Abstract
The aim of the study was to investigate the association of the extracellular inhibitors of Wnt/β-catenin signalling sclerostin and Dickkopf-1 (Dkk-1) with carotid intima-media thickness (CIMT) in type 2 diabetes mellitus (T2DM). We performed a cross-sectional study including 40 T2DM postmenopausal women and 40 healthy controls. CIMT was measured by B-mode ultrasound. Serum sclerostin and Dkk-1 were measured by solid-phase enzyme-linked immunosorbent assay (ELISA). Serum sclerostin and Dkk-1 concentrations were significantly higher in T2DM group than in controls. There was a significant negative correlation between sclerostin and Dkk-1 and CIMT in T2DM (p = 0.0063 and p = 0.0017, respectively). After adjustment for potential confounders, associations remained significant only for sclerostin. These data suggest that sclerostin, an established modulator of the canonical Wnt signalling, may protect against progression of vascular complications in diabetic patients, possibly by attenuating upregulation of β-catenin activity in the vascular cells.
Introduction
Type 2 diabetes mellitus (T2DM) is associated with atherosclerosis and with consequent two to four times increased risk of cardiovascular events.1–3 Atherosclerosis induces morphological and functional alterations of the arterial wall such as proliferation of vascular smooth muscle cells (VSMCs), thickening of the arterial wall, atheroma plaque formation and eventually vascular calcification leading to macrovascular disease in T2DM. 4 T2DM is also associated with an increased risk of fracture that is higher than in the general population at a given bone mineral density. 5 In recent years, evidence from animal and human studies has suggested a relationship between bone turnover modifications and vascular disease. The interest towards this association has increased since the observation that the Wnt/β-catenin signalling pathway is involved in the regulation of both bone and vascular metabolism. 6 A growing body of evidence has in fact confirmed that its activity is not limited to bone formation but is also involved in other metabolic processes. Wnt signalling, although quite novel in comparison to other vascular signal pathways, has the capacity to promote vascular quiescence and stability under pathological conditions, suggesting an emerging function in the vasculature. 7 Data from multiple laboratories have demonstrated that β-catenin activation is a key component of arteriosclerotic physiology, particularly in diabetic arteriosclerosis.8–11 The endogenous regulators of the canonical Wnt signalling include extracellular and intracellular antagonists. Most soluble extracellular inhibitors are secreted factors that interact with either Wnt receptors, such as sclerostin and Dickkopf-1 (Dkk-1), or Wnt ligands, such as secreted frizzled-related proteins (SFRPs) and Wnt inhibitory factor 1/2 (Wif-1). Thambiah et al. 12 reported that circulating levels of Dkk-1 are inversely associated with arterial stiffness in pre-dialysis chronic kidney disease, whereas no significant correlation was found between arterial stiffness and sclerostin in the same patients. Four studies, including one from our group,13–16 reported that circulating sclerostin is increased in T2DM compared to non-diabetic individuals, independent of age and gender. The increase in sclerostin serum levels is consistently associated with a decrease in bone resorption markers. Recently, Morales-Santana et al. 17 reported that in T2DM patient, sclerostin serum levels are independently associated with surrogate markers of atherosclerotic disease such as abnormal carotid intima-media thickness (CIMT) and with carotid plaque and aortic calcification. All these evidences confirm that the Wnt signalling is under complex regulation, which needs to be further explored. The aim of this cross-sectional clinical study, which reports an additional subanalysis of our previous study, 16 was to evaluate in T2DM patients the possible association of circulating sclerostin and Dkk-1 with CIMT, generally considered an important non-invasive indicator of (subclinical) atherosclerosis.18,19
Materials and methods
Study subjects
We enrolled 40 postmenopausal women with T2DM and 40 gender- and age-matched healthy subjects recruited from the general community as a control group. T2DM patients were recruited consecutively from those referred to the Diabetes Unit of our Clinic. Diabetes was defined according to American Diabetes Association criteria. 20 Exclusion criteria were the following: (1) cerebrovascular disease (ischaemic stroke or transient ischaemic attack), coronary heart disease (myocardial infarction, diagnosed angina, coronary revascularization surgery) or peripheral arterial disease; (2) known diseases affecting bone (Paget’s disease, rheumatoid arthritis, hyperparathyroidism, hypercortisolism, renal bone disease); (3) use of drugs affecting bone metabolism in the last 12 months, including bisphosphonates (in the last 3 years), and vitamin D preparations and (4) use of thiazolidinediones in the last 12 months. All patients were on medication for diabetes, including metformin, sulphonylureas, insulin or a combination of drugs. Ethical approval was obtained from the University Hospital Ethical Committee. Informed consent was obtained from all subjects before entering the study, which was conducted in accordance with the Declaration of Helsinki. In all subjects, we measured height and weight and calculated body mass index (BMI).
