Abstract
Objective:
Adiponectin is produced by adipose tissue and regarded as protective hormone for diabetes and coronary heart disease (CHD). Its role in heart failure is discussed controversially.
Methods:
In this study, 1015 consecutive patients admitted for acute (
Results:
Adiponectin was higher in patients with normal glucose tolerance (NGT) (13.65 ± 10.31 mg/l) compared to impaired glucose tolerance (IGT) (11.12 ± 7.5,
Conclusion:
Adiponectin levels are inversely correlated to progressing CHD and glucose intolerance but positively correlated to increasing heart failure.
Introduction
Adiponectin is an adipocyte-secreted protein associated with insulin resistance, increased body weight and cardiovascular dysfunction and may have anti-inflammatory properties. 1 It was originally identified by four independent groups and also called as Acrp30, GBP28, apM1 and AdipoQ.2–5 Low adiponectin levels predict future risk of developing type 2 diabetes. 6 Moreover, adiponectin levels are decreased in patients with coronary heart disease (CHD), 7 and high adiponectin levels predict a lower risk of future myocardial infarction. 8 However, high adiponectin levels are surprisingly associated with increased risk of recurrent cardiovascular events 9 and mortality in patients with myocardial infarction 10 and heart failure.11–13 Therefore, it may be difficult to use adiponectin levels in individual patients to predict risk of cardiovascular disease or mortality.
We have investigated a large cohort (
Research design and methods
Study population
In this study, 1015 consecutive patients admitted for coronary catheterization to the ‘Praxisklinik Herz und Gefässe’, Dresden, Germany, from June 2007 to June 2009 were included. Altogether, 149 patients were admitted for acute coronary syndrome and 866 patients for routine catheterization. Data on patients are published elsewhere. 14 In summary, 319 patients were female, mean age was 68.2 years and mean body mass index (BMI) was 27.4 kg/m2. Patients with known diabetes mellitus (DM) were excluded as were patients with known endocrine, pancreatic, hepatic or chronic kidney diseases [estimated glomerular filtration rate (eGFR) < 50 mL/min]. All patients provided written consent, and the study was performed in accordance with the Declaration of Helsinki (approval by the local ethics committee, Sächsische Ärztekammer registration no. EK-BR-36/06-1).
After oGTT, patients were classified as follows: with normal glucose tolerance (NGT) (
Laboratory analyses
All patients underwent an oGTT on the day after the coronary angiography. After an overnight fast for at least 10 h, a standard oGTT (75 g of glucose in 250 mL of water) was performed between 8:00 a.m. and 10:00 a.m. according to World Health Organization (WHO) recommendations. 15 The diagnosis of diabetes according to the oGTT results was based on the WHO Consulting Group Criteria: 15 venous fasting plasma glucose (FPG) ≥ 126 mg/dL (7 mmol/L) and/or 2-h post-load plasma glucose after an oGTT ≥ 200 mg/dL (11.1 mmol/L). 15 IGT was defined as FPG < 126 mg/dL (7.0 mmol/L) and 2-h post-load plasma glucose ≥ 140 mg/dL (7.8 mmol/L) and <200 mg/dL (11.1 mmol/L) by WHO criteria. 15 IFG was defined as FPG ≥ 100 mg/dL (5.6 mmol/L) and FPG < 126 mg/dL (7 mmol/L) using American Diabetes Association (ADA) criteria. 16 NGT was defined as FPG < 100 mg/dL (5.6 mmol/L) and 2-h post-load plasma glucose < 140 mg/dL (7.8 mmol/L) by WHO criteria. 15
Plasma glucose, HbA1c, lipid levels, creatinine, electrolytes, blood count, lipids and C-reactive protein were analysed in the Central Laboratory of the Municipal Hospital of Dresden-Neustadt by routine methods. Whole adiponectin was measured by enzyme-linked immunosorbent assay (ELISA) kit purchased from Mediagnost GmbH, and NT-proBNP was measured by Enzyme Light Fluorescence Assay of Roche Diagnostics GmbH. Adiponectin levels are demonstrated as mean ± standard deviation (SD) in text and median + 25/75 and 10/90 percentiles in Figures 1 to 4.
