Abstract
We conducted a meta-analysis of cohort and case-control studies to evaluate all-cause and cardiovascular (CV) mortality of patients with type 2 diabetes mellitus (T2DM) who received sulphonylurea (SU) treatment, when compared to any other diabetes treatment. Only studies reporting raw data on mortality during SU treatment were included. Data were combined using random-effects (RE) models. Unadjusted odds ratios (ORs) are presented. Of 4991 publication titles and abstracts reviewed, 20 studies (n = 551,912 patients) were included. For cohort studies (n = 276,050), patients receiving SU monotherapy or combination treatment had significantly higher all-cause and CV mortality risks compared to any non-SU treatment [all-cause, 13 studies: OR = 1.92, 95% confidence interval (CI) = 1.48–2.49; CV, 5 studies: OR = 2.72, 95% CI = 1.95–3.79]. Validity was limited by the high treatment group heterogeneity (I 2 > 90%) and study-inherent biases/design differences. In conclusion, patients receiving SU treatment had increased all-cause and CV mortality risks. However, the meta-analysis was limited by the high heterogeneity of non-randomized studies.
Introduction
Sulphonylureas (SUs) are an established oral therapy for lowering blood glucose levels in patients with type 2 diabetes mellitus (T2DM). SUs are widely used in routine clinical practice, not only in combination with metformin, but also as first-line monotherapy. 1,2 After the initiation of treatment, SUs effectively lower haemoglobin A1c (HbA1c) levels by approximately 1%–1.5%, similar to metformin, thiazolidinediones or glucagon-like peptide 1 (GLP-1) agonists. 2 –4 Treatment costs are low, which is one reason for the widespread use of SUs despite newer treatment approaches that have no class-specific risk of causing hypoglycaemia and/or weight gain. For more than 30 years, there has been discussion whether SU treatment increases the cardiovascular (CV) mortality of T2DM patients, initially generated by the results of the University Group Diabetes Program (UGDP) study. 5 This study concluded that the SU tolbutamide was associated with increased CV mortality. 5,6 Although the ‘United Kingdom Prospective Diabetes Study’ (UKPDS) did not identify an effect of SU monotherapy on CV mortality, 7 patients treated with SUs in combination with metformin were found to have a significantly increased risk of diabetes-related and all-cause mortality. 7 Recent large, non-randomized cohort and case-control studies have provided evidence of an increased CV mortality during SU treatment when compared to metformin, reinforcing the debate. 8 –11 To further elucidate the potential harm of using SU treatment for T2DM, we conducted a systematic review and meta-analysis to evaluate the all-cause and CV mortality of T2DM patients receiving SU versus any other pharmacological diabetes treatment, including insulin.
Methods
Data sources and searches
A systematic search to identify publications based on the following population, intervention, comparison and outcome (PICO) framework was performed:
Population of interest (P): patients with T2DM;
Intervention of interest (I): any anti-diabetic treatment with SU (tolbutamide was excluded because of the findings of the UGDP study 5 ), either monotherapy or any combination treatment;
Comparators of interest (C): any non-SU treatment for T2DM, including insulin;
Outcomes of interest (O): all-cause and CV mortality.
We performed a systematic search of all the relevant electronic databases, especially PubMed/MEDLINE and EMBASE, from inception (1966 for MEDLINE, 1980 for EMBASE) to April 2011, and an additional review of the Cochrane Central Register of Controlled Trials (CENTRAL) up to 5 May 2011, to identify any publications on SU treatment and CV mortality and on SU and mortality. Search terms were ‘sulfonylurea AND mortality’, ‘sulfonylurea AND cardiovascular’ and any relevant variations of these terms. Reference lists from the main reports, recent issues of key journals and relevant reviews were hand-searched for additional studies of interest, and external experts were asked to provide any publications of interest.
Study selection
Studies were eligible if they (1) had at least one comparator arm reporting mortality data during non-SU anti- diabetic treatment; (2) included more than 100 patients – studies with less than 100 patients were excluded because they were assumed not to report mortality data; (3) lasted >12 months to ensure that a relevant cumulative dosage of the SU was given; (4) were written in English; (5) were published in peer-reviewed journals and (6) reported raw data on mortality (i.e. number of patients who died or mortality rates), as required for the analysis. If an otherwise eligible publication did not include the required raw event data, we asked the authors by email to provide them; studies were excluded in case of no response. Randomized controlled trials (RCTs) were not considered; few RCTs lasting >12 months have been published, the RCTs were methodologically different from the cohort and case-control studies and would have required a separate analysis. Since we aimed to explore the effects of SU treatment during real-life daily clinical conditions (the few RCTs identified did not reflect the general T2DM population), we focused the analysis on the cohort and case-control studies.
Data extraction and quality assessment
Two review authors (G.M. and G.S.) independently checked each publication for eligibility and extracted and tabulated all the relevant data using standardized data abstraction forms. One review author (G.M.) worked together with a second review author (A.H.) who provided in-depth clinical expertise. A stepwise review process (title review, abstract review, full text review) was used to exclude irrelevant studies and duplicate publications reporting identical results. If several publications related to overlapping cohorts, the most relevant report was selected for each individual analysis. For data abstraction regarding CV mortality, we relied on the definition of CV mortality given in the included studies. A systematic quality assessment of each publication was not performed; all eligible publications were included because the analysis was based on raw event data. An independent reviewer (Heiko Moryson, masem Research Institute) compared the work of both investigators and noted any differences between ratings. Any disagreements were settled by consensus between all the authors. All the analyses complied with the reporting guidelines for meta-analysis of observational studies in epidemiology. 12
Data synthesis and analysis
Based on the raw mortality data, unadjusted odds ratios (ORs) for all-cause and CV mortality, with respective 95% confidence intervals (CIs), were calculated for the following treatment arm comparisons, if available: (a) any SU (monotherapy or combination) versus any non-SU treatment, (b) SU monotherapy versus non-SU monotherapy and (c) SU monotherapy versus metformin monotherapy. For each comparison, the unadjusted ORs were pooled using random-effects (RE) models.
