Abstract
Diabetes is a significant risk factor for the development of cardiovascular disease (CVD), particularly in the presence of high blood pressure, poor glycaemic control and high total cholesterol. While efforts to control blood pressure or blood glucose beyond levels considered ‘normal’ in patients with diabetes have not produced the expected reduction in CVD, treatment with statins to reduce levels of low-density lipoprotein cholesterol (LDL-C) has been much more successful. However, many patients with diabetes who receive statins (even at high doses) remain at significant residual risk of CVD due to the presence of atherogenic dyslipidaemia. Markers of persisting risk include low levels of high-density lipoprotein cholesterol (HDL-C), high levels of triglycerides (TG) and LDL-C levels above target despite high-dose statin therapy. Combining statins with drugs that target HDL-C and TG, such as fibrates, niacin and omega-3 polyunsaturated fatty acid (PUFA) ethyl esters, may offer further protection from CVD in patients with diabetes.
Abbreviations and acronyms
ACS: acute coronary syndrome
ADA: American Diabetes Association
AHA: American Heart Association
AP: angina pectoris
ARR: absolute risk reduction
CAD: coronary artery disease
CABG: coronary artery bypass graft
CAWA: carotid artery wall area
CHD: coronary heart disease
CIMT: carotid intima media thickness
CKD: chronic kidney disease
CTT: Cholesterol Treatment Trialists
CV: cardiovascular
CVD: cardiovascular disease
DHA: docosahexaenoic acid
EAS: European Atherosclerosis Society
EPA: eicosapentaenoic acid
ESC: European Society of Cardiology
HDL-C: high-density lipoprotein cholesterol
HPS: Heart Protection Study
HR: hazard ratio LDL-C: low-density lipoprotein cholesterol
MI: myocardial infarction
PCI: percutaneous coronary intervention
PTCA: percutaneous transluminal coronary angioplasty
PUFAs: polyunsaturated fatty acids
RRR: relative risk reduction
SCORE: Systematic COronary Risk Evaluation (European Society of Cardiology)
TG: triglycerides
TNT: Treating to New Targets
UKPDS: United Kingdom Prospective Diabetes Study
Key messages
Diabetes is a significant risk factor for the development of cardiovascular disease (CVD).
Treatment of patients with diabetes with statins to reduce low-density lipoprotein cholesterol (LDL-C) has been successful in the reduction of CVD but many such patients remain at significant residual risk of CVD despite statin therapy (even at high doses).
Markers of persisting risk include low levels of high-density lipoprotein cholesterol (HDL-C), high levels of triglycerides (TG) and LDL-C levels above target.
Combining statins with drugs that target HDL-C and TG, such as fibrates, niacin and omega-3 polyunsaturated fatty acid (PUFA) ethyl esters, may offer further protection from CVD in patients with diabetes.
Introduction
Diabetes mellitus is found in almost every human population in the world, with type 2 diabetes accounting for >85% of cases. 1 In 2009, it was estimated that 285 million people aged 20–79 years had diabetes (6.6% of the global population). 1 This figure is expected to rise to 438 million (7.8%) by 2030. 1 Excess mortality due to diabetes in adults was estimated to be nearly 4 million deaths per year in 2010, and approximately 50–70% of these deaths are thought to be due to cardiovascular disease (CVD).1–3 Of the interventions to reduce CVD in people with diabetes, statins have been highly successful. 2
The value of statins in the reduction of mortality and morbidity in patients with diabetes has been demonstrated by subgroup analyses of trials in broad patient populations and by trials specifically in diabetic populations.
Primary prevention studies
The Collaborative AtoRvastatin Diabetes Study (CARDS) trial compared atorvastatin 10 mg/day with placebo in patients with type 2 diabetes with no previous CVD and low low-density lipoprotein cholesterol (LDL-C) (< 4.14 mmol/L) but with one or more risk factors, such as microalbuminuria or retinopathy. After a median 3.9 years of follow-up there were 127 CVD events in the placebo group compared with 83 in the treatment group, equivalent to a relative risk reduction (RRR) of 37% (p = 0.001). At this point the study reached prespecified efficacy criteria and was terminated early as permitted by the protocol. These results indicated that a statin significantly reduces CV events in patients with diabetes after a relatively short duration of treatment even in the absence of an LDL-cholesterol level considered ‘high’ when the study was designed. 4 However, the Atorvastatin Study for the Prevention of coronary heart disease Endpoints in Non-insulin dependent diabetes mellitus (ASPEN) trial found contrary results in patients with type 2 diabetes. The primary composite endpoint was combined cardiovascular death, non-fatal myocardial infarction (MI), non-fatal stroke, revascularisation, coronary artery bypass graft (CABG) or unstable angina. Compared with placebo, atorvastatin 10 mg/day had no significant effect on the rate of the primary endpoint in the overall population (13.7% vs 15.0%, respectively) or in the primary prevention subgroup (10.4% vs 10.8%, respectively). 5 A Cochrane review of 14 randomised primary prevention trials, however, including several in patients with diabetes, concluded that statins reduce overall mortality and combined fatal and non-fatal CVD endpoints. 6
Secondary prevention studies
A subanalysis of the Treating to New Targets (TNT) study investigated the effect of atorvastatin in patients with diabetes and previous coronary heart disease (CHD) whose LDL-C reached < 3.4 mmol/L after the run-in period of the study when all participants received low-dose atorvastatin. Atorvastatin 80 mg/day significantly reduced the relative risk of a secondary cardiovascular event compared with atorvastatin 10 mg/day (absolute risk reduction (ARR) 4.1%; RRR 25%; p = 0.026 for RRR). 7
The largest subanalysis of statins in patients with diabetes was conducted by the Heart Protection Study (HPS), which investigated the effect of simvastatin in nearly 6000 patients (10% with type 1 diabetes and 90% with type 2 diabetes). Approximately half the population also had a history of vascular disease. The study found that the combined risk of a major vascular event (major coronary event, stroke or revascularisation) was significantly reduced compared with placebo (ARR 4.8%; RRR 22%; p < 0.0001). The risks of individual events were reduced by a similar degree. Results in diabetic patients were not significantly different from those in non-diabetic patients with occlusive arterial disease (p = 0.60). 8
Subgroup analyses of the 4S trial, the Lescol Intervention Prevention Study (LIPS) and the GREek Atorvastatin and CHD Evaluation (GREACE) study found that statins significantly reduce the risk of major vascular events in patients with diabetes and previous CHD.9–11 However, in the ASPEN study, atorvastatin 10 mg/day had no significant effect on the primary endpoint in patients with prior MI or coronary intervention (26.2% on atorvastatin vs. 30.8% on placebo). 5
Intensive therapy
Intensive therapy in type 2 diabetes patients using high doses of statins to achieve very low LDL-C levels may further reduce CVD risk when compared with standard doses. The main TNT study, 12 the Pravastatin or Atorvastatin Evaluation and Infection Therapy-Thrombolysis in Myocardial Infarction 22 (PROVE IT-TIMI 22) secondary prevention study 13 and a meta-analysis of trials comparing moderate with intense statin therapy 14 support this hypothesis.
