Abstract
Background:
Diabetes is a risk factor for perioperative complications after cardiac surgery. We studied its effects on mesenteric endothelial function in a cardiopulmonary bypass (CPB) model.
Methods:
Forty Wistar rats were divided into four groups: sham (D-CPB-), cardiopulmonary bypass (D-CPB+), diabetic (D+CPB-) and diabetic that have undergone CPB (D+CPB+). Two samples of mesenteric artery were used for nitric oxide synthase (NOS) Western blot analysis, and two others for assessing contractile response and endothelium relaxations. Nitrite products and tumour necrosis factor-alpha (TNF-α) were assessed as markers of inflammatory response.
Results:
We observed an enhanced contractile response to the α-adrenergic agonist associated with impairment of mesenteric vasorelaxation in D+CPB+ rats. Western immunoblot analysis of D+CPB+ highlighted an additive effect of hyper-expression of inducible NOS. A significantly increased inflammatory response was observed after CPB in diabetic animals.
Conclusions:
This work confirms the potential deleterious impact of diabetes on the mesenteric endothelium during CPB in cardiac surgery.
Keywords
Introduction
Cardiopulmonary bypass (CPB) is associated with a systemic inflammatory response, mainly caused by blood interactions with the artificial surface of the extracorporeal circuit.1–4 This inflammatory response may be critically important for perioperative outcome.5,6 We have previously discussed the fact that one of the major triggers of CPB-induced production of pro-inflammatory agents may be the selective alteration of endothelial function within the intestinal vasculature. 7 Impairment of gastrointestinal perfusion during CPB, induced by primary endothelial dysfunction, predisposes the intestine to hypoxic injury, which may initiate bacterial translocation and inflammatory mediator release, and promote multiple organ failure as a pathological consequence.
Diabetes is a well-known major risk factor in patients undergoing cardiac surgery.8–13 The diabetic population represents more than 38% of patients undergoing cardiac surgery procedures, especially coronary revascularisation. 14 It has been assumed that the greater surgical risk associated with diabetes mellitus might be a consequence of an altered inflammatory response to CPB, 15 which is characterised by increased secretion of cytokines (interleukin-6 [IL-6]) and tumour necrosis factor-alpha (TNF-α), oxidative stress16,17 and endothelial dysfunction,8,18 especially in the mesenteric vasculature.
All of these assumptions are related to the metabolism of nitric oxide (NO), which appears to play a major role in diabetic patients’ morbidity after cardiac surgery.11,17 It has been shown that NO production from inducible nitric oxide synthase (iNOS) and superoxide anion (O2−) levels are already increased in patients with diabetes under basal conditions.19–21 Similarly, CPB is associated with enhanced production of oxidative stress 22 and NO 11 from iNOS and endothelium-constitutive NOS (eNOS).1,23–25 The cumulative effects of diabetes and CPB on oxidative stress and NO metabolism, as well as their impact on mesenteric endothelial function, could partially explain the morbidity and mortality associated with diabetes in cardiac surgery.
In this context, the goal of this study was to evaluate the impact of diabetes on CPB-induced mesenteric endothelial dysfunction and on eNOS and iNOS protein expression, nitrite production and the inflammatory process in a rat model.
Materials and methods
Animals and groups
All animals received humane care in compliance with the Guide for the Care and Use of Laboratory Animals prepared by the National Institute of Health (NIH Publication no. 86-23, revised 1996).
Forty male Wistar rats (weight 400 g) were randomly divided according to two stratification criteria: diabetes and CPB. Four groups of animals (n = 10 for each group) were used: sham (D-CPB-), CPB only (D-CPB+), diabetes only (D+CPB-) and diabetic animals that have undergone CPB (D+CPB+). The two CPB groups underwent CPB for 90 min and were then monitored for 90 min after terminating CPB. In the non-CPB groups, the animals were cannulated and monitored for 180 min, but a CPB was not performed (Figure 1).
Schematic diagram of experimental procedure and blood-sampling points.
Induction of experimental diabetes
Diabetes was induced in rats by injecting 60 mg/kg of streptozotocine (STZ) (Sigma-Aldrich®) intraperitoneally. Age-matched non-diabetic groups received an equivalent injection of vehicle (0.1 M sodium citrate, pH 4.0). The animals were then housed in a temperature- and light-controlled environment and were allowed access to food and water ad libitum. STZ-treated rats were considered diabetic and retained for the experiments if their blood glucose levels, on day 7 after the initial injection, were higher than 17 mM. Blood glucose was measured using glucose test strips (OneTouch® Ultra®) read on a glucometer (OneTouch® Ultra®, LifeScan, Inc.). The experiments were performed eight weeks after the STZ injections.
Surgical procedure
Anaesthesia was introduced by intraperitoneal administration of ketamine (90 mg/kg) and chlorpromazine chlorhydrate (3.5 mg/kg). Reinjections were performed every hour. Rats were placed in the supine position. The lungs were ventilated with 100% oxygen through a 16-G tracheotomy tube at a tidal volume of 10 ml/kg and a respiratory rate of 60 breaths per minute. A 3-French catheter was placed into the left common carotid artery. Immediately after, 500 UI/kg of heparin was given. The various injections in addition to monitoring of heart rate (HR) and mean arterial pressure (MAP) were performed through the carotid-artery catheter. Arterial (22-G) and venous (16-G) cannulations were performed directly into the left femoral vessels. The CPB circuit was composed of a roller pump (Gambro®, Fresenius, France), a membrane oxygenator (Micro-1 Rat Oxygenator, Dongguan Kewei Medical Instrument Co., Ltd, Guangdong, China), 4 a venous reservoir (5 ml syringe, TERUMO®) and tubing lines (polyvinyl chloride, internal diameter 2.5 mm). All circuits were sterile and used only once. The CPB circuit was primed with 10 ml of 4% Gelofusine (B-Braun Medical Ltd, Boulogne, France). The perfusion flow rate was maintained at 100 ml/kg per min. Blood temperature and rat-body temperature were maintained with heating lights and controlled by monitoring rectal temperature. No blood components were transfused during the experiment. The remaining CPB solution was infused gradually after termination of CPB. Total aseptic conditions were maintained during the experiment.