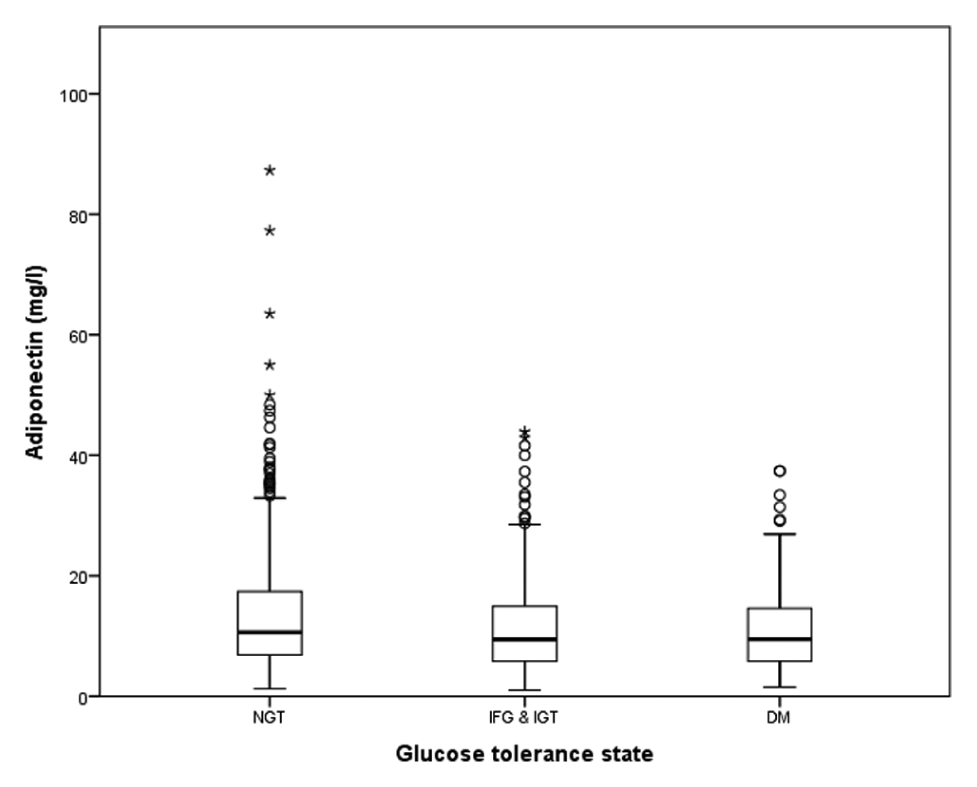
Adiponectin and glucose tolerance. Adiponectin levels in patients with normal glucose tolerance (NGT), impaired glucose tolerance [IGT, including patients with impaired fasting glucose (IFG), see ‘Research design and methods’ section] and patients with diabetes mellitus (DM).
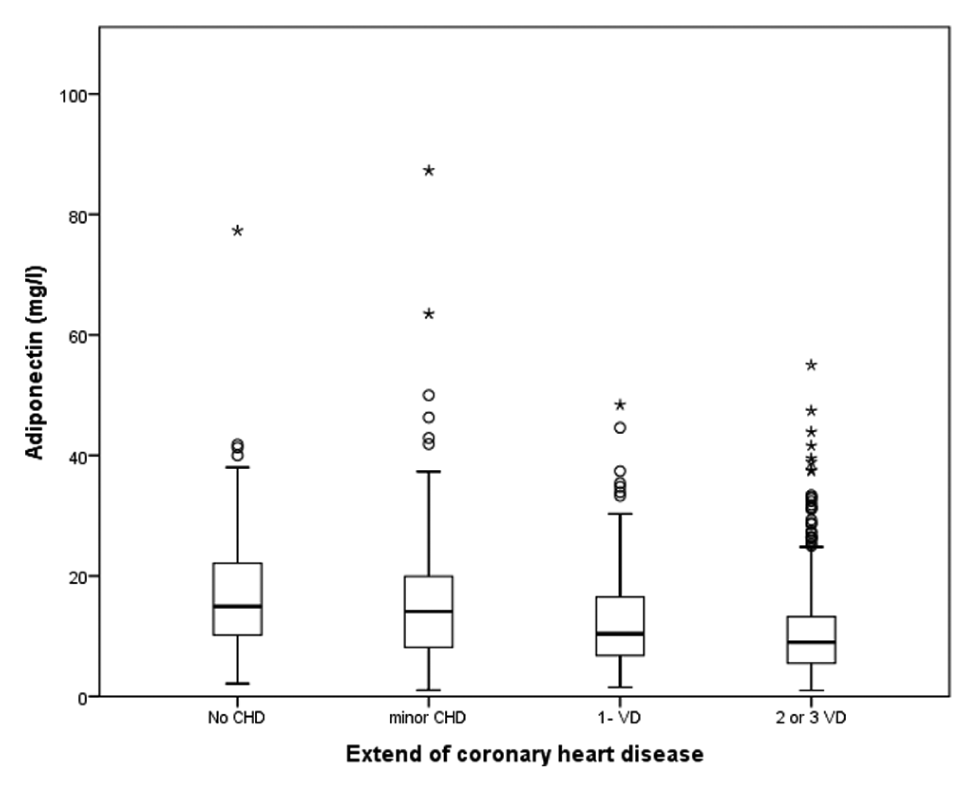
Adiponectin and stage of coronary heart disease. Adiponectin levels in patients with no coronary heart disease (no CHD), minor coronary heart disease (minor CHD), single-vessel disease (1-VD) and double- or triple-vessel disease (2- or 3-VD).