Although some studies did not show a benefit of statins in patients with diabetes, this can sometimes be explained by the trial design or execution. For example, the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack (ALLHAT-LLT) study of pravastatin versus usual care in patients with hypertension did not meet its primary endpoint of all-cause mortality overall or in patients with diabetes. 15 This may have been due to the modest differences in total cholesterol and LDL-C between the pravastatin and control arms (9.6% and 16.7%, respectively for the overall population), together with an increasing proportion of patients in the ‘usual care’ control arm receiving statins. Similarly, the Study of the Effectiveness of Additional Reductions in Cholesterol and Homocysteine (SEARCH) trial reported a non-significant 6% RRR in major CV events overall in survivors of MI treated with simvastatin 80 mg/day compared with 20 mg/day. 16 In this trial, as a result of increasing use of non-study intensive statin regimens in patients randomised to the 20 mg/day dose, the difference in LDL-C between treatment arms was only 0.35 mmol/L.
The Anglo-Scandinavian Cardiac Outcomes Trial-Lipid-Lowering Arm (ASCOT-LLA) study of atorvastatin 10 mg/day versus placebo was stopped early because of the significant reduction in its primary endpoint (non-fatal MI or fatal CHD) in the overall population, but the early trial closure reduced the numbers of events recorded; the benefit of pravastatin, therefore, was not statistically significant in patients with diabetes. 17
In view of these considerations, these studies do not refute the principle that the main lipid target for patients with diabetes should be LDL-C and statins are the best-proven agents for reduction of this parameter. (In some populations, including patients with diabetes receiving haemodialysis18–20 and patients with diabetes and chronic heart failure or reduced left ventricular ejection fractions,21,22 the benefits of statins on CV events are less clear.)
Table 1 summarises the findings of the principal statin studies that specifically described results for patients with diabetes. Overall, these trials included nearly 15,000 patients with diabetes.
Trials of statins to reduce risk of cardiovascular disease that reported results for patients with diabetes.
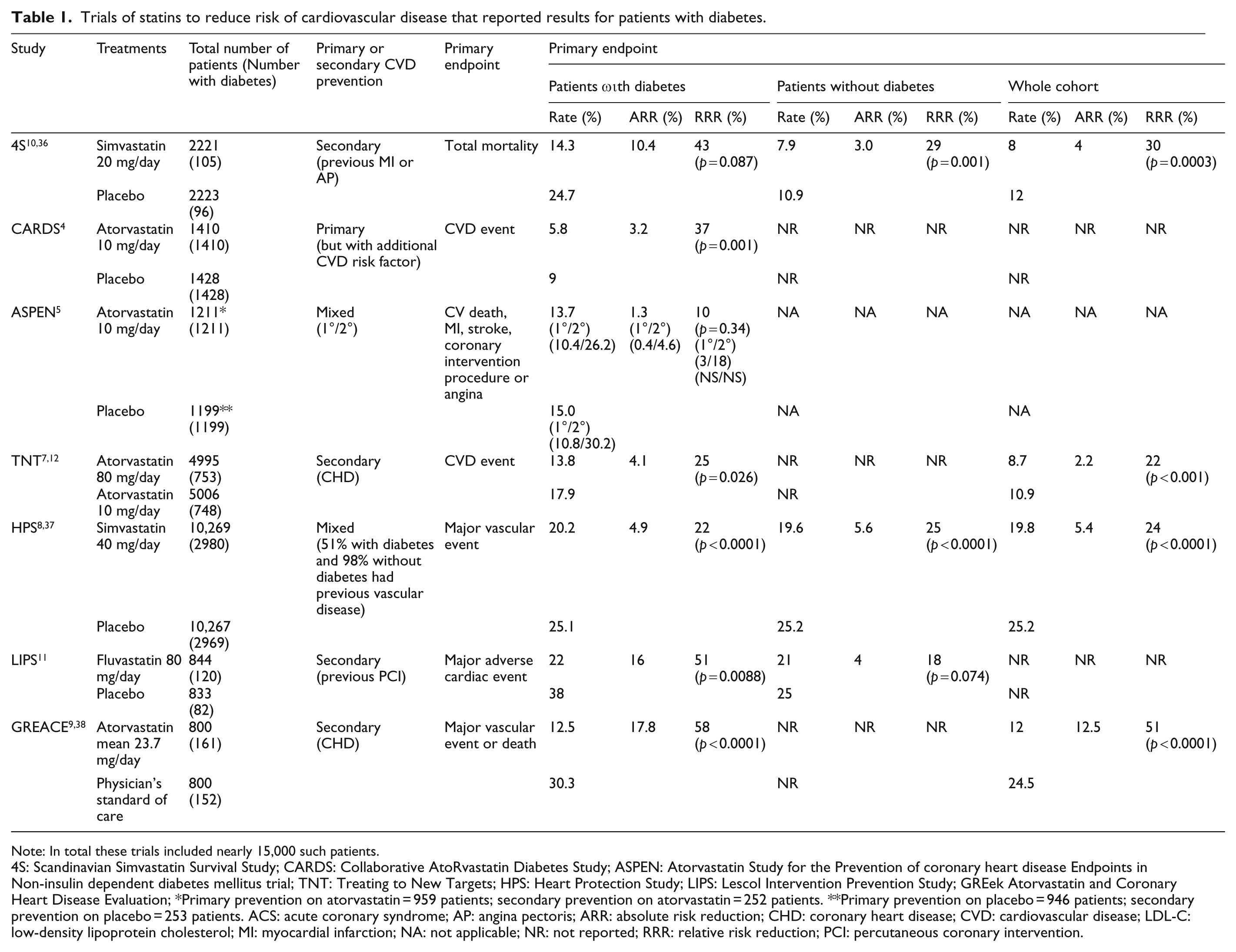
Note: In total these trials included nearly 15,000 such patients.
4S: Scandinavian Simvastatin Survival Study; CARDS: Collaborative AtoRvastatin Diabetes Study; ASPEN: Atorvastatin Study for the Prevention of coronary heart disease Endpoints in Non-insulin dependent diabetes mellitus trial; TNT: Treating to New Targets; HPS: Heart Protection Study; LIPS: Lescol Intervention Prevention Study; GREek Atorvastatin and Coronary Heart Disease Evaluation; *Primary prevention on atorvastatin = 959 patients; secondary prevention on atorvastatin = 252 patients. **Primary prevention on placebo = 946 patients; secondary prevention on placebo = 253 patients. ACS: acute coronary syndrome; AP: angina pectoris; ARR: absolute risk reduction; CHD: coronary heart disease; CVD: cardiovascular disease; LDL-C: low-density lipoprotein cholesterol; MI: myocardial infarction; NA: not applicable; NR: not reported; RRR: relative risk reduction; PCI: percutaneous coronary intervention.
A meta-analysis by the Cholesterol Treatment Trialists (CTT) investigated the effects of statins in 26 trials, including ALLHAT-LLT and ASCOT-LLA, with more than 170,000 participants including more than 32,000 patients with diabetes. 23 It confirmed that patients with diabetes taking a statin experienced a significant reduction in major CV events compared with controls (RRR approximately 18%). The RRR was approximately 20% per 1 mmol/L reduction in LDL-C. The RRR was similar in individuals with or without diabetes (although absolute rates of CV events were higher in the presence of diabetes) and was demonstrated even in a subgroup with a baseline LDL-C < 2.0 mmol/L. Overall, results from individual trials and the CTT meta-analysis show that statins reduce CVD risk in patients with or without type 2 diabetes or prior CVD and over a wide range of baseline cholesterol levels.