Vascular mesenteric analysis
Just before the sacrifice, a laparotomy was performed and a section of mesenteric loop was removed and immediately immersed in cold oxygenated Krebs solution composed of (mM/L): NaCl (118.3), KCl (4.7), CaCl2 (2.5), NaHCO3 (25), MgSO4 (1.2), KH2PO4 (1.2), ethyl diamine tetetric acid (EDTA) (0.02) and glucose (11.1). Four third-order division segments of approximately 2 mm in length of mesenteric artery were dissected under a microscope. Two samples were immediately frozen in liquid nitrogen and stored at −80°C for later eNOS and iNOS Western blot analyses. The other two samples were prepared for assessing mesenteric vasoreactivity.
Myograph studies
Arterial segments of approximately 2 mm in length were mounted in a Mulvany myograph (J.P. Trading, Aarhus, Denmark) containing fresh oxygenated Krebs solution. The Krebs solution was continuously aerated with 95% O2 and 5% CO2. One section was connected to a tension transducer and the other was mounted on a displacement device operated with a micromanometer. After a 30-min stabilisation period, during which time the temperature was progressively increased to 37°C, the optimal internal diameter was set to a tension equivalent of 0.9 times the estimated diameter, at 100 mmHg effective transmural pressure, as described by Mulvany and Halpern. 26 After normalisation, the vessels were allowed to settle for 30 min. The vessels were then exposed to increasing concentrations of phenylephrine (Phe; 10−8 to 3.10−5 mol/L) with or without 15 min of incubation with the NO synthase inhibitor NG-nitro L-arginine (L-NA; 10−5 mol/L). Relaxation with acetylcholine (Ach; 10−8 to 3.10−5 mol/L) or the NO donor sodium nitroprusside (SNP; 10−8 to 3.10−5 mol/L) was assessed in arteries that were precontracted with 10−5 mol/L phenylephrine. Nitroprusside spontaneously forms nitric oxide in solution and, therefore, provides an estimate of the functional response of vascular smooth muscle to NO. Vascular samples were washed three times and allowed to settle for 30 min between each measurement. Concentration-response curves were expressed as the final molar concentration in the chamber. All drugs were obtained from Sigma (la Verpillère, France).
Western immunoblot analysis of iNOS and eNOS expression
Mesenteric arteries were homogenised in cold buffer containing sodium dodecyl sulphate (SDS) (1%), Tris– HCl (10 mmol/L, pH 7.4), aprotinin (5 µg/ml), leupeptin (2.5 µg/ml) and sodium orthovanadate (1 mmol/L). The amount of proteins loaded on the gel was verified by a Bradford assay and was between 35 and 40 µg in each case. The homogenised tissues were separated by SDS–polyacrylamide gel electrophoresis (Mini Gel Protean III System, Bio-Rad Laboratories, Hercules, CA, USA) and transferred onto Hybond ECL membranes (Amersham Biosciences) for 45 min at 100 V (Minitrans-blot Cell, Bio-Rad Laboratories). Membranes were incubated with the following primary antibodies: anti-eNOS (Transduction Laboratories™), anti-iNOS (Transduction Laboratories™) and anti-actin (Santa Cruz Biotechnology). Membranes were washed again and incubated with horseradish-peroxidase conjugated secondary antibody (Jackson Immunoresearch Laboratories, Inc., West Grove, PA). Proteins were then visualised with the Chemiluminescence kit (Lumi Light, Roche). The amount of iNOS, eNOS or actin protein was quantified by an image analyser (Vilber Lourmat® Bio-print 3000) using Bio-Capt and Bio-Profil software.
Measurements
Aliquots of 2 ml of arterial blood were sampled at the following two times: at cannulation (T0) and at the end of the experiment (T1) (Figure 1). Blood samples were immediately centrifuged at 3000 rpm for 5 min at 4°C. Plasma aliquots were stored at -80°C until various measurements were made.
Inflammatory response: TNF-α
Plasma levels of the pro-inflammatory cytokine TNF-α were measured as specific markers of the CPB-induced inflammatory response. TNF-α was assessed using Fluorokine® MultiAnalyte Profiling (F-MAP) kits from R&D Systems (Minneapolis, MN, USA) as previously described. 27 Kits were run on a BioPlex® dual laser Bioanalyzer (Luminex Corp., Austin, TX, USA) according to the manufacturer’s instructions. The minimal detectable dose of TNF-α was less than 3 pg/ml.
Measurement of nitrite products
Nitrite measurements as a marker of NO production were performed using tri-iodide-based chemiluminescence as previously described. 28 The NO signal was quantified using a nitric oxide analyser (NOA) (NOA™ 280, Sievers Instruments Inc., Boulder, CO, USA).
Statistical analysis
All results are expressed as mean ± standard errors of the mean (SEM). In all cases, n refers to the number of animals. All data were analysed, using SYSTAT software, by an analysis of variance (ANOVA) for multiple comparisons followed by the post hoc Tukey’s test. However, there was an exception for comparing samples collected at cannulation to those collected at the end of the experiment within each group for plasma nitrite levels and TNF-α, where a Student’s t test was used. Differences were considered to be statistically significant at p < 0.05.
Results
Characteristics of the animals
The blood glucose levels in the two diabetic groups of rats (25.2 mM ± 1.6; n = 20) were significantly increased (p < 10−4) compared with those in the non-diabetic groups of rats at each point of the experiment (8.1 mM ± 0.5; n = 20) eight weeks after the injection of STZ or vehicle. The weight of the diabetic animals significantly decreased from 388 g ± 3 g to 312 g ± 13 g (p < 10−4) eight weeks after the STZ injection compared with non-diabetic rats, which weighed 567 g ± 8 g after eight weeks.
There were no significant differences during the 180 minutes of the experiments in MAP between the four groups. The heart rate of diabetic animals was significantly slower compared with non-diabetic animals throughout the experimental period (p <10−4).
Myograph studies
The concentration-effect curve of phenylephrine was significantly enhanced in CPB-treated (D-CPB+) rats between 10−5 and 3.10−5 M compared with control rats (D-CPB-) (Figure 2(a)). In the presence of L-NA to inhibit NO synthase, there was no significant difference between the three groups D-CPB-, D-CPB+ and D+CPB- (Figure 2(b) and (d)).
