Abstract
Type 2 Diabetes continues to rise in prevalence throughout the globe, and cardiovascular diseases remain the most common cause of morbidity and mortality among patients. Dipeptidyl-peptidase-4 (DPP-4) inhibitors are a newer class of oral anti-hyperglycemic agents whose effect is mediated through the incretin hormones, GLP-1 and GIP. In this review, we discuss the incretin system, DPP-4 inhibitors and their mechanism of action and, principally, the potential impact of DPP-4 inhibition on the cardiovascular system. Some pre-clinical data, small mechanistic studies and post-hoc analyses of randomized clinical trials suggest a possible beneficial effect on cardiovascular risk. However, the relationship between DPP-4 inhibition and actual cardiovascular outcomes remains unknown. We therefore also review ongoing large, randomized clinical trials examining this very question.
Introduction
The global prevalence of type 2 diabetes mellitus (T2DM) continues to increase at an alarming rate, with cardiovascular disease being the most common cause of mortality and morbidity in affected patients. 1 However, the cardiovascular benefits of correcting the primary and defining abnormality of this disease – namely hyperglycaemia – remain in question.2–4 Accordingly, and despite the nearly one dozen anti-hyperglycaemic drug classes now available to manage T2DM, there remains considerable interest in the development of medicines that might offer cardiovascular advantages in addition to effects on glucose. Indeed, the cardiovascular safety of several currently available anti-diabetic agents has recently been called into question. As a result, from a drug development standpoint, there is a need to ensure that any new medication for T2DM is, at the very least, associated with no deleterious effect on cardiovascular outcomes. Regulatory agencies actually now mandate that all new glucose-lowering medications undergo thorough cardiovascular safety assessment before marketing approval. In this review, we will discuss a newer class of incretin-based therapy, the DPP-4 (dipeptidyl peptidase-4) inhibitors – their mechanism of action, expected results on glycaemic parameters and potential impact on the cardiovascular system.
The incretin system: An overview
Early studies showed that oral carbohydrate consumption was associated with greater pancreatic insulin secretion when compared with an equivalent amount of glucose administered intravenously. 5 This phenomenon was subsequently coined the ‘incretin effect’ and has been estimated to account for up to 50–70% of mealtime insulin output. It was later ascribed to hormones produced in the gastrointestinal tract, which have several systemic effects, including glucose-dependent stimulation of insulin secretion by pancreatic beta-cells. 6 Thus far, two incretin hormones have been identified: glucagon-like peptide-1 (GLP-1), derived from the L-cells of the distal small intestine and large bowel, and glucose-dependent insulinotropic polypeptide (GIP), derived from the K-cells of the proximal small intestine. Additional effects of GLP-1 include suppression of post-prandial glucagon secretion from pancreatic alpha-cells (also glucose-dependent), slowing of gastric emptying through effects on the vagus nerve, and enhancement of satiety at a hypothalamic level leading to reduced food intake. G-protein coupled receptors for GLP-1 are present in several tissues beyond the pancreas, gastrointestinal tract and nervous system, including cardiac myocytes, although their physiological purpose(s) at these other sites remains largely unknown.
The enzyme dipeptidyl peptidase-4 (DPP-4) rapidly degrades both GLP-1 and GIP to their inactive metabolites. Competitive inhibition of DPP-4 increases the half-life and bioavailability of active incretin hormones, enhancing their biological effect (Figure 1). Presently, the available incretin-based therapies for diabetes mellitus include the GLP-1 receptor agonists (which are resistant to DPP-4 degradation) and DPP-4 inhibitors; the mechanisms of action of both involve the potentiation of the endogenous effects of GLP-1. DPP-4 inhibitors, in contrast to GLP-1 receptor agonists, have not been demonstrated to slow gastric emptying, however. DPP-4 inhibitors also increase GIP activity. The other differences between these two drug classes lie in their mode of administration. GLP-1 receptor agonists are injectable peptides whereas the DPP-4 inhibitors are oral agents. In addition, the former, which result in pharmacologically elevated GLP-1 activity, are modestly more efficacious in reducing glucose, typically result in weight loss and are associated with gastrointestinal side effects, mainly nausea. The latter are weight neutral and tend to be better tolerated.
