Abstract

Introduction
Diabetes mellitus (DM) is a grave and growing public health problem, with 25.8 million people estimated to have DM in the United States (8.3% of the population), 1 and 366 million worldwide. 2 The global burden of DM is projected to increase to between 439 million and 552 million people by 2030,2,3 driven primarily by the increase in type 2 diabetes (T2DM), and well in excess of earlier forecasts from just a decade prior.4,5 DM is a major independent risk factor for cardiovascular disease (CVD) and death, with CVD listed as the cause of death in ~65% of people with DM, 6 compared with ~25% in the general US population. 7 However, despite significant advances in drug development for the treatment of T2DM over recent decades, resulting in the present panoply of medications representing 11 drug classes approved in the US for the treatment of hyperglycaemia in patients with T2DM, there is no conclusive evidence to date that glucose lowering therapies reduce CVD risk and mortality in patients with T2DM.
Key to understanding this apparent paradox is the fact that glucose lowering drugs have historically been developed and approved primarily based on their ability to reduce glycosylated haemoglobin (HbA1c), the most widely used integrated clinical measure of glycaemic exposure. HbA1c has been adopted as the principal intermediate marker of DM treatment effectiveness, primarily because reducing hyperglycaemia has demonstrated benefits on DM symptoms and on the incidence of microvascular complications, including diabetic nephropathy, retinopathy and neuropathy. 8 The degree of hyperglycaemia as reflected by HbA1c also tracks observationally with the incidence and prevalence of cardiovascular complications and death,9,10 but the logical converse, that controlling hyperglycaemia should proportionally reduce the incidence of these complications, has not been clearly demonstrated in large, randomised clinical trials.
Three such trials completed in recent years only added to the confusion. The Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) 11 trial, the Veterans’ Affairs Diabetes Trial (VADT) 12 and the Action to Control Cardiovascular Risk in Diabetes (ACCORD) 13 trial examined the effect of ‘intensive’ versus ‘standard’ glycaemic control with various drug combinations on CVD and mortality, in a total of over 23,000 patients with established T2DM participating in these trials. In aggregate, the much discussed results of these trials failed to demonstrate a CVD or survival benefit for intensive glucose control per se and, by virtue of trial designs, did not afford the opportunity to assess effects of specific drugs or drug classes in this regard. Furthermore, increased cardiovascular and all-cause mortality in the intensive treatment group of the ACCORD trial led to premature termination of that trial, raising an important safety concern that has yet to be fully understood. One overarching implication of these study results in aggregate is that the ability of a new drug or drug combination to reduce HbA1c is not an indication of how it may end up affecting cardiovascular outcomes and mortality.
A more optimistic signal came from the 10-year post-trial follow-up of the United Kingdom Prospective Diabetes Study (UKPDS), 14 which reported a significantly lower incidence of myocardial infarction (MI) and all-cause mortality in patients assigned to intensive versus standard glycaemic control, long after discontinuation of the randomised trial protocol treatment and the resulting rapid loss of HbA1c contrast between the groups. A multitude of factors could theoretically underpin the discrepancies between the UKPDS long-term follow-up and ADVANCE / VADT / ACCORD, including the glycaemic intervention in UKPDS initiated earlier in the course of T2DM (i.e. at the time of diagnosis) and longer duration of trial treatment; different drugs used; less intensive HbA1c targets; and much longer follow-up. 15 The key implication of the UKPDS 10-year post-trial follow-up is that cardiovascular risk and mortality reduction are possible with glycaemic control, but may depend greatly on how (with what drugs, to what HbA1c targets), when (how early or late in the evolution of T2DM) and for how long glycaemic control is achieved. Simply showing that a new drug lowers HbA1c below a certain threshold for a set period of time does not capture this complexity, and in no way can predict cardiovascular benefit – or lack of cardiovascular harm.