Laboratory data
Laboratory assays were performed in serum samples obtained in the morning after an overnight fast and stored at −30°C until examination. Serum concentrations of creatinine and glycated haemoglobin (HbA1c) were measured using standard automated laboratory techniques. Estimated glomerular filtration rate (eGFR) was calculated from serum creatinine using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation. 21 Sclerostin and Dkk-1 were measured by enzyme immunoassay using commercially available enzyme-linked immunosorbent assay (ELISA) kits (Biomedica Medizinprodukte, Wien, Austria). Intra- and inter-assay coefficients of variation (CVs) for sclerostin were 5% and 4%, respectively. The detection limit was 2.6 pmol/L. Mean serum concentration in healthy subjects older than 50 years in our laboratory was 43 ± 10.82 pmol/L. The detection limit for Dkk-1 was 0.38 pmol/L. Mean serum concentration in healthy subjects in our laboratory was 9.02 ± 4.44 pmol/L.
Measurement of CIMT
Each subject’s carotid artery ultrasonographic examination was performed at room temperature 22°C–25°C, with subjects in the lying position, with the head elevated 45°–50° and a side tilt of 30° to the right and then the same to the left using the LOGIQ C5 Premium apparatus (GE Healthcare, WI, USA). CIMT was defined as the mean of 10 wall measurements of the viewable distance between the lumen–intima and the media–adventitia interface on both the left and right sides. M-mode examinations were performed 1 cm proximal to the flow divider on the distal wall of the common carotid artery bilaterally at sites free of any discrete plaques. Measurements were taken over three cardiac cycles by a single trained sonographer (I.P.). If a discrepancy was observed in measurement values between longitudinal and vertical sections, the smaller value was selected to avoid overestimation. Reproducibility was high, with a CV value of measurement of IMT as low as 3.4% (Pearson’s correlation coefficient of intraobserver reproducibility of 0.97). The CIMT was measured in millimetres and was defined according to the Atherosclerosis Risk in Communities (ARIC) study. 22 CIMT was defined as pathologic if it was more or equal to 0.9 mm and was considered carotid atherosclerosis if the maximum intima-media thickness of carotid bifurcation (BIF-IMT) was ≥1.2 mm.
Statistical analysis
Statistical calculations were performed using the GraphPad InStat version 4 for Windows (GraphPad Software, Inc., San Diego, CA, USA). Data for continuous variables were expressed as mean ± standard deviation (SD). The normal distribution of values for different parameters was verified using the Kolmogorov–Smirnov test. Comparisons between groups were performed using Student’s t-test or the Mann–Whitney U test, as appropriate. Pearson’s correlation coefficient (normal distribution) or Spearman test (non-normal distribution) was used for univariate analyses in association studies. Correlation coefficients between CIMT and metabolic features were calculated by partial correlation analysis. Multivariate linear regression models were used to estimate the determinants of CIMT. All models were adjusted for age, BMI and fasting glucose. Multicollinearity was examined through calculation of the variance inflation factor (VIF), computed by the following formula: VIF = 1/(1 − R2); a value of VIF ≤ 1 is assumed to exclude multicollinearity for x variables. In all statistical analyses, p < 0.05 was considered to be statistically significant.