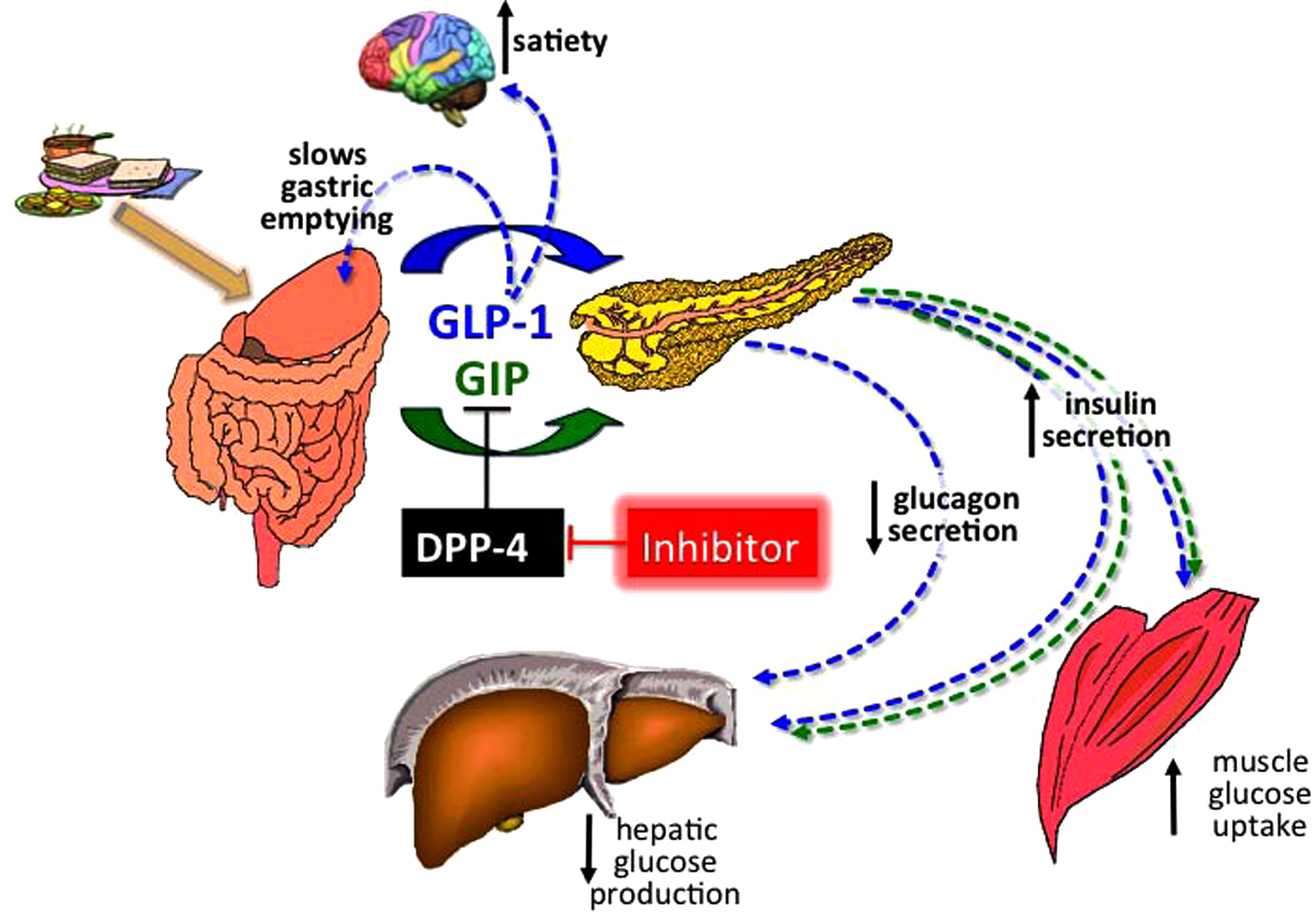
Physiology of the incretin system. After meal ingestion, as blood glucose levels rise, neuroendocrine cells of the intestine secrete the incretin hormones GLP-1 and GIP. Both stimulate insulin secretion by pancreatic beta cells, in a glucose dependent fashion. GLP-1 additionally decreases glucagon secretion from pancreatic alpha-cells, slows gastric emptying and increases satiety, allowing for reduced food intake. Physiologically, the enzyme dipeptidyl peptidase(DPP)-4 rapidly degrades both GLP-1 and GIP to their inactive metabolites. Pharmacological competitive inhibition of DPP-4 increases the half-life and bioavailability of active incretin hormones, enhancing their physiological effect.
