Abstract
Sulphonylureas (SUs) are widely used glucose-lowering agents in type 2 diabetes mellitus (T2DM) with apparent declining efficacy over time. Concerns have been raised from observational retrospective studies on the cardiovascular (CV) safety of SUs but there are few long-term data on CV outcomes from randomized controlled trials (RCTs) involving the use of this class of agents. Most of the observational studies and registry data are conflicting and vary with study population and methodology used for analyses. To address the SU controversy, we reviewed the recently published literature (until end of the year 2011) to evaluate the impact of SUs on CV outcomes in modern, longer-term (≥72 weeks) RCTs where they were compared in a head-to-head fashion versus an active comparator or were used as part of a treatment strategy. We identified 15 trials and found no report of an increase in the incidence of CV events with the use of SUs. However, the available data are limited, and, most importantly, there was no adequately powered formal head-to-head CV outcome trial designed to address CV safety. Since SUs are still being advocated as second-line therapy added-on to metformin, as one of several classes, and in certain circumstances first-line therapy in T2DM management, definitive data from a dedicated RCT addressing the CV safety question with SUs would be informative. Cardiovascular Outcome Study of Linagliptin versus Glimepiride in Patients with Type 2 Diabetes (CAROLINA) is such a trial, ongoing since November 2010, and is currently the largest head-to-head CV outcome trial that involves a comparison of a SU (glimepiride) with a dipeptidyl peptidase-4 (DPP-4) inhibitor (linagliptin) and will provide a unique perspective with respect to CV outcomes with these two commonly used agents.
Keywords
Introduction
Cardiovascular disease (CVD) is a highly prevalent complication and the major cause of premature death in patients with type 2 diabetes mellitus (T2DM), with some estimates suggesting that 70%–75% of deaths may be attributable to cardiovascular (CV) complications. 1 –3 The current global diabetes epidemic combined with the increasing longevity of populations in most nations has led to a continued rise in the overall prevalence of CVD, imposing an increasing burden on health-care systems. 4,5 Action to reduce this burden of diabetes-related CVD is therefore needed and includes the development of new and effective treatments for T2DM that may potentially benefit CV outcomes.
The last two decades have seen the development of a wide variety of new therapeutic options to treat the hyperglycaemia of T2DM. Each class of glucose-lowering agents can show broadly similar efficacy as monotherapy with relatively minor and often non-clinically meaningful differences in glucose-lowering potency (at least in short term), which depend in part on the study design populations and baseline glycated haemoglobin (HbA1c) levels. 6 In contrast, each therapeutic class is characterized by a distinct adverse-event profile that may be related to the specific mechanism of action and/or potential off-target effects. 7 Several of these side effects (in particular hypoglycaemia and weight gain) are clinically meaningful to patients and physicians, and it is conceivable that these adverse events may further increase the CV risk in T2DM or may negate the potential CV benefits of some of the glucose-lowering agents.
Today, new diabetes agents face increased regulatory scrutiny and are required to demonstrate CV safety before, or after, approval. Indeed, the US Food and Drug Administration (FDA) key post-approval criterion to exclude unacceptable CVD risk for new diabetes drugs is an upper bound of the 95% confidence interval (CI) of <1.3 for the hazard ratio (HR) of CV events. 8 On the other hand, these regulatory requirements may also provide the opportunity for some of the drugs in these ongoing CV outcome trials to be tested for CV benefits if the study design allows for sufficient statistical power. This review will cover the current evidence on the long-term risk of CV events with sulphonylureas (SUs), which remain one of the most widely used drug classes in T2DM. It will also review and put into a clinical perspective some of the currently ongoing CV studies of the dipeptidyl peptidase-4 (DPP-4) inhibitors, another class of glucose-lowering agents that are frequently added to metformin and that also stimulate insulin secretion, but are associated with a low risk of hypoglycaemia and do not result in weight gain.