As a result of these trials of statins in patients with diabetes, guidelines for the prevention of CVD from the European Society of Cardiology (ESC), American Heart Association (AHA) and American Diabetes Association (ADA) strongly recommend statin therapy for patients with diabetes and overt CVD irrespective of LDL-C levels.2,24–26 The guidelines published recently by the ESC and European Atherosclerosis Society (EAS) also recommend statin therapy in these patients. 27 For patients with diabetes, guidelines recommend lifestyle intervention with the addition of pharmacological treatment if necessary, with the intention of reducing LDL-C levels to < 2.5 mmol/L (< 100 mg/dL) or, in patients at the highest risk, to < 1.7–2.0 mmol/L (< 70–80 mg/dL).2,24–27 If these targets are not met, the first ‘add-on’ step according to the CTT should be to intensify statin therapy to the highest tolerated (or highest licensed) dose and to consider replacing a ‘weak’ statin with a more potent one. 23 The definitions of highest risk vary between guidelines but generally include established CVD or the presence of additional risk factors such as smoking. In practice, these guidelines imply that statins are the drugs of choice and should be prescribed to the majority of patients with diabetes, either because they have markers of increased risk such as microalbuminuria or because lifestyle intervention alone does not achieve the desired LDL-C level.2,24–27
However, Table 2 shows that the attained mean LDL-C levels of patients receiving statin therapy alone in randomised trials still ranged from 1.99 to 3.17 mmol/L. Therefore, if statin therapy, after appropriate adjustment of the dose or drug, 23 does not achieve the desired lipid goals, the ESC/EAS guidelines recommend addition of a cholesterol absorption inhibitor, a bile acid sequestrant or niacin, while other guidelines suggest the addition of these compounds or a fibrate.2,24–28
Concentration of plasma lipids in trials of statins.
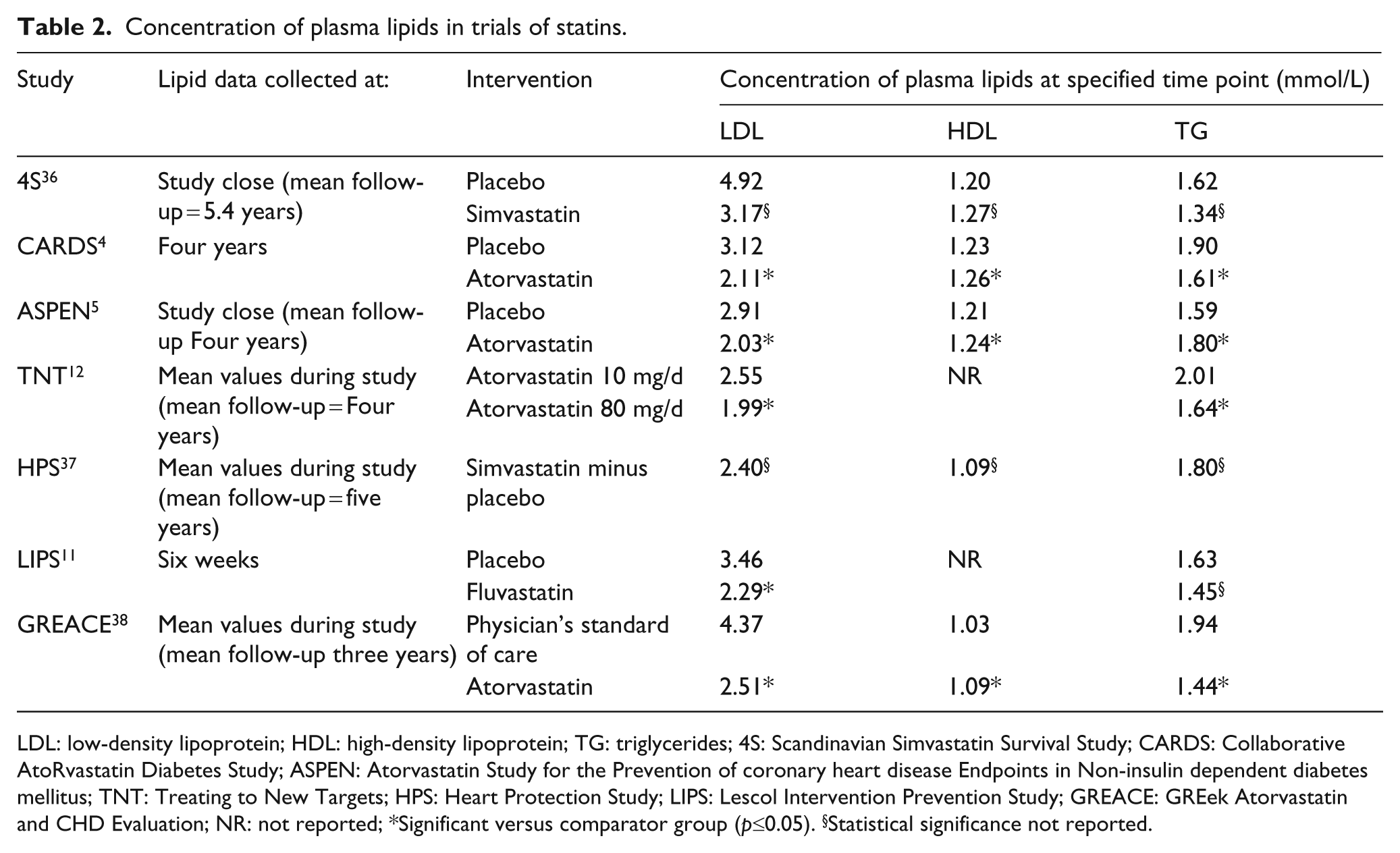
LDL: low-density lipoprotein; HDL: high-density lipoprotein; TG: triglycerides; 4S: Scandinavian Simvastatin Survival Study; CARDS: Collaborative AtoRvastatin Diabetes Study; ASPEN: Atorvastatin Study for the Prevention of coronary heart disease Endpoints in Non-insulin dependent diabetes mellitus; TNT: Treating to New Targets; HPS: Heart Protection Study; LIPS: Lescol Intervention Prevention Study; GREACE: GREek Atorvastatin and CHD Evaluation; NR: not reported; *Significant versus comparator group (p≤0.05). §Statistical significance not reported.