We were unable to adjust the analysis for potential confounders. Individual publications used different adjustments including a wide variety of different confounders. Some publications reported hazard ratios (HRs) rather than ORs, and in many cases, the adjusted analyses were reported for treatment cohorts that did not match with our analysis cohorts. Therefore, we had to use raw event rates as the basis for our analysis, but could not calculate adjustments because the required data were frequently not reported. We considered the RE model more appropriate for the analysis of unadjusted data than a fixed-effects model.
Tau 2 and I 2 were measured to evaluate statistical heterogeneity among the different cohort studies. Based on the data available (Supplementary Table 2), we investigated potential sources of heterogeneity by stratification of the all-cause mortality analysis using the following factors (cut-offs defined a priori, based on common medical research criteria): mean duration of follow-up period (≤3 and >3 years), study size (≤10,000 and >10,000 patients), country (Europe, North America, other, international), age (≥65 and <65 years), gender, mean body mass index (BMI; ≥30 and <30 kg/m2) and frequency of macrovascular complications (MVC; <100% and 100% of patients). These variables were chosen because they were relevant for the research question and because these data were available for the majority of studies. Data for other factors that might be relevant, such as HbA1c or the specific SU derivate used, were reported too rarely for a meaningful subgroup analysis. All analyses were conducted using the R open-source statistical program, version 2.13.0 with package metaphor. 13
Results
Figure 1 summarizes the study selection process and most frequent reasons for exclusion of studies. A detailed summary of reasons for exclusions is presented in Supplementary Table 1. Of 5875 publications originally retrieved, 5861 were identified by systematic search, 13 were identified by hand-search and 1 was provided by an external expert. Of these, 8 otherwise eligible publications did not report raw event data. The information was provided upon request for 1 study, 14 the 7 remaining studies were excluded from the analysis. 1,7,11,17 –20
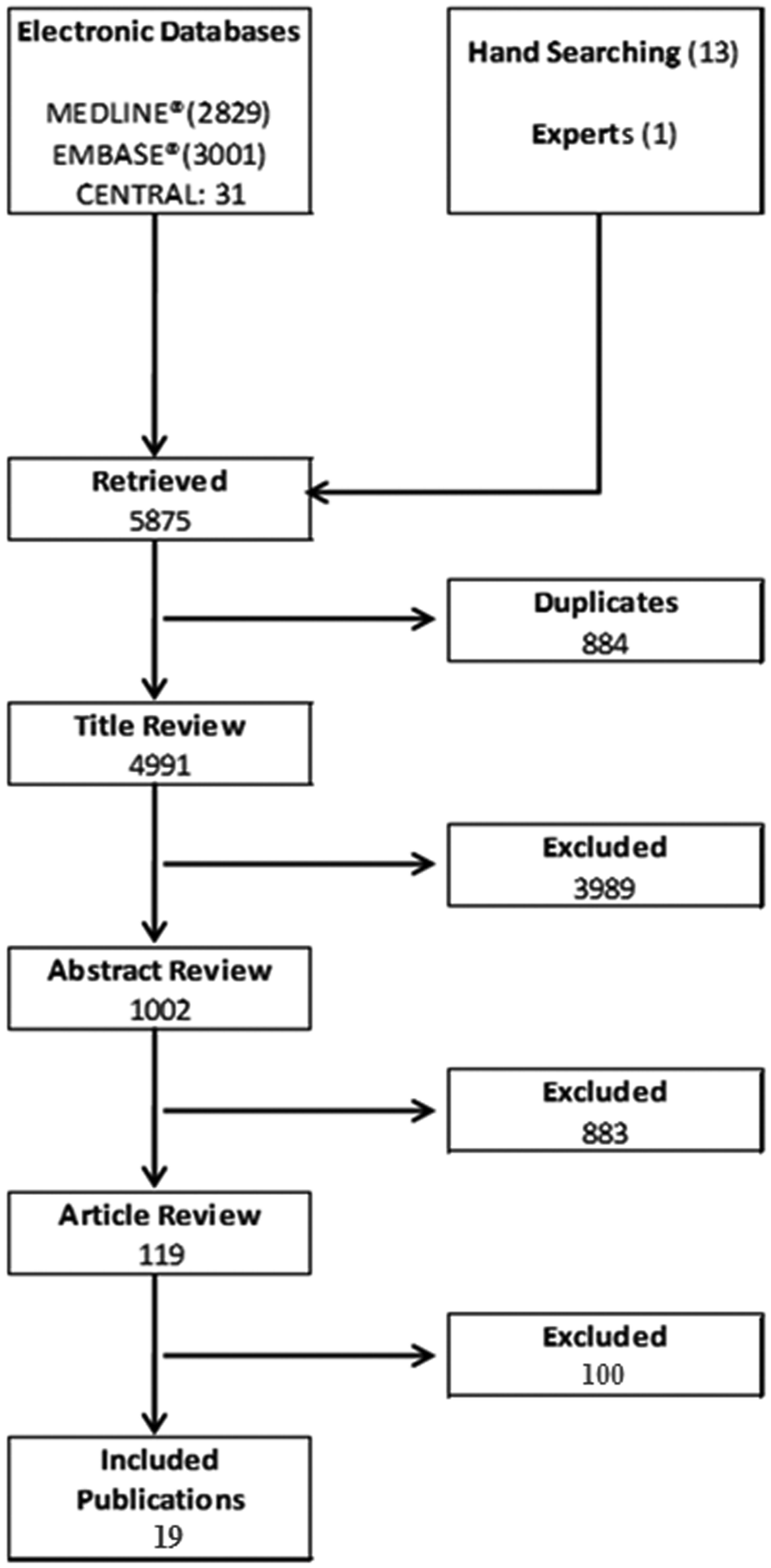
Study selection process. Studies were excluded on the title review level if they were clearly not related to the PICO criteria of this study. The most frequent reasons for exclusion on the abstract level (>1%) were lack of original data (n = 492), review article (n = 71), non-English publication (n = 55), study of <100 patients (n = 45) and study duration ≤12 months (n = 22). The most frequent reasons for discontinuation on the article review level (>5%) were lack of mortality data (n = 26), lack of non-SU comparator arm (n = 15), lack of SU arm (n = 10), review article (n = 8), study duration ≤12 months (n = 8) and lack of raw event data (n = 7). Three publications were excluded reporting duplicate data (Evans et al., 14 Fisman et al. 15 and Johnson et al. 9 ).
