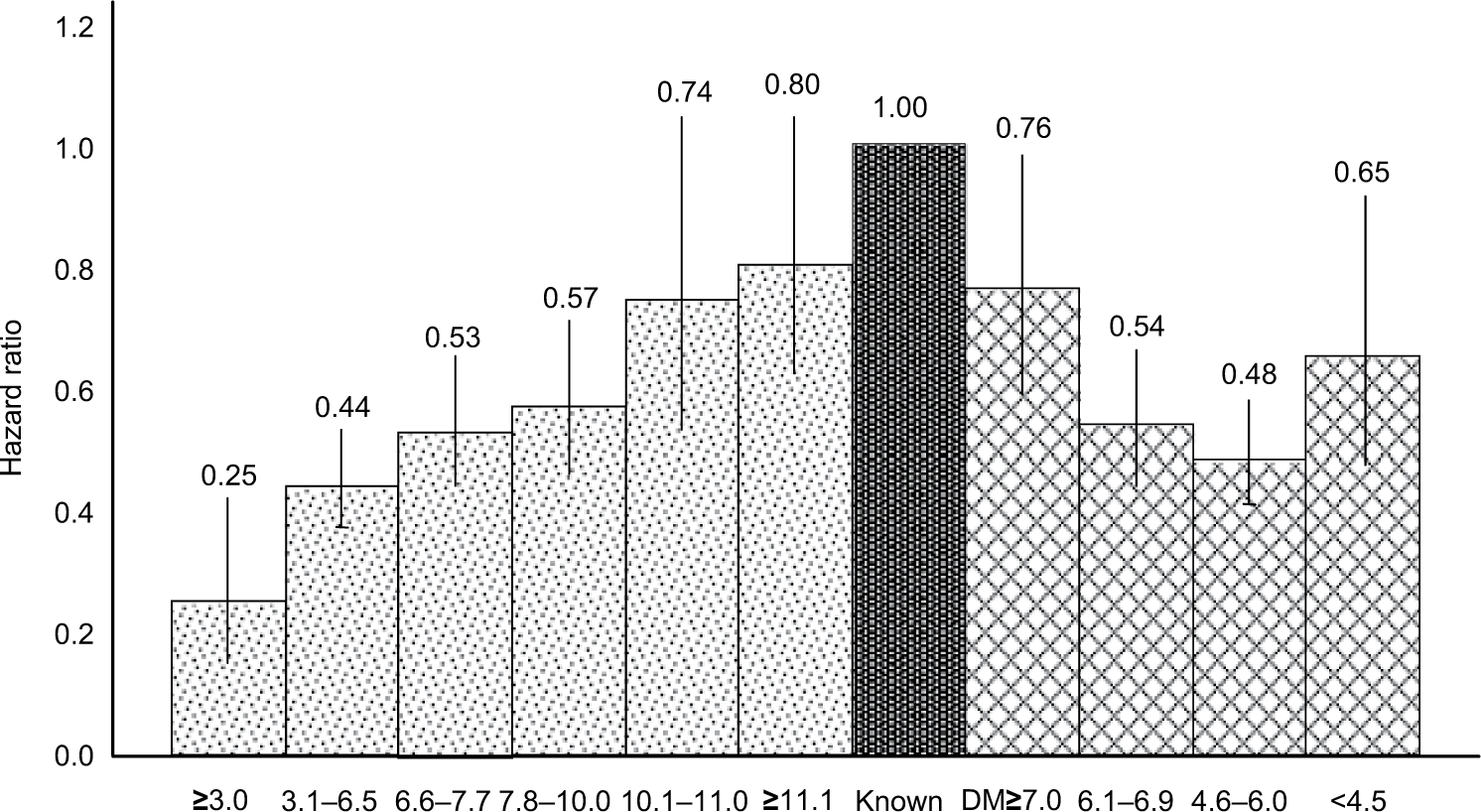
Hazard ratios and 95% confidence intervals (vertical bars) for CVD mortality for FPG (hatched bars) and 2hPG (dotted bars) intervals using previously diagnosed DM (dark bar) as the common reference category. Data are adjusted for age, sex, cohort, body mass index, systolic blood pressure, total cholesterol, and smoking.22,23
Several studies show that increasing HbA1c is associated with increasing CVD risk.25 –27 Studies that compared all three glycaemic parameters (FPG, 2hPG, and HbA1c) for mortality and CVD risk revealed that the association is strongest for 2hPG and that the risk observed with FPG and HbA1c is not significant after controlling for the effect of 2hPG.28,29
A review of the impact of gender on the occurrence of coronary artery disease (CAD) mortality reported that the overall relative risk (the ratio of risk in women to risk in men) was 1.46 [95% confidence interval (CI) 1.21–1.95] in people with DM and 2.29 (95% CI 2.05–2.55) in those without, suggesting that the well-known gender differential in CAD is reduced in DM. 30 A meta-analysis of 37 prospective cohort studies (n = 447,064 DM patients) estimated gender-related risk of fatal CAD and reported higher mortality in patients with DM than those without (5.4 vs. 1.6%, respectively). 31 The relative risk in DM was significantly greater among women (3.50) than in men (2.06). A recent study revealed a greater adverse influence of DM on adiposity, homeostatic model assessment-insulin resistance (HOMA-IR) and downstream blood pressure, lipids, endothelial dysfunction, and systemic inflammation in women than in men, which may contribute to their greater relative risk of CAD. 32 Also, it seems that women put on more weight before developing diabetes and consequently undergo bigger changes in risk factor status. 33
2.5 Delaying conversion to type 2 diabetes
Dietary habits and a sedentary lifestyle are of major significance in the development of T2DM.34,35 Randomized clinical trials (RCTs) demonstrate that lifestyle modification, based on modest weight loss and increased physical activity, prevents or delays progression in high-risk individuals with IGT. 36 People at high risk of T2DM and/or with established IGT should be given appropriate lifestyle counselling (see 4.1). 37 The absolute risk reductions are approximately 15–20 cases per 100 person-years and lifestyle intervention.
Congestive heart failure (CHF) provided to six high-risk individuals for 3 years will prevent one case of DM. 16 A 12-year follow-up of men with IGT who participated in the Malmö Feasibility Study 38 revealed that all-cause mortality among men in the lifestyle intervention group was lower (and similar to that in men with normal glucose tolerance) than that among men who had received ‘routine care’ (6.5 vs. 6.4 per 1000 person-years at risk; P = 0.009). In the Chinese Da Qing study, 39 participants with IGT in the 6-year lifestyle intervention group had, 20 years later, a persist ent reduction in the incidence of T2DM and a non-significant 17% reduction in CVD death while the adjusted incidence of severe retinopathy was 47% lower in the intervention group. 40 In the 10-year follow-up of the Finnish Diabetes Prevent on Study (DPS), total mortality and CVD incidence were no different between the intervention and control groups, but the DPS participants, who had IGT at basel ne, had lower all-cause mortality and CVD incidence compared with a Finnish population-based cohort of people w th IGT. 41
Recommendations for diagnosis of disorders of glucose metabolism.

CVD = cardiovascular disease; HbA1c = glycated haemoglobin A1c;
IGT = impaired glucose tolerance; OGTT = oral glucose tolerance test; T2DM = type 2 diabetes mellitus.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
3. Cardiovascular risk assessment in patients with dysglycaemia
3.1 General risk assessment
There are risk scores developed for people with diabetes but a more simple classification has been advocated by the 2012 Joint European Society Guidelines on CVD prevent on, 42 which advise that patients with DM and at least one other CV risk factor or target organ damage are at very high risk, and all other people with DM at high risk for developing CVD.
3.2 Risk assessment based on biomarkers and imaging
In patients with T2DM albuminuria is a risk factor for future cardiovascular (CV) events, CHF and all-cause mortality after adjusting for other risk factors, 43 and an elevated circulating N-terminal pro B-type natriuretic peptide (NT-proBNP) is a strong predictor of excess CV mortality, independent of albuminuria and conventional risk factors. 44 Coronary artery calcium (CAC) imaging is superior to established risk factor scores for predicting silent myocardial ischaemia (SMI) and short-term outcome. CAC and myocardial perfusion scintigraphy findings were synergistic for the prediction of cardiovascular events. 45 Ankle-brachial index (ABI), 46 carotid intimamedia thickness and detection of carotid plaques, 47 arterial stiffness by pulse wave velocity, 48 and cardiac autonomic neuropathy (CAN) by standard reflex tests may be considered as useful cardiovascular markers, 49 adding predictive value to the usual risk estimates. CAD is often silent in DM and up to 60% of myocardial infarction (MI) maybe asymptomatic, diagnosed by systematic electro-cardiogram (ECG) screening. 50 In asymptomatic patients, routine screening for CAD is controversial and is, for example, not recommended by the ADA, since it does not improve outcomes as long as CV risk factors are treated. 51 This position is, however, under debate and the characteristics of patients who should be screened need to be better defined. 52 Silent myocardial infarction may be detected by ECG stress test, myocardial scintigraphy or stress echocardiography. SMI affects 20–35% of DM patients who have additional risk factors, and 35–70% of patients with SMI have significant coronary stenoses on angiography. SMI is a major cardiac risk factor when associated with coronary stenoses at angiography and the predictive value of SMI and silent coronary stenoses adds to routine risk estimate. 53 Further evidence is needed to support screening for SMI, which may be carried out in those at very high risk (with evidence of PAD, high CAC score or proteinuria), and in subjects who wish to start exercise programmes. 54 In patients with SMI, coronary revascularization may be proposed on an individual basis. However the cost-effectiveness of this strategy needs evaluation.
Recommendations for cardiovascular risk assessment in diabetes.
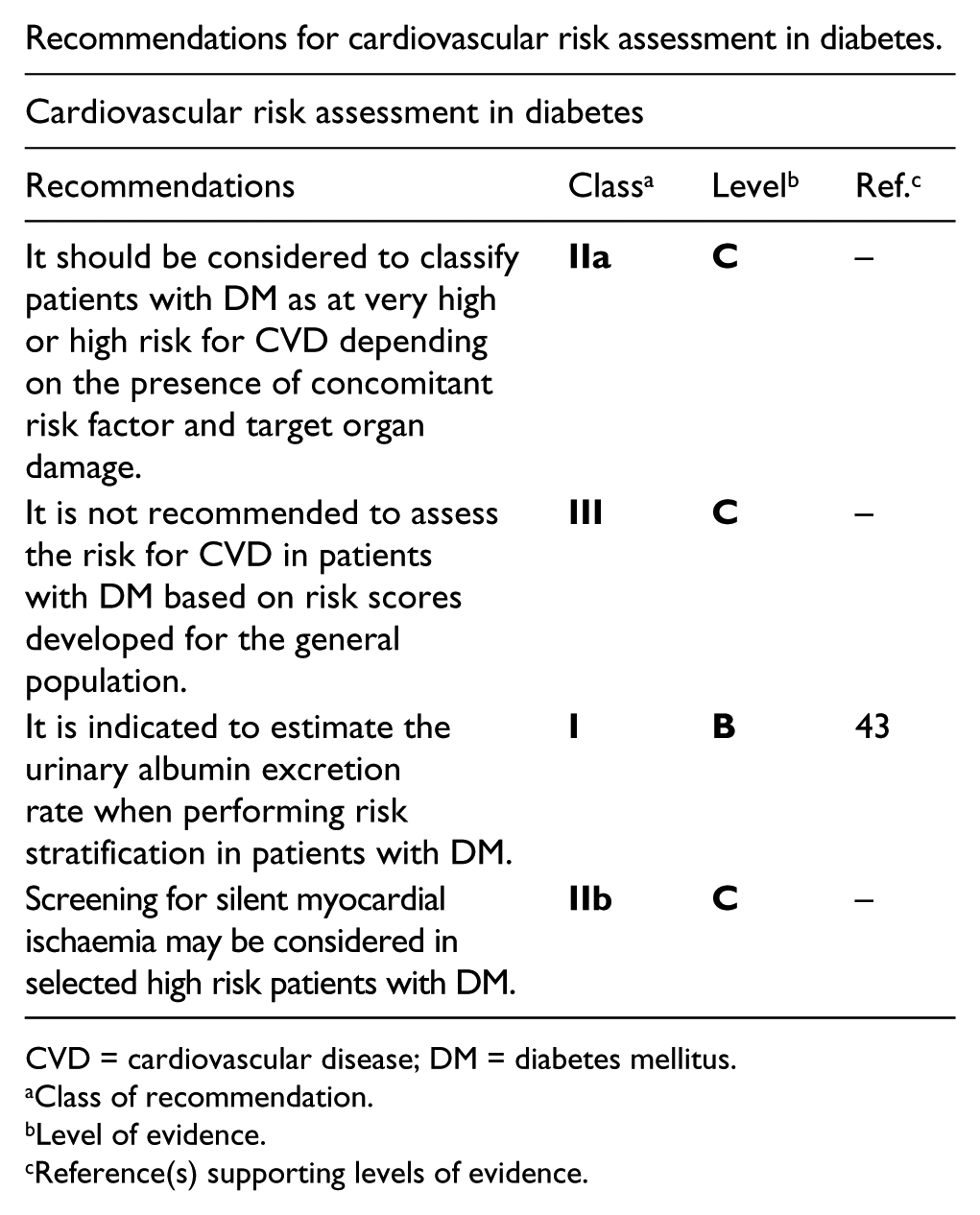
CVD = cardiovascular disease; DM = diabetes mellitus.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
4. Prevention of cardiovascular disease
4.1 Lifestyle
4.1.1 Diet
Dietary interventions recommended by the EASD Diabetes and Nuitrition Study Group are less prescriptive than earlier dietary advice, 34 but emphasise an appropriate intake of total energy and a diet in which fruits, vegetables, wholegrain cereals, and low-fat protein sources predominate. It has been suggested that there is no benefit in a high protein over a high carbohydrate diet in T2DM. 55 Specific dietary recommendations include limiting saturated and trans-fats and alcohol intake, monitoring carbohydrate consumption, and increasing dietary fibre. Routine supplementation with anti-oxidants, such as vitamins E and C and carotene, is not advised. 56 For those who prefer a higher intake of fat, a Mediterranean-type diet is acceptable, provided that fat sources are mainly derived from monounsaturated oils using virgin olive oil. 57
4.1.2 Physical activity
Aerobic and resistance training improve insulin action, plasma glucose (PG) and lipid levels, blood pressure, and card ovascular risk. 58 Regular exercise is necessary for continuing benefit. Little is known about the best way to promote physical activity; however, data from a number of RCTs support the need for reinforcement by healthcare workers.59 –61 Systematic reviews reported that structured aerobic exercise or resistance exercise reduced HbA1c by about 0.6% in T2DM.59,60 Combined aerobic and resistance training has a more favourable impact on HbA1c than aerobic or resistance trainingalone. 62 In a recent meta-analysis of 23 studies, structured exercise training was associated with a 0.7% fall in HbA1c when compared with controls. 59 Structured exercise of >150 min/week was associated with a fall in HbA1c of 0.9%; <150 min/week with a fall of 0.4%. Overall, interventions of physical activity advice were only associated with lower HbA1c levels when combined with dietary advice. 62
Recommendations on life style modifications in diabetes.