Results
As previously reported, 16 there were no differences between T2DM patients and controls for age (63.68 ± 8.39 vs 62.12 ± 7.99 years), menopausal age (49.26 ± 5.05 vs 48.45 ± 4.98 years), smoking habit (2.5% vs 5%), serum level of creatinine (0.92 ± 0.18 vs 0.90 ± 0.13 mg/dL) and eGFR (67.7 ± 5.1 vs 69.5 ± 5.7 mL/min/1.73 m2). As expected, T2DM patients had significant higher BMI (31.16 ± 5.06 vs 26.55 ± 4.77 kg/m2, p < 0.001) and HbA1c serum levels (7.27% ± 0.51% vs 4.94% ± 0.48%, p < 0.001) than controls. Serum sclerostin (53.18 ± 10.94 vs 47.50 ± 12.62 pmol/L) and Dkk-1 (12.90 ± 10.27 vs 9.07 ± 5.68 pmol/L) concentrations were significantly higher in T2DM patients than in controls (p < 0.05 for both comparisons). We found a significant difference in maximal CIMT in T2DM compared to controls (0.867 ± 0.214 vs 0.784 ± 0.107 mm, p < 0.05). In univariate analyses, sclerostin correlated positively with age in both patients and controls (p < 0.001) and with years since diagnosis in T2DM patients (p < 0.001). Partial correlation analysis demonstrated that serum sclerostin was inversely correlated with CIMT in diabetic patients after adjusting for age (r = −0.42; p = 0.006). No significant association between sclerostin and CIMT was observed in controls (p = 0.874) (Figure 1(a) and (b)). Dkk-1 serum