Abstract
The active incretin hormone glucagon-like peptide-1(7-36)amide (GLP-1) is a 30-amino acid peptide that exerts glucoregulatory and insulinotropic actions by functioning as an agonist for the GLP-1 receptor (GLP-1R). In addition to its anti-diabetic effects, GLP-1 has demonstrated cardioprotective actions. Here we review the cardiovascular effects of the GLP-1 analogues currently approved for the treatment of type 2 diabetes, namely exenatide and liraglutide. We discuss their anti-hyperglycaemic efficacy, and offer a clinical perspective of their effects on cardiovascular risk factors such as body weight, blood pressure, heart rate and lipid profiles, as well as their potential consequences on cardiovascular events, such as arrhythmias, heart failure, myocardial infarction and death. Lastly, we briefly review additional GLP-1R agonists in clinical development.
Introduction
In 2010, almost 300,000,000 adults worldwide were reported to be suffering from type 2 diabetes (T2D), a number that is expected to double by 2030. 1 The well-characterised associations between T2D and cardiovascular disease (CVD) are two-fold. First, CVD is the underlying cause of more than 60% of deaths in diabetic patients.2–5 Second, the hyperglycaemia of T2D, as measured by the percentage of glycosylated haemoglobin (HbA1c), is believed to increase the risk of coronary artery disease (CAD) and myocardial infarction (MI).6,7 Given this relationship, physicians managing T2D must remain aware of the cardiovascular effects of their therapeutic choices. Unfortunately, with only a few exceptions in select patient populations, most oral anti- diabetic (OAD) therapies made available to date have not resulted in reduced incidences of MI, stroke or cardiovascular death in diabetic subjects. 8 In this context, the recently introduced incretin hormone targeted therapies may hold greater promise. As a new class of anti-diabetic drug, they have demonstrated potentially cardioprotective effects in both animal models and early clinical studies. 9
The incretin hormone glucagon-like peptide-1 (GLP-1) is derived from a proglucagon precursor and secreted by intestinal L-cells in response to oral nutrient ingestion. The majority of circulating GLP-1 is the 30-amino acid peptide GLP-1(7-36)amide, which acts through a G-protein-coupled receptor (GLP-1R) on pancreatic beta-cells to exert glucoregulatory and insulinotropic actions. 9 However, GLP-1(7-36)amide is short-lived, being rapidly degraded to the non-insulinotropic metabolite GLP-1(9-36)amide, by action of the ubiquitously expressed enzyme dipeptidyl-peptidase-4 (DPP-4). In patients with T2D, secretion of GLP-1 is diminished, and administration of either DPP-4 inhibitors or exogenous GLP-1 analogues represents potential therapeutic approaches. 10
While DPP-4 inhibitors, such as sitagliptin, saxagliptin and vildagliptin, are approved for the treatment of T2D, the current review will focus primarily on DPP-4-resistant GLP-1 analogues, also known as GLP-1R agonists (e.g. exenatide and liraglutide). Meanwhile, the class of DPP-4 inhibitor drugs will be reviewed by Jose and Inzucchi in the same issue of this journal. The first GLP-1R agonist to receive regulatory approval in the USA and Europe was exenatide twice daily (ExBID), marketed under the trade name Byetta™, and indicated for T2D patients inadequately controlled on oral metformin and/or sulphonylurea (SU).11,12 The US Food and Drug Administration (FDA) approved the use of ExBID in 2005, followed by the European Marketing Agency (EMA) in 2006.11,12 In October 2007, the FDA released a warning that post-marketing studies of ExBID may suggest a link between treatment with the drug and acute pancreatitis, and that healthcare professionals should monitor for signs of pancreatitis in patients using ExBID. 11 Between 2005 and 2008, over 7,000,000 prescriptions had been written for ExBID, with reports of only 78 cases of impaired kidney function. 13 The longer-acting, once daily liraglutide, marketed as Victoza®, recently received regulatory approval in both the USA and Europe. Liraglutide was first approved by EMA in June 2009, followed by FDA approval in January 2010.14,15 Currently, a longer acting, sustained-release formulation of exenatide (exenatide once weekly, ExQW, marketed as Bydureon™) is in late stage clinical development and received marketing authorisation in Europe a few months prior to the writing of this manuscript. 16
The current perspective paper reviews clinical data on the cardiovascular effects of currently approved GLP-1R agonists (ExBID, liraglutide, ExQW). We also discuss here the potential cardiovascular benefits and limitations associated with the use of these drugs in T2D patients, either as monotherapy or in combination with other standard OADs.
Methods
We searched PubMed and Embase databases for the period 1989 to present, with the key words: liraglutide, exenatide, cardiovascular, human, clinical, body weight, blood pressure, pulse rate, lipids, MACE, arrhythmia, heart failure, myocardial infarction, death, events. We then selected relevant articles dealing with our topic for review and discussion. We also searched the official clinical trials registry at www.clinicaltrials.gov for studies currently underway with exenatide and liraglutide examining cardiovascular endpoints, using keywords: exenatide, liraglutide, cardiovascular. Lastly, electronic versions of abstracts presented at the most recent EASD conferences from 2009 to 2011 available at www.springerlink.com were consulted for additional information.
Pharmacokinetics
Exenatide
Exenatide is a synthetic version of exendin-4, a 39 amino acid peptide isolated from the venom of the Gila monster (Heloderma suspectum).17,18 This venom has been widely studied, and is known to contain serotonin, hyaluronidase, phospholipases, phosphodiesterases, proteases and some biologically active peptides. In the discovery of exenatide as a therapy for diabetes, researchers first noticed that Gila monster venom could induce insulin secretion in dispersed acini from guinea pig pancreas, with concurrent 50–60-fold increase in cellular cAMP. 18 Exenatide shares 53% homology with native GLP-1, but still binds effectively to GLP-1R on pancreatic beta-cells for its insulinotropic effects. 19 After subcutaneous (s.c.) administration, exenatide reaches peak plasma concentrations in 2.1 h, with a half-life of approximately 2 h. DPP-4-resistant exenatide is measurable in plasma for up to 10 h following s.c. injection, warranting twice-daily dosage for full glycaemic control. The elimination of exenatide from the body is thought to be mainly by glomerular filtration, followed by proteolytic degradation. 20 The pharmacokinetics of exenatide are unaffected by age or ethnicity.