In fact, one does not need to look far back into the history of DM treatments to find cases of drugs that were good at lowering HbA1c and yet were associated with cardiovascular harm and increased mortality, as well as some having pernicious off-target effects. The most prominent recent example is that of rosiglitazone, linked to increased incidence of MI and death in a 2007 meta-analysis that sparked much controversy but has not yet been refuted, 16 eventually leading to severe restrictions put in place by the US Food and Drug Administration (FDA) on the use of rosiglitazone, and to complete withdrawal of the drug in Europe and most other countries around the world. Another recent (and less controversial) example is that of muraglitazar, a dual agonist of the peroxisome proliferator-activated receptors (PPARs) α and γ, for which development was halted in 2006 because of an increased composite risk of heart failure, major CVD events and death in phase II and III clinical trials. 17
Importantly, there is increasing evidence that cardiovascular assessment of individual drugs is not reliably applicable to other members of the same drug class. For instance, the important safety concerns with rosiglitazone cannot be extrapolated to pioglitazone, the other thiazolidinedione presently available. To the contrary, the Prospective Evaluation of Pioglitazone and Macrovascular Events (PROactive) randomised trial suggested that pioglitazone might in fact decrease the risk for a composite of MI, stroke and death in patients with T2DM (this was a secondary endpoint; pioglitazone had a similar but not statistically significant effect on the primary endpoint, a broad CVD composite). 18 Similarly, recent retrospective evidence adds further support to the notion that different sulphonylureas are associated with widely variable outcomes with regard to CVD risk and mortality: tolbutamide, glyburide, glipizide and glimepiride were all associated with worse outcomes when compared with metformin in a large nationwide cohort, while outcomes with gliclazide and metformin were not statistically different.19,20 In this study, extensive statistical adjustment was performed for differences in patient mix across the various drugs analysed as well as adjustment for propensity for prescription of each, and in addition, analyses were stratified by presence or absence of prior myocardial infarction.
In light of cardiovascular safety concerns with drugs that work perfectly well at reducing HbA1c, the FDA issued a non-binding Guidance for Industry in December 2008, recommending that new diabetes drugs be assessed for cardiovascular safety prior to approval. 21 Recommendations include prospective and blinded adjudication of all cardiovascular events in phase II and III trials, and appropriate design of these trials to allow for an adequate meta-analysis of all phase II and III data, which would have to show no evidence of increased cardiovascular risk with the new drug. The FDA will also generally require a post-marketing cardiovascular safety trial of newly approved diabetes drugs if premarketing data indicate a potential but not statistically significant risk (specifically, if the upper bound of the 95% confidence interval for estimated increased risk is greater than 1.3). 21
The perfect storm
Contrary to the outcry from some investigators, health care providers and industry advocates that the new regulatory guidelines put forth in 2008 will thwart development, break the bank and are largely unnecessary, we feel strongly the opposite.
First, increasing awareness of the cardiovascular risk and consequences of diabetes over the last several decades underpins the imperative to assess CVD risk effects of glucose management treatments and strategies.
Second, the global health burden associated with diabetes-related morbidity and mortality continues to expand with no foreseeable resolution, in which case we simply cannot afford, medically, socially or economically to continue to use drugs effective at modulating HbA1c without having some certainty that at the very least CVD risk is not increased, and ideally to identify therapies with which CVD risk may be favourably affected.
Third, we have witnessed a bi-directional invalidation of HbA1c as a putative surrogate for CVD efficacy. For example, metformin had approximately one-half the potency of HbA1c reduction as the insulin provision strategies in the UKPDS, but had superior outcomes. To the contrary, rosiglitazone and muraglitazar with relatively robust HbA1c efficacy had adverse CV safety signals, and much discordance observed in the recent randomised trial results summarised above assessing more intensive versus standard intensity glucose control.
Lastly, as described previously, the explosion of available therapies just over the past decade and a half has resolved most of the urgency to bring any effective therapies to clinical use for such a vulnerable population, from three classes to 11 classes of drugs and from fewer than 10 to greater than 30 formulations presently available.
Though many other factors have contributed to the evolution of the regulatory guidance, we feel that these four key conditions have comprised the ‘perfect storm’ underpinning the imperative for the present focus on and requirement for CVD risk assessment.