DPP-4 inhibitors: Mechanisms and clinical effects
DPP-4 is a 766 amino acid transmembrane glycoprotein, also known as adenosine deaminase complexing protein 2 or CD26. DPP-4 inhibitors are cyanopyrrolidines with key interactions with the DPP-4 complex that allow for competitive inhibition. DPP-4 is expressed on the surface of several cell types, including monocytes and lymphocytes.7,8 It is a serine aminopeptidase enzyme which inactivates GLP-1, GIP and other proteins in vivo via dipeptide cleavage of the N-terminal amino acid. Protein dimerisation is needed for catalytic activity, and physiological function of the enzyme depends on its glycosylation. Importantly, DPP-4 substrates are extensive and include several proline or alanine containing peptides, such as growth factors, chemokines, neuropeptides and vasoactive peptides.6,7 Inhibition of the DPP-4 enzyme also modulates the activity of several cardioactive factors, neuropeptide Y and stromal cell derived factor-1 (SDF-1). 6 Because of its affinity for a wide range of substrates, DPP-4 is not specific for GLP-1 and therefore has the potential to mediate a wide range of pleiotropic effects (both positive and negative), independent of GLP-1. Other examples of enzymes with DPP-4-like activity include fibroblast activation protein, DPP-2, DPP-8 and DPP-9; thus, DPP-4 inhibitors thought to be selective for DPP-4 could in fact be inhibitors of other enzymes similar to DPP-4. 9
The currently available DPP-4 inhibitors include sitagliptin, saxagliptin, linagliptin, vildagliptin and alogliptin. The first three are approved in the USA and throughout much of the world for the treatment of T2DM; vildagliptin has been approved for use in Europe and Latin America and alogliptin for use in Japan. Other members of this class are in phase III clinical trials, and include dutogliptin and gemigliptin.
The various DPP-4 inhibitors are unique in their metabolism, excretion, potency, and, as a result, their recommended daily dosages. Saxagliptin, sitagliptin and vildagliptin are hepatically metabolised and renally eliminated. Linagliptin is primarily excreted via non-renal pathways with minimal hepatic metabolism; therefore dose adjustments are not needed in the setting of renal or liver disease. With daily doses ranging from 100 mg for sitagliptin to 5 mg for saxagliptin and linagliptin, the drugs are all similar in their efficacy in lowering HbA1c levels, safety profiles and patient tolerance. 10 DPP-4 inhibitors result in a mean decrease in A1c ranging between 0.5% and 1%, 11 depending to some degree on the baseline degree of hyperglycaemia. In clinical trials, the most commonly reported adverse effects associated with DPP-4 inhibitor therapy included nasopharyngitis, upper respiratory tract infection, urinary tract infection and headache. Urticarial dermatological reactions and angioedema have also been reported. Rarely, pancreatitis may occur, although it remains unclear whether this is actually an adverse effect of the medications themselves, since this condition is seen with increased frequency in patients with T2DM. Generally speaking, the DPP-4 inhibitors are well tolerated and in contrast to certain other anti-diabetic agents, not associated with hypoglycaemia, weight gain, gastrointestinal symptoms or peripheral oedema.
