Abstract
The second Diabetes Glucose and Myocardial Infarction (DIGAMI 2) study randomised patients with diabetes and myocardial infarction to insulin or oral-based treatment. To determine the effects of insulin-based treatment, the Diabetes Treatment Satisfaction Questionnaire (DTSQ) and the Psychological General Well-Being (PGWB) Index were administered at baseline and 12 months. Insulin-treated patients (n = 197) had a worse risk profile and more co-morbidity at baseline than patients on oral glucose-lowering agents (n = 127). The treatment satisfaction and psychological well-being was similar between insulin and oral groups at baseline [DTSQ: median (first–third quartile) 30 (24–34) vs 31 (27–34), NS; PGWB: 77 (73–82) vs 79 (76–82), NS] and at 12 months [DTSQ: 32 (28–35) vs 34 (30–36), NS; PGWB: 81 (78–84) vs 82 (78–84), NS]. Improvement was significant in both groups. Insulin-based therapy was well accepted and did not decrease treatment satisfaction or psychological well-being compared to oral glucose-lowering treatment in patients with type 2 diabetes and myocardial infarction.
Introduction
Type 2 diabetes is, as defined by the World Health Organisation (WHO), a chronic metabolic disorder characterised by defects in insulin secretion and/or insulin action causing a progressive loss of glycaemic control. It is associated with dysfunction and failure of various organs, mainly due to the development of micro- and macrovascular complications, causing an increase in morbidity and mortality. 1 The global burden of diabetes, whereof type 2 diabetes accounts for about 90%, is huge and rapidly increasing. 2 In a recent report from the International Diabetes Federation (IDF), the number of people with diabetes was 336 million in 2011 with an estimated 64% increase to 551 million within two decades. 3 Diabetes-related mortality, of which at least half is caused by cardiovascular (CV) complications, accounts for 8% of the global all-cause mortality. 3 The presence of diabetes increases the risk to develop CV disease two to three times 2 independent of traditional risk factors. 4 Thus, diabetes imposes an increasing burden on health-care budgets, and improved management should have great potential to reduce these costs. 3
The importance of glycaemic control in reducing or postponing the development of diabetes-related complications is emphasised in current management guidelines.2,5 Insulin is the most powerful agent to efficiently decrease hyperglycaemia. The joint American Diabetes Association (ADA)/European Association for the Study of Diabetes (EASD) recommendations advocate early insulin-replacement strategies to reach an optimal glycaemic target. 5 Still such treatment is often postponed until maximum dosages of a combination of oral agents fail to reach recommended glucose targets, exposing the patients to prolonged periods of inadequate glycaemic control.6,7 The reluctance to start insulin is likely influenced by concerns imagined with insulin administration, including adverse effects on quality of life (QoL). An improved understanding of the patient’s perception of glucose-lowering treatment may help to overcome such barriers and, if favourable, facilitate earlier introduction of insulin.
The aim of the present study, a pre-planned sub-study to the second Diabetes Glucose and Myocardial Infarction (DIGAMI 2) trial, was to investigate whether insulin therapy influenced treatment satisfaction, psychological well-being and self-reported health in patients with type 2 diabetes and myocardial infarction.
Design and methods
DIGAMI 2 investigated three different glucose-lowering strategies in 1253 patients with type 2 diabetes admitted for suspected acute myocardial infarction as previously reported. 8 In brief, patients were included if they had a history of type 2 diabetes or an admission blood glucose > 11.0 mmol/L and were hospitalised due to a suspected acute myocardial infarction based on chest pain >15 min during the preceding 24 h and/or recent electrocardiogram (ECG) signs of new Q-waves and/or ST-segment deviations in two or more leads. Exclusion criteria were inability to cope with insulin treatment, residence outside the hospital catchment area, previous participation in DIGAMI 2 or other studies. Patients were randomly allocated to one of the three glucose-lowering strategies: (1) acute insulin–glucose infusion during at least 24 h followed by multi-dose insulin long term (n = 474), (2) insulin–glucose infusion acutely followed by standard therapy long term (n = 473), and (3) control patients given glucose-lowering treatment according to local practice (n = 306). No statistically significant difference in glucose control, CV event rates or mortality was noticed between the treatment groups during the median follow-up period of 2.1 years.