ExQW has been formulated by embedding a 2 mg dose of exenatide within poly-(D,L-lactide-co-glycolide) microspheres, a biodegradable medical polymer. 21 The latter enables sustained release of exenatide, allowing once-weekly dosing and ensuring continuous threshold-level exposure to exenatide. 22 This method has been used in other long-acting pharmaceutical preparations such as naltrexone (for alcohol and opioid dependence) and risperidone (antipsychotic). 22
Liraglutide
Liraglutide is a potent, long-acting synthetic analogue of the human GLP-1 molecule. It shares 97% homology with native GLP-1(7-37), with the addition of a γ-glutamic acid residue at position 26 to allow attachment of a C-16 fatty acid (palmitoyl) group and substitution of lysine for arginine at position 34 to ensure mono-acylation. 23 Structure–activity studies have reported a relationship between liraglutide acylation and potency for GLP-1R as well as protraction time in vivo. 24 Indeed, the presence of a palmitoyl group on liraglutide facilitates non-covalent binding to plasma albumin, which in turn hinders DPP4 action by shielding the molecule from enzymatic access. As such, due to albumin binding, aggregation at injection site 25 and reduced susceptibility to DPP-4, liraglutide has been reported to have slow absorption (T max: 10–14 h) and a long plasma half-life of 12–13 h following s.c. injection, allowing for once-daily dosing.26,27 More importantly, the acylated modifications of liraglutide do not confer any loss in potency for GLP-1R compared with native GLP-1. 23
Pharmacokinetic studies in vivo revealed that liraglutide circulates in the plasma mainly as the intact molecule (≥ 89%), alongside two minor metabolites (≤ 11%), following a single s.c. injection of 0.75 mg to healthy males. 28 It is believed that both DPP-4 and neutral endopeptidase (NEP) are involved in the degradation of liraglutide in vivo. However, due to absence of intact liraglutide in urine and faeces, and the low level of metabolites in plasma, it is speculated that liraglutide is fully degraded within the body and that its degradation products are either recycled into new proteins and lipids, or eliminated by the liver and kidney, or even excreted from the body as carbon dioxide, urea and water.28,29 Subsequently, in a separate study, it was found that T2D patients with renal impairment did not experience increased exposure and did not require dose-adjustments when given once-daily liraglutide (0.75 mg s.c.). 30 Moreover, the pharmacokinetics of liraglutide was unaffected by age and gender (after adjusting for body weight) following a single injection of 1.0 mg s.c. to healthy subjects. 31
Efficacy of treating hyperglycaemia
Exenatide
ExBID has demonstrated efficacy in glycaemic control, based on large randomised, controlled trials evaluating ExBID either as add-on therapy to metformin and/or SU or compared with insulin, in approximately 2731 T2D patients (Table 1). As adjunct therapy to existing OAD, ExBID (10 µg) decreased HbA1c (to ≤ 7%) in a significantly larger proportion of patients than placebo after 30 weeks of therapy.32–34 This effect seems to last over time, as ExBID exposure caused sustained reductions in HbA1c (−1.0%) over three years, with 46% of patients achieving the targeted ≤ 7% HbA1c. 35 In T2D patients sub-optimally controlled on metformin and SU, addition of ExBID caused similar reductions in HbA1c (−1.1%) as insulin glargine after 26 weeks of treatment (Table 1). 36 Similarly, in comparison with biphasic insulin aspart, ExBID induced equivalent reductions in HbA1c, after 52 weeks in combination with existing metformin and SU. 37 However, ExBID was much more effective in controlling postprandial glucose excursions than insulin glargine or aspart.36,37
Exenatide (ExBID) studies.
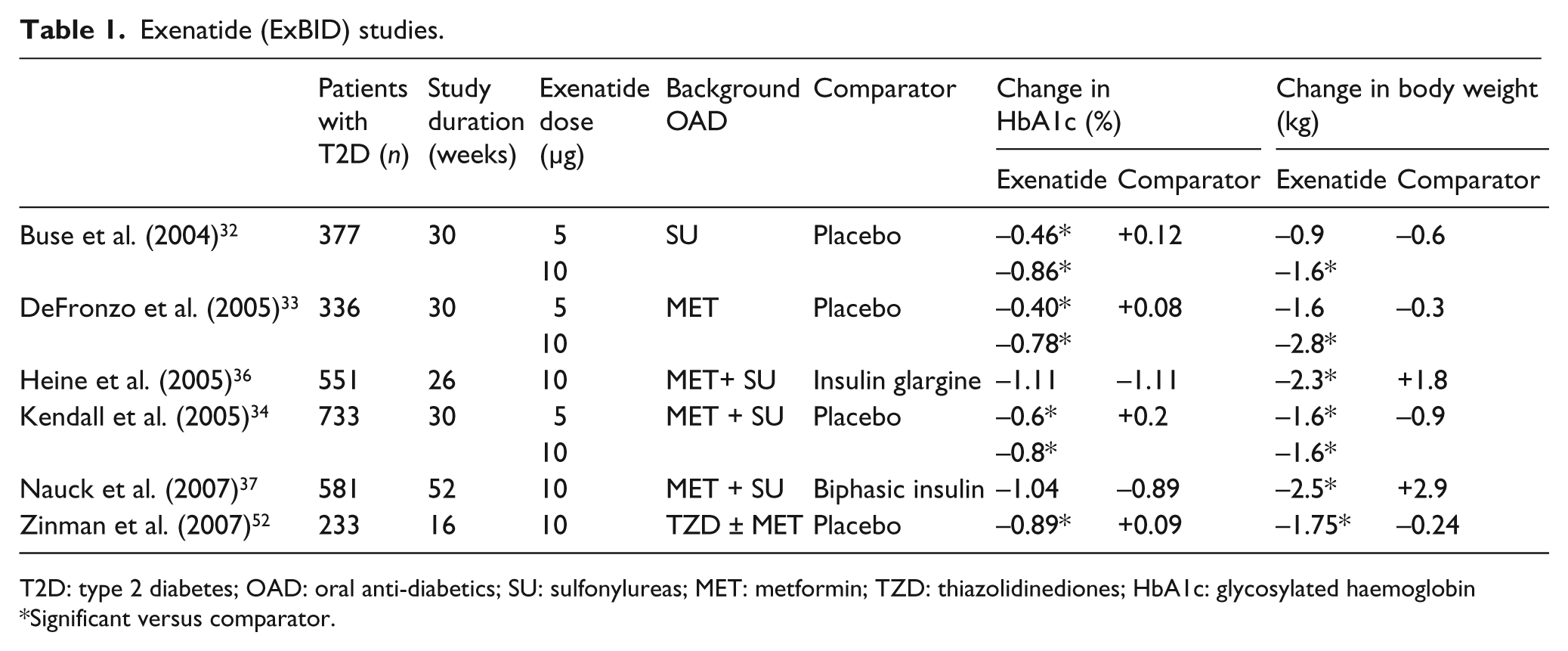
T2D: type 2 diabetes; OAD: oral anti-diabetics; SU: sulfonylureas; MET: metformin; TZD: thiazolidinediones; HbA1c: glycosylated haemoglobin
Significant versus comparator.