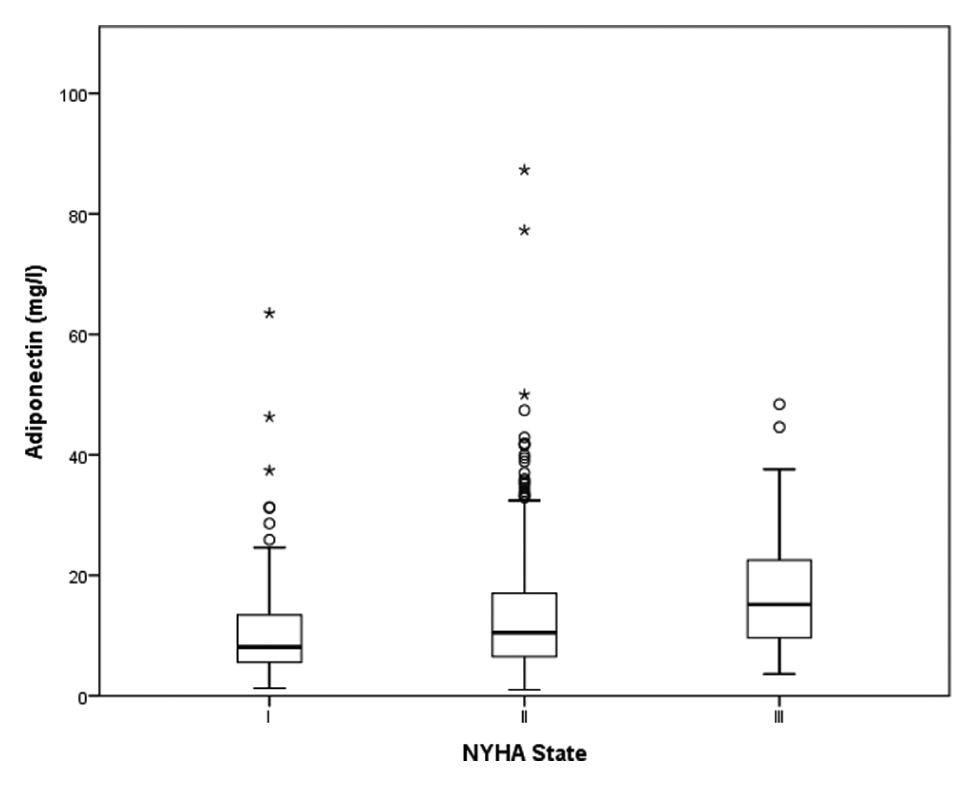
Adiponectin and NYHA state of heart failure. Adiponectin levels in patients with NYHA states I, II and III as indicated.