Data from recent longitudinal studies (e.g. European Prospective Investigation into Cancer and Nutrition (EPIC)-Netherlands, EPIC-Potsdam, Hoorn and Action in Diabetes and Vascular Disease: Preterax and Diamicron-MR Controlled Evaluation Study (ADVANCE)) seem to indicate that the risks of middle-aged cohorts with diabetes developing CHD and CVD have dropped to around 3–5% and 6–8% respectively over follow-up of four to 10 years.29–31 The reasons for these long-term trends may be manifold and most likely include the much wider use of statins (50–80% of patients) in these cohorts. However, despite treatment with statins, patients with diabetes remain at a twofold increased risk of CVD compared with the general population. 32 In addition, the CTT meta-analysis found that even following statin therapy, 4.6% of participants with diabetes died of CHD compared with 3.1% of those without diabetes. 33 Moreover the RRR achieved with statins is about 20–40%, leaving 60–80% of CVD events not prevented: the remaining CVD events have been described as ‘residual risk’. 34 Therefore, although statin therapy has helped markedly to reduce the risk of CVD, there remains a residual risk of CV events in patients with type 2 diabetes. In this context, it is important that the emphasis be on the individual risk and not simply cholesterol targets. 35
Despite the demonstrated benefits of statins for the prevention of CVD, further measures may be needed in the treatment of more vulnerable cohorts such as patients with diabetes. The objective of this review is to discuss the mechanisms of persisting CVD risk in patients with diabetes receiving statins and to investigate other interventions that could help to reduce this risk further.
Indicators of CVD risk beyond LDL-C
Other risk factors contribute to CVD risk in addition to LDL-C levels. The Prospective Cardiovascular Münster (PROCAM) score uses eight independent risk factors to determine the risk of an acute coronary event. In order of importance these are: age; high LDL-C levels; smoking; low HDL-C levels; high systolic blood pressure (BP); a family history of MI; diabetes; and high TG levels. 39 Hyperglycaemia and hypertension are established risk factors for CVD in type 2 diabetes. The United Kingdom Prospective Diabetes Study (UKPDS) found that a 1% reduction in glycosylated haemoglobin (HbA1c) resulted in a 21% reduction in the risk of death related to diabetes and a 14% reduction in the risk of MI.40,41 Chronic kidney disease (CKD), in particular a reduced glomerular filtration rate, is also associated with an increased risk of CVD, and patients with diabetes are more likely to develop CKD than the general population. 42
The level of HDL-C (or total cholesterol: HDL-C ratio) is included in a number of other risk scores including QRISK, 43 the UKPDS risk engine 44 and the Framingham risk score. 45 However, these scores do not incorporate TG levels. The original publication of the ESC Systematic COronary Risk Evaluation (SCORE) did not include either HDL-C or TG levels in the calculation of CV risk, as this would be difficult to show on the simple charts it uses. 46 A more recent publication from the SCORE group suggested that inclusion of HDL-C might be useful in some settings. 47
Lipid risk factors for CVD in patients with type 2 diabetes
In patients with type 2 diabetes, low HDL-C and high TG levels are of particular importance because these patients often have a particular form of atherogenic dyslipidaemia characterised by small dense LDL-C particles, low HDL-C levels and high TG levels. 34