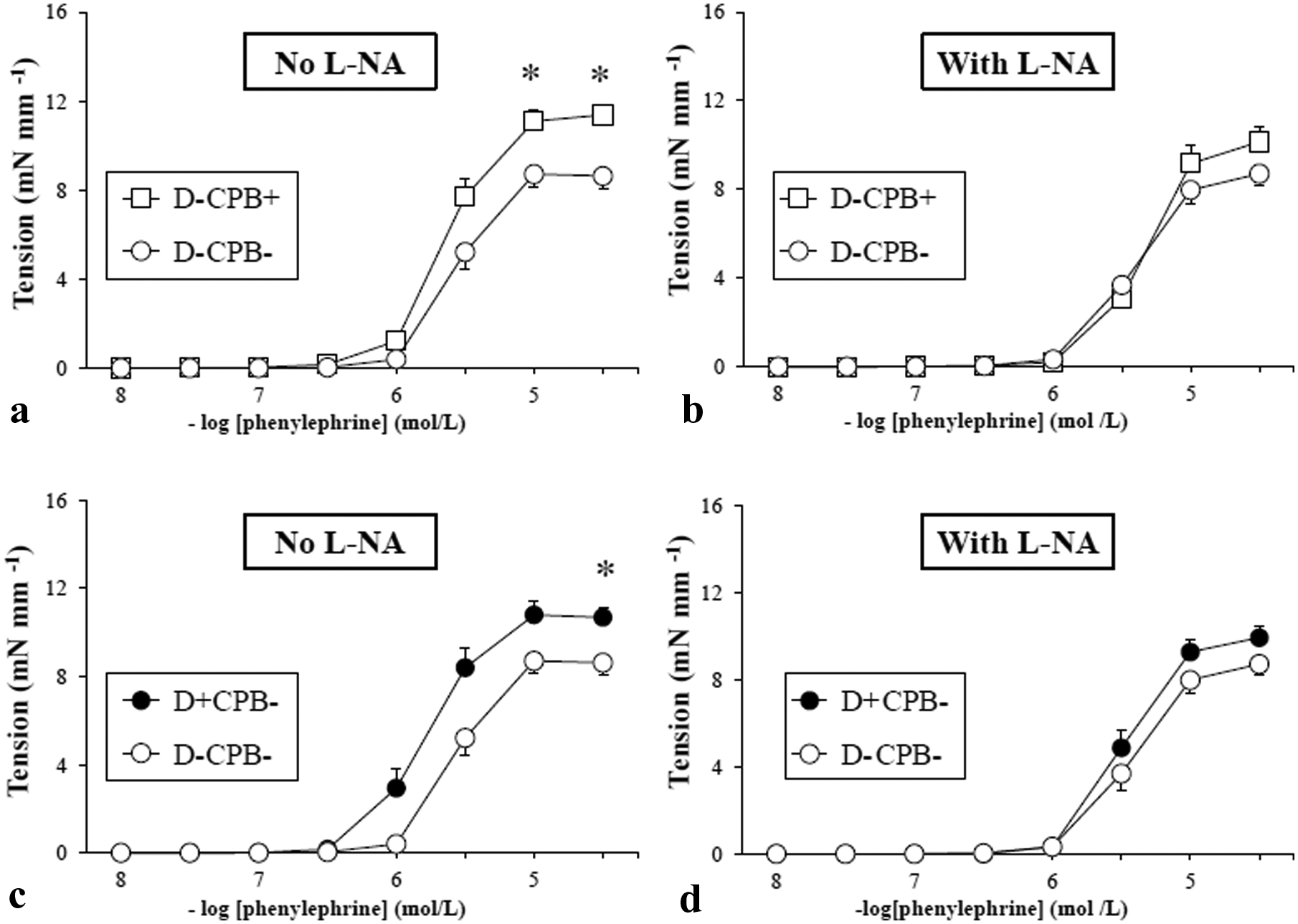
(a), (b), (c), (d), in vitro responses to phenylephrine with and without L-NA (mean ± SEM). *p < 0.05. White circles: D-CPB- group; white squares: D-CPB+ group; black circles: D+CPB- group; black squares: D+CPB+ group.
In rats subjected to CPB (Figure 3(a)), diabetic rats (D+CPB+) showed a higher contractile response of between 10−6 and 3.10−5 M to phenylephrine compared with non-diabetic rats (D-CPB+) (p < 10−4). In diabetic animals (Figure 3(c)), rats subjected to CPB (D+CPB+) showed a mesenteric vascular contractile response of between 10−6 and 3.10−5 M to phenylephrine concentration, which was significantly higher compared with animals that did not undergo CPB (D+CPB-) (p < 10−4).