Overall, 19 publications reporting 20 individual studies (17 cohort studies, 3 case-control studies) were included in the analysis. The cohort study publication by Schramm et al. 10 reported all data separately for patients with and without prior myocardial infarction. The two different populations were kept as separate studies to enable separate evaluation for the subgroup analysis. These studies included data for 551,912 T2DM patients overall and for 329,775 patients who received any SU treatment (monotherapy or combination therapy).
Table 1 summarizes the individual studies and patient characteristics included in the meta-analysis (see Supplementary Table 2 for details by treatment arm). Study types (cohort and case-control studies) were analysed separately with the major focus on the 17 cohort studies, in which data were collected between 1991 and 2007 for a total of 276,050 patients, including 168,267 patients on any SU treatment. The number of patients per cohort study ranged from 152 to 105,039. 10,31 Mean ages ranged from 52.5 years to 76.0 years per treatment arm 10,21 and mean follow-up durations ranged from 12 months to 124.8 months. 27,33
Overview of studies included in the analysis.

BMI: body mass index; CV: cardiovascular; MVC: macrovascular complications; Non-SU: any non-sulfonylurea anti-diabetic treatment, including insulin; N: number of patients; NR: not reported; SU: sulfonylurea.
Mean or median, as reported.
All-cause mortality was reported in all but one of the 17 cohort studies, 29 and three additional studies 21,27,34 were excluded from the all-cause mortality analysis because they were based on patient cohorts overlapping with cohorts reported in other studies (see Table 1). Figure 2 depicts the unadjusted ORs for all-cause mortality for the remaining 13 studies and the pooled OR derived from RE models, comparing patients on any anti-diabetic treatment that includes SUs with patients on any anti-diabetic treatment that excludes SUs. Patients who received any SU treatment had a higher OR for all-cause mortality of 1.92 (95% CI = 1.48–2.49) than those who did not receive any SU treatment. When assessing monotherapies only, patients on SU monotherapy had a higher OR for all-cause mortality, 2.48 (95% CI = 1.95–3.16), when compared with any other anti-diabetic monotherapy including insulin (Figure 3), and an OR of 2.63 (95% CI = 2.08–3.04) when specifically compared with metformin (based on 10 studies where patients on monotherapy were reported separately).
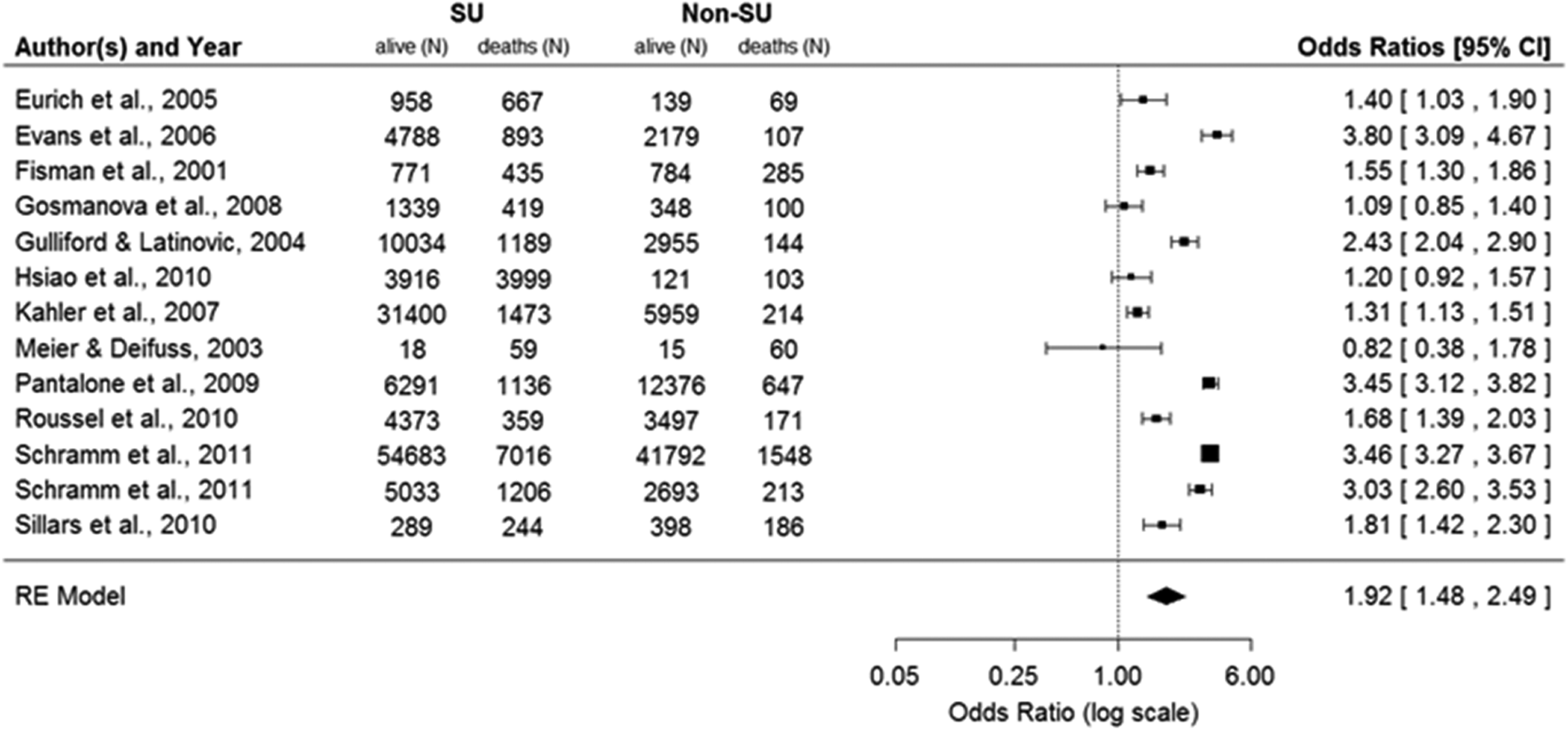
Unadjusted odds ratios for all-cause mortality during SU treatment: any SU (monotherapy or combination) versus any non-SU treatment.