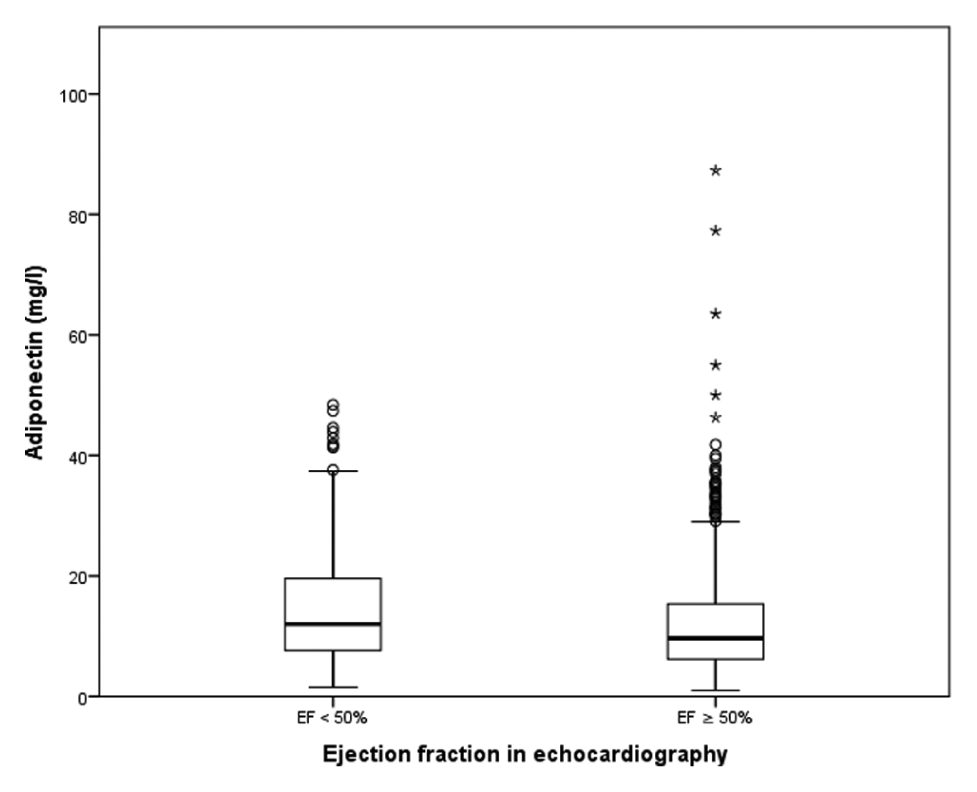
Adiponectin and ejection fraction in echocardiography. Adiponectin levels in patients with ejection fraction (EF) < 50% and > 50% in echocardiography as indicated.
Coronary angiography
Coronary angiography was performed by the Judkins method or modified Sones technique with direct puncture of either brachial or radial artery in the catheterization laboratory of the Praxisklinik Herz und Gefaesse, Dresden. CHD was defined based on the results of the coronary angiography as follows – no CHD: normal luminal diameter of any coronary artery, minor CHD: <50% stenosis of any coronary artery, 1-VD: >50% of any coronary artery disease, 2-VD: >50% stenosis of two different main coronary arteries and 3-VD: >50% stenosis of three different main coronary arteries. Coronary angiography findings were classified by independent visual assessment of two experienced interventional cardiologists and, at the 50% diameter stenosis threshold, by computer-based quantitative coronary angiography (QCA) as follows: no angiographic evidence of obstructive CHD, minor CHD with lesions of <50% diameter narrowing, 1-VD, 2-VD or 3-VD. 17
Statistical methods
Because of the sample size, a normal distribution of the means could be assumed; the analysis of variance (ANOVA) with the Tukey–Kramer post hoc test and the student’s
Results
Adiponectin and glucose tolerance
Adiponectin was higher in patients with NGT (13.65 ± 10.31 mg/L) compared to IFG/IGT (11.12 ± 7.5 mg/L,
Adiponectin and CHD
There was a stepwise decrease in adiponectin levels from no CHD (18.16 ± 12.49 mg/L) to minimal CHD (16.01 ± 11.42 mg/L, non-significant to no CHD) to 1-VD (12.18 ± 8.8 mg/L,
Adiponectin and heart failure
Patients with heart failure NYHA III (17.4 ± 10.27 mg/L) had higher adiponectin levels compared to patients with NYHA II (12.94 ± 9.41 mg/L,
Correlation of adiponectin with known risk factors of cardiovascular disease
We found a positive correlation of adiponectin to whole cholesterol (
As adiponectin levels differ according to gender (men = 9.99 ± 6.71, women = 17.81 ± 11.23,
Adiponectin dependence from glucose tolerance (IGT/IFG or DM compared to NGT).