Currently, the drugs are predominately used as second- or third-line agents, typically after metformin or the combination of metformin-sulfonylurea or metformin-thiazolidinedione. Fixed-dose combination tablets with metformin are now available for several of the aforementioned compounds. They are also approved for use as monotherapy, mainly used in this setting in those who cannot tolerate or have active contraindications to metformin (e.g. chronic kidney disease). DPP-4 inhibitors may also have a unique role in elderly patients who face several precautions regarding the use of other anti-hyperglycaemic drugs due to their potential toxicities. Cost remains a major impediment for more widespread use of these agents, given their availability only as branded products.
Cardiovascular effects of GLP-1
There is a growing body of evidence to suggest that GLP-1 – and as an extension of this, GLP-1 based therapy – may have favourable cardiovascular effects. For example, GLP-1 has been found to be cardioprotective in experimental models of heart failure and myocardial infarction (MI). 12 In a clinical study led by Nikolaidis, the investigators compared in-hospital outcomes of 22 high-risk patients with recent acute MI, left ventricular (LV) dysfunction and successful reperfusion. 13 Ten patients in the intervention arm were infused with 72 hours of recombinant GLP-1 added to standard therapy with the control arm treated with standard therapy alone. GLP-1 infusion was associated with an improved LV ejection fraction (from 29 ± 2% at baseline to 39 ± 2% post GLP-1 infusion) and improved contractility, measured 6–12 hours post-infusion. The paucity of published follow-up studies over the past seven years in this area, however, suggests that any initial conclusions regarding the benefits of GLP-1 on the cardiovascular system may have been over optimistic. Several studies have also suggested that GLP-1 agonists ameliorate cardiovascular risk factors, such as blood pressure and lipid profiles.14–16 We would caution, however, that these effects have been proposed to be secondary to the weight loss seen with GLP-1 agonists, an effect not observed with DPP-4 inhibition.
If GLP-1 is beneficial to the heart, the question naturally arises whether pharmacological DPP-4 inhibition might provide any cardiovascular protection through an increase in GLP-1 bioavailability and signalling. However, pre- clinical studies tended to use supraphysiological doses of GLP-1 to explore for vascular effects; DPP-4 inhibition would only increase GLP-1 levels to a mildly supraphysiological range, which may not have a similar cardiovascular impact if one indeed exists. However, it is also tenable that DPP-4 inhibition may have cardiovascular effects independent of GLP-1.
Cardiovascular effects of DPP-4 inhibition
Read et al. tested whether increasing the plasma concentrations of GLP-1 by DPP-4 inhibition could protect the heart from ischaemic left ventricular dysfunction during dobutamine stress echocardiography (DSE) in patients with ischaemic heart disease.
17
Fourteen patients with known coronary artery disease and normal LV function awaiting revascularisation were studied; all had underdone recent coronary angiography. Exclusion criteria included abnormal LV regional wall motion at rest, MI within the previous three months, conduction abnormalities, valvular heart disease, and current insulin therapy. At baseline, there were no differences in the plasma concentrations of GLP-1, glucose, insulin or in LV wall function. Fourteen patients were randomly assigned to either 100 mg of sitagliptin or placebo. DSE was conducted with tissue Doppler imaging at rest, peak and 30 minutes. Seventy-five grams of oral glucose was administered to all participants before DSE to promote GLP-1 secretion, and plasma levels of glucose, insulin, fatty acids and GLP-1 were measured sequentially. The LV response to stress was observed by measuring the ejection fraction and mitral annular velocity, a surrogate marker for systolic function. After treatment with sitagliptin, both parameters improved (ejection fraction 72.6 ± 7.2%
At peak dobutamine stress, the plasma glucose concentration was lower, and the plasma GLP-1 was higher in the sitagliptin arm (mean difference, 6.8 ± 7.3 pg/ml,
In addition to potential cardiac benefits possibly mediated through GLP-1, DPP-4 inhibitors may also possess direct effects on the heart and on cardiovascular risk factors, independent of the incretin system. One such mechanism may involve improved signalling through the bone marrow chemokine known as stromal cell-derived factor (SDF)-1 alpha, also referred to as pre-B cell growth stimulating factor (PBSF). Endothelial progenitor cells (EPCs), derived from the bone marrow, are known to promote vascular repair and neoangiogenesis. When vascular damage occurs, local growth factors and cytokines are elaborated, which ultimately signal the bone marrow to release EPCs targeted to the injured sites. EPCs differentiate into mature endothelial cells and assist in the reconstruction of the vasculature. 7 Importantly, one of the regulators of EPCs is SDF-1 alpha, which actually stimulates their mobilisation. 18 Since SDF-1 alpha is a known substrate for DPP-4, DPP-4 inhibition will increase SDF-1 alpha concentrations, potentially enhancing the delivery of EPCs to injured endovascular sites.