levels were inversely correlated with CIMT in T2DM (r = −0.48; p = 0.001). The association was not significant in controls with p = 0.558 (Figure 1(c) and (d)). No significant correlations were found between CIMT, age, BMI and HbA1c in T2DM patients and controls (Table 1). Sclerostin and Dkk-1 correlated significantly in control subjects (r = 0.35, p = 0.02), but not in T2DM patients (r = −0.13, p > 0.05). In the multivariate linear regression model, CIMT was set as dependent variable, while age, BMI, sclerostin and Dkk-1 were set as independent variables. We found that only sclerostin [β = −0.009602; p = 0.0019; standard error (SE) = 0.002851] was an independent predictor of CIMT in T2DM patients. The calculated VIF shows that all R2 are <0.75, confirming that the x variables are independent of each other, excluding multicollinearity problems.
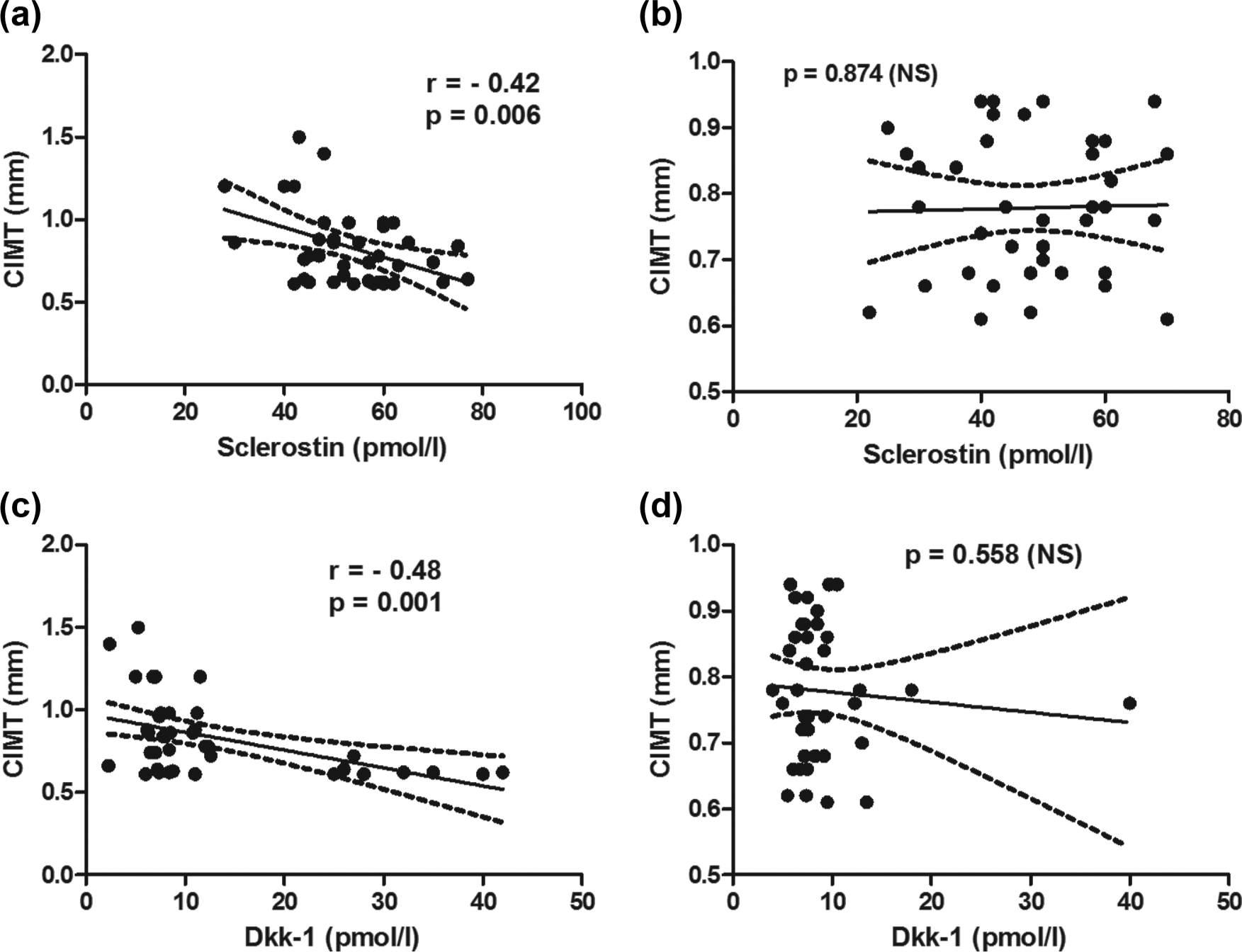
Univariate correlation between sclerostin and CIMT and between Dkk-1 and CIMT in (a, c) T2DM patients and (b, d) controls. Dotted lines indicate 95% confidence intervals.
Correlations of CIMT in T2DM patients and controls.
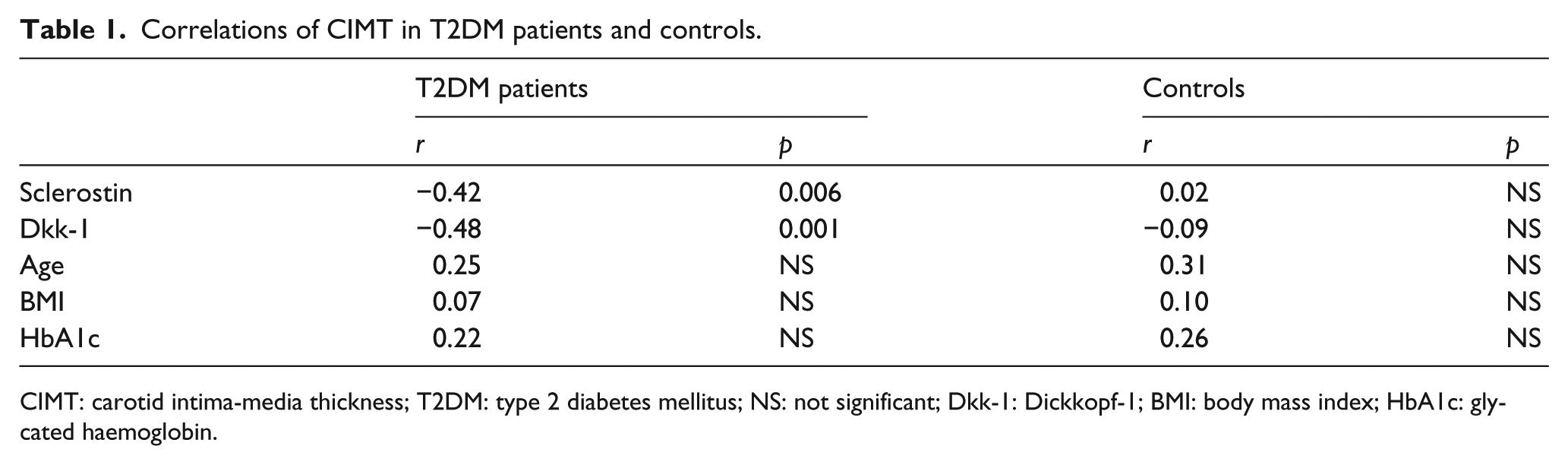
CIMT: carotid intima-media thickness; T2DM: type 2 diabetes mellitus; NS: not significant; Dkk-1: Dickkopf-1; BMI: body mass index; HbA1c: glycated haemoglobin.