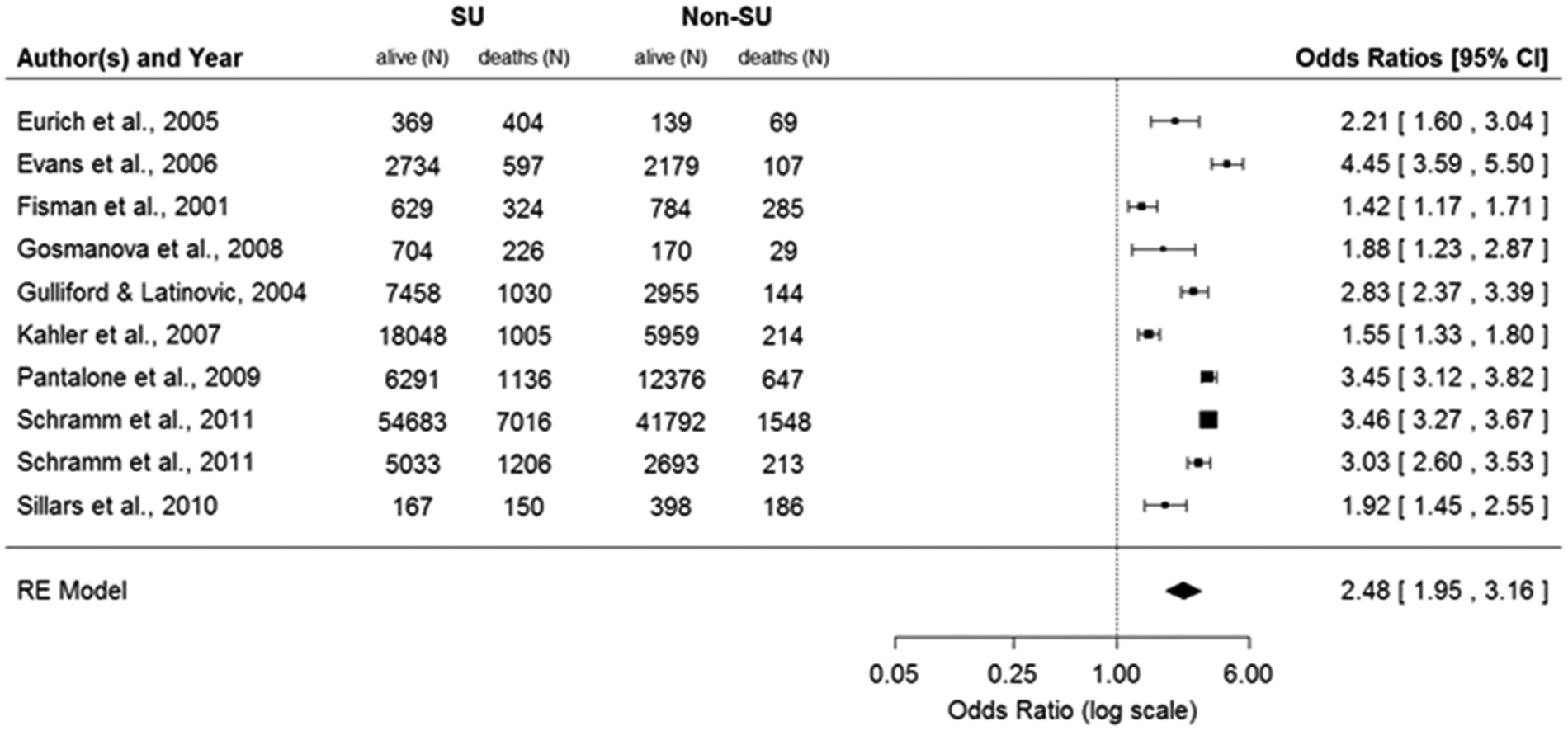
Unadjusted odds ratios for all-cause mortality during SU treatment: SU monotherapy versus non-SU monotherapy.
CV mortality data were reported in only seven cohort studies, and two of these studies 22,34 were excluded from this analysis because they were based on patient cohorts overlapping with cohorts reported in other studies (see Table 1). 16,29 Figure 4 presents the ORs for CV mortality based on the five remaining studies, comparing patients on any anti-diabetic treatment including SU with patients on any non-SU treatment. Patients who received any SU treatment had an overall OR for CV mortality of 2.72 (95% CI = 1.95–3.79) when compared with non-SU patients. Patients on SU monotherapy had an OR for CV mortality of 2.93 (95% CI = 2.16–3.98) when compared with any other anti-diabetic monotherapy including insulin (Figure 5) and an OR of 3.09 (95% CI = 2.30–4.14) when compared specifically with metformin.