The effect of DPP-4 inhibition on this chemokine was addressed by Fadini et al., who conducted a controlled, but non-randomised clinical trial comparing four weeks of sitagliptin versus no additional treatment added to baseline metformin and/or sulfonylurea therapy in 32 patients with T2DM.
19
Circulating concentrations of SDF-1 alpha and EPC counts were measured. There were no differences in baseline characteristics between the sitagliptin and control groups. After four weeks, as compared with controls, patients who received sitagliptin experienced a two-fold increase in EPCs (
Aligned with this concept, another proposed mechanism by which DPP-4 inhibitors might proffer cardiovascular benefits is through a reduction in inflammation, now recognised as an important contributor to the atherosclerotic process. As aforementioned, an effect of sitagliptin in reducing MCP-1 has been demonstrated. Limited animal studies have also suggested that DPP-4 inhibition may reduce the number of inflammatory cells within visceral adipose tissue and halt the progression of atherosclerosis via immunomodulation.20–22
DPP-4 inhibition and effects on cardiovascular risk factors
Several other clinical studies have studied the potential effect of DPP-4 inhibitors on cardiovascular risk factors, such as hypertension and hyperlipidaemia. Here, there are insufficient data to determine whether any possible effects are direct ones or mediated through the modulation of incretin hormone physiology. A handful of in vitro studies have also demonstrated that DPP-4 is expressed on endothelial cells. Most recently, Shah et al. have shown that inhibition of DPP-4 within the microcirculation
23
relaxes vascular tone through the direct mediation of the nitric oxide system.
24
The investigators proposed that the effect of this drug class on vascular relaxation might promote better control of blood pressure, an important cardiovascular risk factor. Yet, in most DPP-4 trials in humans, no consistent effect on blood pressure, when measured routinely, has been demonstrated. In a single small study (
In general, no notable effects of the DPP-4 inhibitors have been demonstrated on circulating lipid concentrations in human clinical trials. Boschman et al., however, have suggested that vildagliptin therapy may augment post-prandial lipid mobilisation and oxidation. 27 Matikainen et al. further assessed the effects of vildagliptin on postprandial lipid and lipoprotein metabolism in patients with T2DM, wherein treatment with the DPP-4 inhibitor improved triglyceride and apolipoprotein B-48 particle metabolism after a fat-rich meal. 28 Clearly, more studies will need to be done to elucidate the balance of effects of DPP-4 inhibition on cardiovascular risk factors.
DPP-4 inhibitors and cardiovascular outcomes
Studies evaluating the effects of DPP-4 inhibitors on actual cardiovascular events are even more limited, and their findings should be considered highly preliminary. Williams-Herman et al. conducted a pooled analysis of 19 double-blinded clinical studies, (
Frederich et al. sought to assess the relative risk of cardiovascular events across eight randomised double-blinded phase II and III trials evaluating saxagliptin in patients with T2DM.