CVD = cardiovascular disease; DM = diabetes mellitus; T2DM = type 2 diabetes mellitus.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
4.1.3 Smoking Cessation
Smoking increases the risk of T2DM, 63 CVD, and premature death, 64 and smoking cessation decreases risk of CVD. 65 Current smokers with DM should be offered a structured smoking cessation programme, including pharmacological support if needed. Detailed instructions on smoking cessation are presented in the 2012 Joint European Prevention Guidelines. 42
4.2 Glucose control
Randomized controlled trials provide compelling evidence that the microvascular complications of DM are reduced by tight glycaemic control,69 –71 which also exerts a favourable—although smaller—influence on CVD, however, apparent first after many years.72,73 Intensive glucose control, combined with effective blood pressure and lipid-lowering, markedly shortens the time needed to show reductions in cardiovascular events. 74
4.2.1 Microvascular disease (retinopathy, nephropathy and neuropathy)
Retinopathy is the most frequent microvascular complication in DM. Although its incidence has declined following the implementation of intensive treatment regimens, vision-threatening proliferative retinopathy affects 50% of subjects with type 1 diabetes mellitus (T1DM), and 29% with T2DM develop vision-threatening macular oedema.75 –77 Rapidly progressive retinopathy indicates increased cardiovascular risk and the combination of retinopathy and nephropathy predicts excess morbidity and mortality; in T2DM advanced retinopathy more than doubles this risk. 78
Intensified glucose lowering, targeting an HbA1c of 6.0–7.0%, (42–53 mmol/mol), 79 has consistently been associated with decreased frequency and severity of microvascular complications. This applies to both T1DM and T2DM, although it is less apparent in T2DM with established complications.80 –84 Analyses from the Diabetes Control and Complications Trial (DCCT) and the United Kingdom Prospective Diabetes Study (UKPDS) demonstrated a continuous relationship between increasing HbA1c and microvascular complications, without an apparent threshold.85,86 In the DCCT, a decrease in HbA1c of 2% (22 mmol/mol) significantly lowered the riskofthe development and progression of retinopathy and nephropathy, 69 although the absolute reduction was low at HbA1c <7.5% (58 mmol/mol).
4.2.2 Macrovascular disease: medium-term effects of glycaemic control
Action to Control Cardiovascular Risk in Diabetes (ACCORD). A total of 10 251 T2DM subjects at high cardiovascular risk were randomized to intensive glucose control. They achieved an HbA1c of 6.4% (46 mmol/mol) or to standard treatment reaching an HbA1c of 7.5% (58 mmol/mol). 81 After a mean follow-up of 3.5 years, the study was terminated due to higher mortality in the intensive arm (14/1000 vs. 11/1000 patients/year deaths), which was pronounced in those with multiple cardiovascular risk factors and driven mainly by cardiovascular mortality. Hypoglycaemia was more common with intensive treatment and in patients with poorer glycaemic control, although the role of hypoglycaemia for the development of CVD events is not entirely clear. Further analysis revealed that the higher mortality may be due to fluctuations in glucose, in combination with an inability to control glucose to target, despite aggressive glucose-lowering treatment. 87 A follow-up of ACCORD did not support severe symptomatic hypoglycaemia as being related to higher mortality. 88
Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE). Eleven thousand, one hundred and forty T2DM subjects at high cardiovascular risk were randomized to intensive or conventional glucose-lowering therapy. 82 The intensive arm achieved an HbA1c of 6.5% (48 mmol/mol), compared with 7.3% (56 mmol/mol) in the standard arm. The primary endpoint (major macrovascular or microvascular complications) was reduced in the intensive arm [hazard ratio (HR) 0.90; 95% CI 0.82–0.98] due to a reduction in nephropathy. Intensive glycaemic control failed to influence the macrovascular component of the primary endpoint (HR0.94; 95% CI 0.84–1.06). In contrast with ACCORD, there was no increase in mortality (HR0.93; 95% CI 0.83–1.06) despite a similar decrease in HbA1c. Severe hypoglycaemia was three times lower in the intensive arm of ADVANCE, compared with ACCORD, and HbA1c lowering to target was achieved at a slower rate. In addition, the studies had a different baseline CVD risk, with a higher rate of events in the control group of ADVANCE.
Veterans Administration Diabetes Trial (VADT). One thousand, seven hundred and ninety-one T2DM patients were randomized to intensive or standard glucose control, reaching an HbA1c of 6.9% (52 mmol/mol) in the intensive-therapy group, compared with 8.4% (68 mmol/mol) in the standard-therapy group. 83 There was no significant reduction in the primary composite cardiovascular endpoint in the intensive-therapy group (HR 0.88; 95% CI 0.74–1.05).
ORIGIN (Outcome Reduction with Initial Glargine Intervention). Twelve thousand, five hundred and thirty-seven people (mean age 63.5 years) at high CVD risk plus IFG, IGT or T2DM were randomized to receive insulin glargine (with a target fasting blood glucose level of 5.3 mmol/L (≤95 mg/dL) or standard care. After follow-up of 6.2 years, CV outcomes were similar in the insulin glargine and standard care groups. Rates of severe hypoglycaemia were 1.0 vs. 0.31 per 100 person-years. Median weight increased by 1.6 kg with insulin-glargine and fell by 0.5 kg with standard care. 89
Conclusion. A meta-analysis of cardiovascular outcomes based on VADT, ACCORD and ADVANCE suggested that an HbA1c reduction of ~1% was associated with a 15% relative risk reduction (RRR) in non-fatal Ml, without benefits in terms of stroke or all-cause mortality. 90 However, patients with a short duration of T2DM, lower baseline HbA1c at randomization, and without a history of CVD seemed to benefit from intensive glucose-lowering strategies. This is supported by ORIGIN, which did not demonstrate either benefit or detriment to cardiovascular endpoints, even though insulin was associated w th ncreased hypoglycaemia. This suggests that intensive glycaemic control should be appropriately applied in an individualized manner taking into account age, duration of T2DM and history of CVD.
4.2.3 Macrovascular Disease: Long-term effects of glycaemic control
Diabetes Control and Complications Trial (DCCT) and Epidemiology of Diabetes Interventions and Complications (EDIC). In DCCT, cardiovascular events were not altered in the intensive-treatment group. 69 After termination of the study, 93% of the cohort were followed for an additional 11 years (EDIC), during which time the differences in HbA1c disappeared. 72 During the 17-year follow-up, the risk of any cardiovascular event was reduced in the intensive group by 42% (9–63%; P < 0.01).
United Kingdom Prospective Diabetes Study (UKPDS). Although a clear reduction in microvascular complications was evident, the reduction in Ml was only 16% (P = 0.052). In the extension phase, a risk reduction in MI remained at 15%, which became significant as the number of cased increased. 73 It should be noted that this study was performed when lipid-lowering and blood pressure were less-effectively managed and it may have been easier to verify a beneficial effect of glucose-lowering agents than in subsequent trials.
Conclusion. DCCT and UKPDS show that in T1DM and T2DM: (i) glycaemic control is importantto reduce long-term macrovascular complications; (ii) a very long follow-up period is required to demonstrate an effect and (iii) early glucose control is important.
4.2.4 Glycaemic targets
An HbA1c target of <7.0% (<53 mmol/mol) to reduce microvascular disease is a generally recommended.69 –71,73,81 The evidence for an HbA1c target in relation to macrovascular risk is less compelling, due to the complexities surrounding the chronic, progressive nature of DM and the effects of metabolic memory.71,73,90 Consensus indicates that an HbA1c of <7% should be targeted but with acknowledgement of individual patient requirements. Ideally, tight control should be instigated early in younger subjects without attendant co-morbidities. Successful glucose-lowering is assisted by self-monitoring of blood glucose, most notably in patients with insulin-treated DM. 91 Although postprandial hyperglycaemia is associated with an increased incidence of CVD events, it is controversial as to whether addressing this is of benefit for CVD outcomes.92 –95 More stringent targets (e.g. HbA1c 6.0–6.5% (42–48 mmol/mol]) might be considered in selected patients with short disease duration, long life expectancy, and no significant CVD, if achieved without hypoglycaemia or other adverse effects. As discussed, the accumulated results from T2DM cardiovasculartrials suggest that not everyone benefits from aggressive glucose management, and it is importantto individualize treatment targets. 96
4.2.5 Glucose-lowering agents
A detailed description of the choice of glucose-lowering agents and the role of combination therapy is beyond the scope of this document and has extensively been reviewed in the recent joint ADA/EASD guidelines. 96
Cardiovascular safety of glucose-lowering agents. The possible adverse cardiovascular effects of irosiglitazone 97 raised quesitions as to the cardiovascular safety of glucose-lowering drugs, particularly in combination. A 10-year post-trial follow-up of UKPDS revealed that patients treated with sulphonylurea–insulin had a risk ratio (RR) for Ml of 0.85 (P = 0.01) and for death of 0.87 (P < 0.007).71,73 The correspond ng RRs for metformin in overweight patients were 0.67 (P = 0.005) and 0.73 (P = 0.002). Although UKPDS indicated that metformin has a beneficial effect on CVD outcomes, there is no other clear evidence to support this view and metformin in combination with sulphonylurea may be detrimental. However, the results of this meta-analysis also suggest a benefit after a long duration of treatment in younger patients. 98 Pioglitazone reduced a secondary vascular composite in the PROactive (PROspective pioglit Azone Clinical Trial In macroVascular Events) study (HR 0.84; 95% CI 0.72–0.98; P = 0.027), 99 however, the primary outcome did not achieve significance and the interpretation of these results remains contentious. Pioglitazone is associated with fluid retention secondary to renal effects and peripheral oedoma, and worsening of established heart failure in susceptible individuals. In the STOP-NIDDM (Study to prevent non insulin-dependent diabetes) trial, acarbose in patients with IGT reduced the number of CVD events, including cardiovascular mortality. 93 Meglitinides have not been formally tested in T2DM but, in high-risk patients with IGT, nateglinide did not reduce fatal or non-fatal cardiovascular events. 100 No outcome data from RCTs have so far been published for glucagon-like peptide 1 agonists, dipeptidylpeptidase-4 (DPP-4) inhibitors, or sodium–glucose co-transporter-2 (SGLT-2) inhibitors but prospective trials are ongoing.
4.2.6 Special considerations
Hypoglycaemia. Intensive glucose-lowering increases the incidence of severe hypoglycaemia three- to four-fold in both T1DM and T2DM.69,84 Impaired hypoglycaemic awareness increases with duration of DM and is a significant risk factor for hypoglycaemia, which must be taken into account when glucose-lowering therapy is considiered. 101 In addition to the short-term risks of cardiac arrhythmia and cardiovascular events, longer-term risks include dementia and cognitive dysfunction.102,103 The outcome of glucose-lowering studies has raised the question as to whether hypoglycaemia is an important risk factor for MI in patients with DM. Frier et al. 102 have extensively reviewed this topic, providing evidence for a number of adverse effects of hypoglycaemia on the CV system, particularly in the presence of autonomic neuropathy. Insulin, meglitinides and sulphonylureas are particularly associated with hypoglycaemia, which is common in both T1 and T2DM.
Glucose-lowering agents in chronic kidney disease. Around 25% of T2DM subjects have chronic kidney disease (CKD) stages 3–4 [estimated glomerular filtration rate (eGFR)] <50 mL/min].
Recommendations for glycaemic control in diabetes.

CVD = cardiovascular disease; DM = diabetes mellitus; HbA1c = glycated haemoglobin A1c; T1DM = type 1 diabetes mellitus; T2DM = type 2 diabetes mellitus.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
Aside from the increased CV risk associated with this, glucose-lowering agents may need to be modified, either because the drug is contra-indicated in CKD or because the dosage needs to be altered. 104 Metformin, acarbose and most sulphonylureas should be avoided in stage 3–4 CKD, whilst insulin and pioglitazone can be used. The DPP-4 inhibitors require dose adjustment with progressive CKD with the exception of linagliptin, which is well tolerated in these circumstances. SGLT2 inhibitors have not been evaluated in CKD.
Elderly subjects. Glycaemic targets for elderly people with long-standing, complicated diabetes should be less ambitious than for younger, healthier individuals. If lower targets cannot be achieved, an HbA1c of <7.5–8.0% (<58–64 mmol/mol) may be acceptable, transitioning upwards as age increases and capacity for self-care, cognitive, psychological and economic status, and support systems decline. 96
Individualized care. The influences on quality of life, adverse effects of polypharmacy and inconvenience of intensified glucose-lowering regimens have to be carefully evaluated for each individual with DM. From a public health perspective, even minor decreases in mean glycaemia may prove advantageous. On the other hand, the intensified glucose-lowering treatment may impose a considerable burden and possible harm on the individual. Each individual should be encouraged to achieve the best compromise between glucose control and vascular risk and, if intensified therapy is instituted, the patients must be informed and understand the benefits and risks.
4.3 Blood pressure
Hypertension and diabetes is a common combination that causes a four-fold increase in CVD risk.105,106 Obesity, aging, and the appearance of renal disease increase the prevalence of hypertension, whilst T2DM doubles CVD risk in men and triples risk in women.
4.3.1 Treatment targets
Randomized, controlled trials in T2DM have shown the positive effects on cardiovascular outcomes of reducing BP below 140 mm Hg systolic and 85 mm Hg diastolic.107 –110 In ACCORD, the relative reduction of the composite endpoint (non-fatal MI, non-fatal stroke, or CVD death) by intensive treatment (achieved mean systolic BP 119 mm Hg) compared with standard treatment (mean systolic BP 134 mm Hg) did not reach statistical significance. 108 The proportion of patients with serious side-effects (hypotension and declining renal function) increased from 1.3 to 3.3% with aggressive treatment. Accordingly, this study does not support a reduction of systolic BP below 130 mm Hg. Bangalore et al. 111 reported a meta-analysis of 13 RCTs in patients with DM, IFG, or IGT who, in the intensive group, had a systolic BP ≤135 mm Hg and in the standard group ≤140 mm Hg. The intensive blood pressure control related to a 10% reduction in all-cause mortality (95% CI 0.83–0.98), a 17% reduction in stroke, but a 20% increase in serious adverse events. Sys tolic BP ≤130 mm Hg related to a reduction in stroke but did not affect other CVD events.
In summary, present evidence suggests that the BP target should be <140/85 mm Hg in patients with DM. A lower BP (systolic <130 mm Hg) may be considered in patients with hypertension and nephropathy with overt proteinuria. Further reduction might be associated with an increased risk of adverse events, especially with advanced age and longer duration of T2DM, and the risk-benefit of intensive BP management needs to be considered on an individual basis.
4.3.2 Managing blood pressure-lowering
Lifestyle intervention. including salt restriction and weight loss is the therapeutic basis for all patients with hypertension; however, it is usually insufficient for adequate BP control.
Pharmacological treatment has only been tested in a few RCTs comparing cardiovascular outcomes with BP-lowering agents, specially targetting patients with DM.107,112,113 However, several RCTs with sizeable DM subgroups reported specifically on the outcome in this subgroup.114 –121 Blockade of the renin-angiotensin-aldosterone system (RAAS), by an ACE-inhibitor (ACE-I) or an angiotensin-receptor blocker (ARB), is of particular value when treating hypertens on in DM at high card ovascular risk.114,115,119 –121 As a primary intervention, BP control using RAAS blockers prevents the on set of microalbuminuria in T2DM,107,109 but not in T1DM.122 –124 As a secondary intervention, intensified BP control using ACE-I slowed progression of kidney disease in T1DM and reduced end-stage renal failure.125,126 In T2DM, high doses of ramipril prevented both renal and cardiovascular outcomes. 127 ARBs reduced progression from microalbuminur a to proteinuria and prevented renal outcomes but not cardiovascular death.128,129 The DIRECT (DIabetic REtinopathy Candesartan Trials) studies investigated the effects of blood pressure-lowering with candesartan on the development and progression of retinopathy and there was a non-significant favourable trend in T1DM and T2DM.130,131
Evidence supports the use of an ACE-I, rather than a calcium channel blocker, as initial therapy to prevent or retard the occurrence of microalbuminuria in hypertensive patients with DM. 132 Dual RAAS blockade, combining an ACE-I with an ARB, did not show further benefit in the ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET) and was associated with more adverse events. In the Aliskiren Trial in Type 2 Diabetes Using Cardio-Renal Endpoints (ALTITUDE), the addition of aliskiren to RAAS-blockadein high-riskT2DM did not result in a decrease in cardiovascular events and may even have been harmful.133,134
Thiazides and beta-blockers are associated with an increased risk of developing T2DM, compared with calcium channel blockers and RAAS inhibitors, 135 but it is not known whether they result in meta bolic adverse events of clinical importance in established T2DM. A recent meta-analys is emphasized the priority of BP lowering over choice of drug. 136 In the absence of cardiac co-morbidity, beta-blockers are not first choice and appropriate BP control often requires combined therapy with a RAAS inhibitor and a calcium channel blocker or a diuretic.119,120 The Avoiding Cardiovascular Events through Combination Therapy in Patients Living with Systolic Hypertension (ACCOMPLISH) trial indicated that the calcium channel antagonist amlodipine is superior to hydrochlorothiazide in combination treatment with an ACE-I. 121
A combination of drugs is needed in most patients. All available drugs can be used but evidence strongly supports the inclusion of an inhibitor of the RAAS (ACE-I/ARB) in the presence of proteinuria. Since DM patients tend to have high BP during the night, administration of anti-hypertensive drugs at bedtime should be considered and deally after evaluat on of the 24-hour ambulatory blood pressure profile of the patient.
