Abstract
We tested whether short-term, low-dose treatment with the fluvastatin and valsartan combination could improve impaired arterial wall characteristics in type 1 diabetes mellitus patients. A total of 44 type 1 diabetes mellitus patients were randomised into the treatment group [n = 22; received a low-dose combination of fluvastatin (10 mg daily) and valsartan (20 mg daily)] and the control group (n = 22; received placebo), both for 30 days. Brachial artery flow-mediated dilation (FMD), pulse wave velocity (PWV) and carotid artery β-stiffness were measured. Significant improvements in FMD (+73.2%), PWV (−7.5%) and β-stiffness (−10.0%) were achieved after 1-month treatment compared to the control group (all p values < 0.001). Three months after therapy discontinuation, important residual improvement in measured parameters was still present. No changes in lipids and blood pressure accompanied the beneficial improvements. We conclude that relatively simple intervention (low-dose, short-term fluvastatin/valsartan combination) produces substantial, long-term improvement of arterial wall characteristics in type 1 diabetes mellitus patients.
Keywords
Introduction
Cardiovascular morbidity in the type 1 diabetes mellitus population remains high, despite intense efforts in prevention and treatment.1–3 Functional and structural arterial wall characteristics become impaired earlier, and their impairment is greater in type 1 diabetes mellitus patients compared to the healthy population of matching age. 4 The mentioned changes of the arterial wall occur mainly as a consequence of numerous metabolic factors that accompany diabetes itself, comprising glycosylation of molecules in the arterial wall, chronic inflammation and oxidative stress.3–7 Arterial wall changes in these patients are characterised mainly by endothelial dysfunction and increased arterial stiffness, both of which were proved in previous studies.3–5,8 Thus, type 1 and 2 diabetes mellitus are associated with accelerated stiffening of the arteries that increases the biological age by approximately 10–15 years of chronological age compared to non-diabetics. 4 Consequent upon arterial wall changes, the cardiovascular risk increases and leads to cardiovascular diseases that are exhibited most evidently in atherothrombotic events.4,8,9
In our previous studies, we have shown the effectiveness of a new approach for improving arterial wall properties in a subgroup of apparently healthy middle-aged males. Namely, short-term, 1-month therapy with low-dose fluvastatin, valsartan and particularly their combination improved arterial wall characteristics.10–12 The effects of this treatment even persisted for several months after its discontinuation. As mentioned, the combination of fluvastatin and valsartan in low dose was evidently the most effective. The results obtained could be ascribed to the pleiotropic effects of the drugs used, since no effects on lipid levels or blood pressure values were observed. In numerous previous studies, the existence of several pleiotropic effects on arterial wall processes was proved for statins as well as angiotensin receptor blockers.13–16 Our above-mentioned studies added to the body of knowledge by showing that even sub-therapeutic, low doses of the drugs used induce significant pleiotropic effects on the arterial wall.
The present study was designed to further evaluate the above-described new approach in patients with type 1 diabetes mellitus. Therefore, its aim was to explore the effectiveness of 1-month treatment with the low-dose fluvastatin and valsartan combination on functional and structural arterial wall characteristics in patients with type 1 diabetes mellitus and to explore the persistence of the possible effects obtained, even after treatment discontinuation.
Methods
Subjects
Forty-four patients of both sexes (25 females and 19 males) with type 1 diabetes mellitus were recruited for this double-blind, randomised study. Inclusion criteria were diagnosis of type 1 diabetes mellitus lasting at least 5 years, age between 30 and 50 years, stable insulin dosage in the last 6 months, levels of HbA1c below 7.5% (reported in the last month) and no history of cardiovascular disease (carotid, coronary and peripheral). Exclusion criteria were smoking, treatment with statins or any drug from the family of renin–angiotensin–aldosterone system inhibitors.
Study design
Patients were randomly divided into control and treatment groups. The control group received placebo, while the treatment group received a low-dose combination of fluvastatin and valsartan (10 mg fluvastatin daily and 20 mg valsartan daily) over the 1-month period. All subjects were informed about the study protocol and gave their informed consent. The study was conducted in compliance with the principles of the Declaration of Helsinki ‘Recommendations guiding physicians in biomedical research involving human subjects’ (Helsinki, Finland, June 1964). This study was approved by The National Medical Ethics Committee of Slovenia.