Study population
The present population comprises 533 patients from Sweden, Norway, Denmark and Finland, who accepted to participate in the QoL sub-study. The participants were allocated to one of the two groups based on glucose- lowering treatment at the time of hospital discharge. Patients prescribed insulin at least once daily were allocated to the insulin group, although they may have been on oral glucose-lowering agents as well. Patients on oral treatment or diet comprised the oral treatment group. All patients received lifestyle advice and concomitant treatment according to international guidelines for myocardial infarction. 9
Glucometrics
Random blood glucose obtained immediately after admission was analysed locally as whole blood glucose in millimoles per litre. HbA1c was analysed by high-performance liquid chromatography in a core laboratory (Department of Laboratory Medicine, Malmö Hospital, Sweden) on capillary-derived blood samples applied on filter paper with an upper normal limit of 5.3%. 10
QoL assessment
QoL was assessed by three distinct measures: Diabetes Treatment Satisfaction Questionnaire (DTSQ), Psychological General Well-Being (PGWB) Index and Rating Scale (RS). All three measures were administered as self-report questionnaires at two occasions: (1) prior to hospital discharge and before starting insulin and (2) at a follow-up visit after 12 months.
DTSQ measures the experience of diabetes treatment including ease of use, side effects and efficacy. 11 This tool can be used in type 1 and type 2 diabetes, for all diabetes treatments and has been validated and recommended by WHO and IDF. 12 The questionnaire consists of six items assessing treatment satisfaction and two items assessing patient perceived frequency of hyper- and hypoglycaemia. Each item is rated on a Likert-type scale graded from 0 to 6. The total score reflecting treatment satisfaction is 36 points. A high score represents high satisfaction and vice versa. The questions assessing hyper- and hypoglycaemia are evaluated separately and a score of 0 means never while a score of 6 means always.
The PGWB is a validated 22-item questionnaire that is widely used in clinical trials and epidemiological research to assess self-perceived psychological well-being or distress. 13 It evaluates six dimensions of mental health: anxiety, depressed mood, vitality, general health, self-control and subjective well-being and is divided equally between positive and negative questions. The experience of symptoms or feelings is rated on a six-graded scale. Self-perceived evaluation of psychological well-being is expressed as a summary score, which can reach a maximum of 132. High scores represent greater well-being.
The RS is a validated tool to assess self-reported health. 14 It is a simple, preference-based instrument for assigning one numerical value for a certain health state in relation to perfect health or death. The RS consists of a straight vertical scale [Visual Analog Scale (VAS)] numbered from 0 to 100, where 0 is death and 100 is perfect health. 14 Patients were asked to mark their current health state on the vertical line in relation to perfect health and death.
Ethics
All patients gave their signed consent to participate following oral and written information about the study. The study conformed to good clinical practice guidelines, and the investigations were carried out in accordance with the Declaration of Helsinki. Local ethics review boards approved this study.
Statistical analysis
Continuous variables are presented as median (first–third quartile). The categorical variables are expressed by counts and proportions. The Wilcoxon–Mann–Whitney rank sum test was used when comparing continuous variables and Fisher’s exact test when comparing categorical variables. Two-sample t-test was used to determine significant difference between scores at baseline and 12 months. A two-sided p-value of <0.05 was accepted as statistically significant.
The DTSQ and PGWB questionnaires were included in the analyses if ≥60% of the questions had been answered. To be included in this study, the requirement of completed answers had to be fulfilled both at baseline and at 12 months. A mean imputation method was used to replace missing values in individual questionnaires with ≥60% of the questions answered by replacing missing values with the mean value from the whole population before calculating the respective individual total score. By methodological reasons, patients who were not treated with any glucose-lowering agents were excluded (baseline: n = 68; 12 months: n = 29) from the analysis of treatment satisfaction (DTSQ), and the same applied for patients who changed treatment from oral to insulin or vice versa during the follow-up period (n = 15). The baseline glucose-lowering treatment or change of such treatment during follow-up does not interfere with the analysis of PGWB and RS. The RS score was included in the analysis if the score was clearly marked with a cross or vertical line across the scale. Patients who used a circle or more than one cross to mark the line were excluded. Self-reported hypoglycaemia was only analysed in patients who were on glucose- lowering medication both at baseline and at 12 months. Microsoft Office Excel 2007 was used for mean imputation. Minitab version 13.32 was used for the rest of analyses.