Glycaemic control and CVD risk
Epidemiological studies have firmly established that the risk of CVD is progressively related to the degree of hyperglycaemia, as indicated by HbA1c levels. 9,10 However, despite the strong epidemiological association of HbA1c with CV events, there is less compelling evidence that intervention studies designed to reduce glycaemia reduce the risk of CV events in patients with T2DM. 11 –13 The United Kingdom Prospective Diabetes Study (UKPDS) showed that a policy aiming for optimal glucose control using a SU or basal insulin was associated with a non-significant lower myocardial infarction (MI) risk versus standard therapy [relative risk (RR) = 0.84; 95% CI = 0.71–1.00] after a median of 10 years of follow-up, while the risk of deaths, stroke or amputation did not change significantly. 14 Interestingly, the CV benefits of improved glycaemic control became evident only after an additional 10 years of follow-up after the end of the controlled intervention component of the study as a ‘legacy effect’, during which time both groups were treated by their primary care physicians and achieved similar glucose control. 15 Clinically relevant post-trial RR reductions in MI (15%, p = 0.01) and death from any cause (13%, p = 0.007) emerged over this additional time, despite no statistically significant differences being observed in the interventional phase of the study. The results of the UKPDS therefore indicate that achieving optimal glucose control early in the course of T2DM in newly diagnosed patients could have long-term benefits, irrespective of treatment modality (SU, insulin or metformin). The Action to Control Cardiovascular Risk in Diabetes (ACCORD) Trial 16 demonstrated a non-significant reduction of 10% in the composite primary CV outcome of non-fatal MI, non-fatal stroke and CV death among 10,251 patients assigned to achieve a target HbA1c of <6.0%, versus those with a less challenging target of 7.0%–7.9%. 16 However, the intensive glucose-lowering component of the trial was stopped early – after 3.5 years – due to an unexplained excess in all-cause mortality among patients receiving intensive therapy [257 deaths (5.0%)] vs 203 (4.0%) for standard therapy; HR = 1.22; 95% CI = 1.01–1.46; p = 0.04]. Importantly, around one-third of patients in the study had experienced a previous CV event, and the authors suggested that intensive glucose-lowering therapy could be deleterious in patients with T2DM with a high risk of CVD. In addition, the intensive treatment group experienced significant weight gain and a clinically meaningful increased risk of severe hypoglycaemia, 16 which could have also contributed to the increased mortality, although these possibilities remain controversial.
A lack of a beneficial effect on CVD outcomes of intensive glycaemic management was also observed in two other large trials: the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) trial 17 and the Veterans Affairs Diabetes Trial (VADT). 18,19 These studies included patients with T2DM of moderate-to-long duration (mean duration at baseline, 8.0 and 11.5 years, respectively) and, despite achieving significant differences in glucose control between the treatment arms, only non-significant reductions in macrovascular events were observed. 17,18 Together, UKPDS, ACCORD, ADVANCE and VADT provide interesting insights into the management of CV risk in T2DM. The observed differences in outcomes between the UKPDS and the other studies could be related, at least in part, to differences in the study populations. In the UKPDS, patients had newly diagnosed T2DM, whereas those in the other studies had more established or advanced disease. Furthermore, in contrast to the UKPDS, ACCORD, ADVANCE and VADT, all recruited patients with increased CVD risk, about one-third of whom had a history of macrovascular disease compared with only 7.5% in the UKPDS. 20 These differences in study populations could account for the relatively more favourable, potential and long-term ‘legacy effect’ of glycaemic control on CV outcomes observed in the UKPDS. The issue and hypothesis surrounding the ‘long-term legacy effect’ of glycaemic control are however in need of clarification and will be further addressed with the post-trial monitoring implemented for ACCORD (ACCORDION – a prospective, observational follow-up study of at least 8000 participants from ACCORD) 21 and ADVANCE [ADVANCE-ON (NCT00949286) targeting 10,000 participants from ADVANCE]. 22 Results from these two follow-up studies are expected in the 2014–2015 timeframe.