Several studies have shown that a low level of HDL-C is an independent risk factor for CVD in diabetes.25,41,48,49 For example, the UKPDS found that low HDL-C was an independent risk factor for coronary artery disease (CAD) and MI. 41 However, the Justification for the Use of statins in Prevention: an Intervention Trial Evaluating Rosuvastatin (JUPITER) found that while there was an inverse relationship between HDL-C level and CV risk in patients receiving placebo, the same was not observed for those treated with rosuvastatin. 50 Data are even less clear for the effect of TG levels on CV risk, especially in patients with diabetes. The UKPDS found that high levels of TG were a risk factor for CAD but this was not independent from other risk factors. 41 However, a meta-analysis of 17 studies investigating CV risk and TG levels in the general population did find a significant association even when results were adjusted for HDL-C levels. 49 As well as contributing to macrovascular risk, low HDL-C levels and high TG potentially contribute to the microvascular complications of diabetes such as retinopathy and nephropathy. 34 Both the ADA and ESC recommend that TG levels should be < 1.7 mmol/L and HDL-C levels should be >1 mmol/L in men and >1.3 mmol/L in women.2,24–27
Interventions to reduce CVD risk beyond statin therapy: clinical trials and current status
The clinical benefits of statins are believed to be principally mediated by reductions in LDL-C levels. 25 Their effects are modest on TG and HDL-C. For example, in the PROVE-IT study median HDL-C levels increased from baseline by 6.5% in the atorvastatin 80 mg/day group and 8.1% in the pravastatin 40 mg/day group. 13 In the TNT study, high-dose atorvastatin (80 mg/day) significantly reduced TG levels from baseline (p < 0.001) but had no significant effect on HDL-C levels. 12 In cases of mixed dyslipidaemias, the ESC/EAS guidelines recommend that, in addition to statins, patients receive drugs that influence HDL-C and TG levels to further reduce CVD risk. 27 Recent clinical trials have investigated these agents in more than 24,000 patients with diabetes; results are summarised below and in Tables 3 and 4. Table 5 shows the average lipid levels attained during these studies or the final lipid levels measured at the end of the study. Although TG levels in the intervention groups were usually decreased to the intended range of ≤1.7 mmol/L, mean LDL-C failed to reach the intended therapeutic target of ≤2 mmol/L in the majority of studies.
Trials of non-statin lipid-modifying therapies to reduce CVD endpoints.
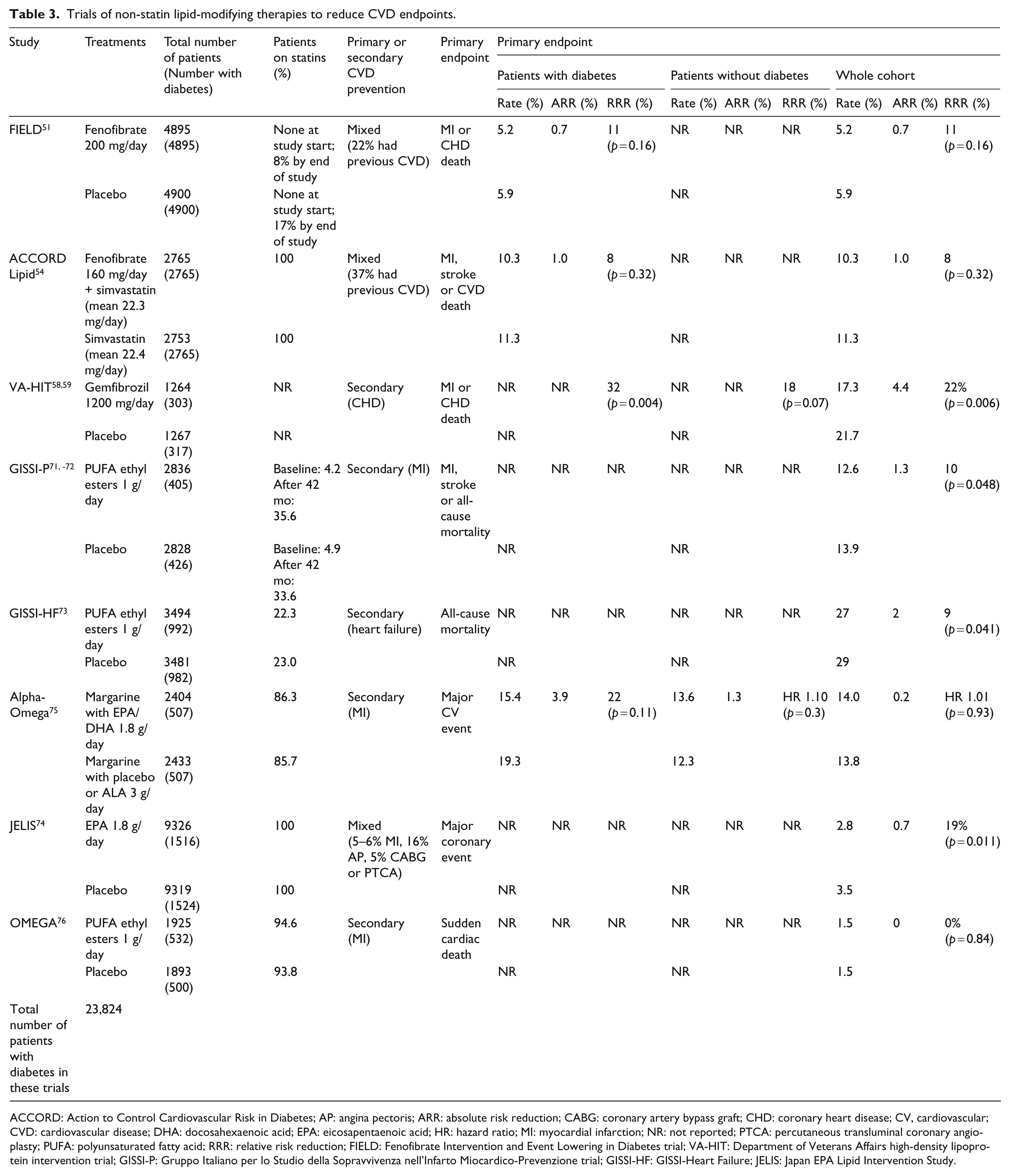
ACCORD: Action to Control Cardiovascular Risk in Diabetes; AP: angina pectoris; ARR: absolute risk reduction; CABG: coronary artery bypass graft; CHD: coronary heart disease; CV, cardiovascular; CVD: cardiovascular disease; DHA: docosahexaenoic acid; EPA: eicosapentaenoic acid; HR: hazard ratio; MI: myocardial infarction; NR: not reported; PTCA: percutaneous transluminal coronary angioplasty; PUFA: polyunsaturated fatty acid; RRR: relative risk reduction; FIELD: Fenofibrate Intervention and Event Lowering in Diabetes trial; VA-HIT: Department of Veterans Affairs high-density lipoprotein intervention trial; GISSI-P: Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico-Prevenzione trial; GISSI-HF: GISSI-Heart Failure; JELIS: Japan EPA Lipid Intervention Study.
Trials of statins plus other lipid-lowering therapies that examined CIMT or CAWA.
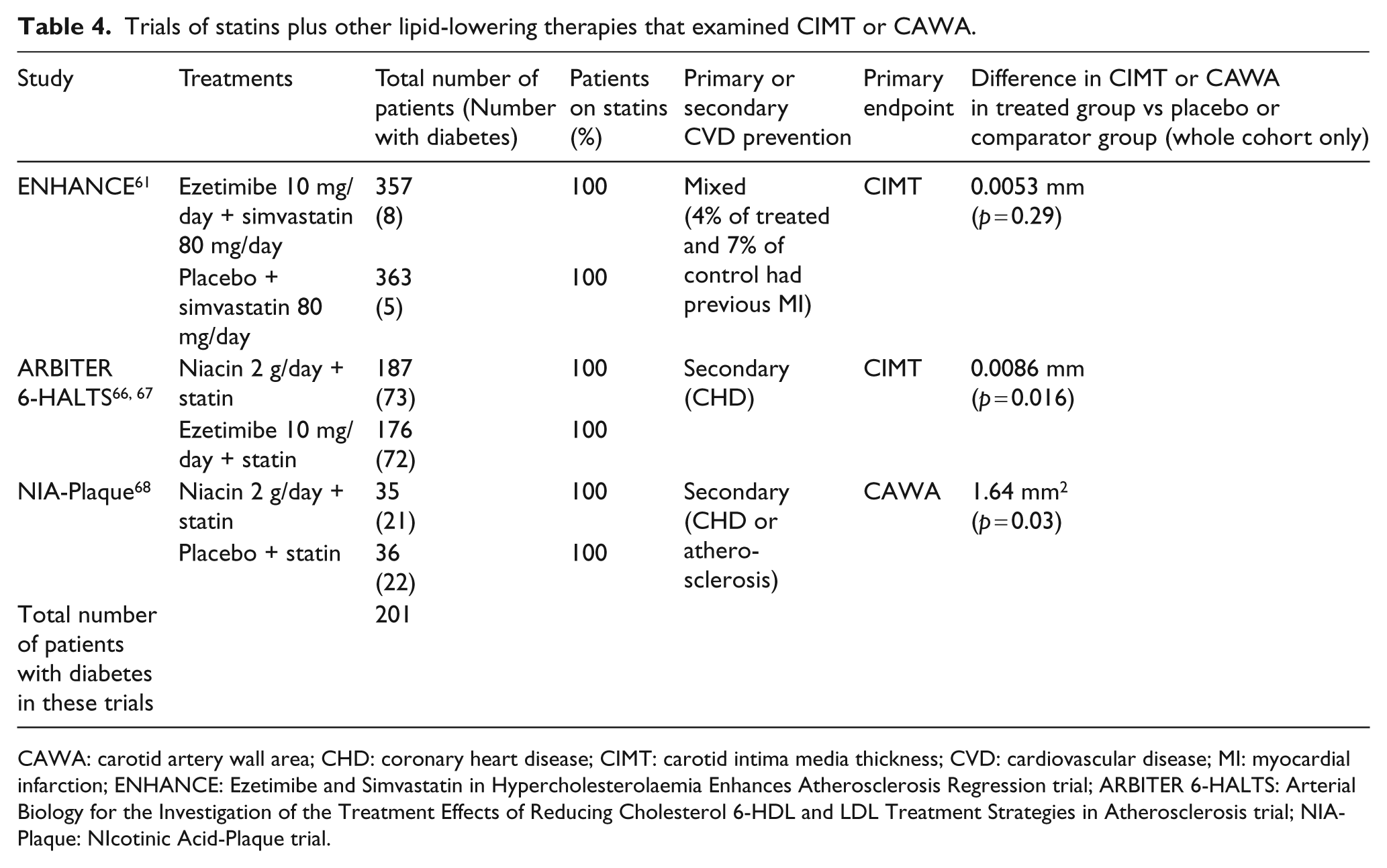
CAWA: carotid artery wall area; CHD: coronary heart disease; CIMT: carotid intima media thickness; CVD: cardiovascular disease; MI: myocardial infarction; ENHANCE: Ezetimibe and Simvastatin in Hypercholesterolaemia Enhances Atherosclerosis Regression trial; ARBITER 6-HALTS: Arterial Biology for the Investigation of the Treatment Effects of Reducing Cholesterol 6-HDL and LDL Treatment Strategies in Atherosclerosis trial; NIA-Plaque: NIcotinic Acid-Plaque trial.
Concentration of plasma lipids in trials of non-statin lipid-modifying therapies.