Recommendations for blood pressure control in diabetes.
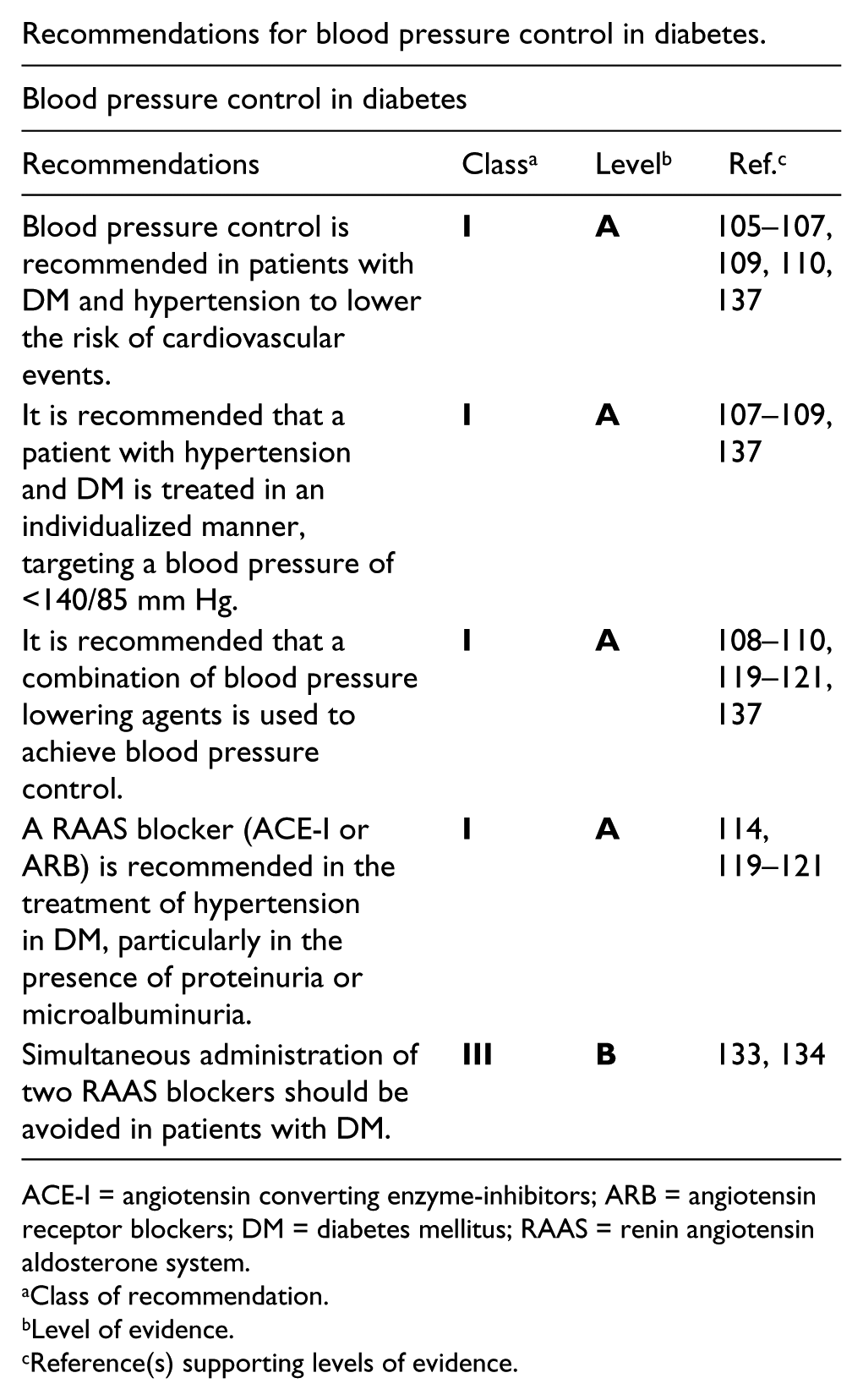
ACE-I = angiotensin converting enzyme-inhibitors; ARB = angiotensin receptor blockers; DM = diabetes mellitus; RAAS = renin angiotensin aldosterone system.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
4.4 Dyslipidaemia
In T1DM serum, triglyceride (TG) is normal and high-density lipoprotein cholesterol C (HDL-C) within the upper normal range or slightly elevated. A cluster of lipid abnormalities accompanies T2DM, the core components of which are a moderate elevation of fasting and non-fasting TGs and low HDL-C. Other features comprise elevations of TG-rich lipoprotein, including chylomicron and very low-density lipoprotein (VLDL) remnants and small dense low-density lipoprotein (LDL) particles. An imbalance between the hepatic import and export of lipids results in excess liver fat accumulation (non-alcoholic fatty liver disease), which drives the overproduction of large VLDL particles in T2DM and assoc ated hypertriglyceridaemia. Increased free fatty acid (FFA) flux comes from both the systemic FFA pools and de novo lipogenesis in the setting of insulin resistance (IR).138,139
Dyslipidaemia and macrovascular disease. A causal association exists between elevation of triglyceride rich particles, low HDL-C, and CVD risk.140,141 Data from statin trials strengthen the position of low high-density lipoprotein (HDL) as an independent CVD risk marker, even when LDL-C level is not elevated.142,143 Data from the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) and ACCORD studies demonstrated that CVD event rates were significantly higher in dyslipidaemia (LDL-C 2.6 mmol/L (100 mg/dL), TG ≥2.3 mmol/L and HDL-C ≤0.88 mmol/L).144,145
In FIELD baseline variables predicting CVD events over 5 years were lipid ratios (non-HDL–HDL-C and total–HDL-C). The power of serum TG to predict CVD events was attenuated by adjustment for HDL-C. 146 The data concur with results from the Emerging Risk Factor Collaboration (ERFC), 147 which reported that a 1SD increase in HDL-C (0.38 mmol/L or 15 mg/dL) was associated with a 22% reduction in risk of coronary heart disease.
Dyslipidaemia and microvascular disease. In FIELD, fenofibrate reduced albuminuria and slowed eGFR loss over 5 years, despite an initial, reversible increase in creatinine in T2DM. 148 Lipid-lowering does not seem to directly affect retinopathy. In FIELD, fenofibrate was associated with a reduction in laser therapy for retinopathy, although this appeared to be independent of lipid levels. ACCORD reported a reduction in progression of retinopathy [odds ratio (OR) 0.60; P < 0.0056] using combined statins and fenofibrate.
4.4.1 Management of dyslipidaemia
Type 2 diabetes mellitus: primary prevention. In the Collaborative Atorvastatin Diabetes Study (CARDS), 2838 patients were randomized to atorvastatin or placebo. 149 and the study was terminated prematurely, due to a 37% reduction (P = 0001) in the primary end-point (first acute coronary heart disease event). In the Heart Protection Study (HPS) sim-vastatin (40 mg/day) reduced the primary endpoint by 33% (P = 0.0003) 150 and in the Anglo-Scandinavian Cardiac Outcomes Trial (ASCOT) DM subgroup, atorvastatin reduced major CVD events and procedures by 23% (P = 0.04). 151
Type 2 diabetes mellitus: secondary prevention. The benefits of statin therapy in DM are seen in all subgroup analyses of major RCTs. 152 A meta-analysis of 14 RCTs, including 18 686 people with DM, reported a 9% reduction in all-cause mortality and a 21% reduction in the incidence of major vascular outcomes per mmol/L of LDL-C lowering (P < 0.0001), similar to non-DM. This was associated with absolute reduction in LDL-C and was seen at an LDL-C as low as 2.6 mmol/L. 153 Data from 10 RCTs reported that initensive statin dosage reduced the composite endpoint of CAD by 10% (P < 0.0001), but did not reduce mortality. 154 Intensive lowering of LDL-C had a beneficial effect on progression of atheroma in DM and non-DM subjects. 155
Intensification of LDL-C lowering can be achieved by adding ezetimibe to a statin. Although there are no RCT data on CVD outcome, a trial is under way (IMPROVE-IT [IMProved Reduction of Outcomes: Vytorin Efficacy International Trial]: ClinicalTrials.gov: NCT00202878). An analysis of pooled safety data comparing the efficacy and safety profile of combination therapy with ezetimibe/statin vs. statin monotherapy in DM and non-DM subjects (n = 21,794) 156 reported that the combination provided greater effects on all major lipid measures. The Study of Heart and Renal Protection (SHARP) trial reported a 17% reduction of major atherosclerotic events in CKD treated with simvastatin plus ezetimibe daily, when compared with placebo. 157 It should be emphasized that, although the relative reduction of events maybe similar for subjects with and without DM, the absolute benefit is greater in DM patients, due to their higher risk.
Type 1 diabetes mellitus. The Cholesterol Treatment Trialists (CTT) analysis of 1466 T1DM patients, most with prior CVD, reported a similar reduction in risk of CVD events (RR 0.79) to that seen in T2DM. 153 Although there are no trial data on statin use in younger T1DM, statins should be considered in those at high risk of CVD, irrespective of LDL-C levels.
Safety of statin therapy. RCTs demonstrate that statins are safe and well-tolerated. 158 Adverse events—other than aching muscles—are rare. In the majority of cases of myopathy or rhabdomyolysis there are drug interactions with a higher-than-standard dose of statin. 159 The combination of gemfibrozil and statins should be avoided, due to pharmacokinetic interaction, but there are no safety issues with fenofibrate and statins.144,145 A meta-analysis including 91,140 participants reported that statin therapy was associated with risk of new-onset T2DM (OR 1.09) 160 that translates to one case of T2DM in 255 patients treated for 4 years. Over the same period, statins would prevent 5.4 CVD events for each mmol/L reduction in LDL-C. A meta-analysis of five statin trials reported that the risk of new-onset DM increased with intensive statin (atorvastatin or simvastatin 80 mg daily) therapy (OR 1.12) compared with moderate (simvastatin 20 mg or pravastatin 40 mg) doses. 161 In the intensive group, two cases of new-onset DM per 1000 patient-years were seen, whilst CVD events were reduced by 6.5 cases. The Food and Drug Administration (FDA) approved label changes for statins (www.fda.gov/downloads/Drugs/DrugSafety/UCM293474.pdf), but emphasized that the small risk of developing DM is outweighed by the reduction in vascular events.161,162 A meta-analysis of 27 randomized trials demonstrated that, in individuals with a five-year risk of major vascular events lower than 10%, each mmol/L reduction in LDL-C produced an absolute reduction in events of 11 per 1000 over five years, without increases in cancer or deaths from other causes. This benefit greatly exceeds any risks from statin therapy. 163
Residual risk in subjects on low-density lipoprotein-lowering therapy. Type 2 DM patients at the LDL-C target remain at high risk of CVD events, 140 and targeting elevated TG (>2.2 mmol/L) and/or low HDL-C (<1.0 mmol/L) may provide further benefits. In the FIELD, fenofibrate did not reduce the primary endpoint (CAD-related death and non-fatal MI), but total CVD events were reduced from 14 to 12.5% (HR 0.9; P = 0.035).144,164 In ACCORD, patients were assigned to fenofibrate plus simvastatin (20–40 mg daily) or placebo without an additional effect on the primary endpoint. In a pre-specified subgroup analysis of subjects with TG >2.3 mmol/L (>204 mg/dL) and HDL-C <0.9 mmol/L (<34 mg/dL), cardiovascular risk was reducod 31% in the fenofibrate-plus-simuastatin group. 145 A subgroup analysis of dyslipidaemic subjects (TG >2.3 mmol/L and HDL-C <0.9 mmol/L) in the FIELD study revealed a 27% reduction in CVD risk. 144 In both FIELD and ACCORD, fenofibrate was associated with a robust (22%) reduction of TG, whereas elevation of HDL-C was less than expected (+2% and +2.4%, respectively). Meta-analyses have confirmed the clinical benefits of fibrates on major CVD events, but not on cardiovascular mortality.165,166
Strategies to elevate high-density lipoprotein cholesterol C. High-density lipoprotein cholesterol C is inversely related to CVD in epidemiological studies and in many statin trials. 218 Low levels of HDL-C are associated with increased levels of triglycerides and are often seen in patients with metabolic syndrome and/or DM. Targeting low HDL-C for CVD prevention is, however, not supported by evidence. Two recently reported RCTs, using the CETP (cholesteryl ester transfer protein) inhibitors torcetrapib and dalcetrapib,167,168 failed to reduce cardiovascular events despite a 30–40% increase in HDL-C. Fenofibrate has trivial efficacy in elevating HDL-C and, whilst niacin increases HDL-C (~15–30%), recent studies have not shown any cardiovascular benefit of niacin, 169 but have been associated with an increased risk of adverse side-effects, 170 which led to withdrawal ofthe marketing licence.
Recommendations on management of dyslipidaemia in diabetes.