Impact of specific diabetes therapies on CVD risk
A large number of diabetes drug classes are available for the management of T2DM, but it remains unclear how specific agents compare with respect to long-term CV risk. 23,24 To date, the large CV outcome trials have essentially assessed the impact of multiple combinations of glucose-lowering agents as part of an overall treatment regimen (e.g. UKPDS, ACCORD, ADVANCE and VADT) or as individual treatments versus placebo [e.g. the Prospective Pioglitazone Clinical Trial in Macrovascular Events (PROactive) 25 ]. There have only been few, 23,24 long-term head-to-head trials that have compared the effects of different diabetes drugs on CV outcomes/CV surrogates [e.g. Carotid Intima–Media Thickness in Atherosclerosis Using Pioglitazone versus Glimepiride (CHICAGO), 26 Pioglitazone Effect on Regression of Intravascular Sonographic Coronary Obstruction Prospective Evaluation (PERISCOPE) 27 and Assessment on the Prevention of Progression by Rosiglitazone on Atherosclerosis in Type 2 Diabetes Patients with Cardiovascular History (APPROACH) 28 ]. Thus, a comparative understanding of the CV impact of the most widely used diabetes drugs is for the most part lacking. 13
Impact of SUs on CVD events in randomized controlled trials
SUs [e.g. glyburide (known as glibenclamide in Europe), gliclazide, glipizide or glimepiride] are frequently recommended as the preferred second-line therapy because of their well-established efficacy and low cost. However, the results of some studies have led to uncertainty about their durability and long-term CV safety, which may potentially be related to the fact that SUs not only bind to the SU receptor (SUR) subunit (subtype SUR1) of the potassium ATP (KATP) channel in the beta-cell membrane, but may also bind to the SUR receptor (subtype SUR2) on cardiac myocytes and on endothelial cells, and thus have direct effects on CV function. 29
The controversy regarding the CV safety profile of SUs started with the highly criticized University Group Diabetes Program (UGDP), conducted in the 1960s that first gave rise to concerns about the safety of the first-generation SU, tolbutamide. In this study, a significantly increased risk of all-cause and CV mortality was observed among participants receiving this SU versus placebo. 30 However, the UGDP was neither designed nor powered to test the hypothesis of inferior CV safety for SU versus placebo. The study has been criticized because the data were not corrected for the higher pre-existing CV risk in the SU-treated patients. 31 Nevertheless, as a consequence of these data, every SU approved for use in the US has in its product label that SU use has been associated with increased CV mortality.
It is unclear whether the findings of the UGDP are applicable to current clinical practice, where modern diabetes management includes a multifactorial approach to reduce the risk of CV complications. 7 Furthermore, it is uncertain whether the UGDP findings apply to all SUs, as discussed in the following.