30
Data from a total of 4607 randomised patients were pooled (
A recent abstract by Johansen et al. presented at the 2011 Scientific Sessions of the American Diabetes Association (ADA) examined the relationship between linagliptin and cardiovascular outcomes. 31 Eight phase III studies with treatment duration of at least 12 months were pooled, with a total of 5239 treated patients. There were 3319 linagliptin patients and 1920 patients treated with placebo, glimepiride or voglibose (an alpha-glucosidase inhibitor). A blinded committee adjudicated all events. The primary endpoint of the study was to calculate the exposure-adjusted incidence rate of adverse cardiovascular events, defined as cardiovascular death, non-fatal MI, non-fatal stroke, or hospitalisation for unstable angina. The incidence rate per 1000 patient-years for the primary endpoint in the linagliptin arm was 5.3 versus 16.8 with comparators (hazard ratio, HR, 0.34 [0.15–0.70]. 31
White et al. also presented data at the 2011 ADA Scientific Sessions, examining the occurrence of MACEs in all phase II and III controlled clinical trials of alogliptin in T2DM. 32 The studies included one 12-week and seven 26-week studies. Cardiovascular end points included adjudicated cardiovascular death, non-fatal myocardial infarction and non-fatal stroke. Across the trials, MACEs occurred in 0.28% of patients in the alogliptin treatment group and 0.50% of patients receiving placebo. Overall, the risk of a cardiovascular event was numerically, but not statistically, lower in those patients receiving alogliptin (HR 0.63 [0.21–1.91]). When the analysis was restricted to those patients at high baseline cardiovascular risk, the HR was similar but with similarly wide confidence intervals, due to overall low number of actual events (HR 0.52, [0.16–1.67]). 32
Finally, a recent meta-analysis by Lamanna
33
and colleagues, presented at the 2011 Annual Meeting of the European Association of the Study of Diabetes (EASD), was conducted to assess cardiovascular safety of the DPP-4 inhibition as a treatment strategy.
33
Fifty-three randomised trials of T2DM patients treated with a variety of DPP-4 inhibitors versus placebo or other comparators, with duration of at least 24 weeks, were identified through publically available databases. A Mantel-Haenszel odds ratio with 95% CIs was calculated for major adverse cardiovascular events; those trials with no events, however, were excluded. There were a total of 20,312 and 13,560 patients in the DPP-4 inhibition and the comparator arms, respectively. DPP-4 inhibitors, compared with placebo or other treatment, were associated with a relatively robust 31% reduced risk of major cardiovascular events (OR 0.69 [95% CI 0.53–0.90],
The numbers of events in these trials, which were designed primarily to assess for antihyperglycaemic efficacy, are obviously small. Yet, the consistent finding of HRs < 1.0 is provocative and suggests the need for larger and longer-term trials.
Ongoing clinical trials
Indeed, several large-scale clinical trials of DPP-4 inhibitors designed specifically to assess cardiovascular effects are currently underway (Table 1). TECOS (
Ongoing clinical cardiovascular outcomes trials of DPP-4 inhibitors in type 2 diabetes.