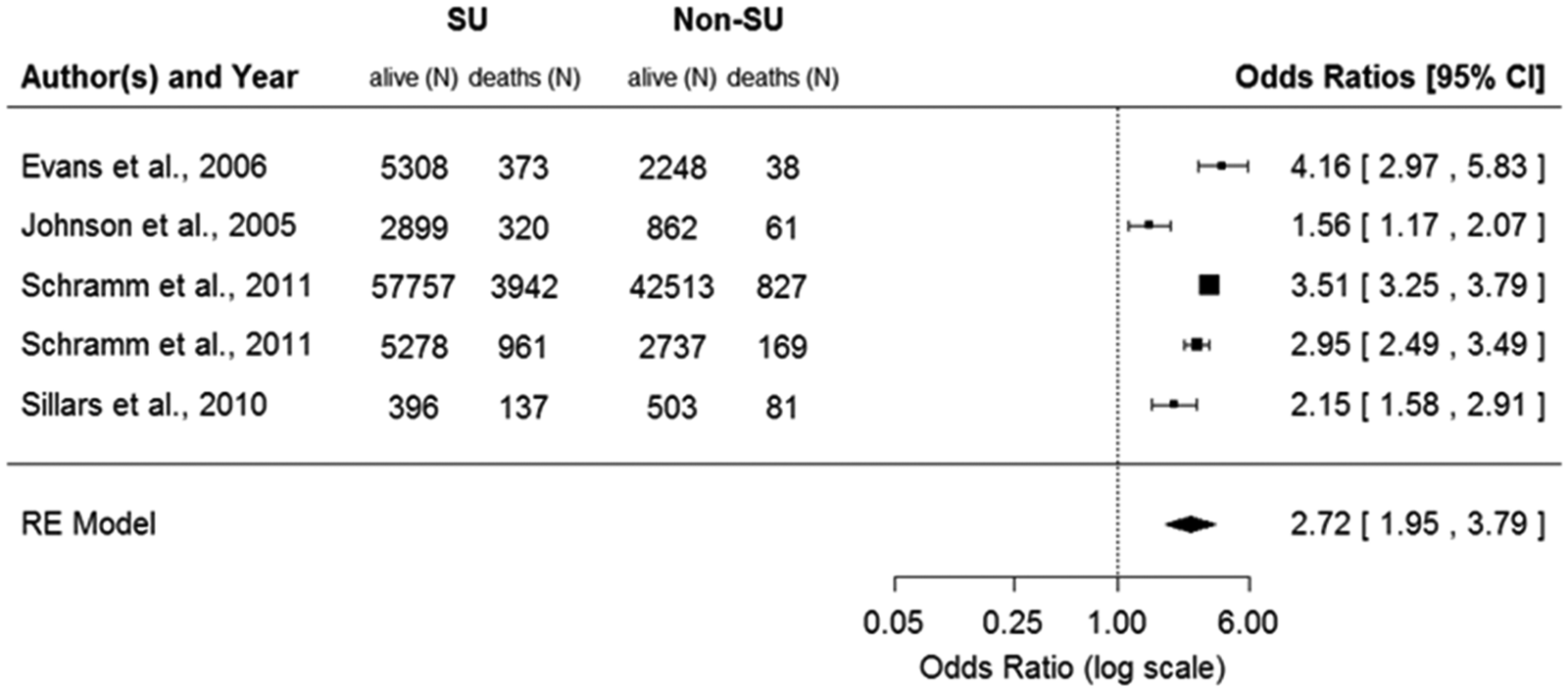
Unadjusted odds ratios for CV mortality during SU treatment: any SU treatment (monotherapy or combination) versus any non-SU treatment.
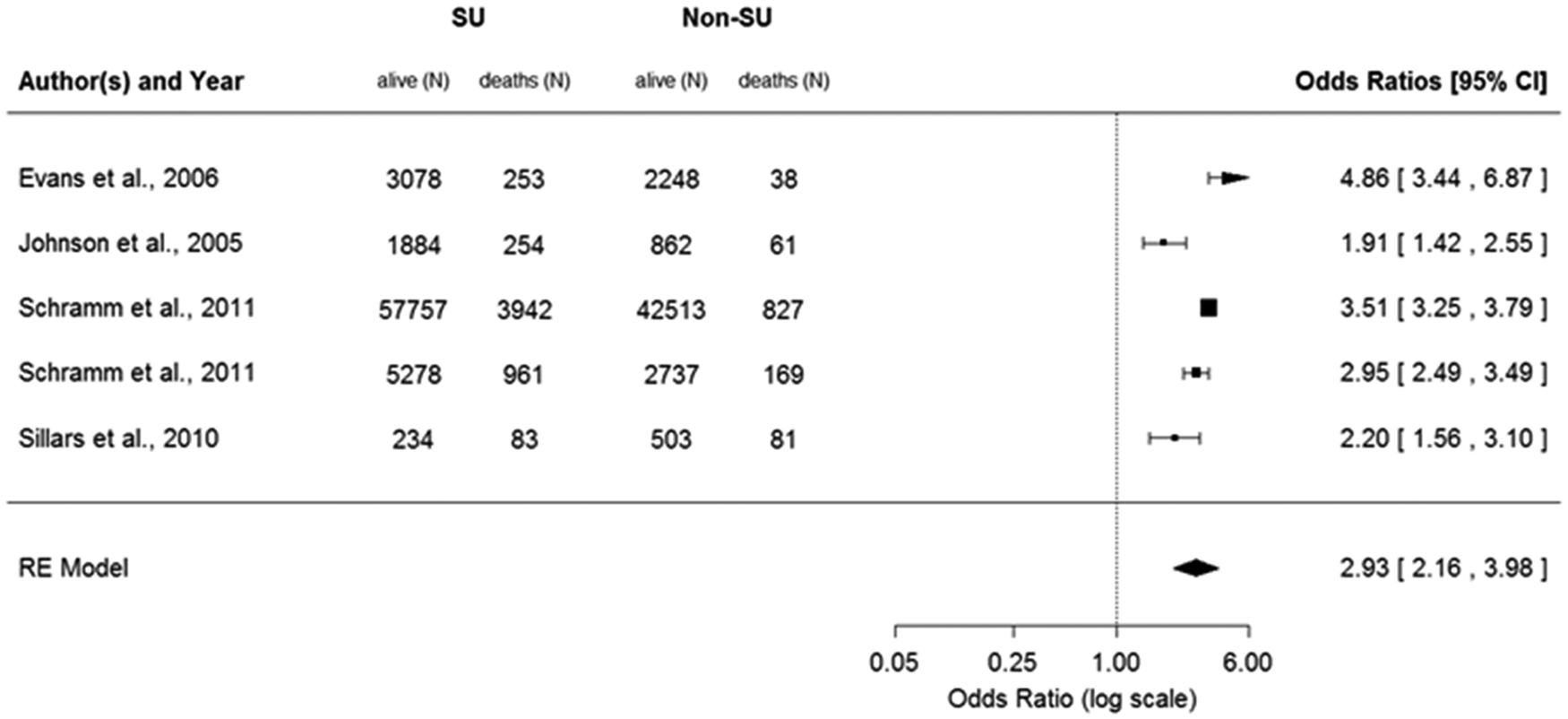
Unadjusted odds ratios for CV mortality during SU treatment: SU monotherapy versus non-SU monotherapy.