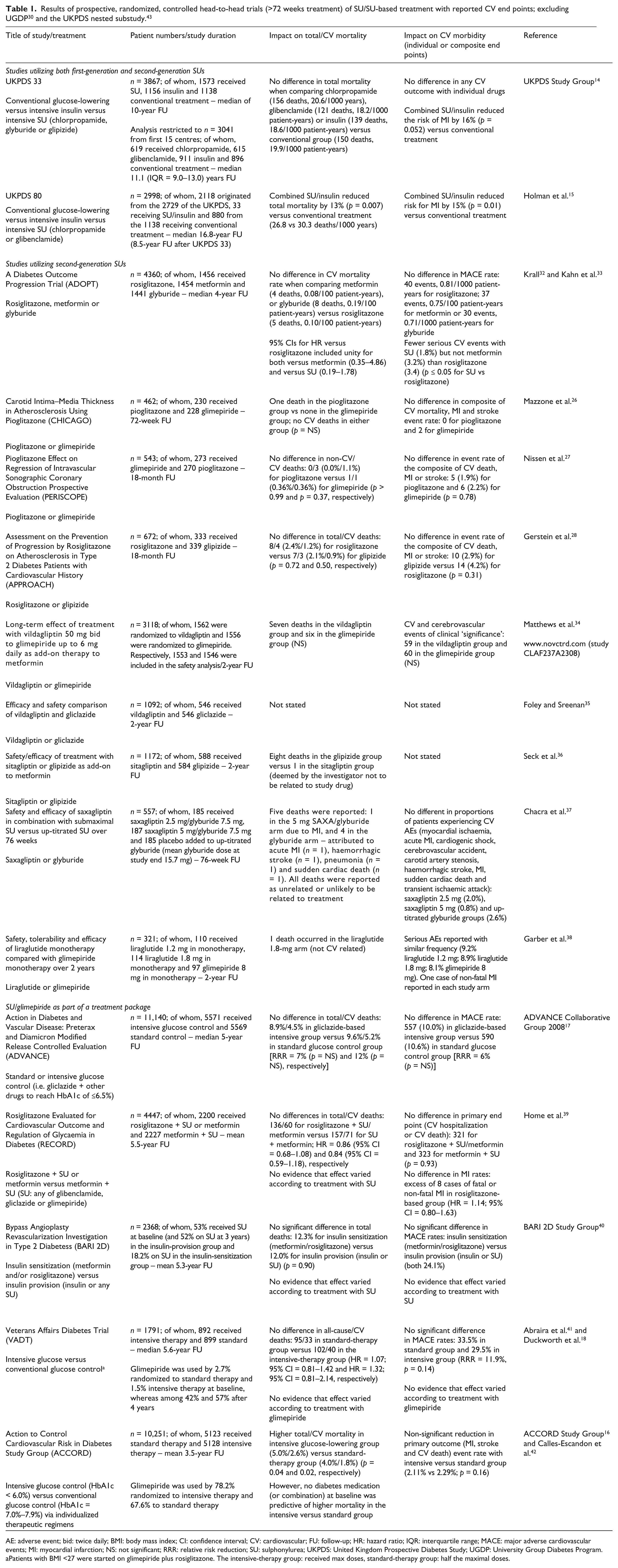
Table 1 lists 15 published randomized controlled trials (RCTs) (identified by searches in PubMed) reported over a 15-year period until the end of 2011 that were of ≥72-week duration and included SU therapy versus an active comparator or as part of a treatment strategy (e.g. comparing a diabetes treatment strategy to reach HbA1c ≤6.5% vs 7.5%). Because of the uncertain relevance of the UGDP trial findings to current day practice, and due to its initiation more than 50 years ago, it is not included in Table 1. Retrospective cohort and observational studies are also excluded, with some discussed in the following. As listed in Table 1, the UKPDS demonstrated that chlorpropamide, glyburide (glibenclamide) and glipizide were not associated with adverse CV events. 14,15 The other 14 long-term RCTs (Table 1), including A Diabetes Outcome Progression trial (ADOPT, n = 4360), 32,33 also indicate that SUs were not associated with an increase in CVD risk when compared head-to-head trial with thiazolidinediones (glitazones), DPP-4 inhibitors, metformin or glucagon-like peptide-1 (GLP-1) analogues. 26 –28,32 –38 The same lack of an adverse outcome was observed when SUs were used as part of a treatment strategy to obtain different glycaemic targets, 14 –19,39 –42 including the ADVANCE (n = 11,140). 17 However, it must be noted that none of the studies targeting ‘similar level’ of glycaemic control in the treatment arms were powered to demonstrate CV safety/benefits, and there were inconsistencies in how the CVD events were reported and adjudicated. A recent meta-analysis of 40 RCTs of glucose-lowering agents found no increased risk of macrovascular events and all-cause mortality with the use of second-generation SUs versus other oral agents or placebo. 23 The authors made special note of the fact that most studies were not designed or powered to examine CV events, and that in addition to the lack of long-term studies, the poor quality and inconsistent reporting of adverse events in the published trials made it difficult to draw conclusions about the CV risk of commonly used diabetes medications. 23 This conclusion also holds true for the studies listed in Table 1, and similar conclusion was reached by the Agency for Healthcare Research and Quality (AHRQ) in the more recent updated systematic review of the comparative effectiveness and safety of oral glucose-lowering medications for treating T2DM. 24
AE: adverse event; bid: twice daily; BMI: body mass index; CI: confidence interval; CV: cardiovascular; FU: follow-up; HR: hazard ratio; IQR: interquartile range; MACE: major adverse cardiovascular events; MI: myocardial infarction; NS: not significant; RRR: relative risk reduction; SU: sulphonylurea; UKPDS: United Kingdom Prospective Diabetes Study; UGDP: University Group Diabetes Program.
Patients with BMI <27 were started on glimepiride plus rosiglitazone. The intensive-therapy group: received max doses, standard-therapy group: half the maximal doses.