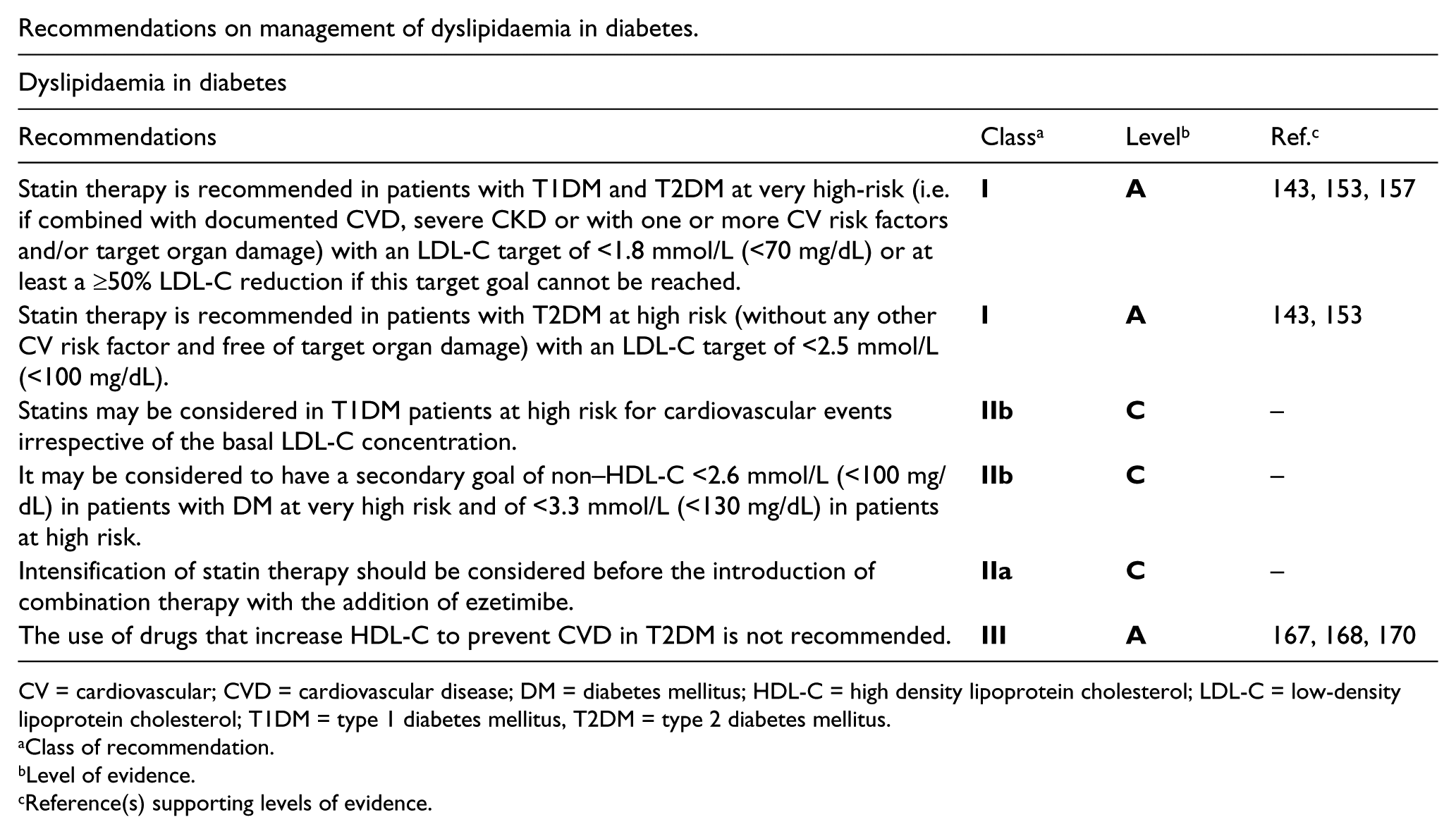
CV = cardiovascular; CVD = cardiovascular disease; DM = diabetes mellitus; HDL-C = high density lipoprotein cholesterol; LDL-C = low-density lipoprotein cholesterol; T1DM = type 1 diabetes mellitus, T2DM = type 2 diabetes mellitus.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
4.5 Platelet function
Platelet activation plays a pivotal role in the initiation and progression of atherothrombosis. 171 Abnormalities in platelet aggregation in DM ex vivo have been described by numerous groups, 172 and both post-prandial and persistent hyperglycaemia have been identified as major determinants of platelet activation in the early and late phases of the natural history of T2DM.173,174
4.5.1 Aspirin
Aspirin inhibits thromboxane (TX) A2-dependent platelet activation and aggregation through irreversible inactivation of platelet cyclooxygenase 1 (COX-1) activity. 175 There are no outcome studies of dose- and time-dependence of aspirin’s antiplatelet effect in T2DM and it is currently recommended at 75–162 mg daily (as used in subjects without DM).175,176 However, daily administration of low-dose aspirin may be associated with incomplete inhibition of platelet COX-1 activity 177 and TXA2-dependent platelet function,178,179 perhaps due to increased platelet turnover in DM. 180 There is emerging evidence of sustained efficacy using twice-daily aspirin in subjects with DM and CVD.180,181
Secondary prevention. The first collaborative overview of the Antiplatelet Trialists’ Collaboration found that antiplatelet therapy (mostly with aspirin) is similarly effective among patients with pre-existing symptomatic CVD, regardless of the presence of DM. 182 They analysed individual data on ‘serious vascular events’ (non-fatal MI, non-fatal stroke, or vascular death) from approximately 4500 patients with DM in the randomized trials and found that treatment with antiplatelet drugs produced a proportional reduction of about one-quarter. 182 Therefore there is no reason to treat patients with DM and CVD differently from non-DM patients and low-dose aspirin is uniformly recommended for both the acute treatment of ischaemic syndromes and their secondary prevention. 175
Primary prevention. Low-dose aspirin is recommended by several North American organizations for the primary prevention of cardiovascular events in adults with DM.176,183 However, direct evidence for its efficacy and safety in this setting is lacking—or at best inconclusive.184,185 Thus, in the most up-to-date meta-analysis, which includes three trials conducted specifically in patients with DM and six other trials in which such patients represent a subgroup within a broader population, aspirin was found to be associated with a non-significant 9% decrease in the risk of coronary events (RR 0.91; 95% CI 0.79–1.05) and a non-significant 15% reduction in the risk of stroke (RR 0.85; 95% CI 0.66–1.11). 176 It should be emphasized that the total number of patients with DM enrolled in these nine trials was 11,787, with 10-year extrapolated coronary event rates ranging from as low as 2.5% to as high as 33.5%. 176 These results have been interpreted as suggesting that aspirin probably produces a modest reduction in the risk of cardiovascular events but the limited amount of available data precludes a precise estimate of the effect size. Consistent with this uncertainty, antiplatelet therapy with aspirin in adults at a low CVD risk is not recommended by the Fifth Joint Task Force of the European Society of Cardiology and Other Societies on CVD Prevention in Clinical Practice. 42
The risk-benefit ratio of aspirin. In a meta-analysis of six primary prevention trials, aspirin was associated with a 55% increase in extracranial (mainly gastro-intestinal) bleeding in both DM and non-DM patients. 186 In terms of the risk-benefit balance in primary prevention, these results probably represent a best case, as those at increased risk of gastro-intestinal bleeding were excluded, and elderly subjects were under-represented. 186 In this analysis, DM at baseline was associated with a two-fold increase in vascular events and a 50% increased risk of major extracranial bleeds. 186
The ADA/AHA/ACCF Scientific Statement and the Endocrine Society Clinical Practice Guideline favour aspirin use in adults with DM when the 10-year risk of cardiovascular events is >10%.176,183 However, relatively little emphasis is placed on the need to evaluate bleeding risk. The annual risk of cardiovascular events is increased in people with compared to those without DM, 176 but this has to be balanced against the annual risk of upper gastro-intestinal bleeding which varies considerably depending on age and history of peptic ulcer disease.175,187
4.5.2 P2Y12 receptor blockers
Clopidogrel, an irreversible blocker of the adenosine diphosphate (ADP) receptor P2Y12, is a valid alternative for patients who are aspirin-intolerant or have symptomatic peripheral vascular disease.188,189 Clopidogrel (7 mg once daily) produces additive cardio-protective effects when combined with low-dose aspirin (7–160 mg once daily) in patients with acute coronary syndrome (ACS) and those undergoing percutaneous coronary intervention (PCI). 188 However, evidence from the CHARISMA (Trial to assess improvement in therapeutico by optimizing platelet inhibition with prasugrel–thrombolysis in myocardial infarction) study indicates that clopidogrel added to aspirin may have deleterious effects in patients with advanced nephropathy. 190 More effective P2Y12 blockers include prasugrel and ticagrelor, which is reversible. 188 In TRITON-TIMI (Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction) 38, prasugrel showed superiority over clopidogrel in post-ACS prevention of recurrent ischaemic events: however prasugrel carried a risk of increased thombosis in myocardial infarction (TIMI) major bleeding. 191 In a DM sub-study, a similar reduction in recurrent ischaemic events was seen, but this was not accompanied by an increase in bleed ing. 192 Ticagrelor was also more effective than clopidogrel in reducing12-month mortality post-ACS, 193 and decreased ischaemic events in DM patients withoutincreased bleeding. 194 Ticagrelor was superiorto clopidogrel in ACS with renal impairment. 195 There is no convincing evidence that clopidogrel or the newer drugs are any more or less effective with DM than without. 188
Recommendations for antiplatelet therapy in patients with diabetes.
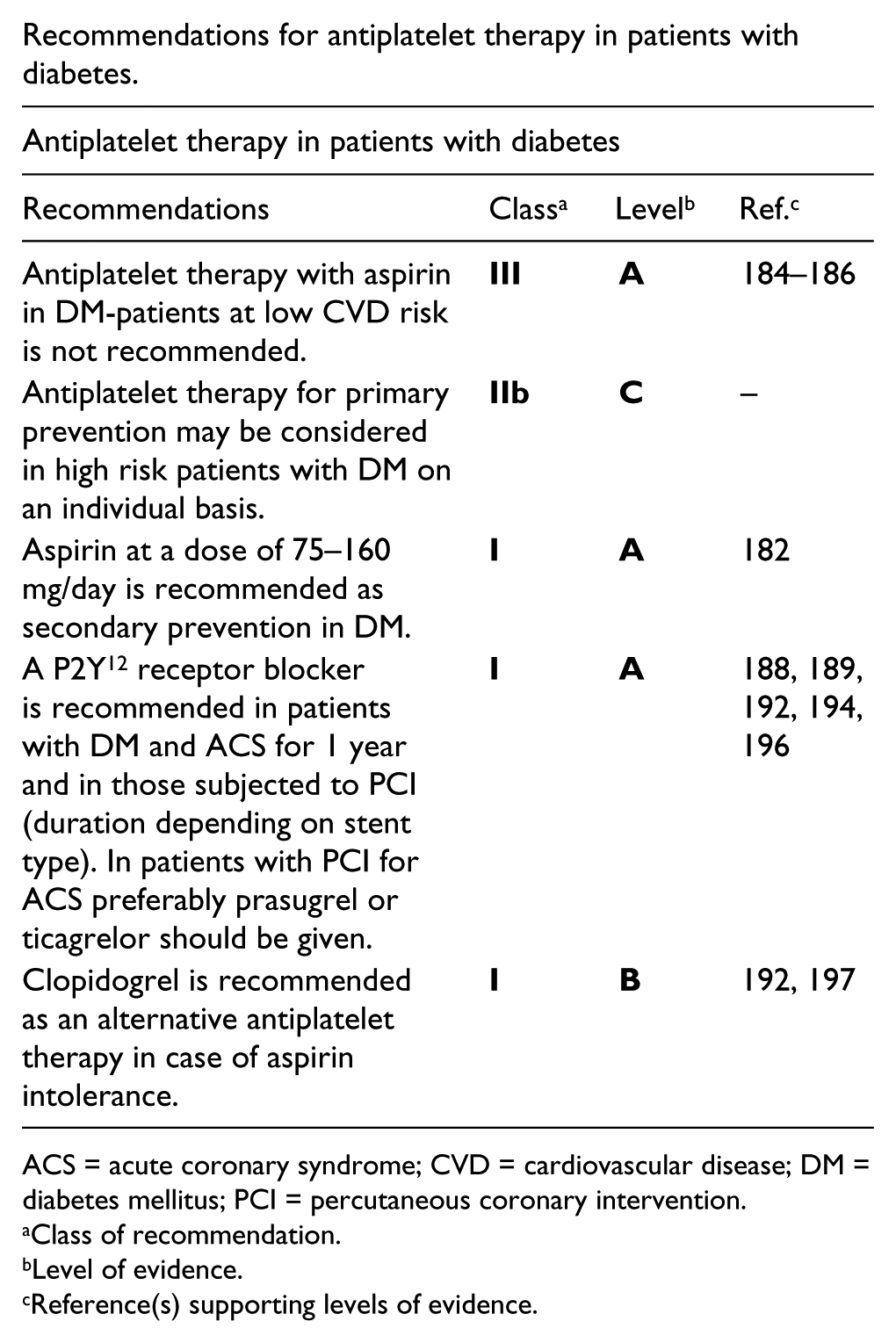
ACS = acute coronary syndrome; CVD = cardiovascular disease; DM = diabetes mellitus; PCI = percutaneous coronary intervention.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
4.6 Multifactorial approaches
Patients with glucose perturbations are in need of early assessment of (i) risk factors (e.g. lifestyle habits including smoking, hypertension, and dyslipidaemia); (ii) micro- and macrovascular disease and autonomic dysfunction; (iii) co-morbidities (e.g. heart failure and arrhythmias); (iv) inducible ischaemia by means of exercise testing, stress echocardiography, or myocardial scintigraphy and (v) myocardial viability and LV function by means of echo-Doppler and/or magnetic resonance imaging. 198 The level of reliability of exercise testing, stress echocardiography, or myocardial scintigraphy is of particular concern in the detection of ischaemia in DM. Confounders are a high threshold for pain due to autonomic dysfunction, multivessel coronary disease, ECG abnormalities, co-existence of PAD and use of multiple medications. Treatment should be target-driven (Table 2).
Summary of treatment targets for managing patients with diabetes mellitus or impaired glucose tolerance and coronary artery disease.

CVD = cardiovascular disease; DM = diabetes mellitus; HbA1c = glycated haemoglobin A1c; IGT = impaired glucose tolerance; LDL = low density lipoprotein; T2DM = type 2 diabetes mellitus.
Diabetes Control and Complication Trial standard.
The value of a multifactorial intervention in patients with DM and established microalbuminuria was demonstrated by Steno 2 which, in a highly specialized setting, randomized 160 subjects to intensive, target-driven multifactorial therapy or to conventional management. The targets in the intensively treated group were HbA1c <6.5%, total cholesterol <4.5 mmol/L (175 mg/dL), and blood pressure <130/80 mm Hg. All patients in this group received RAAS blockers and low-dose aspirin. Although treatment targets were not always attained in the intensive-treatment group, their management was considerably better. This resulted in a reduction in microvascular and macrovascular events by about 50% after 7.8 years of follow-up. The cholesterol target was most successfully attained making the role of statins crucial.199,200 Subsequently, target-driven therapy was recommended to patients in both groups, who were followed for 13 years. By that time, patients originally allocated to the intensively managed group had an absolute mortality reduction of 20% and the HR for death, compared with the conventional group, was 0.54 (P < 0.02). The absolute reduction in cardiovascular events was 29%. In addition there was a substantial reduction in diabetic nephropathy and progression of retinopathy. 74 A health-economic analysis reported intensive management as more cost-effective than conventional care. 201 Data from the Euro Heart Survey on Diabetes and the Heart support a multifactorial approach. Among 1425 patients with known T2DM and CAD, 44% received a comprehensive evidence-based therapy (a combination of aspirin, beta-blockade, RAAS inhibitors and statins). Patients on these combinations had significantly lower all- cause mortality (3.5 vs. 7.7%; P = 0.001) and fewer combined cardiovascular events (11.6 vs. 14.7%, P = 0.05) after one year follow-up. 202
Recommendations for multifactorial risk management in diabetes.