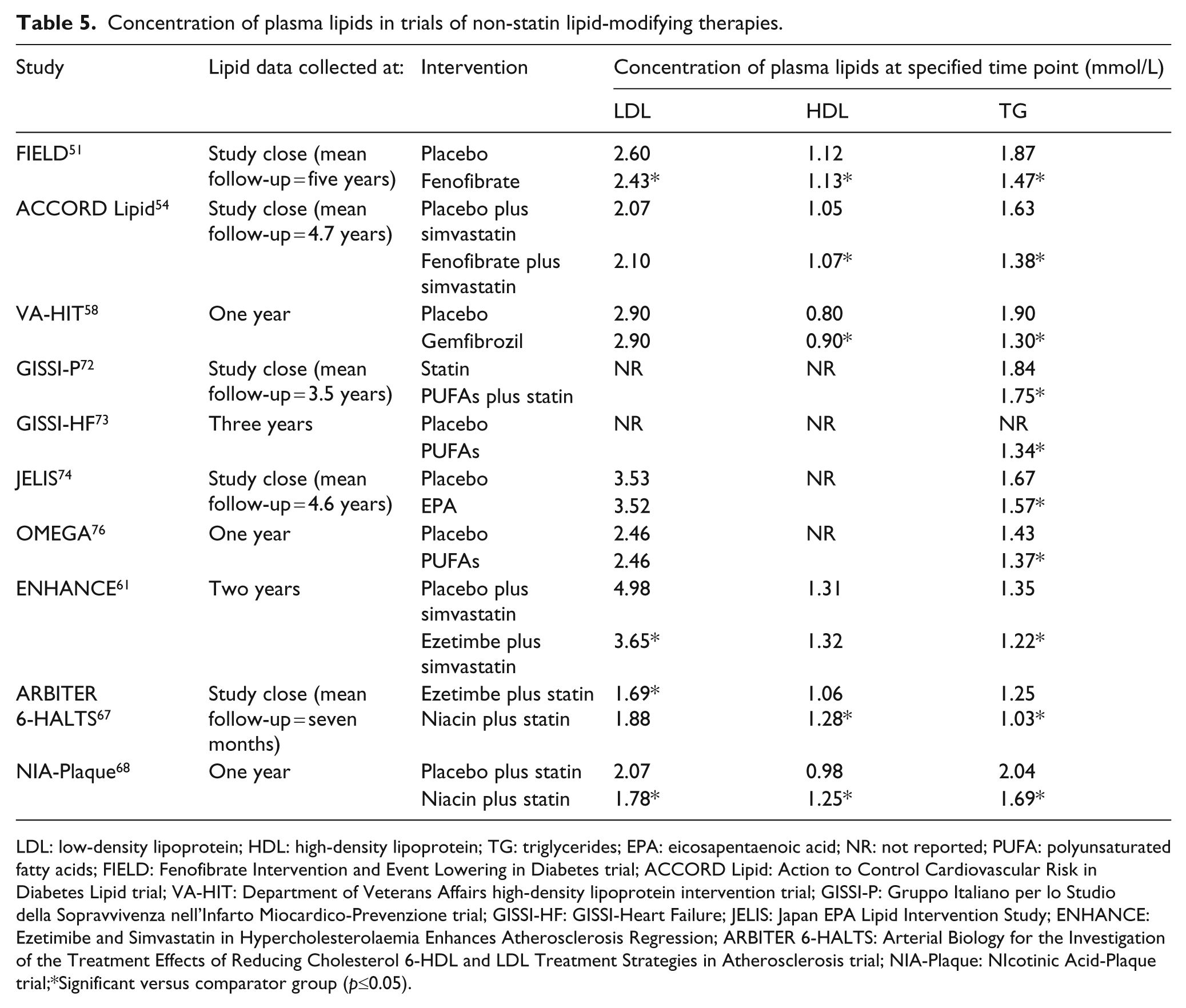
LDL: low-density lipoprotein; HDL: high-density lipoprotein; TG: triglycerides; EPA: eicosapentaenoic acid; NR: not reported; PUFA: polyunsaturated fatty acids; FIELD: Fenofibrate Intervention and Event Lowering in Diabetes trial; ACCORD Lipid: Action to Control Cardiovascular Risk in Diabetes Lipid trial; VA-HIT: Department of Veterans Affairs high-density lipoprotein intervention trial; GISSI-P: Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico-Prevenzione trial; GISSI-HF: GISSI-Heart Failure; JELIS: Japan EPA Lipid Intervention Study; ENHANCE: Ezetimibe and Simvastatin in Hypercholesterolaemia Enhances Atherosclerosis Regression; ARBITER 6-HALTS: Arterial Biology for the Investigation of the Treatment Effects of Reducing Cholesterol 6-HDL and LDL Treatment Strategies in Atherosclerosis trial; NIA-Plaque: NIcotinic Acid-Plaque trial;*Significant versus comparator group (p≤0.05).
Key trials of non-statin lipid-lowering therapies
Fenofibrate
The Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study investigated the effect of raising HDL-C and lowering TG levels with fenofibrate 200 mg/day in 9795 patients with type 2 diabetes. 51 Patients were not taking statins or other lipid-lowering drugs at the start of the study. Over the entire trial, average use of non-study lipid-lowering therapies, predominantly statins, was significantly greater in the placebo group (17%) than the fenofibrate group (8%; p < 0.0001).
Fenofibrate improved lipid parameters including total cholesterol, LDL-C, HDL-C and TG. The combined primary endpoint of non-fatal MI or death due to CHD was directionally positive, but not significantly different for the fenofibrate group compared with the placebo group (RRR 11%; p = 0.16). However, there was a significant reduction with fenofibrate for non-fatal MI (RRR 24%; p = 0.01) and the combined secondary endpoint of CVD death, non-fatal MI, non-fatal stroke or coronary or carotid revascularisation procedure (RRR 11%; p = 0.035) . 51 Furthermore, in a predefined subgroup analysis in fenofibrate-treated patients with low baseline HDL-C (< 1.03 mmol/L in men and < 1.29 mmol/L in women) who seem to be a logical target group for such therapy, there was a significant reduction in the primary endpoint versus placebo (RRR 14%; p = 0.02) . 51 In patients who had both high TG (≥2.3 mmol/L) and low HDL-C levels at baseline, fenofibrate reduced the relative risk of CVD events by 27% compared with placebo (p = 0.005) . 52 Fenofibrate also reduced the number of subsequent CV events in patients who had a previous asymptomatic or ‘silent’ MI (RRR 78%; p = 0.003). 53
The Action to Control Cardiovascular Risk in Diabetes (ACCORD) study investigated the effects on CVD outcomes of intensive control of blood glucose, BP or plasma lipids in three separate study arms. In the ACCORD lipid study arm, simvastatin monotherapy was compared with simvastatin plus fenofibrate (starting dose 160 mg/day) in patients with type 2 diabetes, 37% of whom had experienced a previous CVevent. 54 No significant difference in the combined primary endpoint of MI, stroke or death due to CVD was seen between the monotherapy and combination therapy groups. However, as in the FIELD study, there was a benefit of combination therapy with fenofibrate for patients with high TG and low HDL-C levels at baseline: in this group the primary endpoint occurred in 17.3% of patients on monotherapy versus 12.4% on combination therapy (p = 0.057 for interaction). 55
Administration of fenofibric acid, the active form of the compound, eliminates the need for hepatic and intestinal conversion of fenofibrate to the active acid. Clinical trials of fenofibric acid 135 mg/day in combination with simvastatin, atorvastatin or rosuvastatin at various doses have shown additional benefits on lipid levels compared with statins alone.56,57
Gemfibrozil
The Veterans Affairs HDL Intervention Trial (VA-HIT) investigated the efficacy of gemfibrozil 1200 mg/day in reducing the combined primary endpoint of CHD death, MI and stroke in a secondary prevention setting. One year after randomisation, mean HDL-C was 6% higher in the gemfibrozil group than the placebo group (p < 0.001) and TG levels were 31% lower compared with placebo (p < 0.001). The mean LDL-C level never differed significantly between the groups. 58
Gemfibrozil reduced the relative risk of the primary endpoint of non-fatal MI or death from CHD by 22% (p = 0.006 vs placebo). 58 A subsequent subgroup analysis reported that the relative risk of non-fatal MI, death from CHD or stroke was reduced by 32% in patients with diabetes (p = 0.004 vs placebo) and by 18% in those without diabetes (p = 0.07 vs placebo): the difference in benefit between the two subgroups was not significant (p = 0.26). 59 Patients with diabetes also obtained significant (p < 0.05) reductions in the relative risks of CHD and stroke compared with the placebo group. 59 As gemfibrozil had no effect on LDL-C levels, the reductions in CVD risk may have resulted from raising HDL-C and lowering TG levels.