The recent Phase III DURATION (Diabetes therapy Utilisation: Researching changes in A1C, weight and other factors Through Intervention with exenatide ONce weekly) clinical development programme investigated the efficacy and safety of ExQW versus ExBID, sitagliptin, pioglitazone and titrated insulin glargine as adjunct treatments to diet and exercise, metformin monotherapy or combination OADs in T2D patients.38–41 In DURATION-1, -2, -3 and -5 studies, ExQW consistently and significantly reduced HbA1c (−1.5% to −1.9%) after 24–30 week treatments, as compared with active controls (ExBID, sitagliptin, pioglitazone, and titrated insulin glargine) (Table 2). A pooled analysis of the four DURATION trials examining data from 804 patients treated with ExQW for at least 20 weeks revealed that 46% of these patients reached a target HbA1c of ≤ 6.5%, and 65% achieved HbA1c < 7% at study end point, which were significantly greater than active comparators. 42 Of note, although the open-label design of the DURATION trials may have allowed for patient and investigator bias, ExQW consistently improved glycaemic control versus active comparators across all four DURATION studies.
DURATION studies (ExQW).
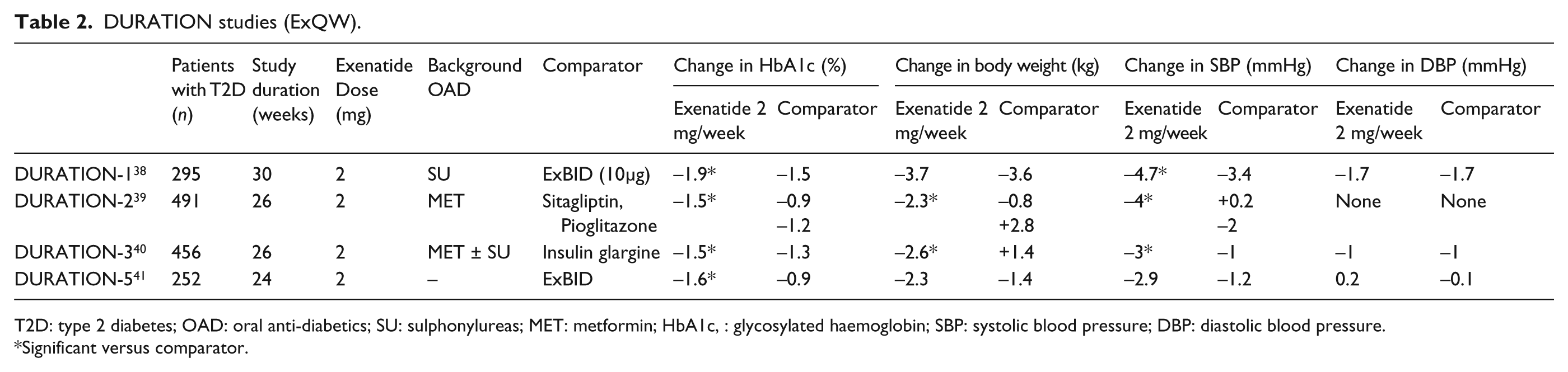
T2D: type 2 diabetes; OAD: oral anti-diabetics; SU: sulphonylureas; MET: metformin; HbA1c, : glycosylated haemoglobin; SBP: systolic blood pressure; DBP: diastolic blood pressure.
Significant versus comparator.
Liraglutide
In addition to a favourable pharmacological profile in terms of long half-life and duration of action, liraglutide has demonstrated efficacy in glycaemic controls of patients with T2D. The Liraglutide Effect and Action in Diabetes (LEAD) study is a worldwide, global development programme of six randomised controlled Phase III trials in > 4000 patients, designed to investigate the efficacy of liraglutide at each step in the treatment continuum of T2D care, from monotherapy to combination with one or two OADs (Table 3).43–48 With HbA1c as the primary efficacy endpoint, the LEAD studies demonstrated that liraglutide was effective both as first-line and combination therapy in lowering blood glucose in T2D patients. For example, reductions in HbA1c occurred quickly within eight weeks after initiation of liraglutide monotherapy (1.2 and 1.8 mg) in patients previously on a diet and exercise regimen. 45 At the higher dose of 1.8 mg, an HbA1c < 7% was sustained and remained stable through the 52 weeks of treatment.
Liraglutide studies – cardiovascular outcomes.