A complete medical examination of patients was performed at inclusion in the study. Ultrasound measurements were performed at inclusion (before the beginning of the treatment period) and at the end of the 1-month treatment period. The following ultrasound measurements were performed: brachial artery flow-mediated dilation (FMD), pulse wave velocity (PWV) on the common carotid artery and local stiffness of the common carotid artery (β-stiffness). All ultrasound measurements were repeated 3 months after therapy discontinuation (4 months from inclusion in the study). Blood pressure was measured with an automated sphygmomanometer (Wellch Allyn Speidel & Keller).
Ultrasound measurements were performed in the afternoon after a 6-h fast following their normal breakfast and morning insulin under standardised conditions in a quiet, temperature-controlled environment. Patients rested in a supine position for 10 min prior to the ultrasound measurements, which were performed by a single examiner using an Aloka Prosound Alpha-10 ultrasound machine with an integrated high-resolution eTracking system.
Ultrasound measurements
Brachial artery FMD measurement
Brachial artery FMD was assessed in accordance with the guidelines.17,18 The diameter of the brachial artery was continuously visualised and recorded 5–10 cm above the antecubital fossa. After monitoring the baseline brachial artery diameter (1 min), the right forearm blood pressure cuff was inflated to 50 mmHg above systolic pressure for 4 min, producing arterial occlusion. The occlusion period was followed by rapid cuff deflation, thus inducing reactive hyperaemia. The brachial artery diameter was then recorded for another 3 min.
The echo machine continuously tracked and recorded the brachial artery diameter during the whole procedure and at the end of the measurement automatically provided the value of FMD (the percentage of change from the baseline diameter of the brachial artery during reactive hyperaemia).
Assessment of arterial stiffness parameters
Arterial stiffness parameters were measured on the right common carotid artery in a supine position with a head elevation of around 45° and side tilt of 30° to the left. The Aloka ultrasound device automatically determined the stiffness parameters through the analysis of pulse waves. The echo tracker’s cursor pair was set onto the anterior and posterior walls approximately 1.5–2 cm proximal to the bifurcation of the common carotid artery. Pressure waveforms were obtained noninvasively using arterial diameter change waveforms calibrated automatically on the basis of systolic blood pressure values. At the end of the measurements, the ultrasound machine automatically calculated the values of PWV and carotid artery local stiffness (β-stiffness).
Blood sample laboratory analysis
Fasting venous blood samples were obtained at inclusion in the study and at the end of the 1-month treatment period with the low-dose fluvastatin and valsartan combination or placebo. Concentrations of blood glucose, electrolytes, total cholesterol, high-density lipoprotein (HDL) cholesterol and triglycerides were measured in blood serum using a VITRO 5.1FS Chemistry System (Ortho Clinical Diagnostics, Inc.). The values of low-density lipoprotein (LDL) cholesterol were calculated using the Friedewald equation.
Statistical analysis
All values were expressed as means ± standard error of the mean (SEM) and were normally distributed. Differences between absolute values were assessed by the one-way analysis of variance (ANOVA). When a significant interaction was present, the Bonferroni post-test was performed. When comparing the relative changes during the treatment period between the treatment and control groups, the unpaired t-test was used. A p value of less than 0.05 was considered significant. All statistical analyses were performed using GraphPad Prism 5.0 software.
Results
Patient characteristics
The average age of participants at inclusion in the study was 35.6 ± 2.1 years in the control group and 36.3 ± 1.7 years in the treatment group. Body mass index (23.3 ± 0.5 kg/m2 vs 25.2 ± 0.9 kg/m2; p = 0.10), waist–hip ratio (0.9 ± 0.1 vs 0.9 ± 0.1; p = 0.64), HbA1c values (6.5 ± 0.5 vs 6.3 ± 0.6; p = 0.71) and duration of diabetes mellitus (15.3 ± 2.1 years vs 18.4 ± 2.4 years; p = 0.35) values did not differ between the control and treatment groups.