Currently, data from recent longer-term RCT (Table 1) and observational studies 44 –48 remain discordant regarding the CV safety of SUs. Several, but not all, 49,50 observational studies have shown that all-cause and CV mortality are increased with either SU monotherapy 44 –47 or when a SU is combined with metformin, 47 as compared to metformin monotherapy. Furthermore, a meta-analysis of observational studies showed that SU–metformin combination therapy versus either monotherapy was associated with a significant increase in the RR of a composite of CV hospitalization or mortality (overall RR = 1.43; 95% CI = 1.10–1.85), but was not associated with a difference in either outcome alone that is difficult to explain. 48 Recent RCTs versus two older RCTs also conflict. Apart from the aforementioned UGDP, 30 a nested substudy of the UKPDS showed that the addition of metformin to a SU in obese and non-obese patients (n = 268) who were not reaching glycaemic targets was associated with an increased risk of diabetes-related death (96% increased risk, p = 0.039) and all-cause mortality (60% increase, p = 0.041) compared with SU monotherapy (n = 296), which contradicts the parallel UKPDS report showing CV benefits with metformin. 43 However, the UKPDS investigators commented that these subgroup results might be a result of extremes of chance, and epidemiological analysis of the possible association of death from diabetes-related causes showed no increased risk in patients treated with SU/metformin combination therapy. 43 To date, no follow-up results have been published on this study arm, and the findings of this substudy are also excluded from the summary in Table 1. In light of the discordant views about the safety of SUs (especially tolbutamide and chlorpropamide) and other glucose-lowering agents, there is clearly a need to explore the safety of these drugs in future trials.
New diabetes therapies and CVD risk: present studies involving DPP-4 inhibitors
In response to concerns about the CV safety of diabetes drugs, at the end of 2008, the FDA issued a directive that clinical trials of new agents for the treatment of T2DM should demonstrate that new medications are not associated with an increased CV risk. 8 The guidance also states that in order to obtain sufficient end points to allow a meaningful assessment of risk, studies should include patients at increased risk of CV events, such as those with relatively advanced disease, elderly patients and patients with some degree of renal impairment. Similar guidance has also been issued by the European Medicines Agency (EMA), which notes that the possibility of a relationship between some diabetes drugs and cardio/cerebrovascular disorders cannot be confirmed or excluded, and that the concerns raised by existing data require further in-depth evaluation. 51
DPP-4 inhibitors are a newer class of oral agents for lowering of blood glucose and improve glycaemic control by inhibiting the inactivation of the incretin hormones, GLP-1 and glucose-dependent insulinotropic polypeptide. 52 These incretin hormones are short-lived intestinal peptides released in response to food ingestion, which have an inhibitory effect on glucagon (thereby reducing hepatic glucose synthesis) and an enhancing effect on insulin secretion, when glucose is elevated (not when it is normal or low). The first agent approved in this class (sitagliptin) was introduced for clinical use in 2006. Of interest is that there are other physiological substrates of DPP-4 (e.g. stromal-derived factor-1α – a chemokine that stimulates bone marrow mobilization of endothelial progenitor cells), which may or may not contribute to non-glycaemic effects of DPP-4 inhibitors, as discussed in the following section.
Three DPP-4 inhibitors, sitagliptin, 53 saxagliptin 54 and linagliptin, 55 are currently available in the United States and elsewhere. A fourth inhibitor, vildagliptin, 56 is available in Europe and other countries, but not in the United States, and a fifth inhibitor, alogliptin, is available in Japan. 57 As a class, the DPP-4 inhibitors provide clinically meaningful reductions in HbA1c compared with placebo and convey a low risk of hypoglycaemia without weight gain. 6
A range of potential CV benefits of DPP-4 inhibitors has been suggested based on findings from animal models, small proof-of-concept studies, and meta-analyses of clinical studies. Data from the meta-analyses based on phase III trials published to date indicate that the use of some DPP-4 inhibitors is associated with reductions in CV outcomes. Significant reductions were reported in a recent meta-analysis on linagliptin 5 mg (HR = 0.34; 95% CI = 0.16–0.70) 58 and in a previous analysis of saxagliptin 2.5–10 mg (HR = 0.43; 95% CI = 0.23–0.80). 59 In contrast, risk estimates were not significant for sitagliptin 100 mg (RR = 0.68; 95% CI = 0.41–1.12), 60 vildagliptin 50 and 100 mg (RR = 0.84; 95% CI = 0.64–1.14 and RR = 0.88; 95% CI = 0.37–2.11, respectively) 61 or alogliptin (HR = 0.61; 95% CI = 0.24–1.56). 62 How these possible benefits are mediated is not fully clear, but it would appear that both GLP-1 and non-GLP-1-related pathways are involved that potentially improves endothelial function and inflammation and cardiac function, reduces infarct size and atherosclerosis and enhances post-ischaemic recovery. 52
Although the results of the different meta-analyses of DPP-4 inhibitors are not entirely comparable (due to differences in primary composite end points and CV adjudication methods), all provide support to the hypothesis that DPP-4 inhibitor treatment may have a beneficial effect on CV outcome compared with other glucose-lowering treatments for T2DM that need to be tested in RCTs. Indeed, there are several ongoing CV safety trials comparing different DPP-4 inhibitors versus placebo added to conventional therapy in patients with T2DM who are at different stages of disease and receiving diverse glucose-lowering therapies.