Results
Clinical characteristics
The invitation to participate in this study was accepted by 533 patients, and of those, 324 had QoL data at both baseline and the 12-month visit. Pertinent characteristics from the participating patients are shown in Table 1. The diabetes duration was longer; HbA1c was higher and history of hyperlipidaemia, hypertension, previous myocardial infarction, previous heart failure and coronary artery bypass grafting (CABG) was more prevalent in the insulin-treated patients than among those prescribed oral glucose-lowering agents. Insulin was prescribed to 197 (61%) of the total patient population at the time of hospital discharge, most frequently in multiple dosages. Sulphonylureas and metformin were the most commonly used glucose-lowering drugs among patients treated with oral agents.
Patient characteristics. Data are expressed as median (first–third quartile) or number (%).
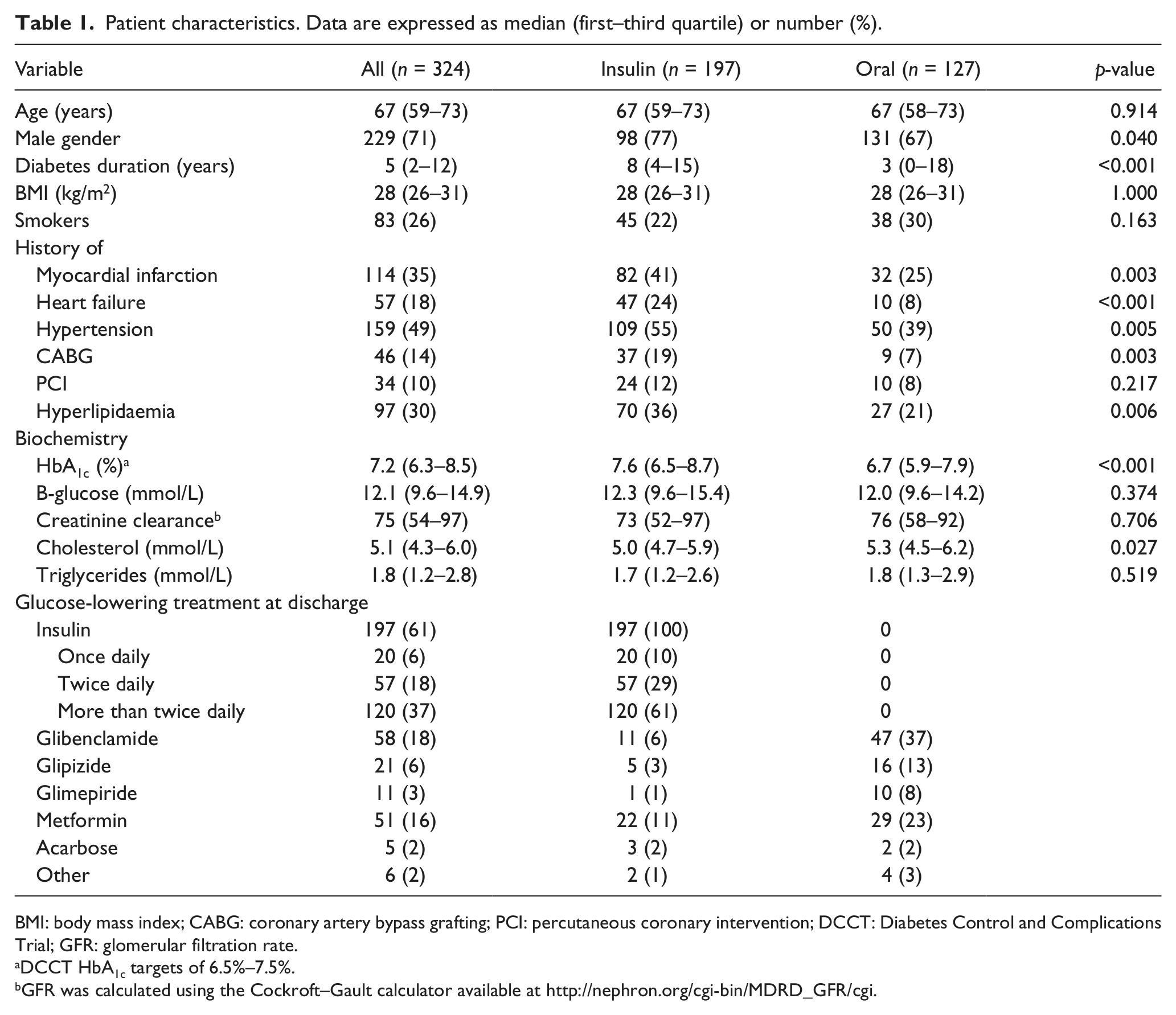
BMI: body mass index; CABG: coronary artery bypass grafting; PCI: percutaneous coronary intervention; DCCT: Diabetes Control and Complications Trial; GFR: glomerular filtration rate.