The combination of fenofibric acid with a statin in these studies was generally well tolerated. 57 Concern has been raised about rhabdomyolysis, a rare but serious adverse effect of statin therapy that can be exacerbated by addition of fibrates. Fibrates, especially gemfibrozil, and statins share metabolic and elimination pathways and competition for these increases plasma concentrations of both drugs, so increasing the likelihood of adverse effects. The Food and Drug Administration (FDA)’s adverse-event reporting system suggests that a combination of fenofibrate and any statin is associated with fewer reports of rhabdomyolysis than when gemfibrozil is combined with statins (0.58 vs 8.6 cases per million prescriptions, respectively). 60 These results suggest that a combination of fenofibrate or fenofibric acid and statins may be better tolerated than gemfibrozil and statins, although there are no direct comparative trials between the two combinations. Interestingly, the ESC/EAS guidelines recommend avoiding the combination of gemfibrozil with statins because of the risk of myopathy. 27
Ezetimibe
The Ezetimibe and Simvastatin in Hypercholesterolaemia Enhances Atherosclerosis Regression (ENHANCE) trial examined changes in carotid intima-media thickness (CIMT) in patients with familial hypercholesterolaemia. The patients were treated with simvastatin 80 mg/day alone or simvastatin 80 mg/day plus ezetimibe 10 mg/day. At study end there was no significant difference in CIMT between treatment groups even though the reduction in LDL-C levels and the increase in HDL-C levels were significantly greater with combination therapy than with simvastatin alone (p < 0.01 and p = 0.05, respectively). However, this study included only 13 (1.8%) patients with diabetes. 61
The Study of Heart and Renal Protection (SHARP) randomised patients with advanced CKD (22% with diabetes) to ezetimibe 10 mg/day plus simvastatin 20 mg/day versus placebo versus simvastatin 20 mg/day. The latter arm was rerandomised at one year to ezetimibe 10 mg/day plus simvastatin 20 mg/day versus placebo. 62 At one year, treatment with ezetimibe plus simvastatin was associated with significant (p < 0.0001) reductions in total cholesterol, LDL-C, TG and apolipoprotein B1 compared with placebo. 62 These changes were numerically greater than those seen with simvastatin alone but the differences were not analysed statistically. The effects of the combination regimen on lipids were maintained at two and one-half years. 62 After a median of 4.9 years of follow-up, the combination demonstrated a 17% relative risk reduction (95% confidence interval (CI) 0.74–0.94) in major atherosclerotic events compared with placebo. It should be noted that at this time point all patients on active treatment were receiving ezetimibe plus simvastatin and therefore this result cannot discriminate the impact of ezetimibe independently of a ‘statin-effect’ in these patients. 63
The Improved Reduction of Outcomes: Vytorin Efficacy International Trial (IMPROVE-IT) trial is currently investigating the effect of simvastatin (40 mg/day with the possibility to increase to 80 mg/day) with or without the addition of ezetimibe 10 mg/day in patients with acute coronary syndrome. 64 The study is expected to report the effect of treatment on CVD death, non-fatal stoke and non-fatal MI in 2013. 65
Niacin
The ARterial Biology for the Investigation of the Treatment Effects of Reducing cholesterol 6 – HDL and LDL Treatment Strategies (ARBITER 6-HALTS) trial compared ezetimibe 10 mg/day with niacin (target dose 2 g/day) in patients with CHD and low HDL-C levels who were already taking a statin.66,67 Compared with ezetimibe, niacin was associated with a greater rise in HDL-C and a greater reduction in TG whereas the reductions in LDL-C and total cholesterol were greater in the ezetimibe group (p≤0.01 for all comparisons).66,67 The trial was terminated early because niacin was associated with significant (p≤0.001) reductions in mean and maximal CIMT compared with baseline whereas these measurements did not change significantly in the ezetimibe group. Major CV events occurred in 5% of the ezetimibe group compared with 1% of the niacin group (p = 0.04).66,67
The NIcotinic Acid-Plaque (NIA-Plaque) trial investigated the effect of niacin 2 g/day versus placebo on carotid artery wall area (CAWA) in patients with type 2 diabetes and CHD and in patients with carotid/peripheral atherosclerosis. Both patient groups were on stable statin therapy. Niacin significantly increased HDL-C levels (p < 0.001) and reduced LDL-C and TG levels (p = 0.01 and p = 0.02, respectively) compared with placebo, and after one year CAWA was significantly reduced in the niacin group compared with the placebo group (p = 0.03). 68
Atherothrombosis Intervention in Metabolic syndrome with low HDL/high triglyceride and Impact on Global Health outcomes (AIM-HIGH) was a placebo-controlled trial of niacin 2 g/day added to simvastatin in patients aged ≥45 years with established vascular disease. This trial was discontinued because of an interim analysis finding that it was unlikely to demonstrate a difference in outcomes with niacin, and continuation would have been futile. 69 Some safety concerns were also identified, suggesting a possible increased stroke risk vs placebo. AIM-HIGH may have been underpowered, but the larger Heart Protection Study 2: Treatment of HDL to Reduce the Incidence of Vascular Events (HPS2-THRIVE) (clinicaltrials.gov identifier NCT00461630) is comparing the effect of niacin (2 g/day) plus laropiprant (a flush inhibitor) versus placebo on CVD events in patients with a history of CVD and receiving simvastatin 40 mg/day plus ezetimibe 10 mg/day if necessary. It is expected to report results in 2013 (clinicaltrials.gov identifier NCT00461630).