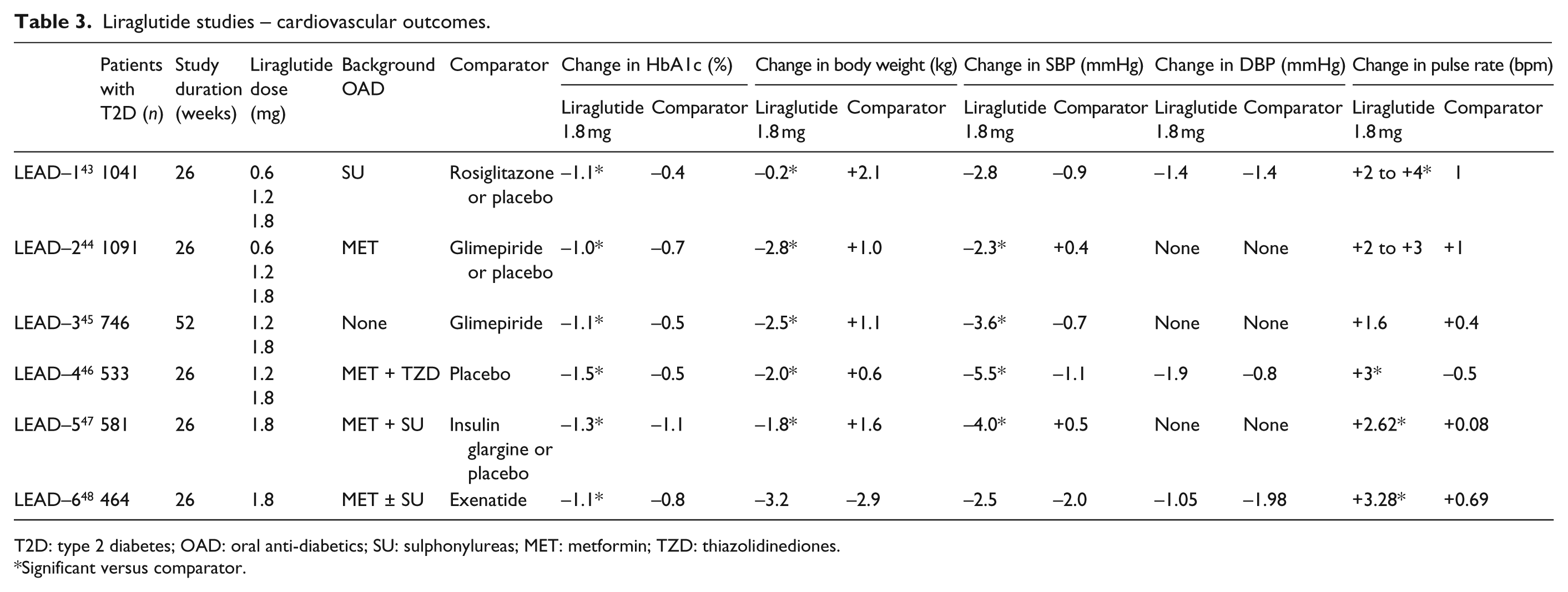
T2D: type 2 diabetes; OAD: oral anti-diabetics; SU: sulphonylureas; MET: metformin; TZD: thiazolidinediones.
Significant versus comparator.
Cardiovascular safety
T2D is a multi-factorial disease often manifesting end-organ complications, primarily CVD, but also nephropathy and retinopathy, leading to increased morbidity and mortality. Importantly, other risk factors that contribute to CVD, including weight gain, hypertension, and hyperlipidaemia, also frequently manifest in T2D. Therefore, in addition to adequate glycaemic control, anti-diabetic treatment regimens should ideally be accompanied by favourable cardiovascular safety profiles and not result in weight gain. Indeed, the clinical development programmes for exenatide and liraglutide have evaluated cardiovascular safety outcomes as secondary endpoints, including body weight, systolic blood pressure (SBP), diastolic blood pressure (DBP), heart rate (HR) and cardiovascular events (arrhythmias, heart failure, MI, or death). Each of these cardiovascular endpoints is individually reviewed below for each of the GLP-1 receptor agonists, ExBID, liraglutide and ExQW.
Body weight
Obesity is associated with an increased risk for the development of T2D, hypertension and CVD including coronary atherosclerosis, congestive heart failure, cardiomyopathy, stroke, arrhythmias and sudden death. 49 Several current anti-diabetic agents such as insulin, SU and thiazolidinediones (TZDs) predispose to weight gain in T2D subjects.50,51
Exenatide treatment has been linked to weight loss in several Phase II and III clinical studies, examining more than 2000 patients. Patients on background metformin and/or SU therapy lost on average 1.6 kg when treated with ExBID compared with placebo (Table 1).32–34 Similarly, ExBID addition to TZD therapy resulted in significant weight losses of −1.75 kg versus placebo. 52 Compared with insulin glargine or aspart, ExBID caused even greater significant weight changes in the range 2.3–2.5 kg in patients sub-optimally controlled on metformin and SU, with glargine and aspart groups gaining 1.8 and 2.9 kg respectively (Table 1).36,37 More recently, patients on background metformin experienced −3.5 kg weight loss with ExBID, compared with +0.3 weight gain with insulin glargine, without any change in bone mineral density. 53 Reductions in body weight with ExBID have been durable, with 101 patients losing an average of 5 kg and maintaining this loss over 1.4 years. 54 Another study in 299 T2D patients showed significant weight loss with ExBID in females with a higher baseline body mass index (BMI) accompanied by a decrease in waist circumference. 55
In the DURATION clinical development series, patients on ExQW consistently lost on average 2–4 kg across all trials (Table 2).38–41 The weight loss achieved with ExQW was not significantly different from ExBID.38,41 On the other hand, while patients on pioglitazone and insulin glargine gained on average 1–3 kg, those with ExQW lost approximately 2 kg, resulting in the treatment difference of 4–5 kg (Table 2).39,40 Moreover, body weight reductions were significantly larger for ExQW than for subjects in the sitagliptin treatment arm (−2.3 kg vs. −0.8 kg respectively) (Table 2). 39
While early Phase II clinical trials demonstrated no increase in body weight associated with liraglutide, later Phase III trials revealed meaningful weight loss. Phase II clinical studies showed that, compared with placebo, T2D patients receiving as low as 0.45 mg of liraglutide experienced 1.2 kg reductions in body weight after 12 weeks, while another study showed 2.99 kg reductions with liraglutide 1.90 mg, revealing a dose-dependent effect of liraglutide.56,57 While another placebo-controlled study in T2D subjects did not show any change in body weight with liraglutide 0.6 mg for eight weeks, there was a non-significant trend towards decreased total fat mass and significant increase in lean body mass, all accompanied by no change in 24 h energy expenditure. 58 There was no significant difference between T2D patients on metformin or liraglutide 0.045–0.75 mg in terms of loss in body weight after 12 weeks. 59 Moreover, addition of liraglutide 0.9 mg to SU monotherapy for 24 weeks did not cause any relevant change in body weight compared with SU alone. 60
Alternatively, the LEAD trials revealed significant body weight reductions with liraglutide 1.8 mg monotherapy or adjunct therapy with OADs in people with T2D, as compared with standard therapy (LEAD 1–5). In the LEAD-2 study, a quarter of patients lost an average of 7.7 kg with liraglutide 1.8 mg + metformin. 44 Moreover, weight loss with liraglutide varied as a function of baseline BMI. While all BMI subgroups (< 25; ≥ 25 < 30; ≥ 30 < 35; and ≥ 35; kg/m2) experienced weight loss, the greatest decrease in body weight occurred in subjects with BMI ≥ 35 kg/m2. 61
Some of the central and peripheral biological actions of GLP-1 include delayed gastric emptying and reduced food intake. Having said this, the exact mechanisms governing body weight reduction with exenatide or liraglutide are not known and further studies are warranted. Meanwhile, the weight reduction observed with liraglutide is believed to occur independent of its GI effects. In a substudy of LEAD-2 and LEAD-3, in which body composition was monitored by computerised tomography, the weight loss associated with liraglutide (either as monotherapy or in combination with metformin) was primarily due to reductions in fat mass, mainly visceral adipose tissue. 62
Blood pressure and heart rate
Hypertension, defined as SBP > 140 mmHg and DBP > 90 mmHg, is a frequent comorbidity in T2D contributing to CVD risk. Stringent blood pressure control has been shown to reduce CV events and all-cause mortality.63,64 A reduction of 5.6 mmHg resulted in an 18% decrease in the risk of death from CV disease in patients with diabetes. 65 Accordingly, SBP and DBP are important considerations in the continuum of care in T2D.