Sensitivity analyses revealed large heterogeneity for all six of the individual analyses reported above (I 2 = 89.1%–97.1%). Exclusion of the smallest and largest studies neither changed the pooled OR nor reduced the heterogeneity to a relevant degree. To explore potential sources of this heterogeneity, additional subgroup analyses were performed on the cohort studies. The comparison of all-cause mortality associated with any SU treatment versus all-cause mortality with any non-SU treatment was used for this analysis because it provided the largest patient numbers. There were few differences in pooled ORs between the subgroups evaluated (Figure 6), leaving most of the heterogeneity unexplained. The association between SU treatment and increased all-cause mortality tended to be smaller for shorter follow-up times and older age. BMI data were available from six cohort studies only, but the association between SU treatment and all-cause mortality was significantly increased in obese patients [BMI ≥ 30 kg/m2, OR (95% CI): 3.52 (3.21–3.85); BMI < 30 kg/m2, OR (95% CI): 1.47 (1.23–1.76)].
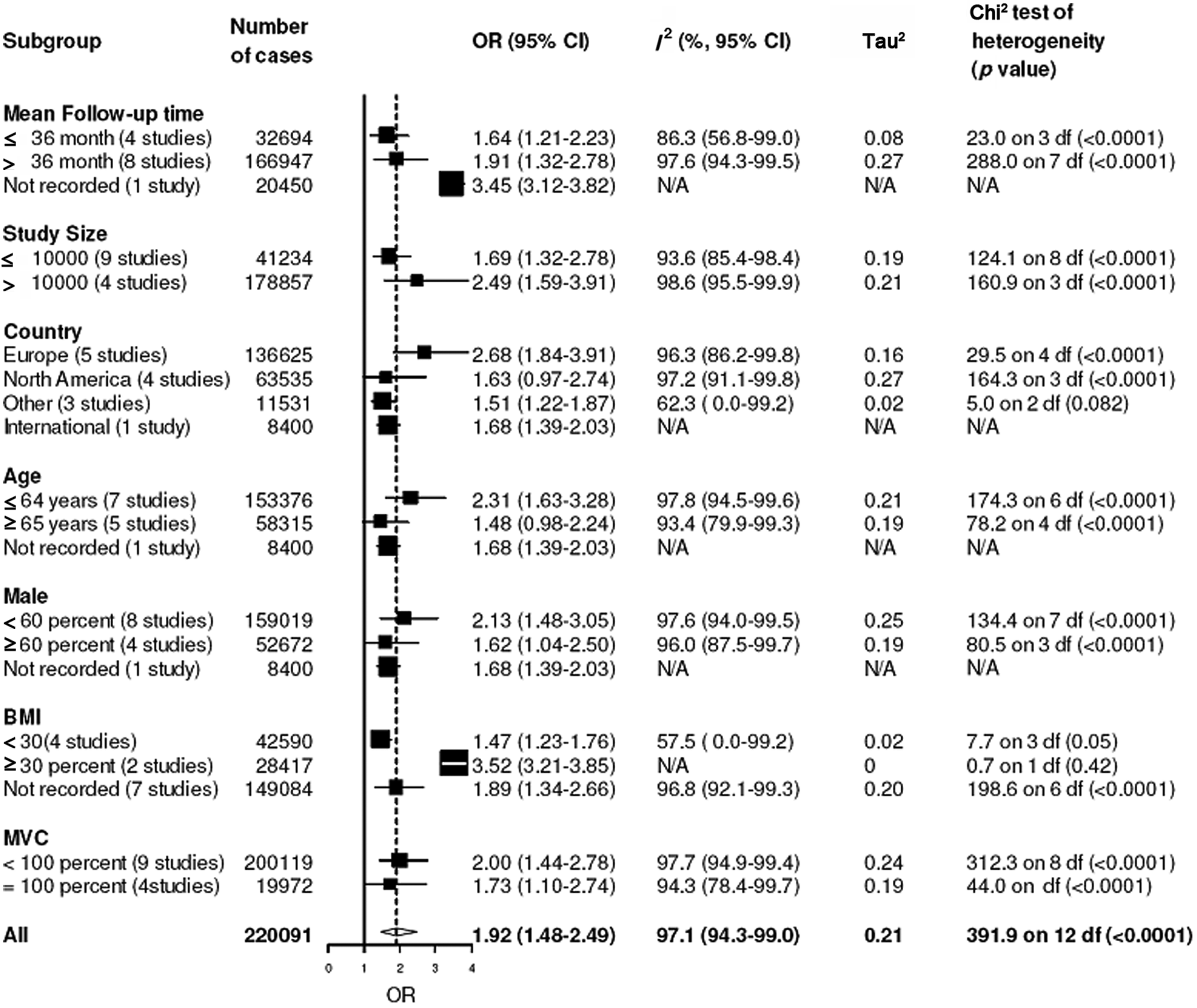
Subgroup analyses of unadjusted odds ratios for all-cause mortality during SU treatment: any SU treatment (monotherapy or combination) versus any non-SU treatment.