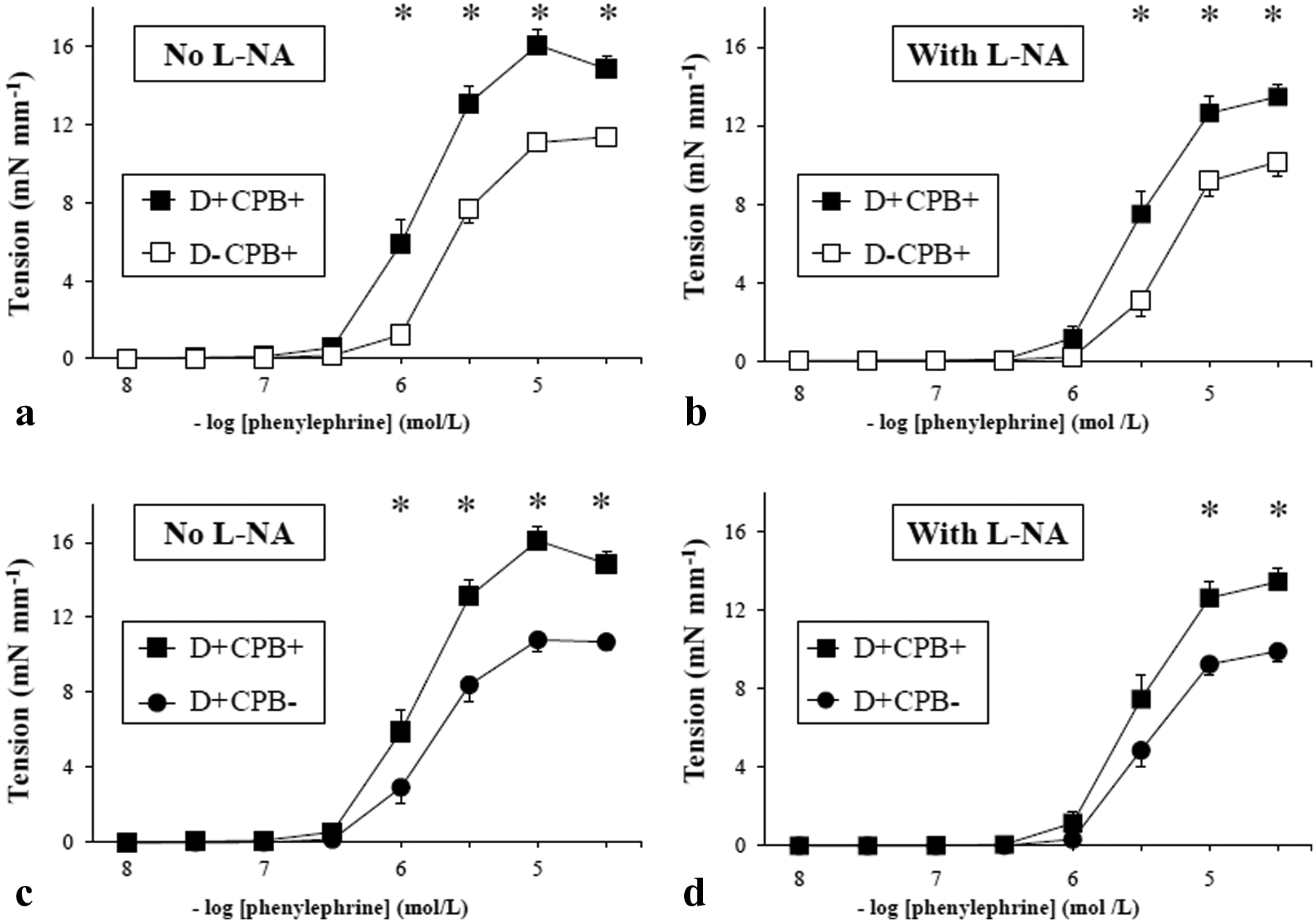
(a), (b), (c), (d), in vitro responses to phenylephrine with and without L-NA (mean ± SEM). *p < 0.05. White circles: D-CPB- group; white squares: D-CPB+ group; black circles: D+CPB- group; black squares: D+CPB+ group.
In the presence of L-NA, the contractile response to phenylephrine, of between 10−5 and 3.10−5 M, was significantly increased in the D+CPB+ group compared with the other groups (p < 10−4) (Figure 3(b) and (d)).
Compared with control rats (D-CPB-), the responses to acetylcholine were significantly altered in the CPB group (D-CPB+) at concentrations ranging from 10−7 to 3.10−7 M and in the diabetic group (D+CPB-) at concentrations ranging from 3.10−8 to 3.10−5 M (Figure 4(a) and (c)) (p < 0.05). The relaxation response to SNP was similar in the three groups D-CPB-, D-CPB+ and D+CPB- (Figure 4(b) and (d)).
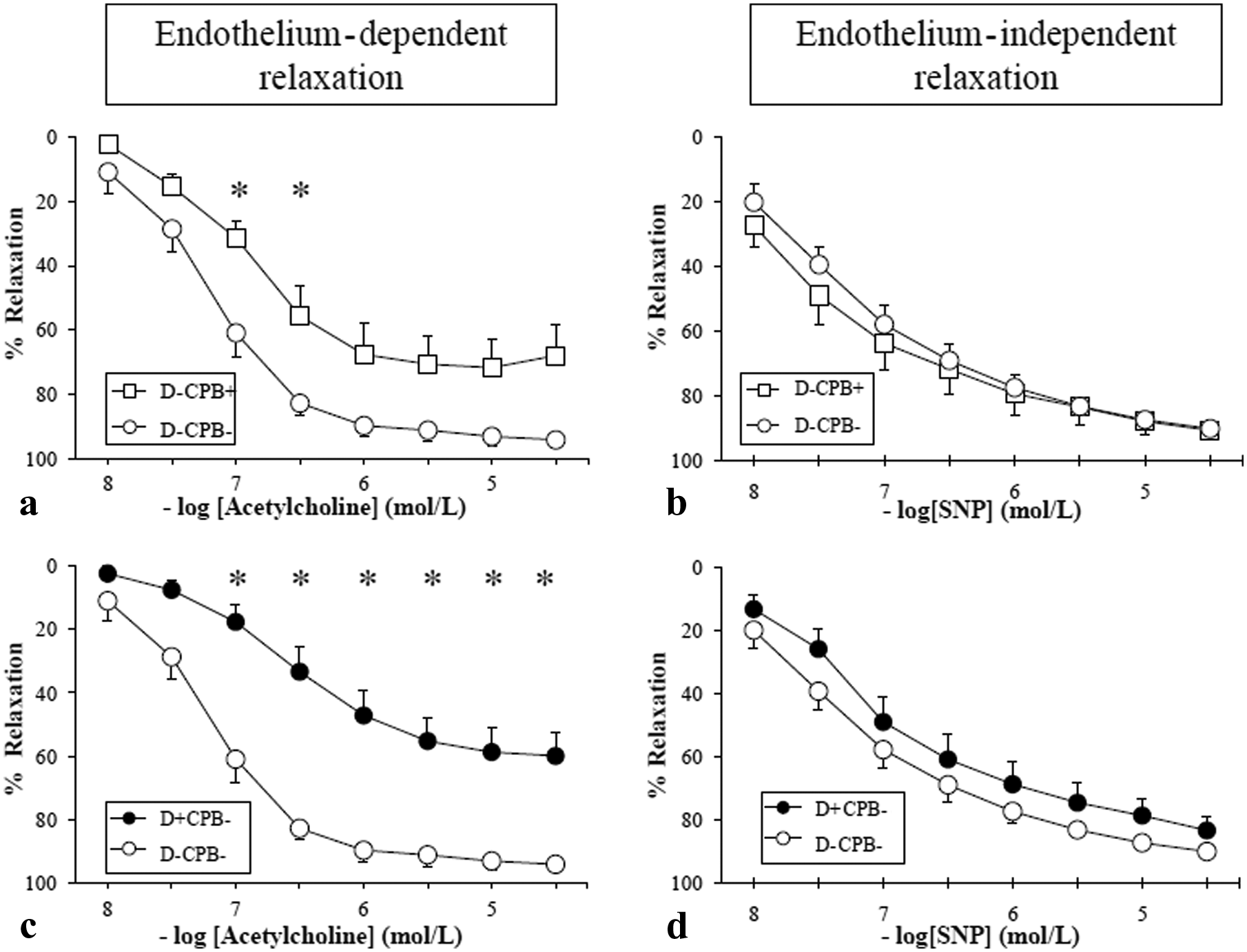
(a), (b), (c), (d), relaxation responses of the precontracted mesenteric artery to acetylcholine (endothelium-dependent relaxation) and sodium nitroprusside (SNP) (endothelium-independent relaxation). Responses are expressed as percentage relaxation of phenylephrine-induced precontraction (mean ± SEM). *p < 0.05. White circles: D-CPB- group; white squares: D-CPB+ group; black circles: D+CPB- group; black squares: D+CPB+ group.