Exenatide, both as ExBID and as ExQW, has a favourable effect on blood pressure, primarily SBP. A pooled data analysis from six clinical trials investigating ExBID in approximately 2171 T2D patients for six months revealed significantly greater reductions in SBP with ExBID compared with placebo (difference of −2.8 mmHg) or insulin (difference of −3.7 mmHg). 66 This decrease was greater in patients with a baseline SBP of ≥ 150 mmHg. There were no significant changes in DBP, although a small correlation was found between weight loss and reductions in SBP (r = 0.09, p = 0.02). 66 In a smaller double-blind placebo-controlled pilot study comparing ExBID twice daily versus placebo for 12 weeks in patients already on metformin and/or a TZD, significant reductions in 24 h, daytime and night-time SBP were observed in patients receiving ExBID without accompanying changes from baseline in DBP or mean 24 h HR. 67
ExQW significantly reduced SBP in the DURATION trials, with mean reductions ranging from −3 to −5 mmHg observed across trials (Table 2).38–40 Active comparators sitagliptin, pioglitazone and insulin glargine failed to significantly reduce SBP from baseline, whereas the ExBID group had similar reductions to ExQW (Table 2). Neither ExQW nor active comparators caused any changes in DBP across the three DURATION studies (Table 2). Meanwhile, mean HR increased by four beats per minute (bpm) compared with baseline in patients receiving ExQW in the DURATION-3 trial, but not in the insulin glargine group. 40 It remains unclear whether this magnitude of change in HR holds any clinical significance in patients treated with this agent.
While the DURATION investigators noted no association between changes in HR and SBP, it remains unclear whether the observed increases in HR are in any way related to the mechanisms that cause reduced SBP. The latter may include improved endothelial function, natriuresis and/or diuresis. In a small pilot study, 28 patients with impaired glucose tolerance or early onset T2D manifest improved post-prandial endothelial function after a single injection of exenatide 10 µg versus placebo, following a high-fat meal. 68 Improved endothelial function has also been demonstrated in small human studies using native GLP-1. 69 A pre-clinical study in rats has demonstrated that exenatide can induce natriuresis and diuresis with accompanying increases in glomerular filtration rate and renal blood flow. 70 Although these mechanisms have yet to be confirmed in humans, one small clinical study with native GLP-1 supports the notion that GLP-1 receptor activation can reduce renal sodium absorption. 71
Whether used as a monotherapy or in combination with other agents, liraglutide consistently reduced SBP across the LEAD 1–5 trials, with SBP falling by means of 2.7 to 6.6 mmHg from baseline with liraglutide 1.2 and 1.8 mg doses.43–48 This reduction was even evident in patients with well-controlled blood pressures at baseline (min–max: 128/76–134/81 mmHg). The same trend was reported from a meta-analysis of all six Phase III LEAD studies examining the specific effect of liraglutide on SBP. 72 In this analysis, the magnitude of SBP reductions observed with liraglutide was greatest in subjects with the highest baseline SBP. For subjects in the highest quartile of baseline SBP (> 140 ≤ 190 mmHg) the fall in SBP was 11.4 mmHg with liraglutide 1.2 and 1.8 mg doses versus 7.7 mmHg with placebo (p < 0.0001). This effect on SBP occurred early and was maintained over time. In a model (adjusted for quartile of baseline SBP) examining longitudinal effects, reductions in SBP began within two weeks of treatment onset, preceded any weight loss (which occurred after eight weeks of therapy) and were maintained over the entire 26 weeks of the study periods.72,73
The reductions in SBP observed in the LEAD trials were not accompanied by any statistically significant changes in DBP.43–48 While patients in the six LEAD trials did experience small reductions in DBP across all treatments groups, e.g. 0.7 to 1.4 mmHg in LEAD-1, there were no significant reductions between groups. 43 Like ExQW, liraglutide once daily resulted in small but statistically significant increases in HR.44–48 Addition of liraglutide to combination therapy resulted in HR increases ranging from 2 to 3 bpm, as compared with active comparator or placebo.
Again, both the clinical significance and mechanisms underlying the effects of exenatide and liraglutide on HR and SBP in diabetic humans remain to be established. To date, one pre-clinical study employing spectral analysis of HR telemetry in mice has suggested that the ability of exenatide to depress HR variability in mice is due in part to reduced central parasympathetic outflow to cardiac vagal neurons. 74 More mechanistic clinical studies are required to explore this and other cardiovascular physiological consequences of GLP-1 receptor activation in humans.
Lipid profile
Another biomarker associated with CV risk is the lipid profile, including total cholesterol (TC), low density lipoprotein (LDL), high density lipoprotein (HDL), free fatty acids (FFAs) and triglycerides (TGs).
Liraglutide also displays a favourable serum lipid profile versus active comparators. A meta-analysis of all six LEAD trials revealed that, after 26 weeks of treatment, liraglutide significantly decreased TC, LDL, FFA and TG levels compared with baseline (p < 0.01 for all). 75 Compared with TZD, insulin or placebo, liraglutide registered a significantly greater reduction in TC of – 0.13 mmol/L (p < 0.01) (Table 4). The LDL level was reduced by – 0.2 mmol/L (p < 0.05) compared with TZD, glimepiride or insulin, although HDL levels were also decreased with all treatment arms except TZD.75 Two additional biomarkers of CV risk, namely brain natriuretic peptide and high-sensitivity C-reactive protein, were also reduced with liraglutide (p < 0.001), while levels of lipoprotein apoB did not change with any treatment. 75
Liraglutide studies – lipid profiles.