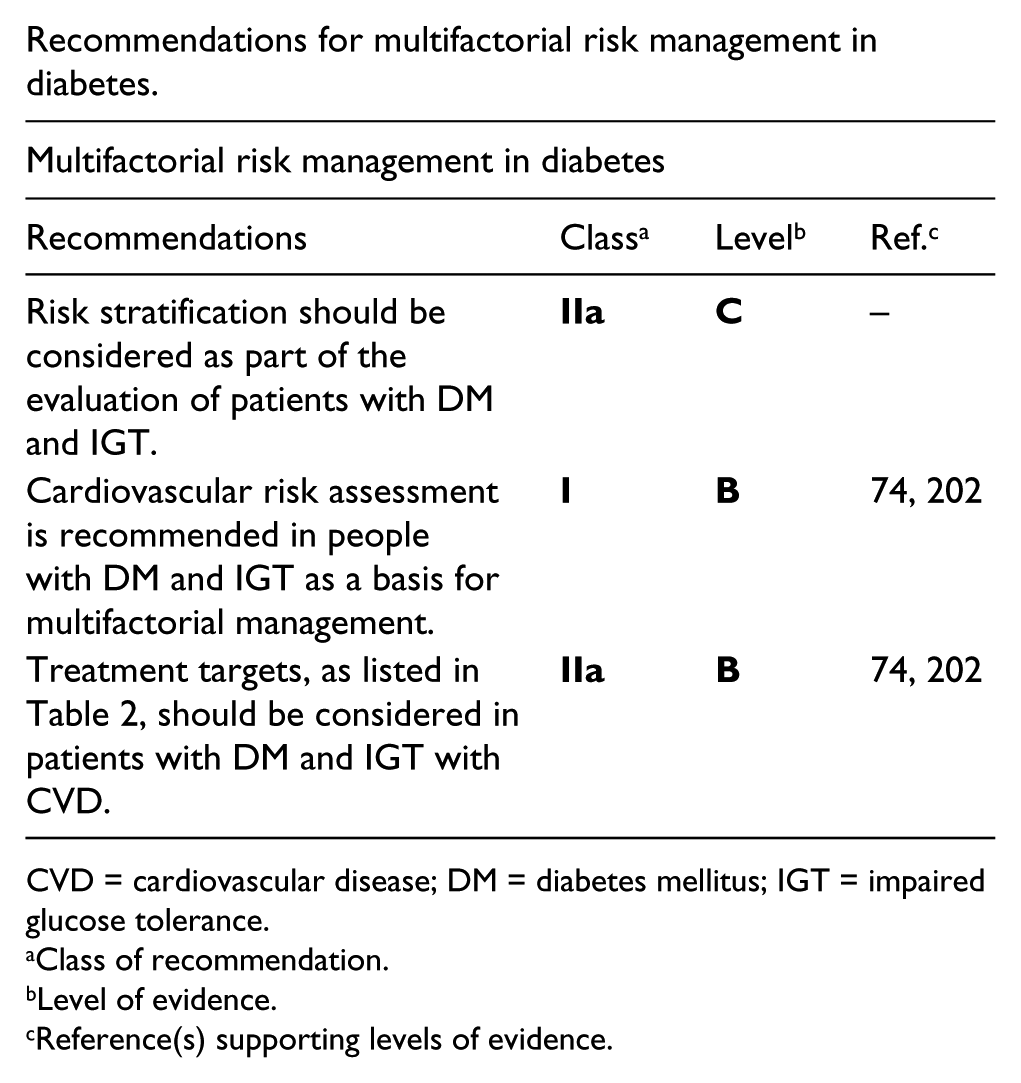
CVD = cardiovascular disease; DM = diabetes mellitus; IGT = impaired glucose tolerance.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
5. Management of stable and unstable coronary artery disease
5.1 Medical management of coronary artery disease
Patients with CAD, without previously known glucose perturbations, should have their glycaemic state evaluated. Elevated levels of HbA1c and FPG may establish the diagnosis of DM, 203 but a normal value does not exclude glucose abnormalities. Accordingly, the appropriate screening method is an OGTT,2,21 which should not be performed earlier than 4–5 days after an ACS to minimize false positive results.204,205 In-hospital and long-term mortality after MI has declined but outcome is still poor in DM, probably due to a higher prevalence of complications and a lack of evidence-based treatments.206,207 Available information favours a proportionately similar efficacy of cardiovascular risk management in DM and non-DM patients but, due to their higher absolute risk, the number needed to treat (NNT) to avoid one cardiovascular event is lower in patients with DM. 202
5.1.1 Pharmacological treatment
β-Adrenergic blockers. As outlined in current European Guidelines β-blockers are advocated for the whole spectrum of CAD with different levels of recommendations and different levels of evidence. 208 – 212 β-Blockers relieve symptoms of myocardial ischaemia (angina pectoris) in patients with stable CAD and may provide prognostic benefits suggested by retrospective analysis of placebo-controlled trials. 209 β-Blockers are effective in improving prognosis in post-MI patients with DM by reducing the likelihood of reinfarction, sudden death and ventricular arrhythmias.213,214 β-Blockers may have negative metabolic effects by increasing IR and masking hypoglycaemic symptoms and there seems to be a difference between non- vasodilating, β-1 antagonists (e.g. metoprolol and atenolol) and β-blockers with vasodilating properties (e.g. the (β/α-adrenoblockers, carvedilol and labetalol, and b1-blockers which modulate NO synthesis, such as nebivolol). 215 Overall, the positive effects of β-blockade on prognosis far outweigh the negative glucometabolic effects.
Blockers of the renin-angiotensin-aldosterone system. Treatment with ACE-I or ARB should be started during hospitalization for ACS and continued in patients with DM and LVEF <40%, hypertension, or chronic kidney disease,208,210,211 and considered in all patients with ST-elevation myocardial infarction (STEMI). Patients with DM and stable CAD are also recommended to receive an ACE-I. 209 The Heart Outcomes Prevention Evaluation (HOPE) study showed a 25% reduction in MI, stroke, or cardiovascular death for patients with known vascular disease or DM, randomized to placebo or ramipril. This finding was consistent in the pre-specified subgroup of patients with DM. 216 A proportionately similar trend towards benefit was observed in the subgroup of DM in the European trial on Reduction Of cardiac events with Perindopril in stable coronary Artery disease.
(EUROPA). 217 In the ONTARGET trial, telmisartan was equivalent to ramipril as regards a primary composite of death from cardiovascular causes, MI, stroke or hospitalization for heart failure, while combining the two drugs caused adverse events without further benefit. 134
Lipid-lowering drugs. The beneficial effects of statins are established as described (see 4.4.1).
Nitratesand calcium channel blockers. There is no evidence for a prognostic impact of nitrates but they may be used for symptomaticirelief.208,210,211
Calcium channel blockers. are efficacious in relieving ischaemic symptoms, and verapamil and diltiazem may prevent re-infarction and death. 208 – 211 These drugs may be appropriate for long-term use in patients without heart failure, as an alternative to β-blockers or when β-blockers may be a less attractive choice, e.g. due to obstructive airways disease. The combination of these drugs and β-blockers should be avoided with bradycardia, atrioventricular conduction disturbances or compromised LV function. An alternative is the use of a dihydropyridine calcium channel blocker, such as amlodipine, felodipine or nicardipine.
Ivabradine. This specific, heart rate-lowering anti-anginal drug inhibits the If current—the primary modulator of spontaneous diastolic depolarization in the sinus node. Ivabradine is indicated in the treatment of chronic stable angina in CAD patients with a contraindication or intolerance to (β-blockers, or in combination with β-blockers if the patient remains symptomatic or has a heart rate >70 bpm, especially if there is also LV dysfunction. It can be used in selected patients with non-ST-elevation ACS in the event of β-blocker intolerance or insufficient heart rate reduction despite maximal tolerated β-blocker dose.209,210
Antiplatelet and antithrombotic drugs. In secondary prevention, antiplatelet therapy in the form of low-dose aspirin (75–60 mg) or clopidogrel (separately or in combination) reduces risk of stroke, Ml, or vascular death although the benefits are less in DM (see 4.5.1). 218 Thienopyridines (ticlopidine, clopidogrel, prasu grel and ticagrelor) reduce cardiovascular events when added to aspirin in patients with ACS.196,208,211 In the Clopidogrel vs. Aspirin in Patients at Risk of Ischaemic Events (CAPRIE) study, the annual event rate in DM was 5.6% with clopidogrel and 7.7% with aspirin, i.e. an absolute risk reduction of 2.1 %, which corresponds to a relative risk reduction of 13% with less bleeding. Due to the elevated event rates in patients with DM, the absolute benefit of clopidogrel is amplified. 197 In TRITON, DM subjects tended towards a greater reduction in ischaemic events with prasugrel than clopidogrel, without an increase in major bleeding. 192
5.1.2 Glucose control in acute coronary syndromes
Elevated PG during ACS is associated with a more serious prognosis in DM.219 –223 Glycaemic control has been tested in the Diabetes and Insulin–Glucose Infusion in Acute Myocardial Infarction (DIGAMI) 1 and 2 and Hyperglycaemia: Intensive Insulin Infusion in Infarction (HI-5) trials. 224 , 225 , 226 The first DIGAMI trial randomized 620 patients with DM and acute Ml to a ≥24 h insulin–glucose infusion followed by multi-dose insulin, or to routine glucose-lowering therapy. 224 Mortality after 3.4 years was 33% in the insulin group and 44% (P = 0.011) in the control group. 227 In contrast, DIGAMI 2 failed to demonstrate prognostic benefits, 225 and a plausible explanation for this is that admission HbA1c decreased by 1.5% from a higher starting value of 9.1% in DIGAMI 1,224,228 compared with a fall of only 0.5% from 8.3% in DIGAMI 2. 225 In addition, the use of β-blockade, statins and revascularization was more extensive in DIGAMI 2. The difference in glucose level between the control and insulin groups In the HI-5 study was also small and there was no reduction in mortality with insulin. 226 Pooled data from the three studies confirmed that insulin–glucose infusion did not reduce mortality in the absence of glucose control in patients with acute MI and DM (RR 1.07; P = 0.547). 229 The Heart2D (Hyperglycaemia and its effect after acute myocardial infarction on cardiovascular outcomes in patients with Type 2 diabetes mellitus) compared the effects of prandial (pre-meal insulin three times daily; n = 557) vs. basal glycaemic control (long-acting insulin once or twice daily; n = 558) on cardiovascular events in T2DM. Glucose targets were a post- prandial glucose (PPG) of 7.5 mmol/L (135 mg/dL) and an FPG of 6.7 mmol/L (121 mg/dL), respectively. The study was stopped after an average follow-up of 963 days, due to lack of efficacy. 94
Some registry studies suggest there is a J- or U-shaped relationship between PG and prognosis,220,222,223 with the implication that both hypoglycaemia and hyperglycaemia are unfavourable. Compensatory mechanisms induced by hypoglycaemia, such as enhanced catecholamine release, may aggravate myocardial ischaemia and provoke arrhythmias.230,231 Recent data indicate that hypoglycaemic episodes identify patients at risk for other reasons (e.g. heart failure, renal dysfunction and malnutrition) and hypoglycaemia does not remain as an independent risk factor after correcting for such variables.232,233
A reasonable conclusion is that DM and acute Ml will benefit fromglycaemic contol if hyporlycaemia is significant (>10 mmol/L or >180 mg/dL). An approximation towards normoglycaemia, with less stringent targets in those with severe co-morbidities, is a reasonable goal but exact targets are still to be defined. Insulin infusion is the most efficient way to achieve rapid glucose control under these circumstances.
Recommendations for the management of patients with stable and unstable coronary artery disease and diabetes.
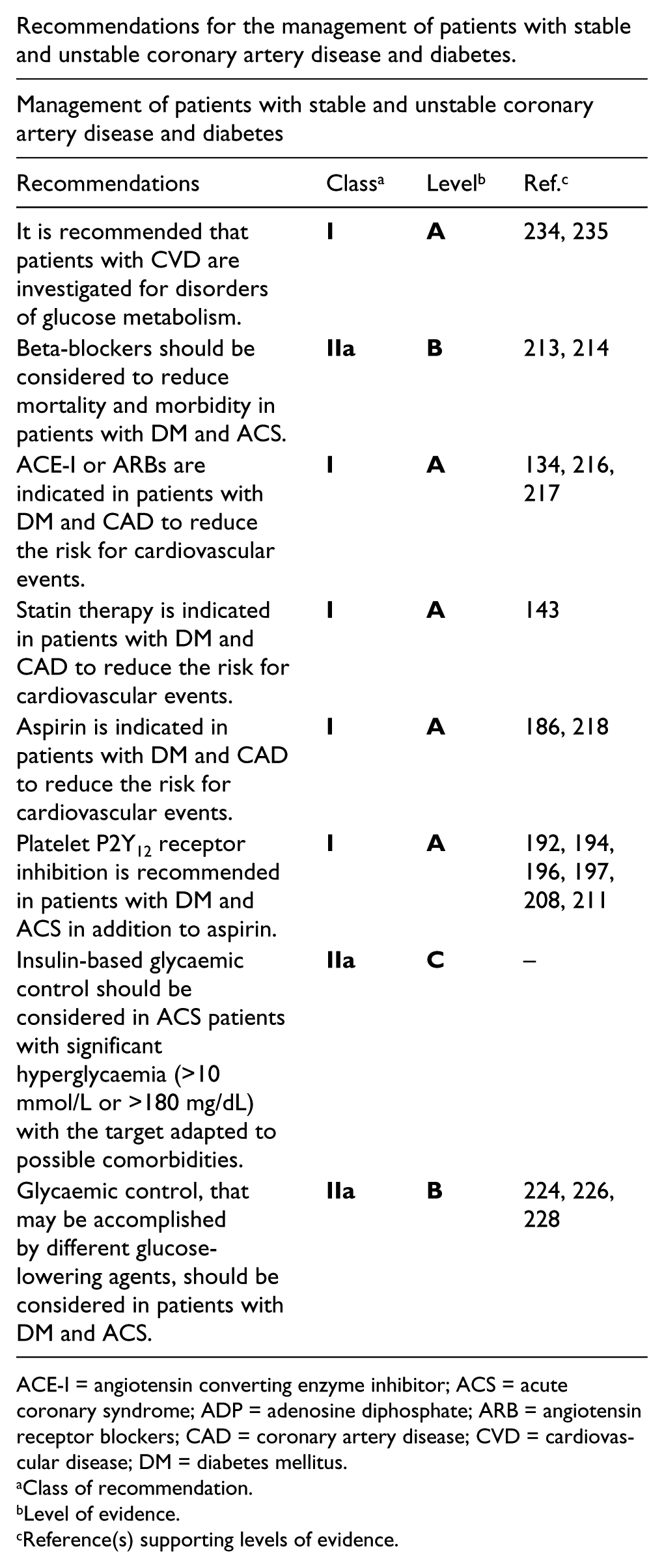
ACE-I = angiotensin converting enzyme inhibitor; ACS = acute coronary syndrome; ADP = adenosine diphosphate; ARB = angiotensin receptor blockers; CAD = coronary artery disease; CVD = cardiovascular disease; DM = diabetes mellitus.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
5.2 Revascularization
A quarter of revascularization procedures are performed in DM, which is challenged by a more diffuse atherosclerotic involvement of epicardial vessels, a higher propensity to develop restenosis after PCI and saphenous graft occlusion after coronary artery bypass graft surgery (CABG), and unremitting atherosclerotic progression causing new stenosis. 236 This results in a higher risk and long-term mortality than in non-DM patients, irrespective of revascularization modality. 237
5.2.1 Myocardial revascularization in stable and unstable coronary artery disease
Stable coronary artery disease. A randomized comparison of myocardial revascularization—either with CABG or PCI—against optimal medical treatment (OMT) in DM patients considered eligible for these treatments, was performed in the Bypass Angioplasty Revascularization Investigation 2 Diabetes (BARI 2D) trial. 238 After five years, no significant differences were noted in the combined endpoint of death, MI, or stroke between the OMT (12%) and revascularization (12%) arms. In the surgical group, freedom from major adverse cardiac and cerebrovascular events (MACCE) was significantly higher with CABG (78%) than with OMT alone (70%; P = 0.01), but there was no difference in survival (CABG 86%; OMT 84%; P = 0.33). In the PCI group, made up of patients with less-extensive CAD than in the CABG stratum, there were no significant differences in MACCE or survival between PCI and OMT. During subsequent follow-up, 38% of patients assigned to OMT underwent at least one revascularization for symptomatic reasons, compared with 20% in the revascularization stratum, showing that an initial conservative strategy with OMT saved about 80% of interventions over the next five years. Overall, except in specific situations such as left main coronary artery stenosis ≥50%, proximal LAD stenosis or triple-vessel disease with impaired LV function, myocardial revascularization in patients with DM did not improve survival, compared with medical treatment. It is noteworthy is that patients were excluded if they required immediate revascularization or had left main coronary disease, a creatinine level >2.0 mg/dL (>177 µmol/L), HbA1c >13.0%, Class III–IV heart failure or if they had undergone PCI or CABG within the previous 12 months.
Acute coronary syndromes. No interaction between the effect of myocardial revascularization and the presence of DM has been documented in trials in non-ST-elevation ACS. An early invasive strategy improved outcomes in the overall population, with a greater benefit in patients with DM in the Treat angina with Aggrastat and determine Cost of Therapy with an Invasive or Conservative Strategy-Thrombolysis In Myocardial Infarction (TACTICS-TIMI 18) trial.239 –241 In STEMI patients, a pooled analysis of individual patient data, from 19 RCTs comparing primary PCI with fibrinolysis, showed that patients with DM treated with reperfusion had an increased mortality, compared with those without DM. The benefits of a primary PCI, compared with fibrinolysis were, however, consistent in patients with and without DM. 242 Patients with DM had significantly delayed initiation of reperfusion treatments and longer ischaemic time, but the reduction in 30-day mortality observed in PCI-treated patients was most pronounced in this group. Due to a higher absolute risk, the NNT to save one life at 30 days was significantly lower for DM (NNT 17; 95% CI 11–28) than non-DM patients (NNT 48; 95% CI 37–60).