These trials of DPP-4 inhibitors designed to evaluate CV outcomes in patients with T2DM are currently recruiting or have recently completed recruitment of participants (Table 2). The Trial Evaluating Cardiovascular Outcomes with Sitagliptin (TECOS) is planned to last for 4–5 years, with a projected completion date of December 2014 (NCT00790205). A similar trial is the Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus–Thrombolysis in Myocardial Infarction 53 (SAVOR-TIMI-53) trial, which will evaluate whether saxagliptin will reduce the risk of CV events when used alone, or added to other glucose-lowering therapies (NCT01107886). 63 The Examination of Cardiovascular Outcomes: Alogliptin versus Standard Care in Patients with Type 2 Diabetes and Acute Coronary Syndrome (EXAMINE) trial will investigate the effect of alogliptin on CV outcomes in subjects with T2DM and acute coronary syndrome. It will last for up to 4.75 years and is expected to be completed in May 2015 (NCT00968708). 64 Of note is that all of the aforementioned trials are placebo controlled when added to background therapy and thus cannot address the clinically relevant question of whether a DPP-4 inhibitor is a more suitable second-line therapy than a SU (Table 2). To gain the greatest possible insight into whether DPP-4 inhibitors hold advantages over other specific diabetes drugs in terms of CV outcomes, we believe it would be best to undertake head-to-head active comparator controlled trials. Such studies would largely extend our current knowledge, since existing data are mainly derived from post hoc pooled analyses of studies of limited duration.
Key features of ongoing CV safety studies of DPP-4 inhibitors.
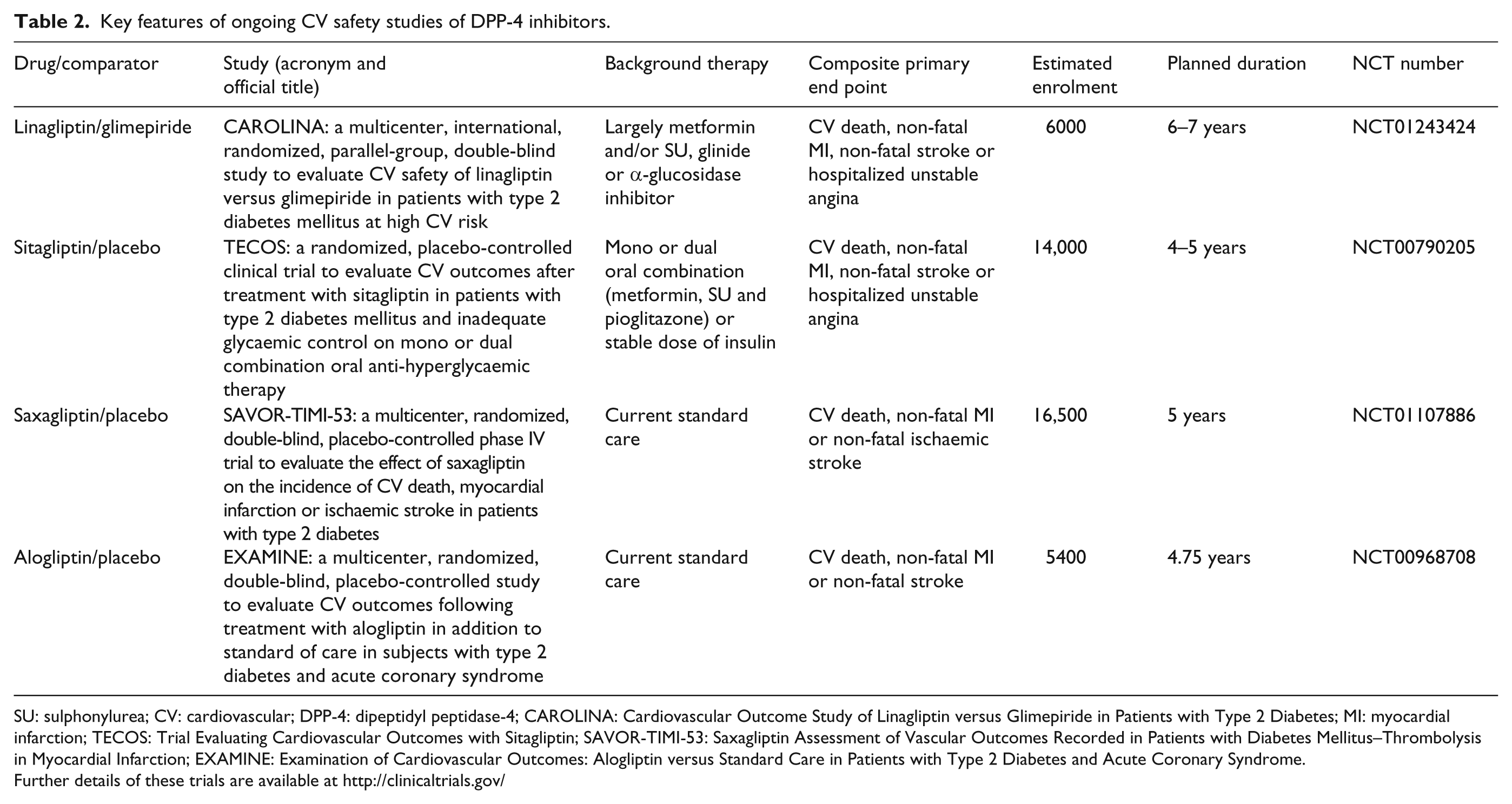
SU: sulphonylurea; CV: cardiovascular; DPP-4: dipeptidyl peptidase-4; CAROLINA: Cardiovascular Outcome Study of Linagliptin versus Glimepiride in Patients with Type 2 Diabetes; MI: myocardial infarction; TECOS: Trial Evaluating Cardiovascular Outcomes with Sitagliptin; SAVOR-TIMI-53: Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus–Thrombolysis in Myocardial Infarction; EXAMINE: Examination of Cardiovascular Outcomes: Alogliptin versus Standard Care in Patients with Type 2 Diabetes and Acute Coronary Syndrome.