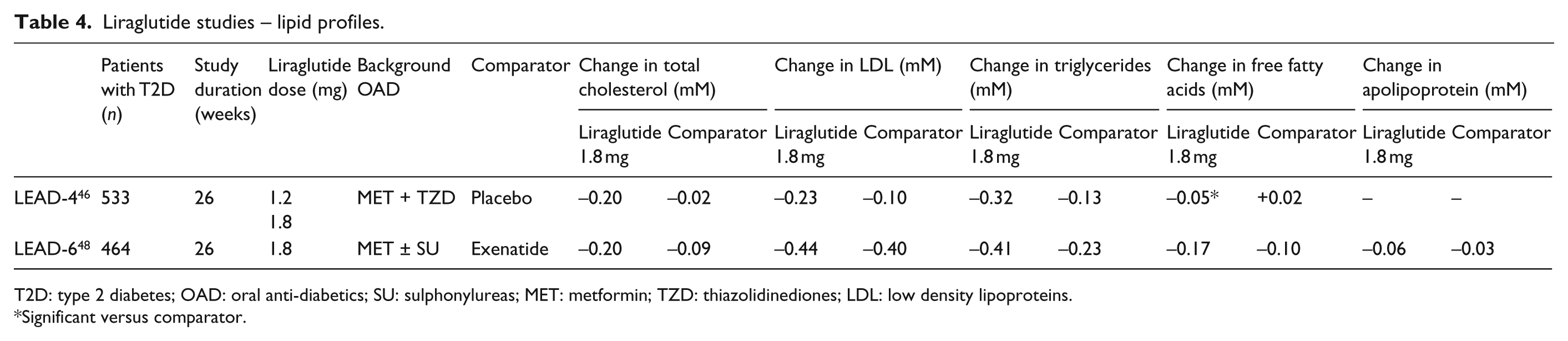
T2D: type 2 diabetes; OAD: oral anti-diabetics; SU: sulphonylureas; MET: metformin; TZD: thiazolidinediones; LDL: low density lipoproteins.
Significant versus comparator.
The long-term effect of ExBID on CV risk factors was studied in an open-ended, open-labelled clinical trial enrolling patients from three placebo-controlled ExBID trials and their open-labelled extensions and comparing ExBID with placebo on background metformin and/or SU for ≥ 3 years. 35 Results of this study showed that TG decreased 12% (p = 0.0003), total cholesterol decreased 5% (p = 0.0007), LDL cholesterol decreased 6% (p < 0.0001) and HDL cholesterol increased 24% (p < 0.0001) in patients treated with ExBID versus placebo.
ExQW was also associated with favourable changes in serum lipids. ExQW reduced mean TC from baseline values by −0.12 to −0.90 mmol/L in three of the four DURATION trials (Table 5).38,40,41 Compared with ExBID, the improvement in TC with ExQW was significantly greater.38,41 The only trial that reported no change in TC for ExQW was DURATION-2, where pioglitazone was associated with a significant increase in TC (Table 5). 39 ExQW significantly reduced LDL cholesterol in two trials,38,41 while no other comparator was associated with any change in LDL (Table 5). ExQW had no significant effect on HDL cholesterol in any of these three DURATION studies.
DURATION studies – lipid profiles.
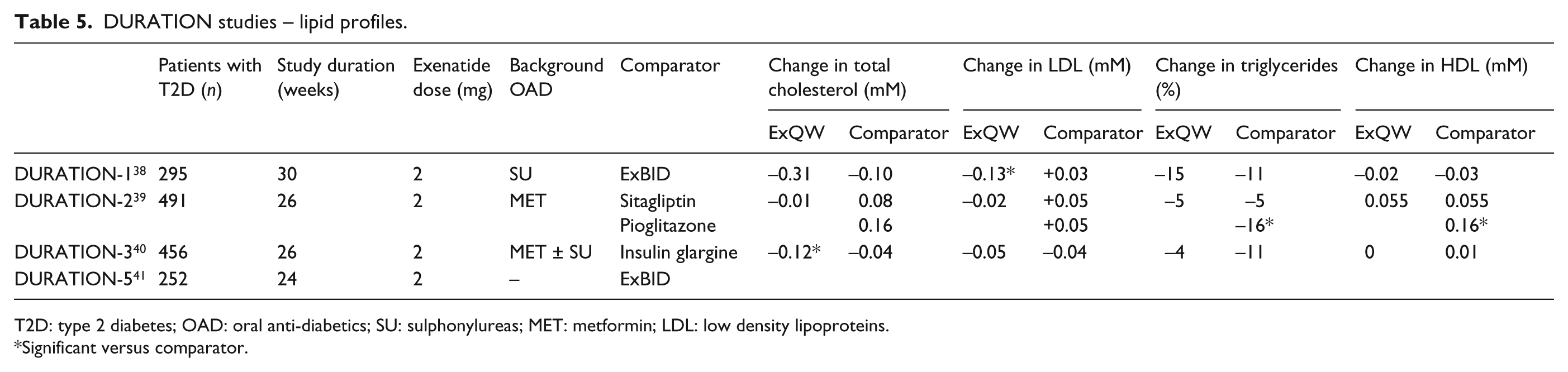
T2D: type 2 diabetes; OAD: oral anti-diabetics; SU: sulphonylureas; MET: metformin; LDL: low density lipoproteins.
Significant versus comparator.
Cardiovascular events
Arrhythmias
Assessment of the QT interval is an important CV safety outcome to consider for drugs under clinical development. The QT interval is defined as the time it takes from the start of ventricular depolarisation (i.e. the start of the Q or R wave on the surface electrocardiogram (ECG)) to the end of ventricular repolarisation (i.e. the end of the T wave on the ECG). QT prolongation can predispose to malignant ventricular arrhythmias (e.g. torsade des pointes), which are often drug-induced. As such, thorough QT studies are used to assess the potential for developing such arrhythmias after long-term exposure to new therapeutics.
A study specifically examining the effects of exenatide on QT interval has appeared recently. 76 This randomised, placebo-controlled study of 70 healthy subjects included three cross over treatment periods with exenatide (10 µg) and placebo. QT intervals corrected for HR (QTcF) and individually (QTcI) were analysed for change between pre- and post-treatment values. Based on both QTc assessments, exenatide did not show a clinically significant prolongation in QT as compared with placebo. 76
An earlier study on the effects of liraglutide on QT intervals reached the same conclusions for this GLP-1R agonist. 77 This placebo-controlled, double-blind cross over study with 51 healthy participants assessed the effects of liraglutide 0.6, 1.2, and 1.8 mg doses versus placebo once daily for seven days each. Four correction methods were used to assess the change in QTc interval from baseline. The study concluded that liraglutide did not cause any significant increase in QTc interval. 77
Heart failure
In an abstract presented at EASD 2011, Best et al. described a retrospective cohort study in which 778,408 patients with T2D prescribed ExBID, insulin (IN) and/or other treatments (OTH) from January 2005 to September 2010 were assessed for the incidence of heart failure, defined as diagnosis of ICD-9 code 428 or brain natriuretic peptide > 100 pg/ml. 78 Patients on exenatide were randomly matched 1:1 with patients on other treatments. Without adjustment for the Charlson Comorbidity Index (CCI), a disease severity measure, the rate of heart failure for patients on ExBID + IN + OTH was lower than for those on IN + OTH (odds ratio, OR = 0.41; 95% confidence interval, CI = 0.33–0.50). After CCI adjustment, the rate of heart failure remained lower in patients receiving ExBID (OR = 0.49; 95% CI = 0.46–0.52).