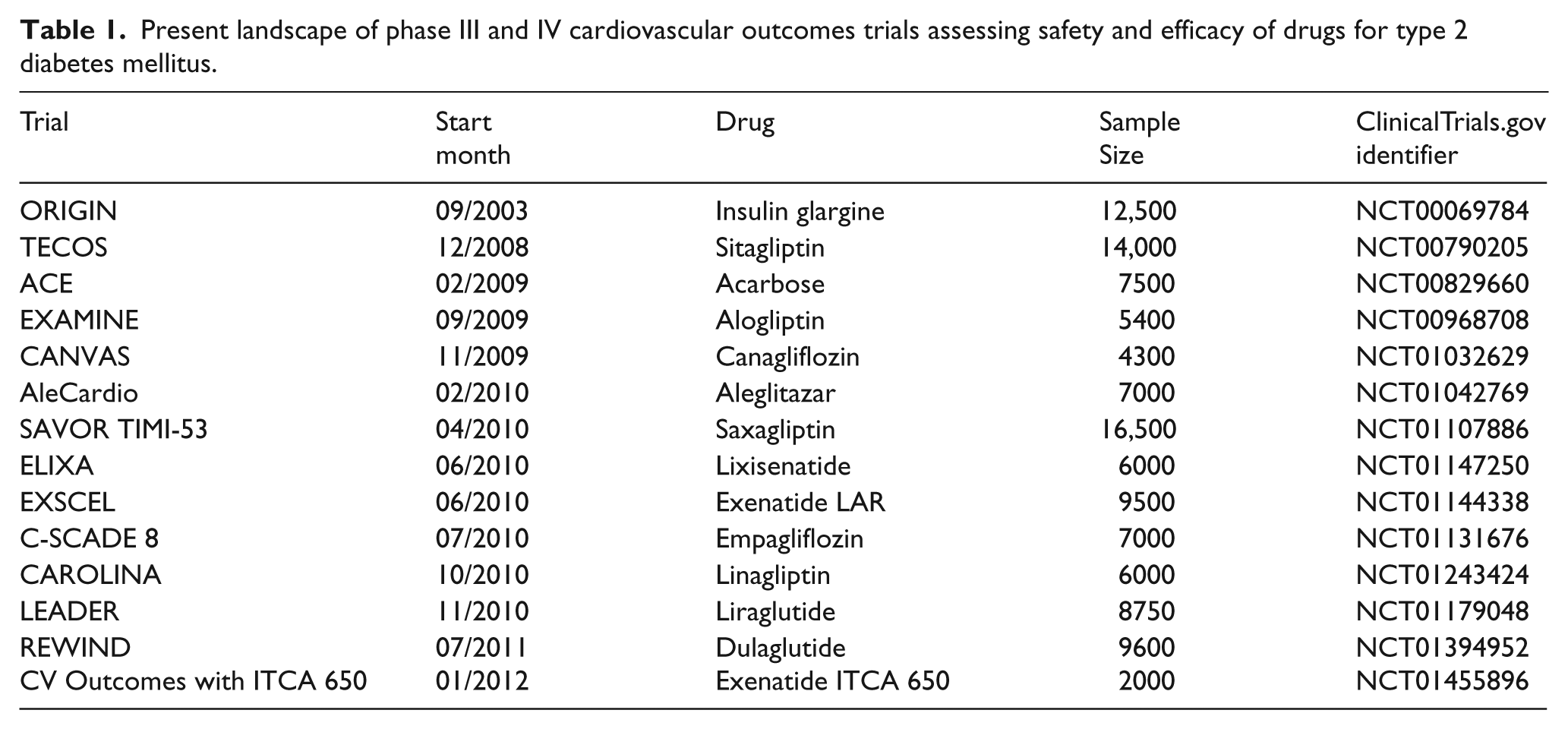
When the FDA published its Guidance for Industry at the height of the 2008–9 global financial crisis, it was indeed conceivable that the added burden of demonstrating cardiovascular safety would slow down or even halt the development of new T2DM drugs. Three years later, although the exact metrics for a scientific assessment of such a trend are not readily available, there is no overt evidence for any slowing of diabetes drug development. Numerous cardiovascular outcome trials of T2DM drugs are presently underway or in advanced stages of planning (Table 1) with aggregate planned enrolment of over 115,000 patients with T2DM, and at least 16 novel therapeutic targets being probed by investigational compounds in clinical registration programs from phase I to III. These projects will yield exponential growth of the available evidence for cardiovascular safety and efficacy of drugs for T2DM over the next decade and beyond and, hopefully, will lead to better treatment strategies and better cardiovascular outcomes for our patients.
Present landscape of phase III and IV cardiovascular outcomes trials assessing safety and efficacy of drugs for type 2 diabetes mellitus.
In this issue of this journal and the next forthcoming, we will publish a series of articles reviewing the state-of-the-art knowledge with regard to various drugs and drug classes in development or use for T2DM, focusing on their known and anticipated effects on cardiovascular intermediates and outcomes, and more detailed descriptions of relevant trials presently underway. These papers represent much of the influence of the evolved regulatory guidance for T2DM drug evaluation and reflect a truly exciting time for science, medicine and, most importantly, for patients with type 2 diabetes.
Footnotes
Funding
This work received no specific support from any funding agency in the public, commercial, or not-for-profit sectors. MOG is supported by a training grant from the U.S. National Heart, Lung and Blood Institute (T32-HL007360).