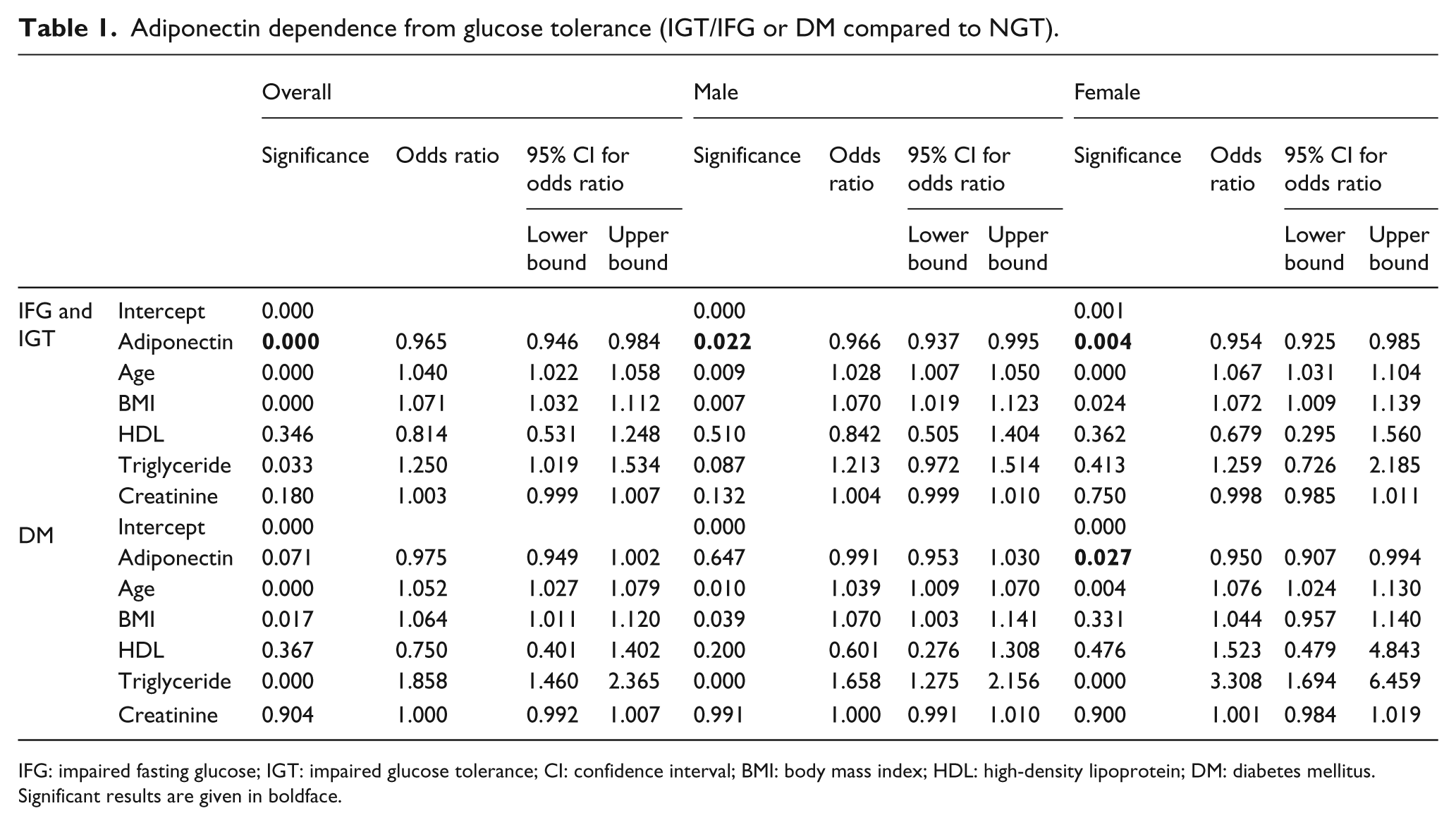
IFG: impaired fasting glucose; IGT: impaired glucose tolerance; CI: confidence interval; BMI: body mass index; HDL: high-density lipoprotein; DM: diabetes mellitus.