Myocardial infarction
In a large, retrospective study of 39,275 patients with T2D on exenatide and 381,218 patients on other glucose-lowering treatments, those on exenatide were found to be more likely to have prior ischaemic heart disease, obesity, hyperlipidaemia, hypertension and other comorbidities at baseline. 79 Despite these factors, patients on exenatide were found to be less likely to have a CVD event than those not treated with exenatide (hazard ratio 0.81; 95% CI = 0.68–0.95; p = 0.01) as well as lower rates of CVD-related hospitalisation (0.88; 0.71–0.98; p = 0.02) and all-cause hospitalisation (0.94; 0.91–0.97; p < 0.001). 79
The preclinical observation that GLP-1, its analogues and perhaps even its metabolites exert cardioprotective effects in the setting of myocardial ischaemia80–82 has recently been tested in a clinical trial. 83 In this study, 172 patients with ST elevation MI were randomized to receive intravenous exenatide or placebo 15 min prior to and 6 h following revascularization with primary angioplasty. Infarct size in relation to myocardial area at risk, as well as absolute infarct size, were decreased in patients receiving exenatide. In this relatively small study, no changes in LV function or 30-day clinical event rates were observed as a consequence of treatment assignment. 83
Death
The long-term cardiovascular mortality associated with GLP-1 receptor agonists has not been extensively evaluated due to the relatively recent availability of these drugs on the market. One study set in the United Kingdom evaluated the life-expectancy associated with exenatide versus insulin glargine, as add-on therapies in T2D patients inadequately controlled on combination OADs after 35 years of treatment. 84 Using a computer simulation model of diabetes (CORE model) and data from a published head-to-head 26-week trial of exenatide versus insulin glargine in 549 patients, it was demonstrated that exenatide was associated with both improvements in life expectancy and quality-adjusted life-expectancy. When compared with insulin glargine, life expectancy improved by 0.057 years with exenatide, while quality-adjusted life years improved by 0.442. The authors attributed the difference between life expectancy and quality-adjusted life expectancy to the impact of exenatide’s body-weight lowering effect on improving lifestyle. 84
The long-term cardiovascular morbidity and mortality effects of treatment with liraglutide were also simulated using the CORE diabetes cohort model, with data extracted from LEAD-1. 85 Simulating a hypothetical cohort of 5000 patients per treatment (liraglutide 1.2, 1.8 mg, rosiglitazone 4 mg, all added to glimepiride), this model estimated that the survival rates would be 15% and 16% higher for liraglutide 1.2, 1.8 mg respectively than rosiglitazone after 30 years of treatment. Projected rates of cardiovascular death would be 69.7%, 68.4% and 72.5% for liraglutide 1.2, 1.8 mg and rosiglitazone respectively. 85
Major adverse cardiovascular events
In a pooled analysis of 15 Phase II–III clinical trials of liraglutide, including a total of 6638 patients, of which 4257 were exposed to liraglutide, the rates of reported major adverse cardiovascular events (MACEs) comprising cardiovascular death, myocardial infarction and stroke, favoured liraglutide versus standard OAD comparators. 86 MACEs were assessed using Medical Dictionary for Regulatory Activities (MEDRA) search terms combined with serious adverse events reported by investigators. Of note, these trials represented relatively short periods of follow-up, with a low total number of adjudicated MACEs (n = 39). Thus while the incidence ratio of 0.73 revealed by this analysis favoured liraglutide over comparators (with the upper 95% confidence limit of 1.41), larger studies with longer durations of follow-up are needed to more fully establish the cardiovascular safety of this agent.
Similarly, in a meta-analysis of 12 studies of ExBID including only 395 T2D patients representing 1070 patient-years of exenatide exposure (with an average exposure of 24 weeks), no increase in CV risk was associated with ExBID use. The exposure adjusted incidence rate of MACEs for patients treated with exenatide was 18.73 per 1000 patient-years, and 23.17 per 1000 patient-years for the pooled-comparator group (placebo or insulin treated). 87 Again, while the hazard ratios (0.7) and relative risk reductions suggest that exenatide may improve cardiovascular outcomes, such small and short duration studies were not designed to test the longer term safety profiles of this class.
In 2008, the FDA issued recommendations that drugs being investigated for diabetes therapies should display an upper bound, two-sided, 95% CI of < 1.8 for MACE estimated risk ratios versus comparator. 88 If the upper bound is between 1.3 and 1.8, the cardiovascular safety of the study drug must be demonstrated in Phase IV cardiovascular outcomes trials. At present, the LEADER trial is currently underway, examining the long-term effect of liraglutide 1.8 mg on CV death, non-fatal MI and stroke as primary outcomes. 89 This multi-national, multi-centre, randomised, double-blind, placebo-controlled Phase III efficacy/safety study began recruitment in 2010 of 8754 patients with T2D. These subjects are being randomised to receive liraglutide or placebo in a 1:1 ratio, and will be followed for a period of five years to determine the cardiovascular safety of long-term treatment with liraglutide. 89 Similarly, the EXCELL study, which also began recruitment in 2010, will test the cardiovascular safety of ExQW by examining MACEs in a randomised, double-blind, placebo controlled study of 9500 patients with T2D over a follow-up period of 5.5 years. 90
With these large FDA-mandated studies not expected to report results until 2016 or beyond, earlier insights into the cardiovascular safety of GLP-1R agonists may depend on smaller mechanistic studies. For example, a small Phase II trial is planned to investigate the efficacy of liraglutide on heart function in T2D patients with co-morbid congestive heart failure. 91 With an estimated enrolment of 80 subjects, this randomised study will compare left ventricle function and functional reserve with tissue Doppler echocardiography in a longitudinal manner with 18 weeks of liraglutide+metformin vs. glimepiride+metformin.