In the CPB groups (Figure 5(a)), relaxation with acetylcholine was markedly impaired in diabetic rats (D+CPB+) at concentrations ranging from 10−7 to 3.10−5 M (p < 10−4). Regarding diabetic rats (Figure 5(c)), animals that underwent CPB (D+CPB+) showed no significant impairment of relaxation to acetylcholine compared with CPB-free diabetic animals (D+CPB-).
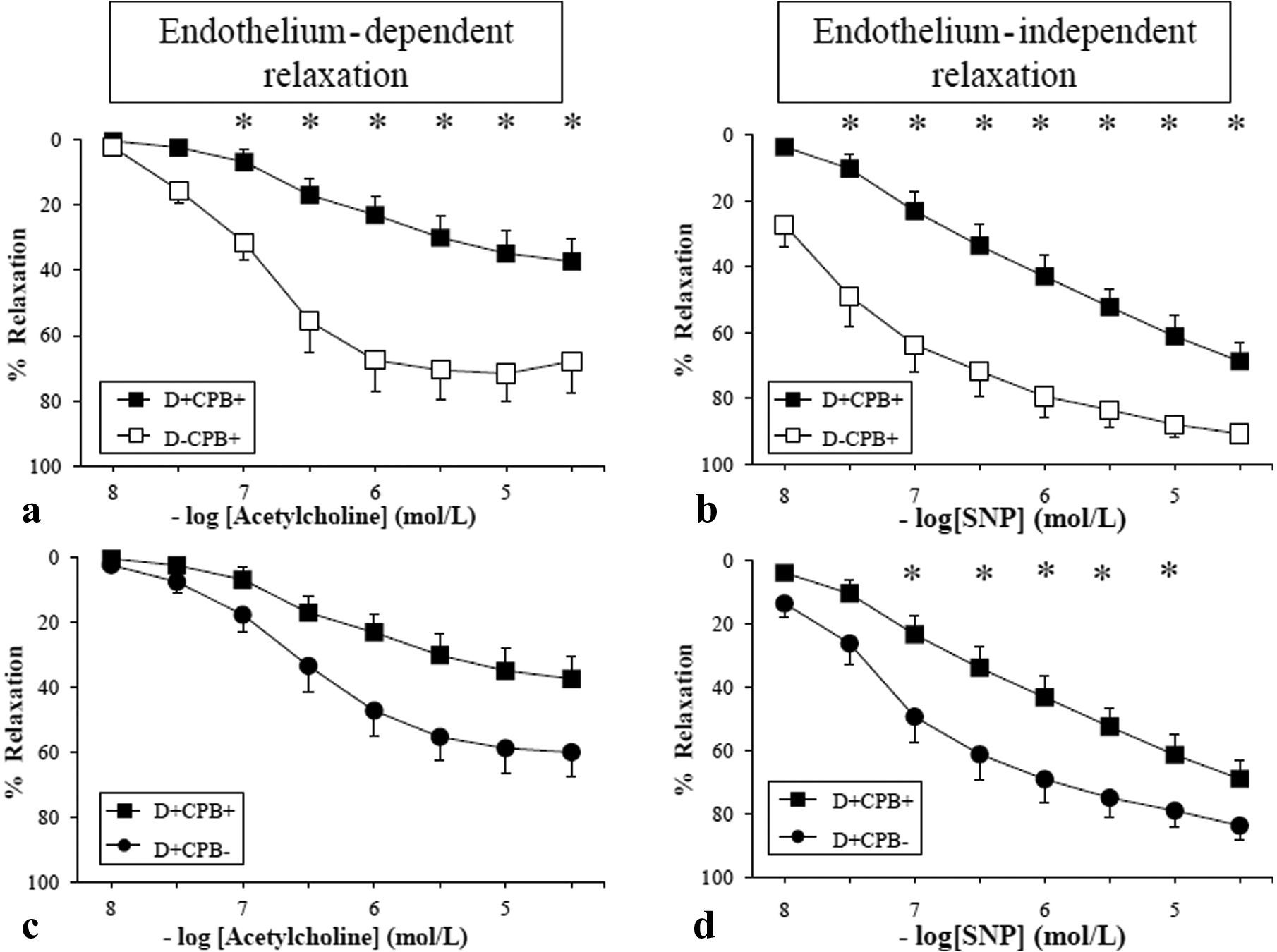
(a), (b), (c), (d) relaxation responses of the precontracted mesenteric artery to acetylcholine (endothelium-dependent relaxation) and sodium nitroprusside (SNP) (endothelium-independent relaxation). Responses are expressed as percentage relaxation of phenylephrine-induced precontraction (mean ± SEM). *p < 0.05. White circles: D-CPB- group; white squares: D-CPB+ group; black circles: D+CPB- group; black squares: D+CPB+ group.