Significant results are given in boldface.
Adiponectin dependence from extent of CHD (minor CHD, 1-VD and 2- or 3-VD compared to no CHD).
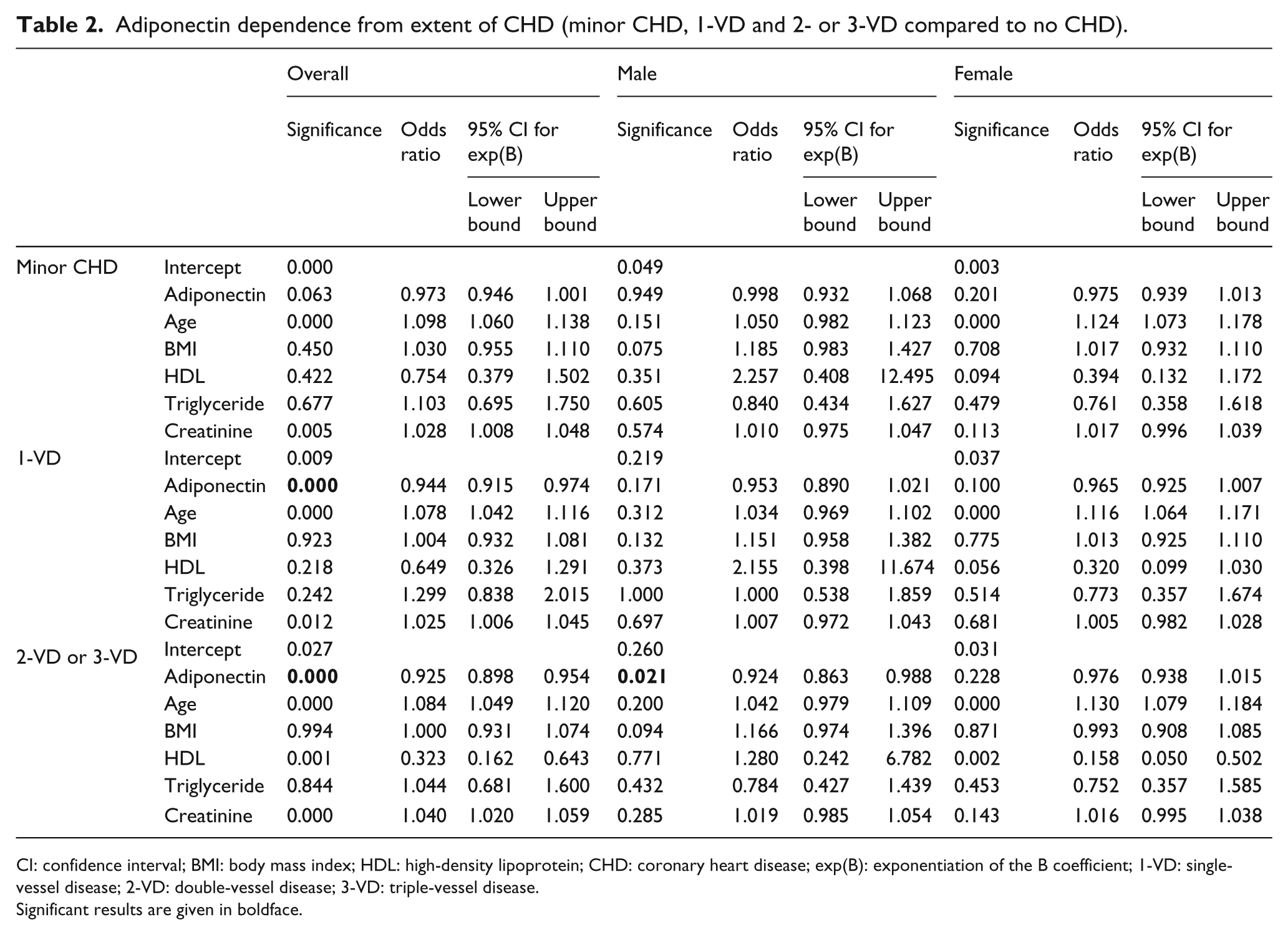
CI: confidence interval; BMI: body mass index; HDL: high-density lipoprotein; CHD: coronary heart disease; exp(B): exponentiation of the B coefficient; 1-VD: single-vessel disease; 2-VD: double-vessel disease; 3-VD: triple-vessel disease.