GLP-1R agonist versus DPP4 inhibitor
When compared with another incretin-based OAD such as the DPP4 inhibitor sitagliptin (100 mg), the GLP-1R agonist liraglutide (1.8 mg) caused greater weight loss (−1.16 kg vs. −3.68 kg; p < 0.0001). 92 This head-to-head study, with both treatments adjunct to metformin, did not reveal any beneficial effects of liraglutide over sitagliptin in improving cardiovascular risk factors. Indeed, while liraglutide proved superior to sitagliptin in achieving meaningful reductions in HbA1c without concomitant hypoglycaemia, there was no significant difference between the drugs in terms of lipid profiles. Both liraglutide and sitagliptin lowered SBP slightly, but the magnitude of reduction for liraglutide was not comparable to that achieved in the LEAD1-6 studies. Rather, minor reductions in DBP were unexpectedly greater with sitagliptin than with liraglutide 1.8 mg (−1.78 vs. 0.07 mmHg; p = 0.0210), with HR increasing slightly with both liraglutide 1.2 and 1.8 mg, but not sitagliptin.
GLP-1R agonist versus GLP-1R agonist
Liraglutide and exenatide were evaluated head-to-head in an open-label, parallel-group study as part of the LEAD trials. 48 T2D patients inadequately controlled on metformin and/or sulphonylurea were randomised 1:1 to receive either liraglutide 1.8 mg once daily or exenatide 10 μg twice daily for 26 weeks as add-on to their existing OADs. The reduction in mean HbA1c (±SE) at week 26 was greater with liraglutide than exenatide (−1.12 ± 0.08 vs. −0.79 ± 0.08; p < 0.0001). Liraglutide was also more efficacious in achieving a targeted HbA1c of < 7% (53% vs. 43%; p = 0.0015). Meanwhile, both drugs were well-tolerated and caused similar weight reductions approximating 3 kg.
Taspoglutide
Taspoglutide is a GLP-1 receptor agonist that was developed to have a longer duration of action than liraglutide, to minimise the number of injections necessary and increase patient compliance. This recombinant GLP-1 analogue (with α-aminoisobutyric acid substitutions at position 8 in the N-terminus and position 35 in the C-terminus) binds the GLP-1 receptor with the same properties as native GLP-1, and has demonstrated sustained plasma levels lasting for at least 14 days, allowing once-weekly dosing.93,94 Early Phase II studies examining the once-weekly efficacy of this agent in T2D patients inadequately controlled on metformin revealed improved glycaemic control and reduced body weight.95,96 In Phase II safety studies, high doses of once-weekly taspoglutide (20–40 mg) were generally well tolerated, with GI complaints reported as the most common adverse events. The multi-national, multi-centre Phase III series of eight clinical trials designated as T-emerge 1–8 evaluated the efficacy and safety of taspoglutide versus placebo or active comparators including exenatide, sitagliptin, insulin glargine or pioglitazone in over 6000 patients. At the 2010 ADA meeting in Orlando, results from some of the T-emerge trials were presented, demonstrating the efficacy of taspoglutide in achieving glycaemic control, with accompanying reductions in the risk of hypoglycaemia and decreased body weight. However, in June 2010, a press release from the manufacturer Roche announced an uncommon (<1%) but higher than expected incidence of hypersensitivity related to taspoglutide. 97 This hypersensitivity manifested most commonly as skin reactions and gastrointestinal symptoms and a potential association with anti-drug antibodies was identified. In September 2010, the taspoglutide clinical trial programme was suspended for further analysis of the immunogenic safety of this compound.
Lixisenatide
Lixisenatide is another GLP-1 receptor agonist currently under evaluation for the treatment of T2D. 98 This compound shares structural similarity with exendin-4, with modifications at the C terminus including addition of six Lys residues, and the deletion of one Pro residue. Lixisenatide binds the GLP-1 receptor with four times higher affinity than native human GLP-1.98,99 In early Phase II trials, lixisenatide, compared with placebo, was well tolerated and improved glycaemic control in T2D patients inadequately controlled on metformin. 99 Phase III trials evaluating lixisenatide versus active comparators or as add-on to standard OADs has been completed and results are expected shortly. Currently underway is the Phase III ELIXA trial, a placebo-controlled study assessing primary cardiovascular endpoint of composite CV death, non-fatal MI, non-fatal stroke and hospitalisation for unstable angina. This study is expected to enrol 6000 T2D patients with a recent acute coronary syndrome event, with an estimated completion by May 2014. 100
Conclusion
Here we have reviewed the published literature, including recent meetings abstracts, to provide a predominantly clinical perspective on the pharmacokinetics, anti-hyperglycaemic efficacy and cardiovascular risk factor and outcomes profiles of exenatide and liraglutide, the two widely available GLP-1R agonists currently in use for the treatment of T2D. These powerful and well tolerated injectable agents effectively lower hyperglycaemia in patients with T2D, without increasing the incidence of hypoglycaemia. They have additional salutary effects such as reductions in body weight and SBP, while generally improving lipid profiles and other biomarkers of cardiovascular disease. Although they likely achieve these additional effects through a combination of mechanisms that include appetite suppression, reduced body fat, natriuresis, diuresis and improved endothelial function, further studies are needed to better understand their clinical significance. Similarly, the clinical significance and physiological mechanisms underlying the small but statistically significant increases in mean resting HR produced by both GLP-1R agonists remain unknown. Having said this, QT intervals are not prolonged by these drugs, and early meta-analyses of the short-term clinical trials conducted to date and retrospective studies of large registry data suggest that the cardiovascular outcomes of diabetic treated with these agents are better than those not treated with them. Finally, promising validation of pre-clinical studies that showed cardioprotective and vasodilatory effects of these drugs is beginning to emerge in clinical studies. Already underway, the large randomised, double-blind, placebo-controlled studies on the long-term cardiovascular safety of these GLP-1R agonists will not be reported on for another five years.
Footnotes
Funding
This work was supported by the Heart & Stroke Foundation of Ontario (HSFO; grant numbers T6757 and CI6824).
MH is also recipient of investigator-initiated research support from Amylin, Merck & Co, and NovoNordisk. However, neither data nor concepts from these industry-supported projects are discussed in this review.
Conflicts of interest
MH has received consulting fees from Merck & Co, NovoNordisk and Roche in regards to their incretin-targeted therapeutics. The other authors have no disclosures.