Further details of these trials are available at http://clinicaltrials.gov/
The only such study to date is the ‘Cardiovascular Outcome Study of Linagliptin versus Glimepiride in Patients with Type 2 Diabetes’ (CAROLINA) (NCT01243424), which began in November 2010 and randomized patients with early T2DM and evidence of CVD or at high CV risk to linagliptin therapy versus glimepiride, predominantly as add-on to metformin background therapy. CAROLINA, which includes 6000 patients, is the first head-to-head CV outcome trial of a DPP-4 inhibitor versus an SU active comparator that is sufficiently powered to demonstrate differences in CV events. Its results may provide firm evidence to aid in the future clinical decision-making when selecting therapy in T2DM. Since in practice a proportion of patients with T2DM will have a renal contraindication or some degree of intolerance to metformin, and local guidelines may indicate other treatments as first-line therapy, the CAROLINA study also includes patients who are treatment-naïve or on other monotherapies (SUs, meglitinides or α-glucosidase inhibitors).
Conclusion
Patients with T2DM are at a substantially increased risk of CVD compared with those without diabetes, and hyperglycaemia is a well-established risk factor for CV events and mortality. The rationale for treatment of T2DM is to reduce hyperglycaemia and concomitantly reduce the risk of CV outcomes using a multifactorial interventional approach. However, several large glucose-lowering CV outcome studies have failed to show the benefits of glycaemic control on CV events and mortality. 16 –18
Overall, current evidence on the CV safety of diabetes drugs, and SUs in particular, remains inconclusive and somewhat controversial in light of discrepancies between RCTs and observational studies. There is, therefore, a need for properly designed, long-term, active controlled studies in T2DM to provide solid evidence on the potential benefit or lack thereof of different glucose-lowering therapies on CV events and mortality. Ongoing studies such as CAROLINA will provide important new information on the CV safety of the DPP-4 inhibitor linagliptin versus the SU glimepiride and will contribute to the decision-making process for selecting the best evidence-based second-line therapy for addition to metformin in T2DM.