Omega-3 fatty acids
Omega-3 PUFAs, especially eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), may have a protective effect on the vasculature, although the mechanism for this effect remains unclear. No trial of PUFAs powered for CVD endpoints and conducted specifically in patients with diabetes has been reported. However, there is an ongoing trial of PUFAs in patients with diabetes (88%), or impaired fasting glucose levels/impaired glucose tolerance (12%). The Outcome Reduction with an Initial Glargine INtervention (ORIGIN) trial is investigating whether insulin glargine or PUFAs, or both, have a beneficial effect on CV events. 70 Results are expected in June 2012 (clinicaltrials.gov identifier NCT00069784).
In the GISSI-Prevenzione (GISSI-P) trial the effect of omega-3 PUFA ethyl esters (EPA/DHA in a ratio of 1:1.2) on death, MI or stroke was studied in patients who had experienced a recent MI. Omega-3 PUFA ethyl esters (1 g/day) were found to significantly (p = 0.048) reduce the risk of the primary endpoint compared with placebo, an effect that was seen after three months of treatment and which lasted until the end of the study (42 months).71,72 The GISSI-Heart Failure (GISSI-HF) trial reported that the same formulation significantly reduced all-cause mortality (p = 0.041) and death or hospitalisation for CV reasons (p = 0.009) compared with placebo in patients with heart failure. 73 In a separate randomisation, rosuvastatin 10 mg/day had no significant benefit on clinical endpoints versus placebo. 22
The Japan EPA Lipid Intervention Study (JELIS) investigated the combination of EPA (1.8 g/day) and a statin compared with statin monotherapy in Japanese patients with high total cholesterol levels at baseline. 74 The addition of PUFAs reduced the relative incidence of a major coronary event by 19% compared with statins alone (p = 0.011).
Two recent secondary prevention studies did not reproduce the benefits of PUFAs seen in the GISSI trials. Patients in the Alpha Omega Trial with a previous MI were given margarines supplemented with EPA/DHA (1.8 g/day), alpha-linolenic acid (ALA) (3 g/day) or placebo margarine. 75 No significant change was seen in the rate of major CV events for either margarine compared with placebo. The OMEGA trial investigated the effect of PUFA ethyl esters 1 g/day versus placebo on sudden cardiac death (SCD) in patients who had experienced a previous MI and were receiving recommended treatments including a statin in the great majority of patients. 76 Addition of PUFA ethyl esters did not significantly alter the rate of SCD (1.5% in both groups); this study, however, was not adequately powered to detect a change in SCD. The differences in outcomes seen in the various PUFA studies may partly result from differences in the omega-3 fatty acids, dose or formulation administered.
Key trials of non-lipid-lowering therapies
Intensive glycaemic control
Intensive glycaemic control was intensely debated after the glycaemic control arm of ACCORD, in which intensive therapy significantly increased mortality leading to early termination of the trial. 77 However, meta-analyses of this and other trials showed a significant benefit of intensive glycaemic control on non-fatal MI and all CHD events, with no effect on mortality.78,79
Recently, the issue of glycaemic worsening by statin therapy has been raised. This can be seen by an increased number of cases of new-onset diabetes while on statins versus placebo reported in some meta-analyses of randomised controlled trials in patients with no previous history of diabetes,80,81 although not in other trials that selected patients with low CV risk. 82 Analysis of five trials of moderate- versus intensive-dose statin therapy reported the latter to be associated with an increased risk of new-onset diabetes (odds ratio (OR) 1.12; 95% CI 1.04–1.22, I2 = 0%). 83 However, the number needed to harm was 489, compared with a number needed to treat of 155 for prevention of CV events.
In patients with diabetes, there is some glycaemic worsening with statin therapy, as observed in the CARDS and AFORRD trials.4,84 The latter study showed a placebo-corrected increase in HbA1c of 0.3 %. Given the enormous benefit of statin therapy, however, it is clear that these adverse effects on glycaemia do not change the indication of statin therapy. However, they do warrant regular monitoring of patient glycaemia, and if necessary, intensification of preventive or glucose-lowering measures, as reflected in the recent FDA changes to the labelling of statins in the US. 85
Intensive blood pressure control
In the BP arm of ACCORD, intensive control had no significant effect on the combined primary outcome of MI, stroke or death due to CVD or on all-cause mortality compared with standard control. 86 In an observational subgroup analysis of the International Verapamil SR Trandolapril (INVEST-BP) study, patients with ‘usual’ control (SBP 130 to < 140 mmHg) had a significantly lower rate of adverse CV events than the ‘uncontrolled’ group (SBP ≥140 mmHg) (12.6% vs 19.8%; p < 0.001), but ‘tight’ control (SBP < 130 mmHg) conferred no further reduction in event rates (12.6% vs 12.7%; p = 0.24) 87 . The Randomised Olmesartan And Diabetes MicroAlbuminuria Prevention (ROADMAP) study randomised patients with type 2 diabetes to olmesartan or placebo, with the addition of other drugs as needed to reduce BP to < 130/80 mmHg. After a median follow-up of 3.2 years, the onset of microalbuminuria (the primary endpoint) was delayed with olmesartan (p = 0.01) but fatal CV events occurred more commonly in the olmesartan group (0.7% vs 0.1%; p = 0.01). 88
Conclusions
Statins clearly reduce the risk of CVD in patients with type 2 diabetes and should be prescribed for most of these patients. However, even when receiving the most appropriate statin therapy by dose and drug, patients with type 2 diabetes remain at increased risk of CVD compared with non-diabetic individuals. Correction of other parameters of dyslipidaemia, such as high TG and low HDL-C levels, is a logical step to further reduce the risk of CVD. Treatments that appear to improve some clinical endpoints (at least in selected patients) include fibrates, niacin and PUFAs.
‘Normalisation’ of blood glucose or BP appears to be difficult and is burdened by adverse effects. It does not seem to greatly reduce CV endpoints and may be associated with increased CV death, especially in patients with pre-existing CVD. Further controlled clinical trials are needed to establish the best add-on therapy to statins for patients with type 2 diabetes and remaining dyslipidaemia. Clearly, therapy should always be adapted to the risk profile and characteristics of each individual patient.
Footnotes
Acknowledgements
Richard Murphy, Ph D, a professional medical writer, provided editorial assistance for the development of this review. This assistance was supported by an unrestricted educational grant from Abbott Products Operations AG, Allschwil, Switzerland. Prof Standl made all decisions regarding its scope and content.
Funding
This article was supported by an unrestricted educational grant from Abbott Products Operations AG, Allschwil, Switzerland. Abbott had no control over the content of this review and Prof Standl received no payment for any part of his work on this article.
Conflict of interest statement
Prof Standl:
Task force: First Joint EASD/ESC Guidelines
DSMBs: PROActive, Navigator, ACE Trials
Investigator: studies initiated by Oxford Trial Unit, Menarini, Novartis, NovoNordisk, Sanofi-Aventis, Servier, MSD/Merck
Lecturing honoraria and consultation fees: Astra Zenica, Bayer, BMS, GSK, Johnson & Johnson, Lilly, Menarini, Merck-Serono, MSD/Merck, Novartis, NovoNordisk, Rambaxi, Roche, Sanofi-Aventis, Servier, Takeda