5.2.2 Type of intervention: coronary bypass graft vs. percutaneous intervention
A meta-analysis based on individual data, from 10 RCTs comparing both types of revascularizations, suggested a distinct survival advantage for CABG in DM patients. 237 Five-year mortality was 20% with PCI, compared with 12% with CABG (OR 0.7; 95% CI 0.6–0.9), whereas no difference was found for patients without DM. A specific comparison of the efficacy and safety of PCI and CABG in patients with DM was performed in theCoronary Artery Revascularization in Diabetes (CARDia) trial. 243 The introduction of drug-eluting stents (DES) coincided with the enrolment period, leading to a mixed use of bare metal stents (BMS) (31%) and DES (69%). After one year there was a non-significantly higher rate of the composite of death, MI, and stroke (driven by a higher rate of MI), and significantly higher rates of repeat revascularization in the PCI group (2 vs. 12%; P < 0.001).
The literature on CABG vs. PCI is confused by confounder bias in registries, the ongoing development of DES and—apart from the FREEDOM trial—a lack of prospective RCTs. The implication is that much of the available information is based on subgroup analyses of trials in which DM patients may be relatively few or selected. As a consequence of increased repeat revascularization in the SYNergy between percutaneous coronary intervention with TAXus and cardiac surgery (SYNTAX) trial, 244 performed in the DES era (using paclitaxel-eluting stents), the rate of MACCE after one year was twice as high with PCI, as compared with CABG. After 5 years, follow-up rates of MACCE were significantly higher in DM, comparing PCI with CABG (PCI 46% vs. CABG 29%; P < 0.001) as well as for repeat revascularization (PCI 35.3% vs. CABG 14.6%; P < 0.001). There was no difference in the composite of all-cause death/ stroke/ MI (PCI 23.9% vs. CABG 19.1%; P = 0.26). It was concluded that, although PCI is a potential treatment option in patients with less complex lesions CABG should be the revascularization choice for DM patients with complex anatomic disease. 245
Data obtained in recent registries support a better outcome for DM treated with CABG, compared with DES even in terms of mortality, at the expense of a higher stroke rate with CABG. 246 In an analysis of 86,244 patients ≥65 years of age undergoing CABG and 103,549 patients undergoing PCI from 2004 to 2008, four-year survival was significantly higher with surgery and the association of surgery with improved survival was most marked in insulintreated DM. 247
The FREEDOM trial randomized 1900 patients, a majority with three-vessel disease, to treatment with CABG or PCI with sirolimus-eluting and paclitaxel-eluting stents. They were all pre- scribed currently recommended medical therapies for the control of LDL-C, systolic BP and HbA1c. The primary results were a composite of total mortality and non-fatal MI or stroke. After a median of 3.8 years the primary outcome occurred more frequently in the PCI group (P = 0.005), with a five-year rate of 26.6%, compared with 18.7% in the CABG group. The benefit of CABG was driven by differences in both MI (P < 0.001) and mortality (P = 0.049) It was concluded that CABG is superior to PCI for patients with DM and advanced CAD (Figure 3). There was no significant interaction based on SYNTAX score, since the absolute differences in the primary endpoint, between PCI and CABG, were similar in patients with a low, intermediate and high SYNTAX score. Given the wide variability of the patients enrolled in Future REvascularization Evaluation in patients with Diabetes mellitus: Optimal management of Multivessel disease (FREEDOM), the trial represents real-world practice. Further analysis revealed that, compared with PCI, CABG was a cost- effective strategy.248,249 An individualized risk assessment and discussion with the patient is mandatory before the type of intervention is decided. 212
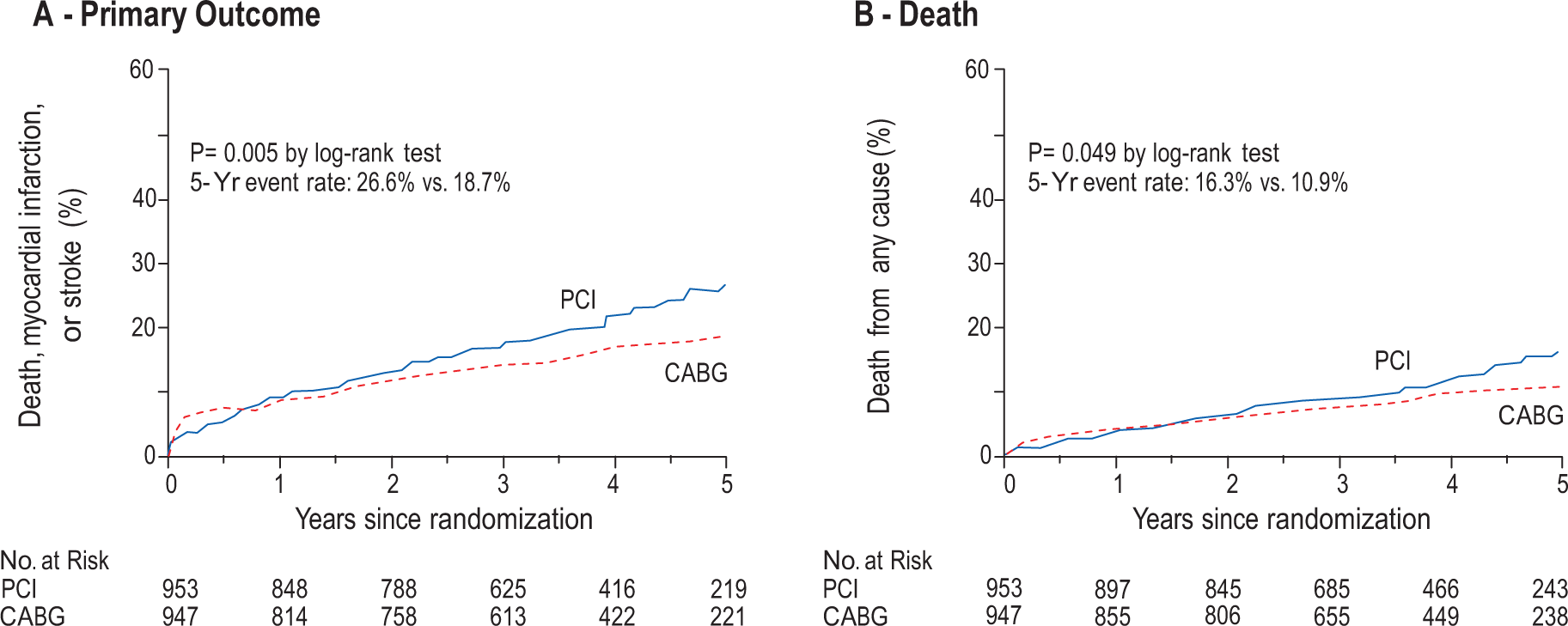
Kaplan-Meier estimates of the primary outcome and death. A: rates of the composite primary outcome of death, myocardial infarction or stroke and B: death from any cause truncated at five years after randomization. The P-value was calculated by means of the log-rank test on the basis of all available follow-up data. Reproduced by permission from Farkouh et al. 248
5.2.3 Specific aspects of percutaneous and surgical revascularization in diabetes mellitus
The DIABETES trial (the diabetes and sirolimus-eluting stent trial) demonstrated a substantial reduction in target vessel revascularization in DM patients treated with sirolimus-eluting stents (7%) vs. BMS (31%). 250 This finding received further support from a meta-analysis of 35 trials comparing DES with BMS, 251 which revealed a similar efficacy of sirolimus-eluting and paclitaxel-eluting stents in this regard (OR 0.29 for sirolimus; 0.38 for paclitaxel) provided dual antiplatelet therapy after DES implantation was continued for >6 months. The risk of death associated with sirolimus-eluting stents was more than twice that associated with BMS in eight trials with dual antiplatelet therapy during less than six months. In contrast, there was no increased risk associated with the use of DES in 27 trials with dual antiplatelet therapy maintained for more than six months. An analysis of the National Heart, Lung and Blood Institute Dynamic Registry data revealed that, compared with BMS, DES were associated with fewer repeat revascularizations—to a similar extent in insulin-treated or non-insulin-treated DM. 252 Finally, the secondgeneration everolimus-eluting stents were not superior in terms of target lesion failure after one year of follow-up in a head-to-head comparison with paclitaxel-eluting stents, while zotarolimus-eluting stents were inferior to sirolimus-eluting stents in patients with DM.253,254
Antithrombotic treatment in DM patients undergoing coronary revascularization for stable angina or ACS is no different from those without DM.255–257 Initial trials in glycoprotein IIb/IIIa inhibitors reported an interaction with DM, but this was not confirmed in the recent Intracoronary Stenting and Antithrombotic Regimen: Rapid Early Action for Coronary Treatment (ISAR-REACT 2) trial. 258 Prasugrel is superior to clopidogrel in reducing the composite endpoint of cardiovascular death or MI or stroke without excess major bleeding. Similarly, ticagrelor, in comparison to clopidogrel in the PLATO (Platelet inhibition and patient outcomes) trial, reduced the rate of ischaemic events in ACS patients, irrespective of the presence of DM, without an increase in major bleeding events.192,194
Patients with DM who undergo CABG often require multiple grafts. There is no randomized evidence regarding the use of one vs. two internal thoracic artery (ITA) conduits in DM. Although observational evidence suggests that using bilateral ITA conduits improves patient outcome without compromising sternal stability, the use of bilateral ITA conduits is still debatable, given the higher prevalence of wound infection and mediastinitis with DM. 259 A recent meta-analysis has shown that ITA harvesting by skeletonization (without the satellite veins and fascia) reduces the risk of sterna wound infection in DM patients undergoing bilateral ITA grafting, 260 although there are no randomized studies on this subject.
5.2.5 Glucose-lowering treatments and coronary angiography and interventions
Few trials have addressed interactions between hypoglycaemic medications and coronary angiography or myocardial revascularization in DM. There is no scientific support for the frequent practice of stopping metformin prior to angiography or PCI, and more recent recommendations are less restrictive. 212 Rather than stopping metformin, a reasonable approach is to withhold metformin for 48 h if renal function deteriorates and until renal function has resumed to its previous level.
Observational data reported concern over the use of sulphonylureas in patients treated with primary PCI for acute MI; this has not been confirmed by post hoc analysis of the DIGAMI-2 trial, although the number of patients undergoing primary PCI in this trial was low. 261 Arrhythmias and ischaemic complications were also less frequent in patients receiving gliclazide/glimepiride. 262 Thiazolidinediones might be associated with lower restenosis rates after PCI with BMS, 263 but carry an increased risk of heart failure due to fluid retention.
No trial has demonstrated that insulin or glucose-insulinpotassium (GIK) improves PCI outcome after STEMI. Observational data in CABG suggest that continuous intravenous insulin infusion achieving moderately tight glycaemic control (6.6–9.9 mmol/L or 120–180 mg/dL) is independently associated with lower mortality and major complications, than has been observed after tighter (<6.6 mmol/L or <120 mg/dL) or more lenient (>9.9 mmol/L or >180 mg/dL) glycaemic control. 264 In the BARI 2D trial, outcomes were similar in patients receiving insulin sensitization vs. insulin provision to control blood glucose. In the CABG stratum, insulin use was associated with more cardiovascular events than insulin-sensitization.238,265
Recommendations for coronary revascularization of patients with diabetes.

BMS = bare-metal stent; CABG = coronary artery bypass grafting; CAD = coronary artery disease; DES = drug-eluting stent; DM = diabetes mellitus; LAD = left anterior descending coronary artery; PCI ¼ percutaneous coronary intervention; STEMI = ST-elevation myocardial infarction.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
6. Heart failure and diabetes
6.1 Heart failure in type 2 diabetes
Prevalence and incidence of heart failure in diabetes mellitus. The prevalence of heart failure in a general population is 1–4% and 0.3–0.5% of the patients have both heart failure and T2DM. Studies in heart failure populations reveal a prevalence of T2DM from 12–30%, rising with age.270,271 In the Framingham study, the age-adjusted relative risk of heart failure in patients with T2DM (age 45–74 years) was 2.2 for men and 5.3 for women. 272 The high incidence of heart failure in patients with T2DM was confirmed in the National Health and Nutrition Examination Survey, with an HR of 1.85 (95% CI 1.51–2.28) in T2DM compared with non-DM. 273 Boonman-de Winter et al. 274 studied 581 T2DM patients (age >60 years) and reported that 28% had previously-unknown heart failure. The prevalence increased rapidly with age and heart failure with preserved left ventricular ejection fraction (LVEF) was more common in women than men. Left ventricular (LV) dysfunction was diagnosed in 26% and diastolic dysfunction in 25%.
Prevalence and incidence of diabetes mellitus in heart failure. The prevalence of DM in a general population is 6–8%. 10 It is higher in subjects with symptomatic heart failure (12–30%) increasing towards 40% among hospitalized patients. 275 In an elderly Italian population, new-onset DM occurred in 29% during 3 years of follow-up compared with 18% in controls without heart failure. 276 When subjects with two or more visits in the Reykjavik study n = 7060) were followed over 30 years DM and heart failure did not predict each other independently although fasting glucose and body mass index (BMI) were significant risk factors, both for glucose disturbances and heart failure. 277
Diabetes cardiomyopathy: Long-standing hyperglycaemia may independently affect myocardial tissue and reduction of LV compliance—an early sign of DM cardiomyopathy—may be detectable early in the course of DM. 278 The frequent co-existence of hypertension and DM makes the contribution of the glucometabolic state to diastolic dysfunction difficult to isolate. The pathogenic mechanisms involve accumulation of advanced glycation products, collagen formation and interstitial fibrosis, leading to impaired calcium homeostasis and impaired myocardial insulin signalling, all of which increase myocardial stiffness and reduce myocardial compliance.279,280 Diastolic dysfunction is identified by quantitative estimation of LV diastolic properties, using conventional Doppler parameters of the transmitral inflow of blood and tissue Doppler imaging of the mitral annulus. 281
6.2 Morbidity and mortality
Heart failure was a major cause of hospitalization inpatients with T2DM in the Hypertension, Microalbuminuria or Proteinuria, Cardiovascular Events and Ramipril (DIABHYCAR) trial, investigating hospitalizations in T2DM patients with albuminuria. 282 On the other hand, T2DM increased the risk of hospitalization in patientswith heart failure in the BEta blocker STroke trial (BEST) 283 (RR1.16; P = 0.027). In the Metoprolol CR/XL Randomised Intervention Trial in Congestive HeartFailure (MERIT-HF), 284 patients with heart failure and T2DM had 1-year hospitalization of 31%, compared with 24% forthose free from DM. In the DIABHYCAR study, the combination of heart failure and T2DM resulted in a mortality rate 12-times-as great as in patients with T2DM but without heart failure (36 vs.3%). 282 BEST and Studies Of Left Ventricular Dysfunction (SOLVD)283,285 reported T2DM as an independent predictor of mortality, mostly in ischaemic heart failure.
6.3 Pharmacological treatment
Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers. improve symptoms and reduce mortality and are indicated in T2DM and heart failure. In the SOLVD trial, the ACE-I enalapril significantly reduced mortality in DM patients with heart failure. 285 Mortality risk reduction in the high-dose vs. low-dose lisinopril group was 1% in DM and 6% in non-DM patients in the Assessment of Treatment with Lisinopril And Survival (ATLAS) trial. 286 Subgroup analyses of clinical trials indicate that the beneficial effects of ARBs are equivalent to those of ACE-Is.287 –290An ARB can therefore be used as an alternative in ACE-I-intolerant patients. ACE-I and ARB should not be used in combination in patients with an LVEF <40%, who remain symptomatic despite optimal treatment with an ACE-I and a β-blocker. According to the 2012 ESC heart failure Guidelines, such patients should be prescribed a mineralocorticoid receptor antagonist (see below), which causes a greater morbidity and mortality reduction than the addition of an ARB. 281 When ACE-Is and ARBs are used in patients with DM, surveillance of kidney function and potassium is mandatory, since nephropathy is frequent.