DCCT HbA1c targets of 6.5%–7.5%.
GFR was calculated using the Cockroft–Gault calculator available at http://nephron.org/cgi-bin/MDRD_GFR/cgi
QoL
Treatment satisfaction and psychological well-being were similar between treatment groups at baseline, improved significantly during the follow-up and did not differ between the treatment groups at 12 months (Table 2 and Figure 1). The analysis of the six dimensions within PGWB did not show any differences between groups (data not shown). According to RS, insulin-treated patients perceived their health lower than patients on oral treatment at baseline and at 12 months; however, both groups showed significant improvement in the measure over the course of the study. Patients with newly introduced insulin rated similar treatment satisfaction and perceived health but higher psychological well-being as patients who continued previously instituted insulin (Table 2). Body mass index (BMI) [kg/m2; median (first–third quartile)] was similar between the two treatment groups at baseline (Table 1) and 12 months [insulin group: 29 (26–32) vs oral: 28 (26–31), p = 0.468], despite a significant increase in BMI in insulin treatment group over time [28 (26–31) vs 29 (26–32), p = 0.038]. QoL scores were similar between patients with increasing and decreasing BMI at 12 months (data not shown).
Comparisons of QoL scores at baseline and 12 months by treatment group. Data are expressed as median (first–third quartile)
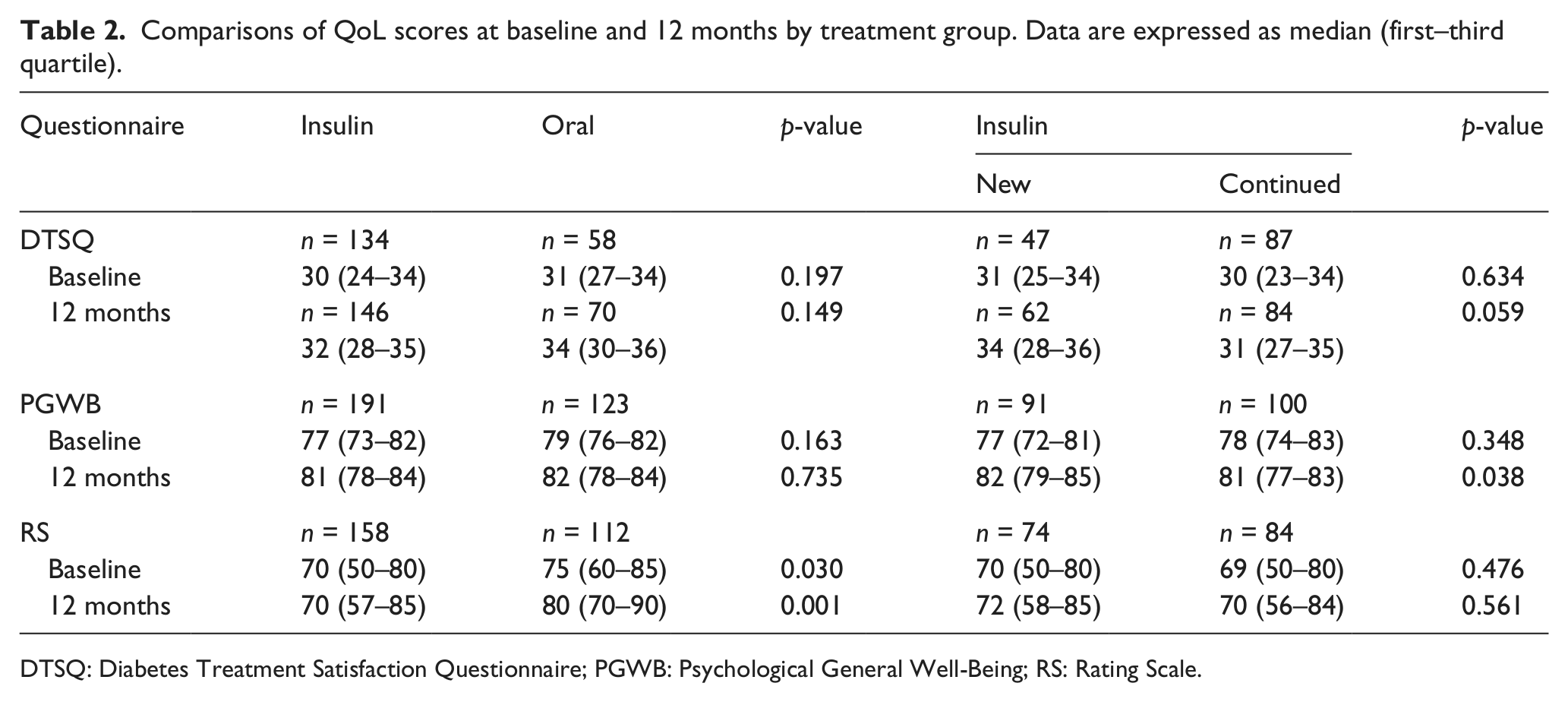
DTSQ: Diabetes Treatment Satisfaction Questionnaire; PGWB: Psychological General Well-Being; RS: Rating Scale.
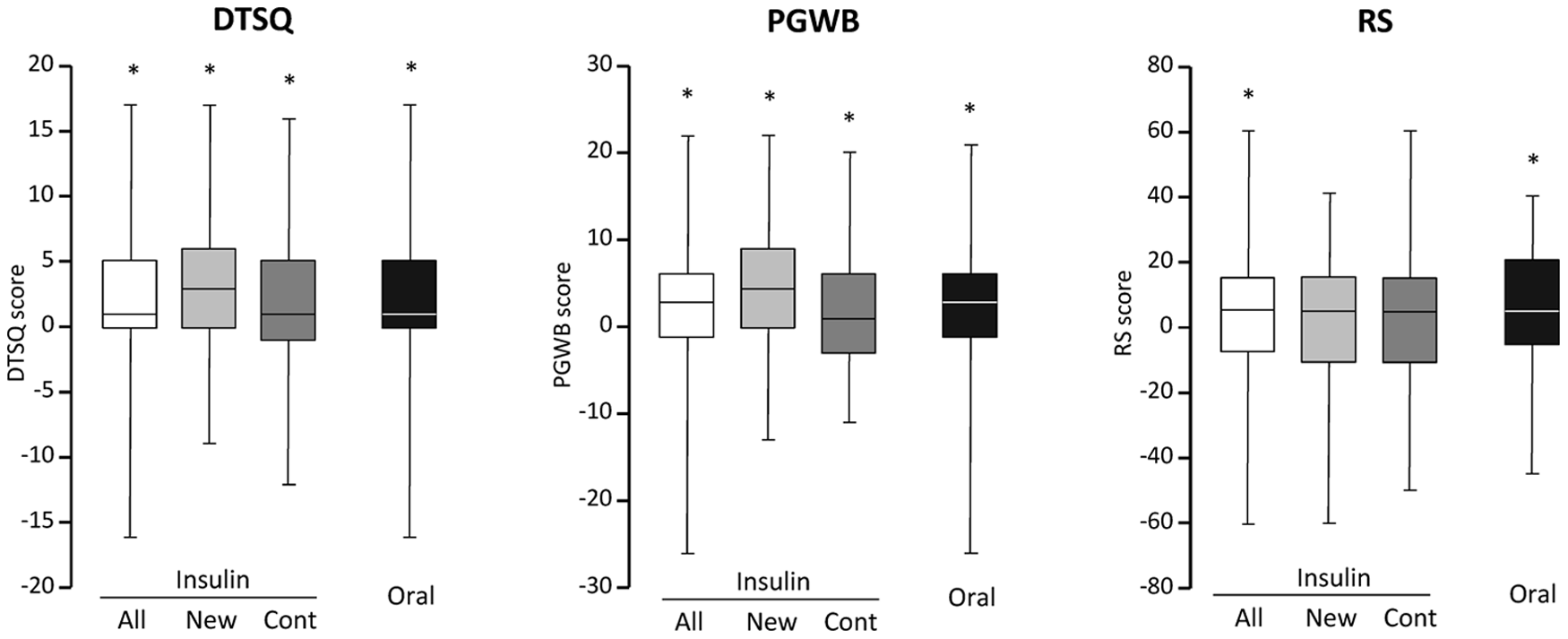