Significant results are given in boldface.
NYHA II compared to I indicated, in the overall analysis, significances in adiponectin, age and BMI (all OR > 1). Those three variables and creatinine showed significant results in the male patient group (all OR > 1). No factor had a significant influence on the female patient group. On comparing NYHA III and I levels, adiponectin, age and BMI presented significant effect in the overall and in the male group (all OR > 1). Adiponectin, age and creatinine had significant impact in the female group (all OR > 1) (for results, see Table 3).
Adiponectin dependence from NYHA state (II and III compared to I) each with confounders of age, BMI, HDL, triglyceride, creatinine and split into men/women.
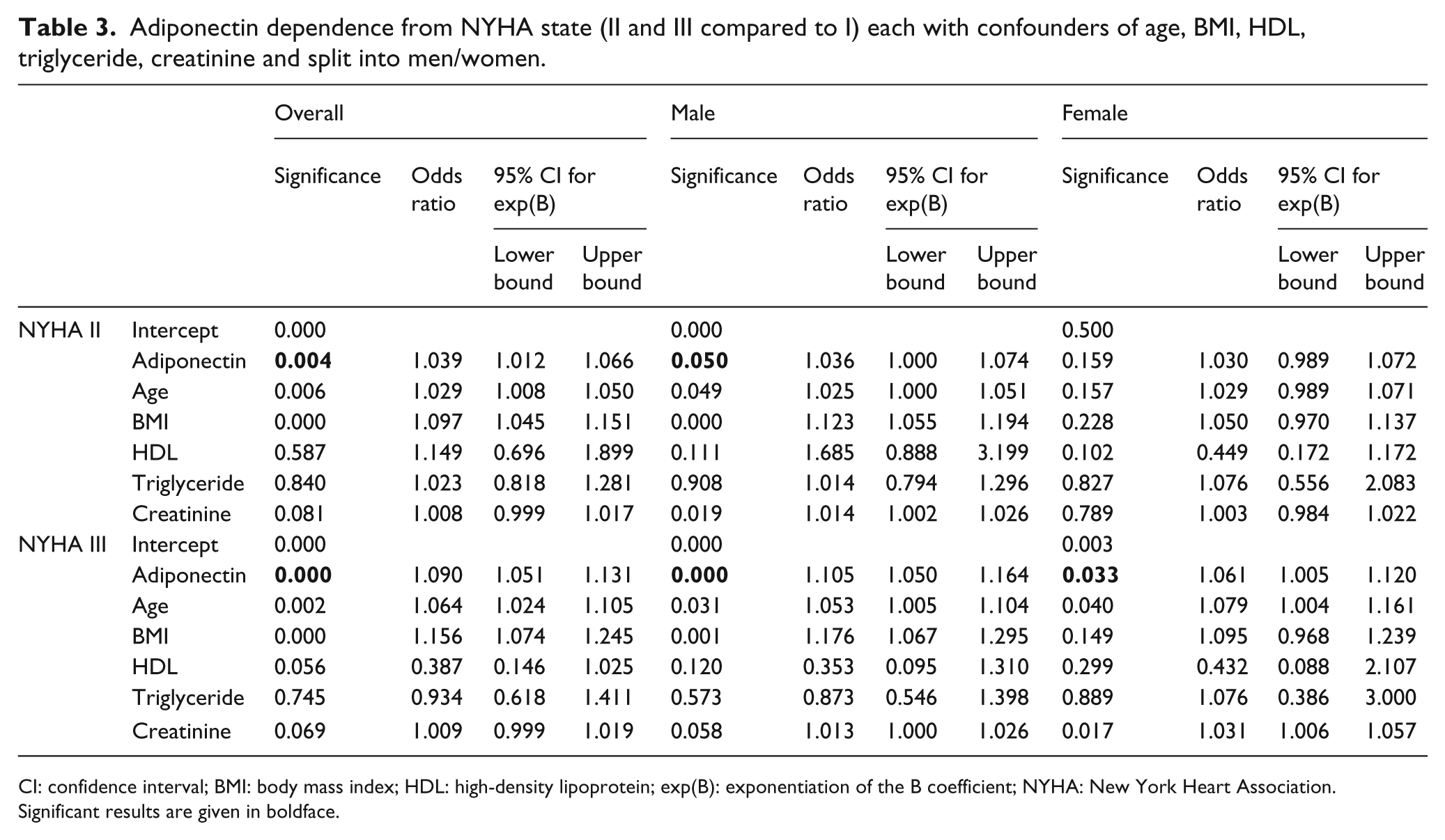
CI: confidence interval; BMI: body mass index; HDL: high-density lipoprotein; exp(B): exponentiation of the B coefficient; NYHA: New York Heart Association.