Beta-blockers. In addition to an ACE-I (or, if not tolerated, an ARB) a β-blocker should be given to all patients with an LVEF 0≤40%. A subgroup analysis of the MERIT-HF trial showed that β-blockers reduce mortality and hospital admission and improve symptoms without significant differences between DM and non-DM. 284 Further meta-analyses of major heart failure trials indicate that the RR of mortality in patients with DM receiving a β-blocker was significantly improved (0.84 vs. 0.72).291,292 β-Blockers also reduce hospitalizations for heart failure in both DM and non-DM.283,284,293,294 Despite this, T2DM subjects are less likely to be discharged from hospital on a β-blocker than non-DM with heart failure. 295 β-Blockers recommended in heart failure andT2DM are: slow release metoprolol succinate (MERIT-HF), bisoprolol [Cardiac Insufficiency Bisoprolol Study (CIBIS II)] and carvedilol [Carvedilol Prospective Randomized Cumulative Survival (COPERNICUS) and Carvedilol Or Metoprolol European Trial (COMET)].293,294,296,297
Mineralocorticoid receptor antagonists. Low-dose mineralo-corticoid receptor antagonists (MRA) are indicated in patients with persistent symptoms [New York Heart Association (NYHA) Class II–IV] and an LVEF ≤35%, despite treatment with an ACE-I (or, if not tolerated, an ARB) and a beta-blocker. 298 The mortality benefits of spironolactone and eplerenone did not differ between patients with and without T2DM and heart failure.299,300 Surveillance of renal function is mandatory because of the increased risk of nephropathy in DM.
Diuretics. The effect of diuretics on mortality and morbidity has not been investigated, but these drugs are useful for the relief of dyspnoea and oedema in heart failure with fluid overload, irrespective of the ejection fraction (EF). Loop diuretics are recommended rather than thiazides, which have been shown to promote hyperglycaemia.
Ivabradine. In a placebo-controlled trial of 6558 patients (30% with T2DM) with heart failure in sinus rhythm and heart rate ≥70 bpm, ivabradine demonstrated a significant reduction in composite endpoints of cardiovascular death and hospital admission for worsening heart failure. The beneficial difference was similar in a prespecified subgroup analysis of patients with and without DM. 301
6.4 Non-pharmacological therapies
Cardiac resynchronization therapy and implantable cardioverter defibrillators. Cardiac resynchronization therapy reduces mortality in patients in NYHA function Class III–IV, with an LVEF ≤35% despite optimal pharma-cological treatment, in sinus rhythm and with a prolonged QRS duration (≥120–130 ms). 302 There is no reason to believe that the effect of resynchronization therapy should be different in patients with DM.
Cardiac transplantation is an accepted treatment for end-stage heart failure. The presence of DM is not a contra-indication, but stringent selection criteria are in place. DM was an independent risk factor for decreased 10-year survival in a registry study of 22,385 patients transplanted between 1987 and 1999. 303
6.5 Glucose-lowering treatment
The impact of various glucose-lowering drugs on T2DM patients with heart failure has been reviewed by Gitt et al. 340 The only drugs addressed by RCT were thiazolidinediones, while evidence on other compounds is largely based on subgroup analyses of larger intervention studies in systolic heart failure, observational studies or registries. The use of metformin has been considered to be contra-indicated because of concerns regarding lactic acidosis. This drug has, however, been associated with lower mortality, lower all-cause hospital admission, and fewer adverse events.305,306 When studied, accumulation of lactic acidosis was not verified. 307 In a nested case-control study including newly diagnosed heart failure and DM, the use of metformin [adjusted OR 0.65 (0.48–0.87)] or metformin with or without other agents [0.72 (0.59–0.90)] was associated with lower mortality, while other oral glucose-lowering agents or insulin were neutral in this respect. 308
Recommendations on sulphonylureas and heart failure are based on observational data. No relationship was seen between sulphonylurea and heart failure mortality in UKPDS, 70 but in the Saskatchewan Health database, mortality (52 vs. 33%) and hospitalizations (85 vs. 77%) were higher among patients treated with sulphonylureas than with metformin during an average 2.5 years of follow-up. 309 A similar difference was not confirmed in another study, which concluded there was no association between sulphonylurea or insulin use and mortality. 307
The thiazolidinediones induce sodium retention and plasma volume expansion, and the resulting fluid retention may provoke or worsen heart failure and cause increased hospitalization.99,310,311 There is a lack of information on the impact of GLP-1 analogues or DPP-4 inhibitors in patients with heart failure, although experimental and early clinical observations indicate favourable effects on myocardial performance. 312 A retrospective cohort study in 16,417 patients with DM and a primary diagnosis of heart failure did not reveal any association between the use of insulin and mortality (HR 0.96; 95% CI 0.88–1.05) when compared with several other classes of glucose-lowering drugs. 307 In the ORIGIN trial, subjects at high CVD risk plus IFG, IGT or T2DM received insulin glargine or standard care, which mainly included metformin and sulphonylurea treatment. During the 6.2-year-long follow-up period there was no difference in hospitalizations for heart failure. 89
Recommendations for management of heart failure in diabetes.

ACE-I = angiotensin converting inhibitor; ARB = angiotensin receptor blocker; LVEF = left ventricular ejection fraction;MRA = mineralocorticoid receptor antagonist; NYHA = New York Heart Association; T2DM = type 2 diabetes mellitus.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
7. Arrhythmias: atrial fibrillationand sudden cardiac death
7.1 Diabetes mellitus and atrial fibrillation
Individuals with atrial fibrillation (AF) are at substantially increased risk of stroke and have twice the mortality rate from CVD, compared with those in sinus rhythm.315,316 Community studies demonstrate the presence of DM in 13% of patients with AF, 317 who share common predisposing factors, such as hypertension, atherosclerosis, and obesity. In the Manitoba Follow-up Study of 3983 males, 318 DM was significantly associated with AF with a relative risk of 1.82 in univariate analysis. In the multivariate model, the association with DM was non-significant, suggesting that the increased risk may relate to ischaemic heart disease, hypertension or heart failure. A multicentre study of 11,140 DM patients confirmed that AF is common in T2DM and demonstrated that, when they co-exist, there is a higher risk of all-cause mortality, cardiovascular death, stroke, and heart failure. 319 These findings suggest that AF identifies DM subjects likely to obtain greater benefits from aggressive management of all cardiovascular risk factors. Because AF is asymptomatic—or mildly symptomatic—in a substantial proportion (about 30%) of patients, screening for AF can be recommended in selected patient groups with T2DM where there is any suspicion of paroxysmal or permanent AF by pulse palpation, routine 12-lead ECG, or Holter recordings.
Diabetes and risk of stroke in atrial fibrillation. Two recent systematic reviews have addressed the evidence base for stroke risk factors in AF and concluded that prior stroke/TIA/thromboembolism, age, hypertension, DM, and structural heart disease are important risk factors.320,321
Diabetes and stroke risk stratification schemes: The simplest scheme is the cardiac failure, hypertension, age, diabetes, stroke (doubled) (CHADS2) risk index. The 2010 ESC Guidelines for the management of AF, updated 2012, proposed a new scheme. The use of ‘low’, ‘moderate’, and ‘high’ risk has been re-emphasized, recognizing that risk is a continuum.322,323 The new scheme is expressed as an acronym “CHA2DS2VASc” [cardiac failure, hypertension, age ≥75 (doubled), DM, stroke (doubled)-vascular disease, age 65–74 and sex category (female)]. It is based on a points system, in which two points are assigned for history of stroke or TIA, or age ≥75 years, and one point for the other variables. Heart failure is defined either as clinical heart failure or LV systolic dysfunction (EF <40%), and vascular disease as a history of MI, complex aortic plaque, or PAD.
Antithrombotic therapy in diabetes patients: A meta-analysis of 16 RCTs in 9874 patients reported that oral anticoagulation was effective for primary and secondary prevention of stroke in studies comprising with an overall 62% reduction of relative risk (95% CI 48–72). 324 The absolute risk reduction was 2.7% per year for primary prevention and 8.4% per year for secondary prevention. Major extracranial bleeds were increased by anticoagulant therapy by 0.3% per year. Aspirin reduced risk of stroke by only 22% (95% CI 2–38), with an absolute risk reduction of 1.5% per year for primary prevention and 2.5% per year for secondary prevention. In five trials comparing anticoagulant therapy with antiplatelet agents in 2837 patients, warfarin was more effective than aspirin, with an RRR of 36% (95% CI 14–52). Oral anticoagulation with vitamin K antagonists (VKAs) or one of the new oral anticoagulants (see below) is recommended in patients with AF,322,323 and should be used in DM patients with AF unless contra-indicated and if accepted by the patient. With the use of VKA, an international normalized ratio (INR) of 2.0–3.0 is the optimal range for prevention of stroke and systemic embolism in patients with DM. A lower target INR (1.8–2.5) has been proposed for the elderly, but this is not based on evidence. In the Atrial fibrillation Clopidogrel Trial with Irbesartan for prevention of Vascular Events (ACTIVE W), warfarin was superior to clopidogrel plus aspirin (RRR 0.40; 95% CI 18–56), with no difference in rates of bleeding. 325 The aspirin arm found that major vascular events were reduced in patients receiving aspirin-plus-clopidogrel, compared with aspirin monotherapy (RR 0.89; P = 0.01). 326 Thus, aspirin-plus-clopidogrel therapy may be considered as an interim measure if a VKA is unsuitable, but not in patients at high bleeding risk. Combinations of VKA with antiplatelet therapy do not offer added benefits and lead to more bleeding, 322 and such combinations should be avoided.
Two new classes of anticoagulants have been developed: oral direct thrombin inhibitors (e.g. dabigatran) and oral factor Xa inhibitors (e.g. rivaroxaban, apixaban, edoxiban and betrixiban). These new drugs have the potential to be used as an alternative to warfarin, especially in patients intolerantto, or unsuitable for, VKAs. In analyses of prespecified subgroups in the Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET) trial, patients with DM had a protection similar to the overall study populations. 327
An assessment of bleeding risk should be carried out before starting anticoagulation. Using a cohort of 3978 European subjects with AF from the Euro Heart Survey, a simple bleeding score known as ‘Hypertension, Abnormal renal/liver function (1 point each), Stroke, Bleeding history or predisposition, Labile INR, Elderly (>65), Drugs/alcohol concomitantly (1 point each)’ (HAS-BLED) was developed, 328 which includes hypertension, abnormal renal/ liver function, stroke, bleeding history or predisposition, labile international normalized ratio, elderly (>65 years), and drugs/alcohol as risk factors of bleeding. A score ≥3 indicates high risk and some caution and regular review of the patients is needed following initiation of antithrombotic therapy.
7.2 Sudden cardiac death
General population studies show that subjects with DM are at higher risk of sudden cardiac death, which accounts for approximately 50% of all cardiovascular deaths. The majority are caused by ventricular tachyarrhythmia, often triggered by ACS, which may occur without known cardiac disease or in association with structural heart disease.329,330 In the Framingham study, DM was associated with an increased riskof sudden cardiac death in all ages (almost four-fold) and was consistently greater in women than in men. 331 The Nurses’ Health Study, 332 which included 121 701 women aged 30–55 years, followed for 22 years, reported that sudden cardiac death occurred as the first sign of heart disease in 69% of cases. The incidence of sudden cardiac death in post-infarction patients with DM and a LVEF >35% was equal to that of non-DM patients with an EF ≤35%. T2DM patients with congestive heart failure or post-MI should have their LVEF measured to identify candidates for prophylactic implantable cardioverter defibrillator therapy. Similarly, secondary prophylaxis with implantable cardioverter defibrillator therapy is indicated in DM patients resuscitated from ventricular fibrillation or sustained ventricular tachycardia, as recommended in the Guidelines. 333 All post-infarction patients with heart failure should also be treated with β-blocking drugs, which reduce sudden cardiac death.329,330 Jouven et al. 334 studied the RR of sudden cardiac death in groups of patients with different degrees of dysglycaemia and showed that higher values of glycaemia led to higher risk. Following adjustment, even patients with borderline DM—defined as non-fasting glycaemia between 7.7 and 11.1 mmol/L (140 and 200 mg/dL)—had an increased risk of sudden cardiac death (OR 1.24, compared with patients with normoglycaemia). The presence of microvascular disease and female gender increased risk in all groups. A recent study showed that autonomic markers, such as heart rate turbulence and deceleration capacity from 24-h Holter recordings, predict the occurrence of cardiac death and sudden cardiac death among T2DM patients with recent MI. 335
Recommendations for the management of arrhythmias in patients with diabetes mellitus.
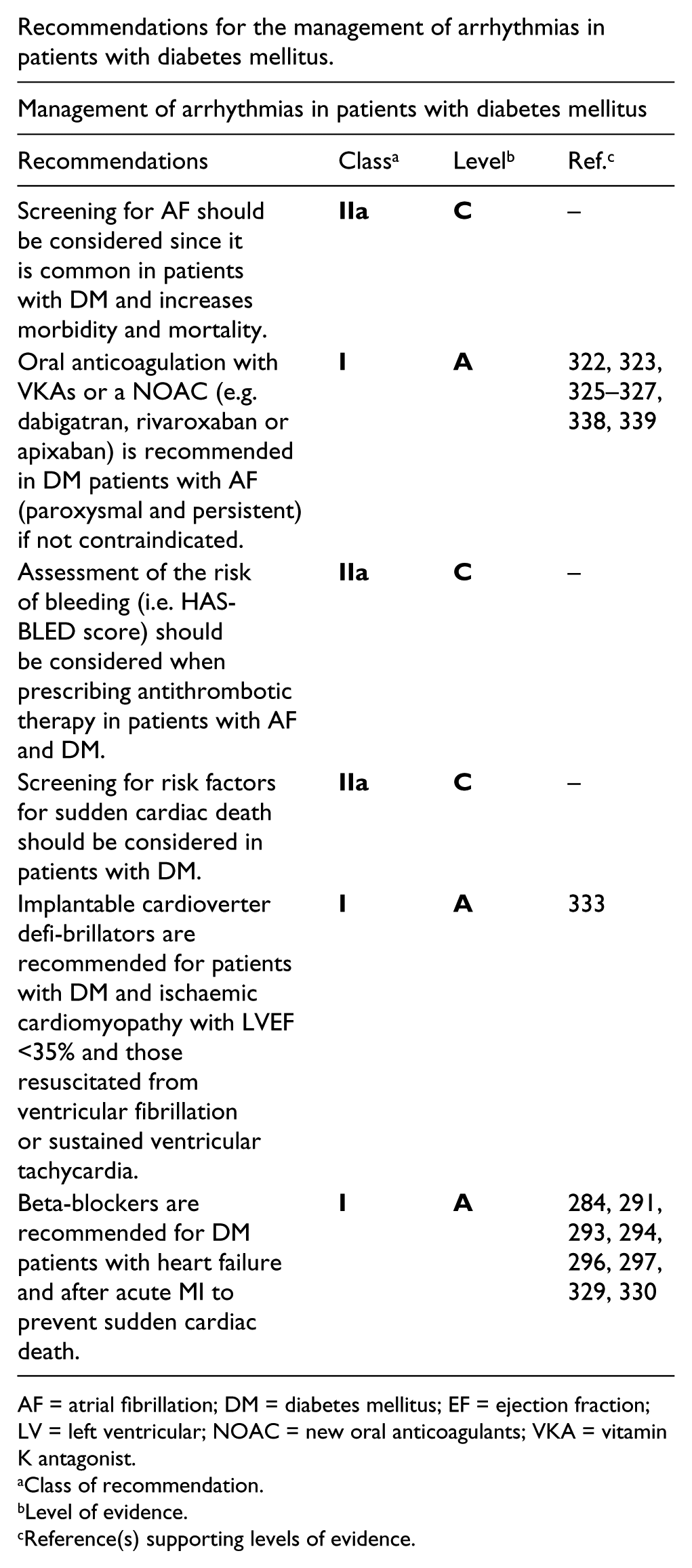
AF = atrial fibrillation; DM = diabetes mellitus; EF = ejection fraction; LV = left ventricular; NOAC = new oral anticoagulants; VKA = vitamin K antagonist.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
Cardiovascular autonomic neuropathy was significantly associated with subsequent mortality in people with DM in a meta-analysis of 15 studies. 336 The MONICA/KORA (World Health Organisation Monitoring Trends and Determinants in Cardiovascular Disease/ Kooperative Health Research in the Region Augsburg) study reported that QTc was an independent predictor of sudden death associated with a three-fold increase in patients with DM and a two-fold increase in those without. 337 Measurements of heart rate variability and QTc may become valuable as predictors of sudden cardiac death in DM patients but evidence to support this as a general recommendation is still lacking.