Other patients’ characteristics at inclusion and at the end of the study are presented in Table 1. No significant differences in systolic and diastolic blood pressure, total, LDL and HDL cholesterol, triglycerides and serum glucose levels were found between control and treatment groups. Treatment with the low-dose fluvastatin and valsartan combination did not significantly influence blood pressure, cholesterol and triglyceride values (Table 1).
Patient characteristics in control and treatment groups at inclusion in the study and after 1 month of treatment.
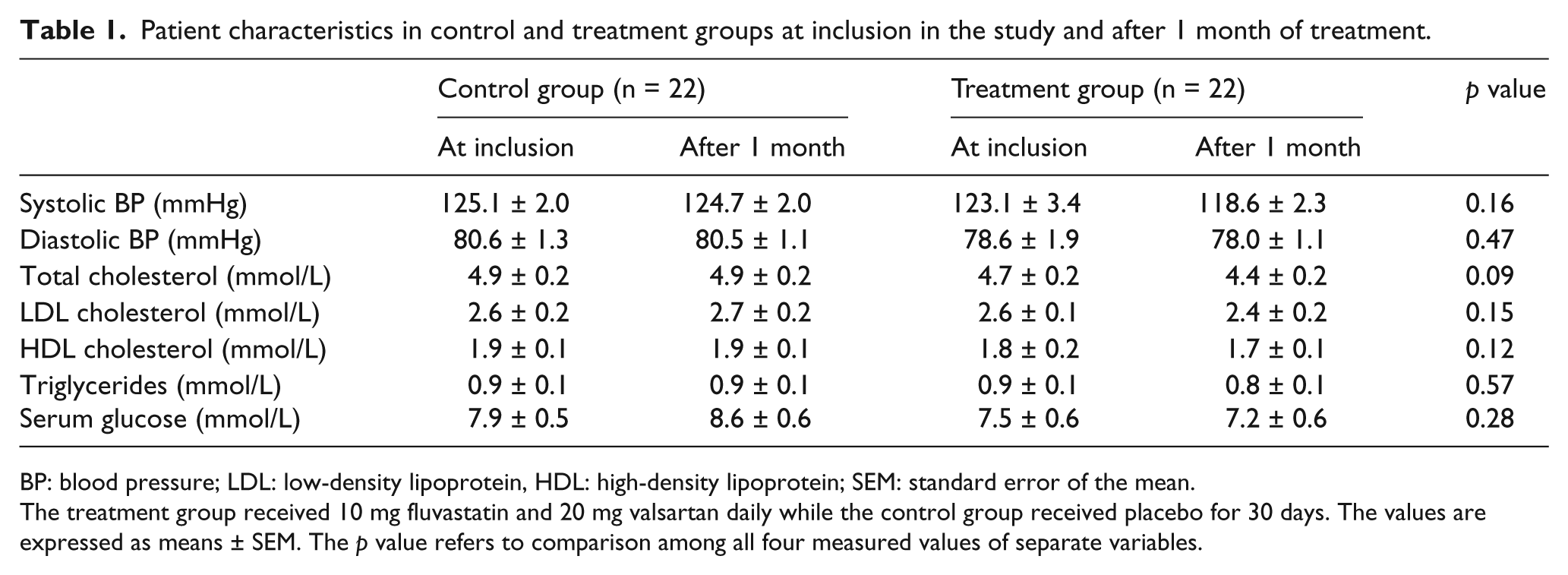
BP: blood pressure; LDL: low-density lipoprotein, HDL: high-density lipoprotein; SEM: standard error of the mean.
The treatment group received 10 mg fluvastatin and 20 mg valsartan daily while the control group received placebo for 30 days. The values are expressed as means ± SEM. The p value refers to comparison among all four measured values of separate variables.
Effect of 1-month intervention on arterial wall properties
In the treatment group, FMD increased from 3.7% to 5.7% (p < 0.05; Figure 1(a)), whereas in the control group no changes were observed. The percentage of FMD improvement after 30 days was significantly higher in the treatment group compared to the control group (73.2%; p < 0.001; Figure 1(b)).