Change in QoL scores over time according to the type of glucose-lowering treatment, insulin (all, newly instituted, continued) or oral. Data are expressed as delta values [median (first–third quartile)]. *Baseline versus 12 months, p < 0.05.
Fifteen patients changed glucose-lowering treatment during the study (insulin to oral: n = 6, oral to insulin: n = 9). In these patients, there was no difference in PGWB, but RS was higher compared to the total cohort [80 (77–90) vs 74 (60–85), p = 0.028].
QoL in relation to glycaemic control
HbA1c, the measure of glycaemic control, was higher in the insulin than in the oral treatment group at baseline and remained higher at the 12-month visit (Table 3). A decrease in HbA1c was noticed in 50% of the patients during the study and of a similar magnitude in the two treatment groups. Patients who had lower HbA1c over time showed improvement in all QoL measures, while patients with stable or increased HbA1c showed improvement in PGWB only (Figure 2(A)).
Glycaemic control expressed as HbA1c at baseline and 12 months by treatment group and change over time [median (first–third quartile)]
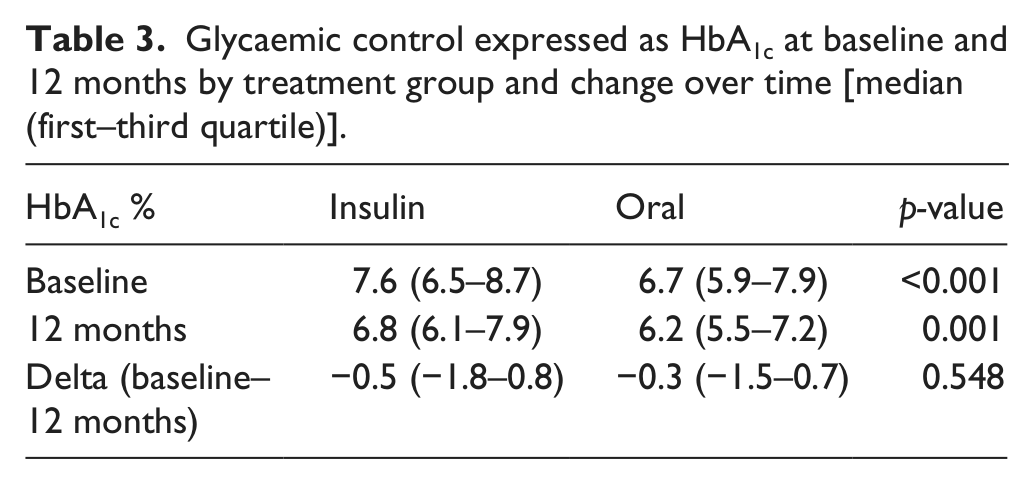
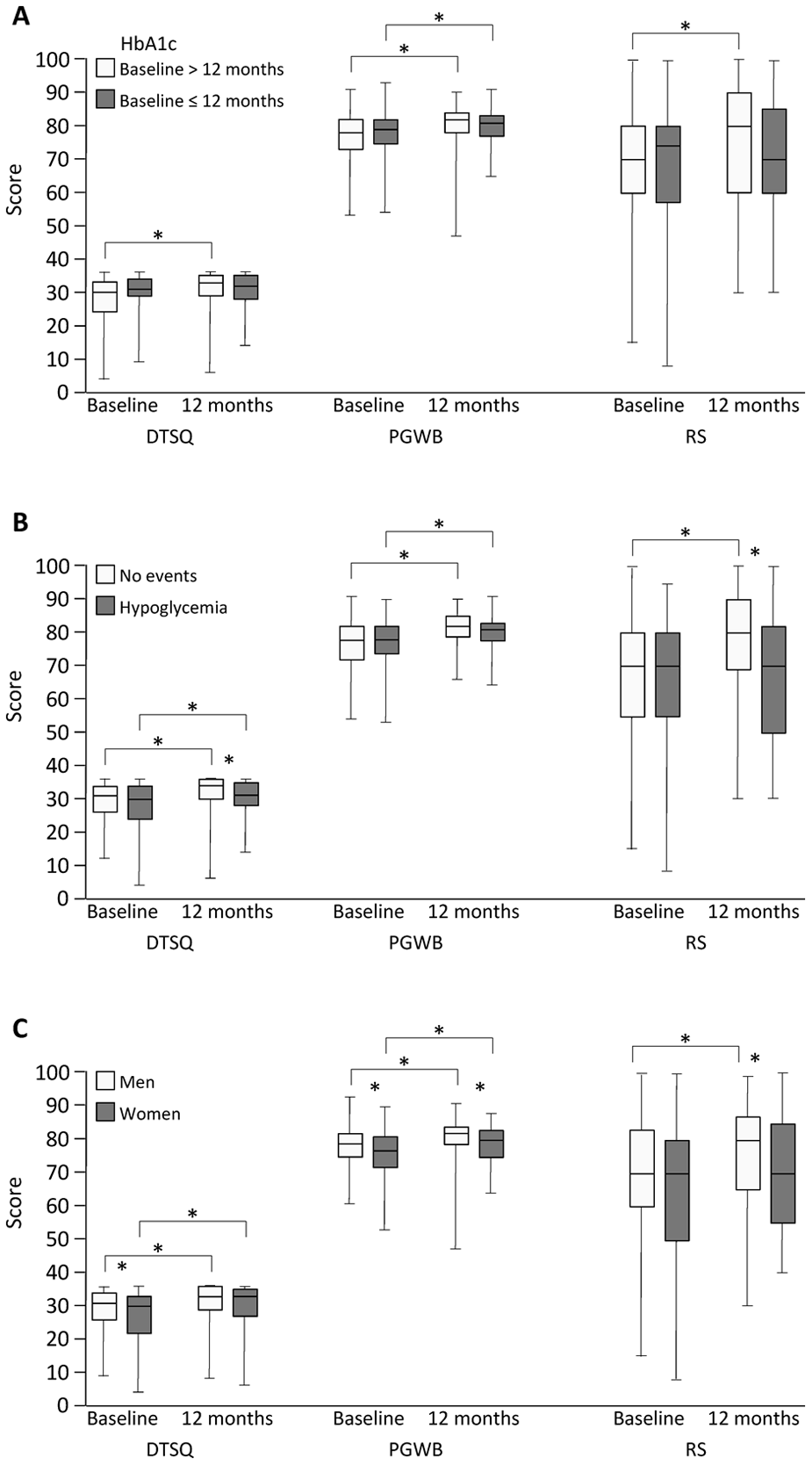
QoL scores at baseline and 12 months by (panel A) change in HbA1c, (panel B) self-reported hypoglycaemia and (panel C) gender. Data are expressed as median (first–third quartile). *p < 0.05.
Self-reported hypoglycaemia was more frequent among patients treated with insulin than oral drugs at baseline [n = 67 (50%) vs n = 16 (30%); p = 0.004] and at 12 months [n = 81 (52%) vs n = 23 (30%); p = 0.002]. Patients who reported hypoglycaemic events also reported lower treatment satisfaction than patients free from events (Figure 2(B)).