Western immunoblot analysis of eNOS and iNOS
No significant differences in eNOS expression were seen between the four groups (Figure 6). On the other hand, iNOS expression was significantly upregulated in the D+CPB+ group compared with the other groups (Figure 7).
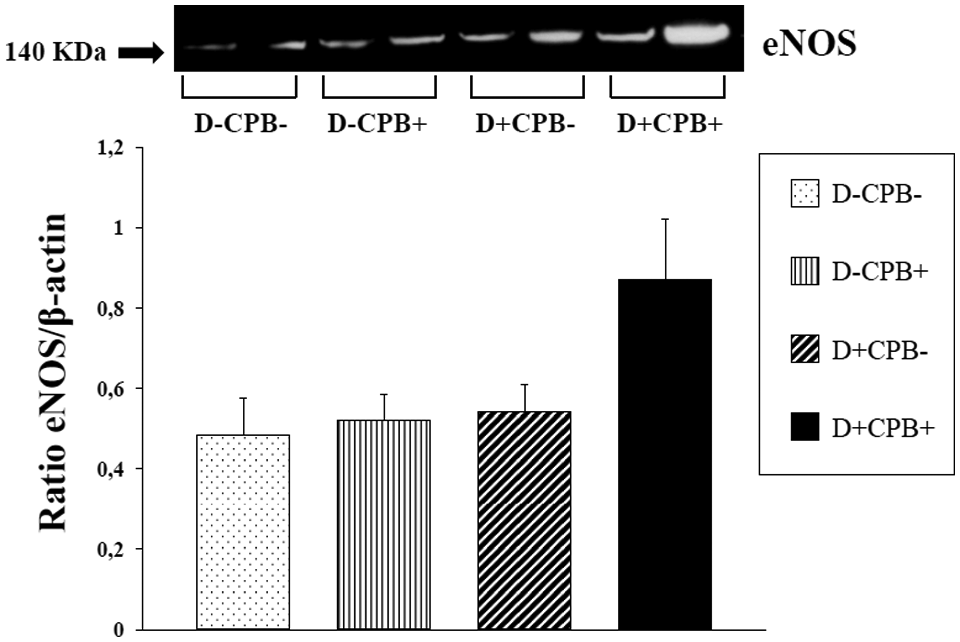
Western immunoblot analysis of eNOS expression in the mesenteric artery. White bars: D-CPB-; dotted bar: D-CPB-; fine vertical stripes: D-CPB+; large oblique stripes: D+CPB-; black bar: D+CPB+.
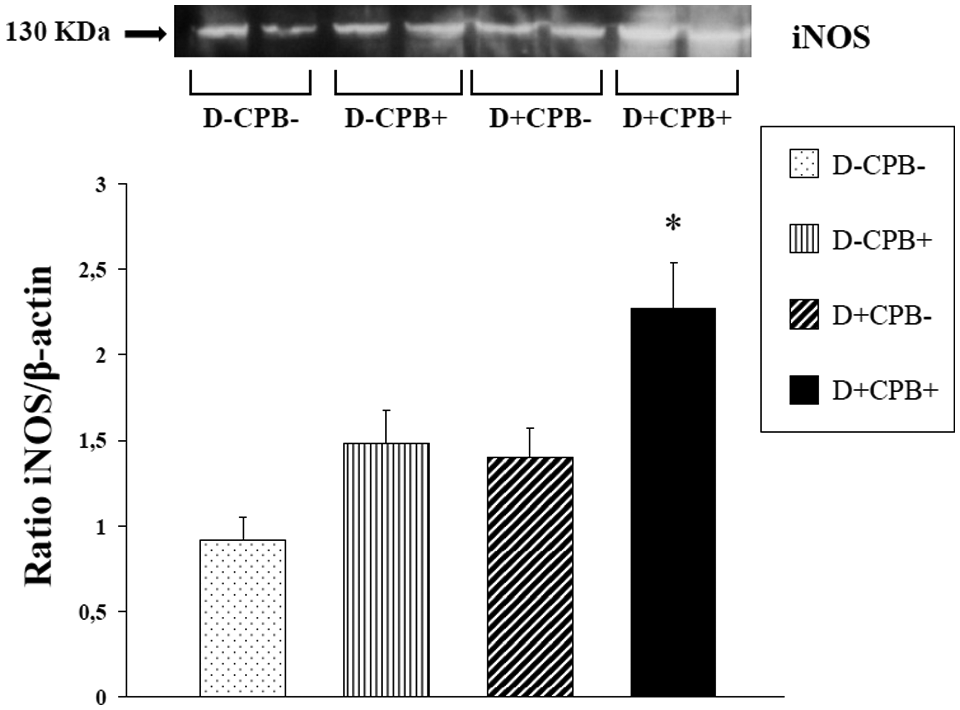
Western immunoblot analysis of iNOS expression in the mesenteric artery. *p < 0.05 vs. all groups. White bars: D-CPB-; dotted bar: D-CPB-; fine vertical stripes: D-CPB+; large oblique stripes: D+CPB-; black bar: D+CPB+.
Plasma nitrite products
There was no significant difference at the time of cannulation in plasma nitrite levels. There was a significant time-dependent difference between cannulation and the end of the experiment (p < 0.05) only in the CPB-exposed groups (D+CPB+ and D-CPB+) (Figure 8). Serum nitrite at the end of the experiment in the D+CPB+ group was significantly higher compared with the D-CPB- (p = 0.05) group.