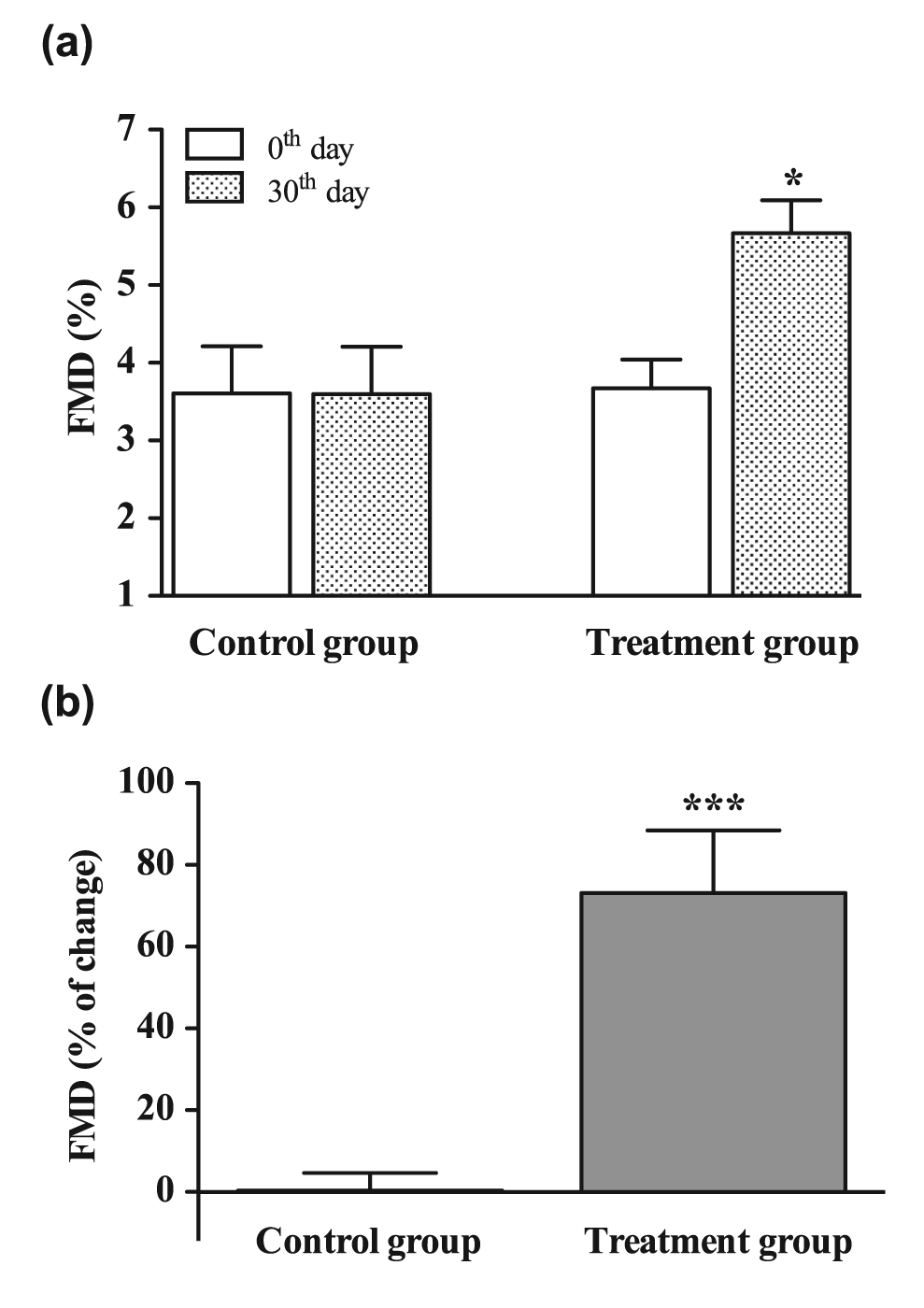
(a) Brachial artery flow-mediated dilation (FMD) values at inclusion in the study (0th day; white columns) and at the end of the treatment period (30th day, patterned columns) and (b) changes (expressed in percent) of FMD in control group (received placebo for 30 days) and treatment group (received 10 mg fluvastatin and 20 mg valsartan daily for 30 days).