For the three case-control studies, 8,35,36 the individual ORs were not combined for the following reasons: two of the three studies were based on patients from the same database (UK General Practice Research Database, GPRD) with potential overlap; 8,36 and in the third study by Lipscombe et al., 35 the publication included inconsistent patient numbers. In each of the two large-scale independent studies, patients who received SU treatment had a significantly higher all-cause mortality risk when compared to patients on any non-SU treatment [Azoulay et al.: 8 92,273 patients, study period 1988–2008 (OR = 1.36, 95% CI = 1.27–1.46); Lipscombe et al.: 35 180,915 patients, study period 2002–2005 (OR = 1.79, 95% CI = 1.74–1.83)]. For the small study by MacDonald et al. 36 (2674 patients, study period 1988–2007) with potential overlap, the unadjusted OR favoured SU (OR = 0.79, 95% CI = 0.67–0.94).
Discussion
In this meta-analysis, based on 17 cohort studies and three case-control studies that in total included more than 550,000 T2DM patients, patients who had received SU treatment had significantly increased risks of all-cause and CV mortality. Study types were analysed separately. RCTs were not included because few long-term RCTs reporting mortality data have been published and those available did not reflect the general T2DM population. We aimed to explore the effects of SU treatment during real-life daily clinical conditions and therefore focused the analysis on the cohort and case-control studies. Based on the cohort study analysis, the all-cause and CV mortality risks of patients receiving any diabetes treatment that includes SUs were 1.9-fold and 2.7-fold higher compared with patients receiving any diabetes treatment that excludes SUs. Of note, patients receiving SU monotherapy even had a 2.6-fold increase in all-cause mortality and a 3.1-fold increase in CV mortality when compared to metformin treatment.
These results are based on unadjusted OR data (see limitations below). The majority of individual cohort studies had reported adjusted HRs or ORs, which can be expected to be less biased. The association between SU cohort and mortality was generally less strong after adjustment, but still pointed into the same direction. In the three largest studies with adjusted data available for all SUs combined, adjusted all-cause mortality risks were all higher with SU monotherapy when compared with metformin monotherapy after adjustment for age, sex and other covariates relating to duration of disease, comorbidities and concomitant medication. 16,21,30 The adjusted all-cause mortality risks associated with SU monotherapy were 1.85 (HR, 95% CI = 1.56–2.17), 1.18 (HR, 95% CI = 1.02–1.33) and 1.15 (OR, 95% CI = 0.91–1.47), respectively. 16,21,30
It is surprising that by today, no prospective randomized controlled clinical trials have been performed addressing the effects of SU treatment on CV end points or mortality rates. Three recent RCTs including a SU treatment arm recorded CV end points at least as secondary end point for safety evaluation. In these studies, no CV harm could be observed during treatment with SUs. 37 –39 However, these trials were conducted in a different patient population with the majority of patients already having MVC. One might hypothesize that this patient population had a high mortality risk anyway, so SU therapy exerted no additional effect. The only exception was the ADOPT trial, which was conducted in a lower risk T2DM population. 37 Other pivotal diabetes trials powered to detect effects on diabetes-related end points, like ADVANCE, VADT, ACCORD and UKPDS 40 only addressed mortality rates during SU within retrospective subgroup analysis. Another recently published RCT in 1500 metformin-treated T2DM patients reported CV safety as secondary end point of glimepiride versus the new DPP-4 inhibitor linagliptin. 41 This study revealed a significant 50% increase in the relative risk of the combined CV end point (CV death, non-fatal myocardial infarction or stroke, unstable angina with hospitalization) in the SU group.
Data for the three case-control studies 8,35,36 were not combined, but SU treatment was associated with increased all-cause mortality in the two large, independent studies. 8,35 Our meta-analysis is additionally supported by several cohort studies not included in the analysis because no raw data were reported. 1,7,11,17 –20 Furthermore, a recent population-based cohort study from Italy also reported an increased risk of macrovascular disease, which progressively increased as the compliance with SU therapy increased, indicating a direct effect of these agents on patient outcome. 42 The pathophysiological processes by which SUs may adversely affect patients’ CV risk and mortality have not been fully elucidated, but there are at least four different pathways that might potentially explain these findings.
First, one side effect of SU treatment is weight gain, and there is evidence for an unfavourable effect of SUs on body composition, that is, an increase of the visceral fat compartment. 3,7,43 –45 The accumulation of visceral fat has been shown to increase insulin resistance and to deteriorate several CV risk markers [lipid profiles, high-sensitivity C-reactive protein (hsCRP)] and may cause high blood pressure and a significant increase in CV morbidity and mortality. The amount of visceral fat identified by computer tomography is an independent predictor of coronary artery disease. 46,47 In our analysis, the all-cause mortality risk associated with SU treatment was particularly high for obese patients (OR = 3.5 for patients with BMI ≥ 30 kg/m2).
Another possible reason for increased CV mortality relates to the fact that SU treatment not only stimulates insulin but also proinsulin release. 48 While the insulin/ proinsulin ratio was introduced as a marker for beta cell functionality and the capability of the beta cell to convert intact proinsulin into insulin and C-peptide, 49 the absolute plasma levels of intact proinsulin were shown to be predictive of CV risk in patients with and without diabetes. 50 –54
Next, SUs have been shown to block the process of ischaemic preconditioning, an endogenous mechanism that protects the heart from ischaemic injury. 55,56 Some SUs have a high selectivity for the pancreatic SU receptors (tolbutamide, chlorpropamide, gliclazide, glipizide), while others, like glibenclamide (glyburide) and glimepiride, also bind to SU receptors on myocardial muscle cells. 57,58 The latter group of SUs would be expected to affect myocardial function. 59,60 This might explain the data from Schramm et al., which suggest that different types of SUs may differ in their association with CV mortality. Gliclazide, for example, showed no association with CV mortality, whereas glimepiride was associated with an increased risk (adjusted HR = 1.26). 10 However, a smaller cohort analysis found no differences in mortality between users of pancreatic- specific and non-pancreatic-specific SUs. 61 In a retrospective analysis of electronic health-care records derived from a clinical data repository at the Cleveland Clinic, treatment with glipizide, glyburide and glimepiride all resulted in a significantly increased mortality rate compared with metformin treatment. 62 However, in this study, the glimepiride arm had a slightly lower mortality rate than the other SUs in the subgroup of patients with a history of coronary artery disease. A subgroup analysis of our data for different SUs was not possible because most of the cohort studies did not report the specific SUs used.