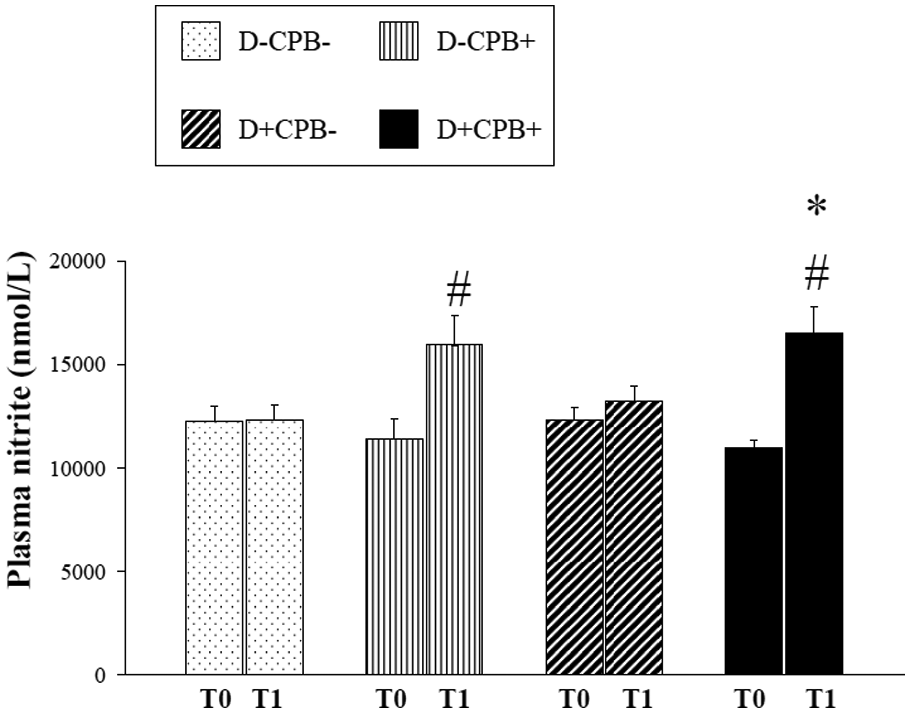
Changes in plasma levels of nitrite. #p < 0.05 vs. T0 within the same group; *p < 0.05 vs. T1 D-CPB-. White bars: D-CPB-; dotted bars: D-CPB-; fine vertical stripes: D-CPB+; large oblique stripes: D+CPB-; black bars: D+CPB+.
Inflammatory response
At the time of cannulation, the TNF-α level was significantly higher in the D+CPB- group than in the D-CPB+ group (p = 0.01) (Figure 9). At the end of the experiment, when compared with the time of cannulation, the TNF-α level was higher in the two CPB groups: D-CPB+ (p = 0.02) and D+CPB+ (p = 0.01). At the end of cannulation, TNF-α concentration was significantly higher in the D+CPB+ group compared with the D-CPB- group (p = 0.03).
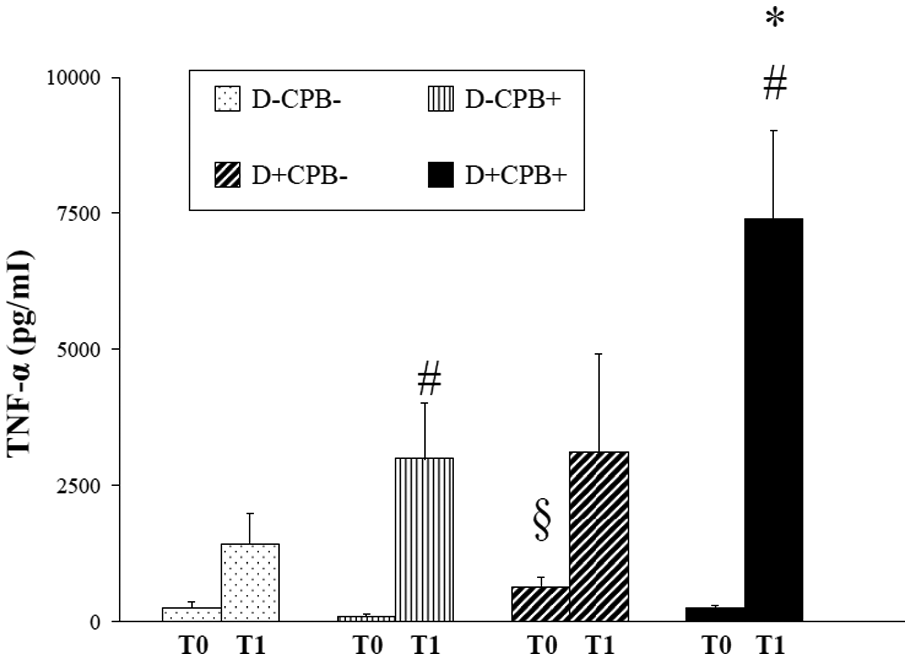
Mean values of TNF-α. #p < 0.05 vs. T0 within the same group; *p < 0.05 vs. T1 D-CPB-; §p < 0.05 vs. T0 group D-CPB+. White bars: D-CPB-; dotted bars: D-CPB-; fine vertical stripes: D-CPB+; large oblique stripes: D+CPB-; black bar = D+CPB+.
Discussion
The major finding of our present study is that diabetes, in a CPB rat model, increases mesenteric endothelial dysfunction as demonstrated by the enhanced contractile response to the α1-adrenergic agonist and a marked impairment of mesenteric vasorelaxation. Endothelial dysfunction was associated with enhanced nitrite and pro-inflammatory cytokine TNF-α production as well as iNOS expression.
Analysis of MAP showed no significant differences between the groups, thereby avoiding any potential impact of haemodynamic changes in our study.
In the present study, as previously described, mesenteric arteries from D+CPB- 29 and D-CPB+ 30 rats showed increased contractile responses to the α1-adrenergic agonist. These increased contractile responses after CPB or diabetes then disappeared after in vitro NO synthase inhibition. A decrease in NO bioavailability, either through loss of NO release or loss of NO biological activity, may explain the discrepancies in contractile response. The D+CPB+ group had marked enhancement of the contractile response to the α1-adrenergic agonist. The persistent significant difference observed in the presence of L-NA could be explained by increased sensitivity to Ca2+ by the contractile proteins 31 or to the increased activation of protein kinase C. 32 These results indicate the high susceptibility of the mesenteric vascular territory after CPB to vasopressor agents. The use of vasopressor agents in the postoperative period after cardiac surgery with CPB may increase mesenteric hypoperfusion and the risk of mesenteric ischaemia, especially in diabetics.