The absolute values of PWV and β-stiffness decreased after 30 days of treatment with low-dose fluvastatin and valsartan, the differences not reaching the level of significance (Figures 2(a) and 3(a)). Comparison of PWV and β-stiffness changes in the separate groups over the 30-day treatment period revealed significant improvements in both PWV and β-stiffness in the treatment group compared to the control group (−7.5% and −10.0%, respectively; both p < 0.001; Figures 2(b) and 3(b)). At inclusion in the study, no significant differences in FMD, PWV and β-stiffness values between the placebo and treatment groups were observed.
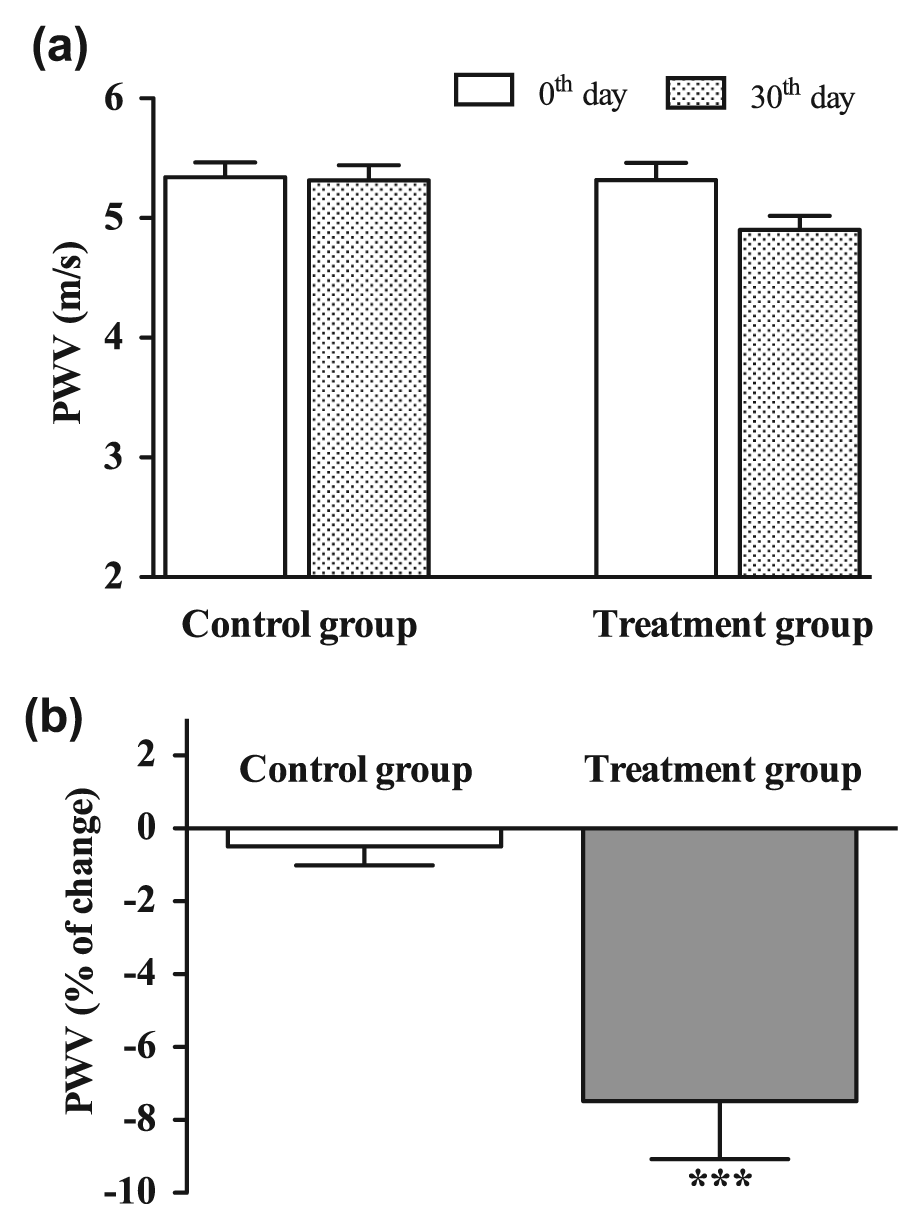
(a) Pulse wave velocity (PWV) on common carotid artery at inclusion in the study (0th day; white columns) and at the end of the treatment period (30th day, patterned columns) and (b) changes (expressed in percent) of PWV in control group (received placebo for 30 days) and treatment group (received 10 mg fluvastatin and 20 mg valsartan daily for 30 days).