8. Peripheral and cerebrovascular disease
8.1 Peripheral artery disease
Diabetes mellitus is a risk factor for the development of atherosclerosis at any vascular site, but particularly for lower extremity artery disease (LEAD), which it increases risk two- to four-fold, and for carotid artery disease. In LEAD, cigarette smoking, DM, and hypertension are important risk factors. Although the association of DM with LEAD is inconsistent on multivariable analysis, it appears that duration and severity of DM particularly affect risk of gangrene and ulceration.340,341 In population studies, the presence of carotid artery stenosis was associated with DM and other classical risk factors, irrespective of age.342 –344 DM is present in a significant proportion of patients with multisite atherosclerosis, who have a worse prognosis than those with a single disease location.345,346 Patients with DM should undergo comprehensive screening for the presence of PAD at different vascular sites. Medical history and physical examination are the cornerstones of diagnostic workup and should include a review of the different vascular beds and their specific symptoms, 347 although many patients remain asymptomatic. Further diagnostic evaluation and treatment should be applied according to the ESC Guidelines on PAD. 347 Briefly, in all DM patients, clinical screening to detect PAD should be performed annually and lifestyle changes encouraged. 348 All patients with PAD should receive adequate lipid-lowering, antihypertensive and antiplatelet treatment,186,349 –351 with optimal glycaemic control.72,200,352
8.1.1 Lower extremity artery disease
Vascular obstructions are often located distally in patients with DM and typical lesions occur in the popliteal artery or in the vessels of the lower leg. In a cohort of 6880 patients over 65 years, one in five patients had LEAD, though only 10% were symptomatic. 353 The incidence and prevalence of LEAD increase with age and duration of DM. The National Health and Nutrition Examination Survey (NHANES II) determined pulse amplitudes in adults, and diminished or absent pulsation of the dorsalis pedis artery was found in 16% of adults with DM aged 35–54 years and in 24% of those aged 55–74. 354 In many older patients, LEAD is present at the time of diagnosis of DM. Progression of LEAD may result in foot ulceration, gangrene and, ultimately, amputation. DM accounts for approximately 50% of all non-traumatic amputations in the United States and a second amputation is common. Mortality is increased in patients with LEAD and three-year survival after an amputation is less than 50%. 351 Early diagnosis is important for the prevention of progression of LEAD and for prediction of overall cardiovascular risk.
Diagnosis. Symptoms suggestive of claudication are walking impairment, e.g. fatigue, aching, cramping, or pain with localization to buttock, thigh, calf, or foot, particularly when symptoms are quickly relieved at rest. An objective measure of LEAD is the ankle-brachial index (ABI), calculated by dividing the systolic blood pressure at the posteriortibial or dorsalis pedal level with the brachial systolic blood pressure. An index of <0.9 is suggestive of LEAD, particularly in the presence of symptoms or clinical findings such as bruits or absent pulses. An ABI <0.8 indicates PAD, regardless of symptoms. Sensitivity of ABI measurement may be increased after exercise. Postexercise ABI may identify significant LEAD, even in subjects with a normal resting ABI. 355 An ABI >1.40 indicates poorly compressible vessels as a result of stiff arterial walls (medial calcinosis) that can impede the estimation of arterial pressure in the artery.
Primary and secondary prevention of LEAD in patients with DM consists of lifestyle changes (addressing obesity, smoking and lack of exercise) and control of risk factors, including hyperglycaemia, hyperlipidaemia and hypertension.
Treatment. In a systematic review of RCTs of exercise programmes in symptomatic claudication, supervised exercise therapy was effective in increasing walking time, compared with standard care. 356 Although cilostazol, naftidrofuryl and pentoxifylline increase walking distance in intermittent claudication, their role remains uncertain. In addition, statin therapy has been reported to be beneficial by increasing walking distance in patients with PAD.347,357 If conservative therapy is unsuccessful, revascularization should be considered. In case of disabling claudication with culprit lesions located at aorta/iliac arteries, revascularization should be the first choice, along with risk factor management. 347
Critical limb ischaemia. (CLI) is defined by the presence of is chaemic pain at rest and ischaemic lesions or gangrene attributable to arterial occlusive disease that is chronic and distinguishable from acute limb ischaemia. Importantly, β-blockers are not contra-indicated in patients with LEAD and DM. A meta-analysis of 11 RCTs found that β-blockers do not adversely affect walking capacity or symptoms of intermittent claudication in patients with mild-to-moderate PAD. 358 At 32-month follow-up of 490 patients with PAD and prior Ml, β-blockers caused a 53% significant and independent decrease in new coronary events. 359 Comprehensive management requires multidisciplinary care to control atherosclerotic risk factors, provision of revascularization where possible, optimization of wound care, appropriate shoe wear, treatment of infection, and rehabilitation. 347 The cornerstone of management is arterial reconstruction and limb salvage. Medical baseline therapy, including platelet inhibitors and statins, should be initiated according to principles outlined elsewhere in this document.347,360,361
The choice of revascularization strategy depends primarily on the anatomy of the arterial lesion. Outcomes of endovascular iliac artery repair in DM have been reported as similar to, or worse than, those without DM, and long-term patency is lower. 362 Long-term patency rates of intravascular interventions in the tibio-peroneal region are low in patients with and without DM, but may be sufficient in the short term to facilitate healing of foot ulcers. 362
The diabetic foot is a specific clinical entity that may involve neuropathy, trauma, arterial disease, infection and inflammation, often in combination. The serious consequences are ulceration, gangrene, and high rates of amputation. In DM patients, LEAD is typically diffuse, and particularly severe in distal vessels. When the ABI is inconclusive, toe pressure, distal Doppler waveform analyses, or transcutaneous oxygen can assess the arterial status. When ischaemia is present, imaging should be used to plan revascularization, employing the same criteria as for CLI. Follow-up includes patient education, smoking cessation, protective shoes, periodic foot care, and recon- structive foot surgery as needed. The management of risk factors and revascularization surveillance are mandatory. 363
8.1.2 Carotid artery disease
Diabetes mellitus is an independent risk factor for ischaemic stroke with an incidence 2.5–3.5 times higher than in non-DM.364,365 The discussion of stroke and transient ischaemic attack (TIA) prevention will be limited to the aspects relating to carotid artery disease, which is causally related to about 20% of all ischaemic strokes. 366 Although DM increases the likelihood of carotid artery disease, it does not change the general diagnostic and therapeutic approach.
Recommendations for management of peripheral artery disease in diabetes.
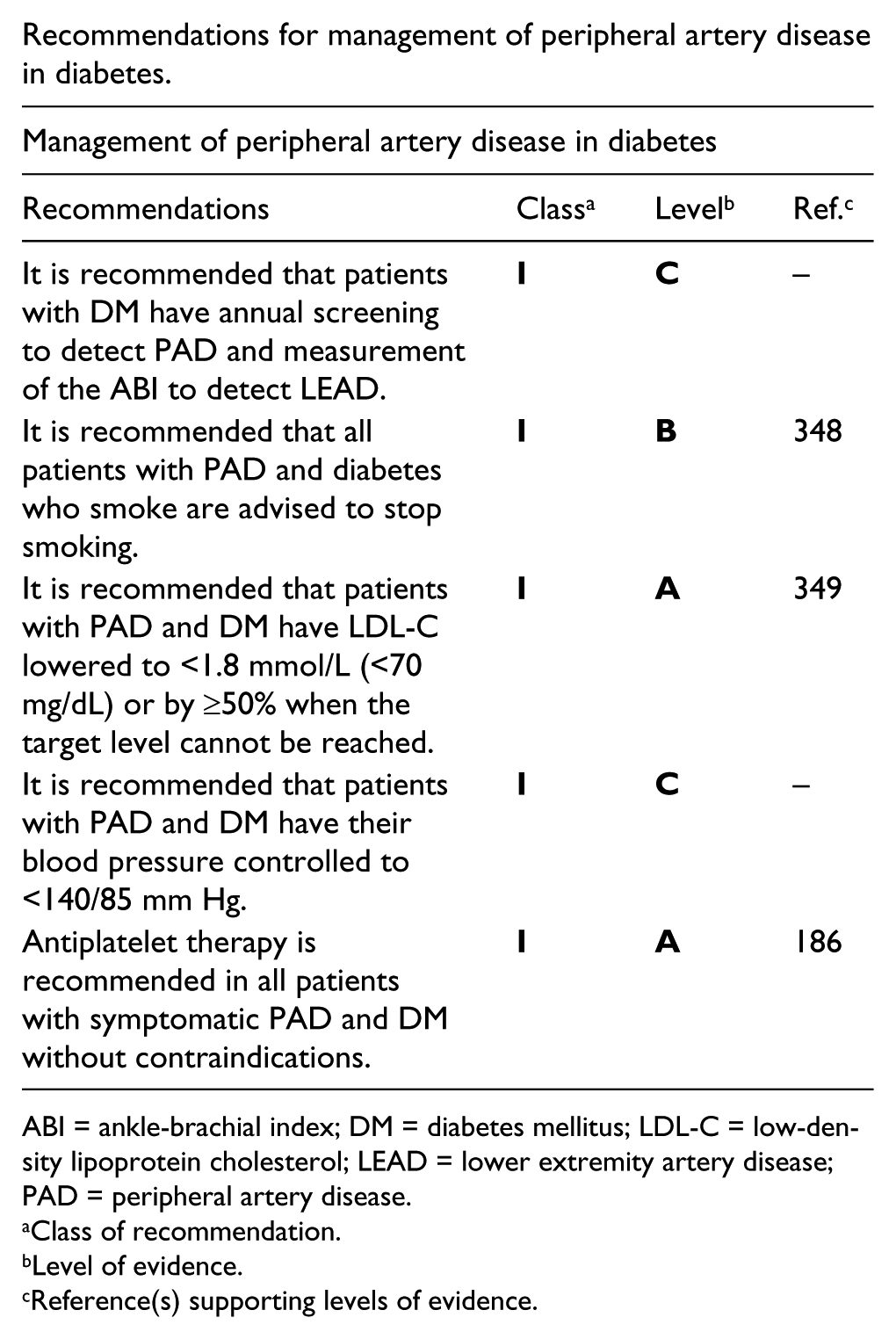
ABI = ankle-brachial index; DM = diabetes mellitus; LDL-C = low-density lipoprotein cholesterol; LEAD = lower extremity artery disease;
PAD = peripheral artery disease.
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
Diagnosis. Carotid bruits are common although many remain asymptomatic, regardless of lesion severity. Although the spectrum of symptoms is wide, only those who have suffered a stroke or TIA within the past six months are regarded as symptomatic.367,368 In this group of patients, the probability of recurrent stroke or TIA is high. 369 Therefore urgent imaging of the brain and supra-aortic vessels is mandatory in patients presenting with TIA or stroke. Duplex ultrasonography, computed tomography angiography, and magnetic resonance imaging are indicated to evaluate carotid artery stenosis.
Treatment. Whilst carotid endarterectomy seems to offer a clear advantage over conservative treatment in patients with symptomatic carotid artery disease, the role of revascularization in asymptomatic patients remains less clear. 347 It needs to be emphasized that most data in symptom-free patients were collected before statins and antiplatelet agents became standard therapy.
9. Patient-centered care
The importance of multifactorial risk assessment and lifestyle management, including diet and exercise, in the prevention and treatment of DM and CVD has been emphasized in earlier sections. However, supporting patients in achieving and maintaining lifestyle changes on an individualized basis, using defined therapeutic goals and strategies, continues to be a substantial challenge.
Recommendations for patient-centred care in diabetes.
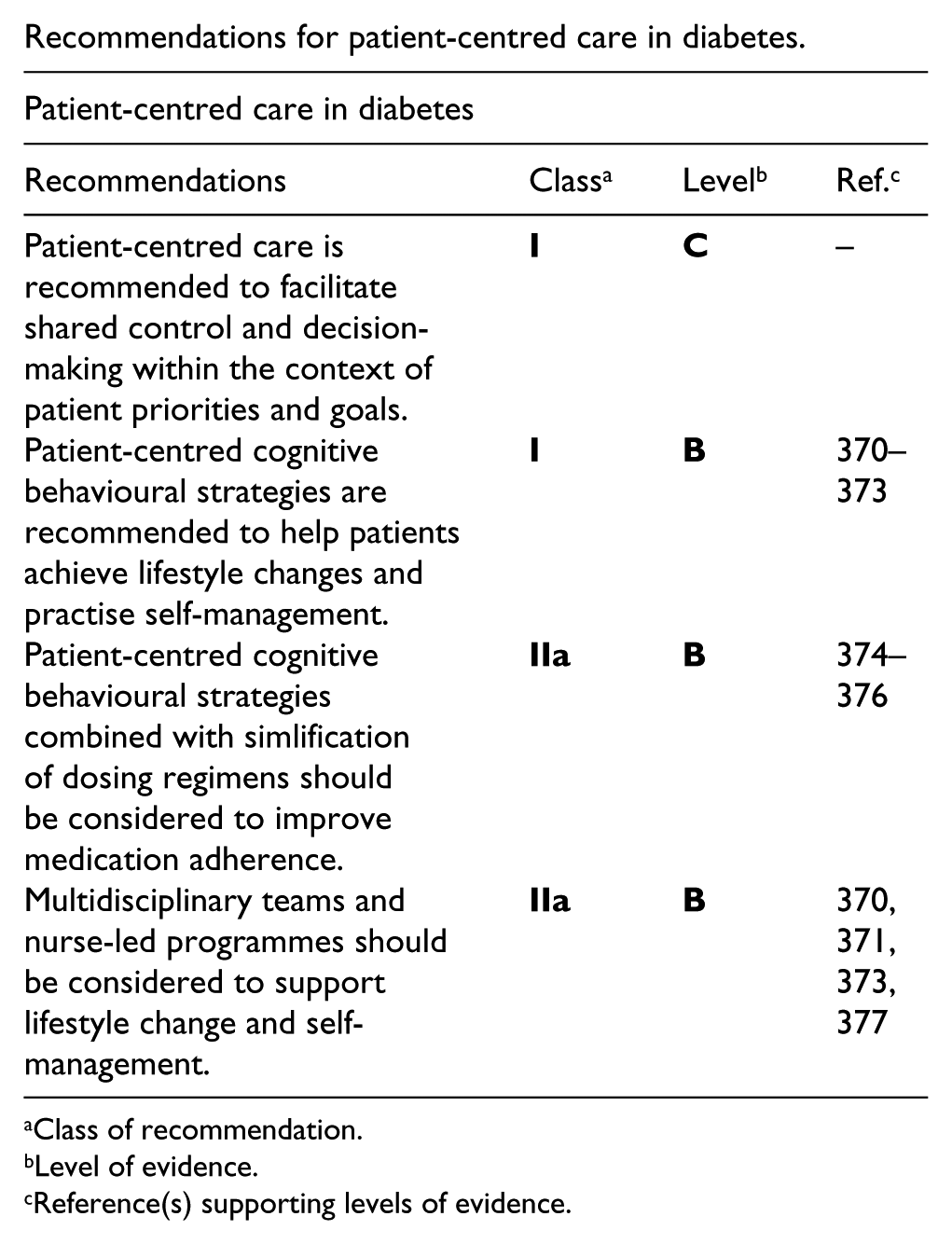
Class of recommendation.
Level of evidence.
Reference(s) supporting levels of evidence.
The CME text ‘2013 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy’ is accredited by the European Board for Accreditation in Cardiology (EBAC). EBAC works according to the quality standards of the European Accreditation Council for Continuing Medical Education (EACCME), which is an institution of the European Union of Medical Specialists (UEMS). In compliance with EBAC/EACCME Guidelines, all authors participating in this programme have disclosed any potential conflicts of interest that might cause a bias in the article. The Organizing Committee is responsible for ensuring that all potential conflicts of interest relevant to the programme are declared to the participants prior to the CME activities.
CME questions for this article are available at: European Heart Journal http://www.oxforde-learning.com/eurheartj and European Society of Cardiology http://www.escardio.org/guidelines.