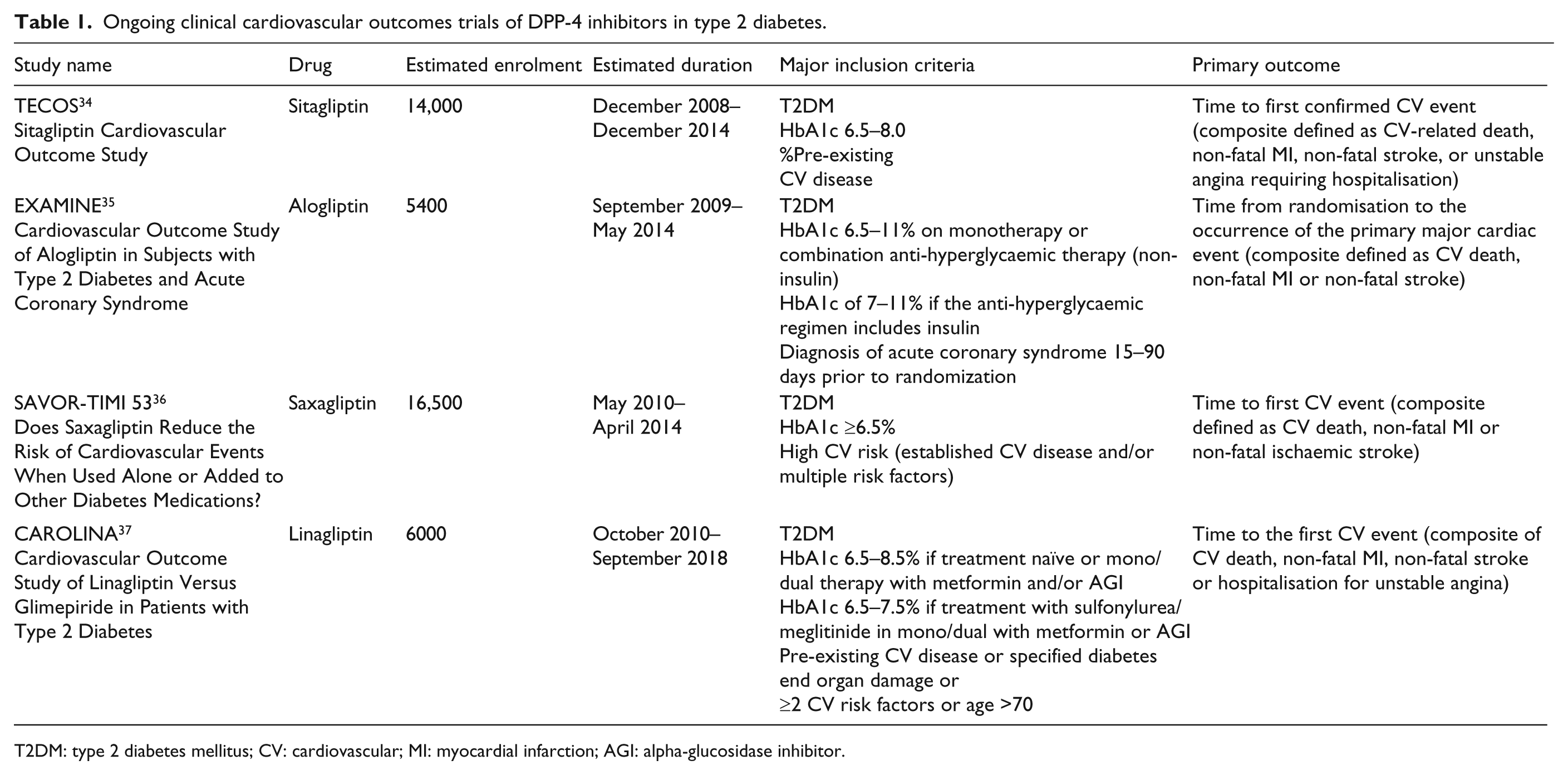
T2DM: type 2 diabetes mellitus; CV: cardiovascular; MI: myocardial infarction; AGI: alpha-glucosidase inhibitor.
Similarly, EXAMINE (
SAVOR-TIMI 53
Finally, in CAROLINA
Conclusion
DPP-4 inhibitors are a newer class of oral anti-hyperglycaemic agents with demonstrated efficacy in the treatment of T2DM. Some pre-clinical data, small mechanistic studies and post-hoc analyses of randomised clinical trials suggest a possible beneficial effect on cardiovascular risk. However, the actual relationship between DPP-4 inhibition and actual cardiovascular outcomes remains unknown. To address this uncertainty, and also to satisfy recent regulatory requirements to ensure cardiovascular safety of all new diabetes drugs, several large clinical trials involving several DPP-4 inhibitors and cardiovascular outcomes are now underway. Together, these trials will allow investigators to learn about the potential cardiovascular impact of these newer glucose-lowering drugs. In addition, their combined data will constitute a huge repository to inform us about their glycaemic efficacy, durability and safety.