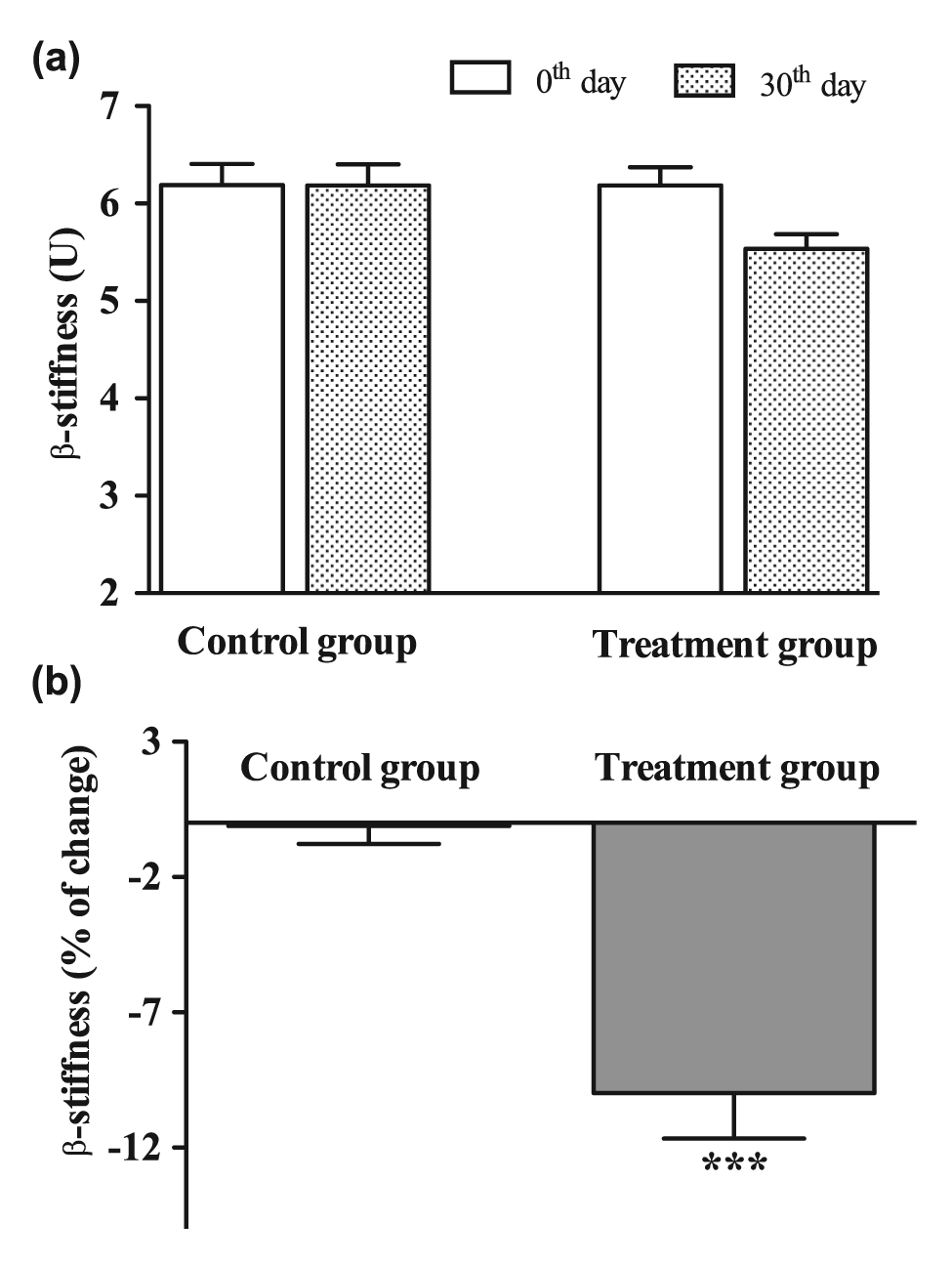
(a) β-stiffness values on common carotid artery at inclusion in the study (0th day; white columns) and at the end of the treatment period (30th day, patterned columns) and (b) changes (expressed in percent) of β-stiffness in control group (received placebo for 30 days) and treatment group (received 10 mg fluvastatin and 20 mg valsartan daily for 30 days).