Key messages
Sulphonylureas (SUs) have been associated with increased cardiovascular (CV) risk, in particular CV mortality, as well as total mortality, in some observational studies and in two questionable randomized controlled trials (RCTs), that is, University Group Diabetes Program (UGDP) and a nested metformin added to SU versus SU alone substudy of the United Kingdom Prospective Diabetes Study (UKPDS).
In 15 well-designed longer-term (≥72 weeks) RCTs using SUs as part of a treatment strategy in type 2 diabetes mellitus (T2DM) [including Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE)] and from RCTs comparing SUs in a head-to-head fashion versus an active comparator [including A Diabetes Outcome Progression trial (ADOPT)], no CV safety signals were detected. However, the data are limited and CV events are inconsistently reported.
Comparative effectiveness studies involving SUs are needed to determine whether SU therapy is linked to increased cardiovascular disease (CVD).
The ongoing Cardiovascular Outcome Study of Linagliptin versus Glimepiride in Patients with Type 2 Diabetes (CAROLINA) trial will provide unique insights into the CV safety profile of the SU glimepiride versus the dipeptidyl peptidase (DPP-4) inhibitor linagliptin.
Footnotes
Acknowledgements
The authors take full responsibilities for the content of this article, were involved at all stages in its development and approved the final submitted version. Editorial assistance (technical editing and formatting) was provided by Giles Brooke PhD and Jennifer Edwards, of Envision Scientific Solutions, during the early-stage preparation of this review.
Conflict of interest
All the authors are involved in the CAROLINA steering committee. Julio Rosenstock has served on scientific advisory boards and received honorarium or consulting fees from Pfizer, Roche, Sanofi, Novo Nordisk, Eli Lilly, MannKind, GlaxoSmithKline, Takeda, Daiichi Sankyo, Johnson & Johnson, Novartis, Boehringer Ingelheim and Lexicon. He has also received grants/research support from Merck, Pfizer, Sanofi, Novo Nordisk, Roche, Bristol-Myers Squibb, Eli Lilly, Forest, GlaxoSmithKline, Takeda, Novartis, AstraZeneca, Amylin, Johnson & Johnson, Daiichi Sankyo, MannKind, Lexicon and Boehringer Ingelheim. Nikolaus Marx served as a speaker for AstraZeneca, Bayer, BMS, Boehringer Ingelheim, Cordis, GlaxoSmithKline, Lilly, MSD, Novartis, Novo Nordisk, Pfizer, Roche, Sanofi-Aventis, as well as Takeda, and as a consultant for AstraZeneca, BMS, Boehringer Ingelheim, Genfit, GSK, MSD, Novo Nordisk, Roche, Sanofi-Aventis and Takeda. Furthermore, he received unrestricted research grants from Boehringer Ingelheim, GSK, MSD as well as Takeda and also participated in clinical trials sponsored by Boehringer Ingelheim and Roche. Steven Kahn has served as a consultant/advisor to Bristol-Myers Squibb, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Intarcia Therapeutics, Johnson and Johnson, Lupin Therapeutics, Merck, Novo Nordisk, Receptos and Takeda. Bernard Zinman has served on scientific advisory boards and received honorarium or consulting fees from Novo Nordisk, Eli Lilly, GlaxoSmithKline, Takeda, Johnson & Johnson, Boehringer Ingelheim, BMS and Sanofi. He has also received grants/research support from Merck, Novo Nordisk and Boehringer Ingelheim. John J. Kastelein has served on scientific advisory boards and received honorarium or consulting fees from Eli Lilly, Takeda, Boehringer Ingelheim, Sanofi, Regeneron, Amgen, Isis, Genzyme, Aegerion, MSD, Pronova, Cerenis, AstraZeneca, Cardoz, CSL Behring, Roche, Novartis and Pfizer. John Lachin serves as a consultant to Boehringer Ingelheim, Gilead Sciences, GlaxoSmithKline, Johnson and Johnson, Eli Lilly, Merck, Novartis and Reata Pharmaceuticals. Erich Bluhmki, Sanjay Patel, Odd-Erik Johansen and Hans-Jürgen Woerle are employees of Boehringer Ingelheim and the manufacturer of linagliptin.
Funding
This study was supported financially by Boehringer Ingelheim.