Hypoglycaemia is a common side effect of SU therapy. 2,3,63 Recent large-scale studies (ACCORD, ADVANCE and VADT) have shown that T2DM patients who experience severe hypoglycaemic episodes are at an increased risk of all-cause and CV mortality. 40,64,65 During acute hypoglycaemia, electrocardiogram (ECG) abnormalities and symptoms of ischaemia may occur; in particular, prolonged corrected QT (QTc) intervals have been reported. 66 –69 Each of these pathophysiological pathways – accumulation of visceral fat, rise in proinsulin levels, inhibition of ischaemic preconditioning and higher frequency of hypoglycaemia – may contribute to the increased mortality risk during SU treatment.
The major limitation of our analysis is that evidence from RCTs is missing (the RCTs identified during the initial literature research did not reflect the general T2DM population and/or real-life daily conditions); we therefore focused on the cohort studies. The validity of the pooled OR estimates in the observational trials analysis was limited by the high level of heterogeneity between the treatment groups of these studies (I 2 > 90%), the inherent biases and differences in study designs, and the potential impact of measured and unmeasured confounders and selection bias because we had to use unadjusted data. Adjustment for confounding factors, such as age, disease duration, comorbidities, concomitant medications, assessment of drug exposure, medication switches and dropout rates, would have been ideal, but options were limited because the required data were frequently unavailable. Therefore, we cannot exclude the fact that patients receiving SU treatment had a higher CV risk than non-SU patients for reasons independent of SU treatment itself. The majority of individual cohort studies had reported adjusted hazard or ORs, but the adjustments used varied widely across studies. The adjusted analyses of the individual studies in general reported less strong associations between SU treatment and mortality when compared to our unadjusted meta-analysis based on the raw data of these studies (see above), suggesting that different treatment options may have been used in different patient populations with different mortalities. However, the adjusted data still pointed into the same direction, and we found consistent results in several subgroup analyses (e.g. by age, gender, BMI or proportion of patients with MVC), suggesting robustness of the association.
With these methodical weaknesses, the results need careful interpretation and should be used for hypothesis generation only. Our approach to systematically review all the available observational studies providing mortality data during SU treatment provides the best evidence currently available. The strength of non-randomized studies is that they reflect the patients’ real-life situation better than RCTs, which often exclude special subpopulations. However, the individual cohort studies that were integrated in this meta-analysis were performed over a wide time span from 1991 until 2007. Because treatment guidelines on the use of SUs have changed over time (SUs are no longer recommended as first-line treatment), the patient populations studied in the older studies may not fully reflect the patients as treated in routine practice today.
It should be emphasized that no extensive, long-term RCTs of SU treatment taken to hard macrovascular end points have been published so far. However, the currently ongoing ‘Cardiovascular Outcome Study of Linagliptin Versus Glimepiride in Patients With Type 2 Diabetes’ (CAROLINA) study, which investigates the impact of long-term treatment with linagliptin versus glimepiride on CV morbidity and mortality in high-risk T2DM patients, will provide further clarification. 70
In conclusion, patients receiving SU treatment had significantly higher all-cause and CV mortalities compared with patients on non-SU treatment in this meta-analysis of non-randomized studies. SUs are an established treatment option, and those are commonly preferred over new treatments because of well-established, long-term safety experience. 1 Based on all available evidence from cohort and case-control studies, and given the lacking evidence from RCTs, the proposed favourable safety profile of SU-based treatment remains questionable. However, while solid long-term data on CV risk and mortality are lacking for established and new T2DM treatments alike, the known evidence for SU treatment with regard to early treatment failure, weight gain and risk of hypoglycaemia is not in favour of SU use. According to a new common position statement of the European Association for the Study of Diabetes (EASD) and the American Diabetes Association (ADA), published in June 2012, glucose lowering treatments in T2DM patients should be pursued within a multifactorial risk reduction strategy. 71,72 Especially in older patients (>65 years) or patients with an unfavourable CV risk profile, treatments associated with weight gain or increased risk of hypoglycaemia should be considered with caution. Based on these considerations, the risks and benefits of SU treatment should be carefully evaluated for each individual patient.
Footnotes
Acknowledgements
Karin Helsberg (PhD), Trilogy Writing and Consulting, Frankfurt, Germany, provided medical writing support for this article. Renate Barharn, Lilly Deutschland GmbH, Bad Homburg, supported the literature search. Heiko Moryson, masem Research Institute supported data validation. Statistical analyses were programmed at the masem Research Institute GmbH, Wiesbaden. Gero Schwenk died in January 2012.
Conflict of interest
Axel Haupt is an employee of Lilly Deutschland GmbH, Bad Homburg, Germany. Thomas Forst and Andreas Pfützner serve as consultants for Lilly Deutschland GmbH, Bad Homburg, Germany. Guido Moeser and Gero Schwenk are researchers of the masem Research Institute GmbH, Wiesbaden, Germany. Markolf Hanefeld has no duality of interest with respect to the meta- analysis. He has worked as Steering committee member for RCTs sponsored by GSK and Sanofi-Aventis. He received honoraria as speaker from Bayer, Takeda, GSK, Sanofi-Aventis, Novartis and Novo Nordisk. Stephan Jacob has received honoraria from Abbott, AstraZeneca, Bayer, Berlin Chemie, BMS, Boehringer Ingelheim, Daiichi Sankyo, Essex, EuMeCom, GSK, LighterLife, Lilly, Merck, MSD, Novo Nordisk, Novartis, Pfizer, Roche, Sanofi-Aventis, Schwarz UCB, Solvay, Takeda and Viatris.
Funding
This study was funded by Lilly Deutschland GmbH, Bad Homburg, Germany.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