Discussion
Our data confirm previous studies reporting that serum levels of sclerostin and Dkk-1 are greater in subjects with T2DM and characterize the relationships between these products and CIMT, which is considered an established measurement method for detection of subclinical atherosclerosis. Adjusting for potential confounders, sclerostin and Dkk-1 levels showed inverse significant association with CIMT. The negative association between sclerostin and CIMT contrasts with the findings by Morales-Santana et al. 17 We used the same kits to measure sclerostin, and therefore, we do not believe that differences in the methodology for measuring sclerostin explain the differences in the findings from our study and that of Morales-Santana. Gender difference may influence association as sclerostin levels are higher in males compared with females. 23 Deterioration of kidney function may be an important contributor to increased sclerostin levels, thus acting as a possible confounder of the association with arterial stiffness. Moreover, we examined only diabetic postmenopausal women; this fact together with the difference in diabetic duration (14.3 years in the subjects studied by Morales-Santana versus 10.03 years in our population) may influence both sclerostin serum levels and its association with CIMT. However, the nature of association between sclerostin and indicators of subclinical atherosclerosis appears controversial. In fact, Hampson et al. 6 recently reported that in postmenopausal women, sclerostin shows a significant positive association with pulse wave velocity, a recognized marker of arterial stiffness, 24 but that this association was lost after adjustment for age, lipid concentration and blood pressure. Although the higher levels of sclerostin observed in T2DM patients suggest a specific role of sclerostin, the biological relevance of serum sclerostin in this condition is not fully understood. Sclerostin plays a physiological role as a negative regulator of the canonical Wnt signalling pathway, whose hallmark is the stabilization of β-catenin in the cytoplasm upon activation. Extracellular inhibitors include Wif 1/2, SFRPs, Dkk-1 and sclerostin. This latter factor binds the coreceptor lipoprotein receptor-related proteins (LRP) 5/6 competitively, inhibiting the binding of Wnt ligands to LRP 5/6. Dkk-1, on the other hand, forms a complex with LRP 6, inducing internalization and degradation of LRP, thereby lowering the availability of LRP for Wnt ligand binding. 25 The logical consequence of any increased availability of both sclerostin and Dkk-1 would be a disruption of the Wnt signalling cascade. In the vascular cells, Wnt signalling also crosstalks with other signalling pathways: β-catenin activity in the vasculature, under control by N-cadherin and reactive oxygen species (ROS), 26 drives VSMC proliferation and neointima formation, 27 giving support to the hypothesis that β-catenin activation is a key component of arteriosclerotic physiology. Atherosclerosis is an actively regulated process, which tends to increase with time, so it is tempting to speculate that high sclerostin levels might be indicative of a sort of defensive mechanism that may attenuate upregulation of the canonical Wnt pathway stimulated by peroxide via an ROS-sensitive nucleoredoxin-dishevelled relay. 28 In this case, sclerostin is likely inversely correlated to CIMT. The behaviour of Dkk-1, which should play almost the same role of sclerostin in defensive response against the activation of the Wnt pathway in vascular cells, seems less clear, as the significance of the negative association between Dkk-1 and CIMT was lost in multivariate analysis. Our observation seems in line with the recent report by Claes et al., 29 who showed that in patients with chronic kidney disease not yet on dialysis, higher circulating sclerostin levels were associated with a lower risk for vascular calcification and that the absence of vascular calcification was an independent determinant of higher sclerostin levels, suggesting that sclerostin may represent a counterregulatory mechanism to retard the progress of vascular calcification. We would like to stress, however, that longitudinal studies characterizing the correlation between sclerostin and the progression of atherosclerosis in T2DM patients are necessary to better understand the nature of this relationship. Limitations of this study include the cross-sectional nature of study measurements, which did not allow us to follow the temporal sequence between concentration of extracellular inhibitors of Wnt signalling and progression of the vascular outcome measures. Second, the sample size is small and might reduce statistical power. Furthermore, the study population consisted of women only. However, the hypothesis that high levels of sclerostin may have a protective effect on vessels, if confirmed by larger studies, underscores the need for future investigation to better clarify the role of the Wnt/β-catenin pathway in the pathogenesis of atherosclerotic disease.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