Significant results are given in boldface.
Conclusion
In the literature, there are data about decreased adiponectin levels in DM and CHD.1,7,8 Low adiponectin levels have been associated with insulin resistance as a key finding in type 2 diabetes and CHD.18,19 Along this line, our study found higher adiponectin levels in patients with suspected CHD and NGT compared to patients with newly diagnosed IGT and type 2 diabetes. In multivariate analysis, this difference holds true for women but not for men.
Although these patients without known diabetes may be expected in an early phase of their disease, many of them had already progressed CHD from catheterization findings. Again in line with the known literature, 20 we found a stepwise decrease in adiponectin levels with progressing CHD in catheterization findings. Currently, it is not clear whether adiponectin is a real protective factor in the coronary disease process or more a bystander reflecting other risk factors. In our population, we found a negative correlation to other risk factors such as smoking habits, obesity, glucose intolerance or triglycerides (or positive correlation to the protective HDL cholesterol). Against expectations, we also found a positive correlation to LDL and whole cholesterol. The high correlation to HDL cholesterol is remarkable and in agreement with previous data. 21 But there is direct evidence for protective influence of adiponectin on cardiac function in the literature.22,23 In multivariate analysis, sex differences were obvious – the significant correlation between adiponectin levels and extent of coronary disease was shown for male but not for female patients possibly due to age being a main confounder in female patients. In contrast, HDL cholesterol was a main confounder in both sexes in agreement with the high correlation described above. 21
Arguing against a pure protective role of adiponectin in CHD, we found a positive correlation of adiponectin to progressing heart failure as judged by NYHA state, NT-proBNP or EF. Similar findings were reported.13,24–26 It is not clear whether this increase in adiponectin levels may be a counter-regulatory process or a pathophysiological detrimental factor. It has been described that NT-proBNP is able to stimulate adiponectin secretion in adipocytes. 25 Also, in our study, there is a positive correlation of NT-proBNP and adiponectin levels as described elsewhere.27,28 Moreover, there is evidence of a local adiponectin secretion in cardiomyocytes, but this secretion seems to be much less than from adipocytes and not measurable in blood plasma.29,30 Some authors blame cachexia in heart failure for increasing adiponectin levels, 28 but this reason is not likely in our patients with early and mild heart failure NYHA II/III or I.
The strength of our study is a large population of clinically well-characterized patients attending a routine catheterization laboratory investigation without previous known IGT. Therefore, these patients seemed to be in an early phase of metabolic–vascular disease although already progressed vascular findings were seen in catheterization. We confirm a negative correlation of adiponectin levels with both progressing glucose intolerance and cardiovascular disease. The interpretation of adiponectin levels for individual patients may be confusing, however, by increase in the adiponectin levels even in earlier states of heart failure (NYHA II or III).
Footnotes
Acknowledgements
The authors thank the staff of the study unit of the Praxisklinik Herz und Gefässe and of the Central Laboratory of the Municipal Hospital of Dresden-Neustadt for organizing patient recruitment and lab analyses.
A.A.G., R.D., S.S., J.S., I.A.Z., O.S., and T.L. are responsible for conception and design, analysis and interpretation of the data; drafting the article; revising the article and for final approval of the version to be published. B.U. is responsible for statistical analysis of data and revising the article critically for important intellectual content. A.B. is responsible for laboratory analysis and also revising the article critically for important intellectual content
Conflict of interest statement
I.A.Z. and V.L. are employees of Roche Diagnostics GmbH, which markets assays of biomarkers for cardiovascular disease such as NT-proBNP. There are no other conflicting interests to declare.
Funding
The ‘Silent diabetes study’ was sponsored by an unrestricted grant of Roche Diagnostics GmbH.