Follow-up ultrasound measurements
After discontinuation of the 1-month treatment with the low-dose fluvastatin and valsartan combination, the percentage improvement of arterial wall properties declined, but was still present after 3 months. Approximately equal residual improvement was observed for FMD and β-stiffness (66.6% and 56.0%, respectively). PWV declined more, though after 3 months 36.1% of basal improvement was still present (Figure 4).
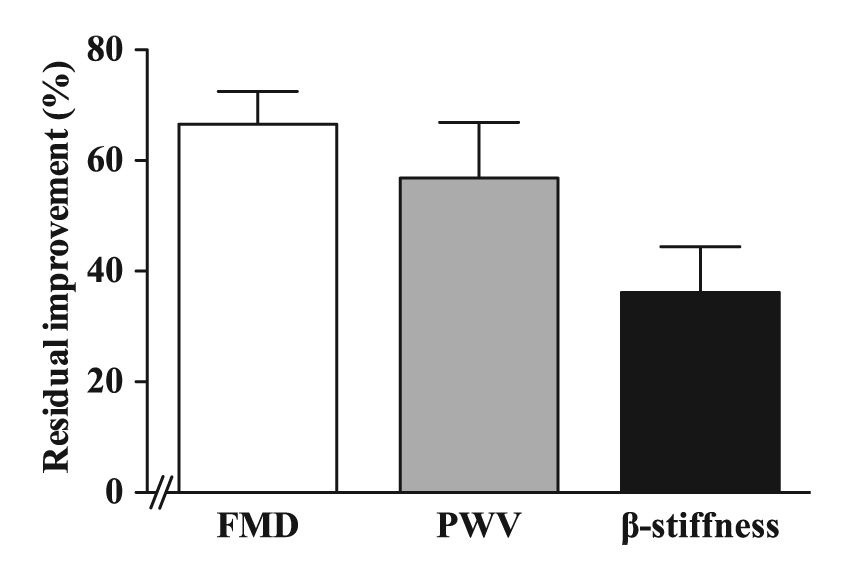
Residual improvement of brachial artery flow-mediated dilation (FMD), pulse wave velocity (PWV) and β-stiffness of common carotid artery 3 months after discontinuation of low-dose fluvastatin and valsartan combination.
Discussion
The present study was pursued in order to evaluate the effectiveness of the low-dose, short-term treatment approach with the fluvastatin and valsartan combination on arterial wall characteristics in patients with long-lasting type 1 diabetes mellitus. In this study, we found that arterial wall characteristics, defined by increase in brachial artery FMD and decrease of PWV and β-stiffness, importantly improved. Furthermore, the beneficial effects lasted, although in lower magnitude, even after therapy discontinuation – at least for three additional months. Since no significant effects on cholesterol levels and blood pressure were observed, these achievements were attributed solely to the pleiotropic effects of the drugs used.
The potential protective approach of the short-term (1-month), low-dose fluvastatin and valsartan combination treatment on arterial wall characteristics was tested in the present study in patients with type 1 diabetes mellitus. The concept was based on the surprisingly encouraging results of the same approach obtained in apparently healthy, middle-aged volunteers. 12 In the present study, 1-month treatment with the low-dose fluvastatin and valsartan combination caused a significant improvement in FMD (+73.2%), PWV (−7.5%) and β-stiffness (−10.0%) compared to the control group. The percentage improvement in arterial wall characteristics was, as expected, larger in apparently healthy, middle-aged participants, where FMD improved by +167.7%, PWV by −10.9% and β-stiffness by −18.8%. 12 These differences could be most likely attributed to the lesser extent of disease in the arterial wall of apparently healthy subjects, where we could expect better improvements of the arterial wall characteristics. Importantly, prolongation of the effect after termination of 1-month treatment was observed in both studied groups. In patients with type 1 diabetes mellitus, 3 months after therapy discontinuation, important residual improvements in FMD, PWV and β-stiffness (66.6%, 36.1% and 56.0%, respectively, compared to initial improvements after 30 days of treatment) were still present, whereas in healthy middle-aged males, much longer lasting effects were observed. Again, induction of obviously more severe and less reversible arterial wall changes by diabetes could be the explanation for the worse results obtained in type 1 diabetes mellitus patients. Nevertheless, although of lower degree, the improvement obtained in type 1 diabetes mellitus patients 3 months after treatment discontinuation was still at a significant level and probably clinically relevant. In both studies, no significant effects of treatment on cholesterol levels and blood pressure were observed.