QoL and gender
Treatment satisfaction and psychological well-being was higher in men than in women at baseline, and men reported higher psychological well-being at 12 months (Figure 2(C)). Both genders showed significant improvement in DTSQ and PGWB over time, while self-rated health only improved in men.
Discussion
Insulin-based glucose-lowering treatment did not affect treatment satisfaction or psychological well-being negatively compared to oral glucose-lowering treatment in patients with type 2 diabetes and myocardial infarction. Improved glycaemic control was associated with improvement in self-reported QoL.
Diabetes is a self-managed disease where the outcome is associated with patient acceptance and adherence to the prescribed glucose-lowering treatment. Thus, satisfaction with treatment and good psychological health may facilitate effective self-management and help patients to achieve recommended glycaemic targets. The present study showed good acceptance of insulin therapy without any negative impact on treatment satisfaction or psychological well-being. Moreover, satisfaction with treatment improved over time, which is in line with previous studies investigating effects of newly introduced insulin.15 –17 Increase in treatment satisfaction was apparent both for newly instituted and continued insulin treatment.
One may speculate that the higher baseline HbA1c in the insulin group, above the recommended target, contributed to an impression of disease severity, and as HbA1c decreased treatment satisfaction increased. It has indeed been suggested that treatment satisfaction is mediated by perceived benefits, while treatment preferences relate to side effects. 18 Improved metabolic control generates positive feedback from health-care providers, which may further enhance treatment satisfaction and well-being. The significant increase in all QoL measures for patients with decreasing HbA1c supports the hypothesis that a negative attitude towards insulin injections may be counterbalanced by an improved glycaemic control. That patients with hypoglycaemic episodes reported a lower treatment satisfaction compared to those without is in line with this assumption and a previous report. 19
PGWB at baseline was similar to what can be expected in a general population of a corresponding age 20 indicating a good mental health in the present cohort. Still, psychological well-being improved over time with no difference between the groups. Patients with newly instituted insulin had higher psychological well-being at 12 months compared with those who already had insulin at baseline. The results are in concordance with previous observations of improvements in psychological domains of QoL21,22 as well as decreased symptoms of depression 23 in insulin-treated patients. Regular visits to health-care providers 21 and good glycaemic control 23 have a positive effect on psychological status. Thus, participation in this study with increased access to health care may have contributed to the improved psychological health.
Worse self-rated health in patients treated with insulin may be an expression of their higher risk factor burden. In line with this are the lower RS scores in patients experiencing hypoglycaemia that may reflect more severe diabetes and its complications rather than relate to glucose-lowering treatment per se. 24 The more pronounced psychological burden and lower self-rated health reported by women at the end of the study is in concordance with previous research. 25
Impaired treatment satisfaction and compromised psychological status are considered to be associated with less-efficient self-care 26 and low adherence to prescribed therapy. Thus, the present findings of similar treatment satisfaction and well-being in patients prescribed insulin and oral glucose-lowering agents may encourage earlier institution of insulin when needed.
Limitations
Due to the variety of questionnaires used in studies measuring effects of insulin therapy on treatment satisfaction and patient well-being, results can be difficult to compare to the present study. Two of the present measures, PGWB and RS, are not disease specific and may be affected by different aspects in life. The high QoL scores may be influenced by socio-economic and demographic factors as well as level of attention paid to the health status of these patients. Thus, study effects cannot be excluded, and the results might be difficult to directly transfer to other populations. Finally, considering the limited number of females in this study, the possibility of a type 2 statistical error cannot be excluded when dealing with gender aspects.
Conclusions
Insulin-based therapy was well accepted and did not decrease treatment satisfaction or psychological well-being compared to oral glucose-lowering treatment in patients with type 2 diabetes and myocardial infarction.
Footnotes
Acknowledgements
The authors are grateful to RN Christina Edman-Jönsson for excellent management of the study logistics.
Conflict of interest
None of the authors have any financial interests or other relationships and affiliations related to the subject of this manuscript.
Funding
This study was supported by the Swedish Heart and Lung Foundation and AFA Insurance and by unconditioned grants from Aventis Sweden and Novo Nordisk Denmark.