The protective effects of statins and sartans on endothelial function and/or stiffness parameters in patients with diabetes mellitus have been explored in several previous studies. In most of these, patients with type 2 diabetes mellitus were recruited and only a few studies included patients with type 1 diabetes mellitus. To the best of our knowledge, in those studies, only therapeutic doses of statins and sartans were tested. No studies employing low doses and producing no decrease in cholesterol or blood pressure were published. In type 1 diabetes mellitus, patients 40 mg atorvastatin daily improved brachial artery FMD and decreased LDL cholesterol after 6 weeks of treatment.19,20 In another study, 40 mg atorvastatin daily after 6 months improved endothelial function and acted anti-inflammatorily and anti-thrombotically in type 1 diabetes mellitus patients. 21 After 3 months of therapy with fluvastatin (30 mg daily) or rosuvastatin (2.5 mg daily), both statins increased brachial artery FMD to a similar extent, rosuvastatin was more potent in decreasing LDL cholesterol compared to fluvastatin and only fluvastatin was effective in decreasing urine isoprostane concentrations in patients with type 2 diabetes mellitus. 22 Valsartan in therapeutic dosage (160 mg daily) was proven to decrease arterial stiffness in type 2 diabetes mellitus patients.23,24 The effects seemed to be independent of blood pressure lowering. 23 Although we performed an extensive literature search, we found no studies where the combination of a statin and sartan, either in therapeutic or in sub-therapeutic doses, was tested on arterial wall characteristics.
It is well known that diabetes mellitus is associated with endothelial dysfunction and accelerated arterial stiffening. Since in diabetes mellitus patients, these changes occur earlier and to a greater extent compared to the age-matched healthy population, their arterial age and risk of atherothrombotic events increase accordingly.4,5 The present study is the first to assess the effectiveness of the short-term, low-dose fluvastatin and valsartan combination on the improvement of endothelial function and arterial stiffness parameters in patients with type 1 diabetes mellitus. Until now, only the separate drugs, namely statins or sartans, in therapeutic doses were mainly studied, mostly in the type 2 diabetes mellitus population.19,20,22–24 It has been shown that the separate drugs in therapeutic dosages could improve brachial artery FMD and arterial stiffness parameters, but always in association with a decrease of lipids and/or blood pressure.
The use of the combined drugs in the present study was based on the proven highest effectiveness of the combination in our previous study performed in apparently healthy, middle-aged males. 12 In addition, in animal experiments, we found that the low-dose combination of a statin and an angiotensin receptor blocker is more effective than the separate drugs.8,25
The advantages of the approach described lie particularly in the low doses used, so there are no side effects, while the short-term dose regimen improves patient compliance. Regular repetition of (1-month) treatment after certain time periods, which remain to be clearly defined (e.g. after at least 3 months or maximally 11 months), would be the cornerstone of a new cardiovascular prevention approach in patients with type 1 diabetes mellitus.
In conclusion, we showed that by a relatively simple intervention using a combination of fluvastatin and valsartan (low-dose, short-term treatment), a substantial long-term improvement of arterial wall characteristics in patients with type 1 diabetes mellitus could be achieved. Consequently, their arterial age and risk of cardiovascular disease decreased. This new approach seems to be promising in achieving a decreasing rate of atherothrombotic events in the type 1 diabetes mellitus population.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by the Slovenian Research Agency (research project L3-2293).