Our model highlights the impairment of mesenteric endothelium-dependent relaxation in the D+CPB- and D-CPB+ groups. The mechanisms underlying this observation could depend on increased degradation of NO, which is secondary to enhanced superoxide production. This reduced endothelium-dependent relaxation in diabetic (D+CPB-) and CPB-exposed (D-CPB+) animals is unlikely to be the result of impairment of the response of the underlying vascular smooth muscle to NO, as nitroprusside induced a similar concentration-dependent relaxation of vessels in diabetic, CPB-exposed and control rats. CPB in diabetes (D+CPB+) revealed impairment of mesenteric endothelium-dependent vasorelaxation, which was greater than that observed in diabetic (D+CPB-) or CPB-exposed rats (D-CPB+). The mechanisms underlying this observation may involve even greater scavenging of NO by increased oxidative stress. Diabetes 33 and CPB 34 have been shown to increase vascular formation of peroxynitrite. It is well established that peroxynitrite avidly oxidises the eNOS cofactor tetrahydrobiopterin (BH4) to form cofactor inactive molecules such as dihydrobiopterin. Under this condition of BH4 deficiency, NOS is in an uncoupled state, resulting in the production of superoxide rather than NO.21,35–37 Thus, upregulated NOS may contribute itself to superoxide production and indirectly contribute to perpetuating and increasing oxidative stress. The additive effect of diabetes and CPB on peroxynitrite and oxidative stress production may partly explain the marked impairment of mesenteric endothelium-dependent vasorelaxation.
Contrary to what was observed in other groups, impairment of mesenteric endothelium-dependent vasorelaxation in the D+CPB+ group also reflects an undeniable major alteration of the relaxation capacity of vascular smooth muscle cells. This alteration of endothelium-independent relaxation observed in only the D+CPB+ group may be explained by an excess of peroxynitrite and major oxidative stress. Independent of the previously recognised scavenging of NO, sGC-activity may be directly downregulated by O2 levels. 38
In the D+CPB+ group, which was associated with a lack of vascular relaxation, we observed a paradoxical increase in the expression of inducible NOS in the vascular wall of mesenteric arteries. Some authors attribute the activation of iNOS to an adaptation in response to reduced NO bioavailability, which is induced by direct superoxide scavenging and counterbalances vasoconstriction. Evidence for this comes from both the diabetic model and the CPB model. In CBP rats, selective inhibition of iNOS during CPB increases serum levels of inflammatory cytokines. 1 Ishikawa et al. 39 showed an increased vasomotor response after selective inhibition of iNOS in a diabetic rat model, and reported that NO produced by iNOS protected the vascular system from mesenteric vasoconstriction. iNOS activation seems to be a double-edged sword. iNOS-derived enhanced NO production may be an adaptive response to limit the development of CPB-induced inflammation, while iNOS-derived excessive NO production may lead to major oxidative stress. Excessive NO from iNOS activation, as we showed in the D+CPB+ group, generates various cytotoxic agents, including inhibition of cellular metabolism by direct reaction of heme and non-heme enzymes, 24 nitration of tyrosine to form peroxynitrite and induction of apoptosis. 40
Characterisation of the systemic inflammatory response in this small animal model of CPB was performed by quantifying plasma cytokine levels. A predictable increase in cytokine levels occurred in the early phase of the CPB-induced inflammatory response. IL-6 and TNF-α reached their peak levels at approximately 2 h after cessation of CPB, before slowly returning to baseline levels within a few hours in the rat model of CPB.7,41 However, IL-6 is a pleiotropic cytokine that characterises all inflammatory processes. TNF-α is the main pro-inflammatory cytokine involved in patients and in the pathophysiology of the CPB-induced inflammatory response.15,16,42,43
We have previously evoked the effect of mesenteric endothelial dysfunction on the inflammatory process. 7 The most striking feature was that, among these CPB+ rats, diabetic rats exhibited an exacerbated inflammatory response. De Lange et al. 44 also showed a significant increase in the pro-inflammatory cytokine IL-6 in CPB-exposed diabetic rats compared with CPB-exposed non-diabetic animals. Similar studies have also found that levels of pro-inflammatory cytokines after CPB are significantly higher in diabetics.15,16,42,43 Matata and Galinanes found lower rates of TNF-α and IL-8 in diabetic patients. 17 The authors stated that the reason for the lack of increased pro-inflammatory cytokines in diabetic patients was unclear, although they suggested that there was possible alteration of gene regulation of inflammatory factors and their signalling pathways.
There were several limitations to the present study. It was a model of only circulatory support, without cardioplegia or aortic clamping. Animals maintained, despite a high flow of assistance, a systolic and diastolic blood pressure. Despite all of the model’s improvements in reducing the priming, we observed an important haemodilution in CPB-exposed animals. However, haemodilution was similar in the two groups that underwent CPB. The method chosen to induce diabetes in the rat abolishes insulin secretion and so is more equivalent to type 1 diabetes, whereas the majority of cardiac surgical patients have type 2 diabetes, in that they are insulin resistant. Complementary experiments should be done in animal models closer to the human model, such as the Zucker rat. Finally, the fact that the response to sodium nitroprusside was altered in diabetic rats that underwent CPB is undoubtedly problematic. The mechanisms of relaxation of vascular smooth muscle involve the phosphorylation and dephosphorylation of many proteins, including myosin, and are dependent on intracellular Ca2+ levels. 45 Dephosphorylation of myosin inhibits myosin-actin interactions, thereby causing relaxation. However, the contraction phase continues after the decrease in intracellular Ca2+. Two mechanisms have been proposed to explain this phenomenon: 46 (i) the formation of actin- myosin bridges where myosin is no longer phosphorylated (hypothesis of ‘locked’ state), and (ii) modulation of different filaments of the cytoskeleton. Interactions between different filament molecules are regulated by the conformational state of constitutive proteins that regulate actin– myosin interactions. Heat-shock protein 20 is central to this modulation, and phosphorylation of this protein inhibits the contraction of smooth muscle. 45 It is difficult to draw conclusions from endothelial function if the underlying smooth muscle response is altered, without explorations of these mechanisms.
We report that experimental CPB in rats with diabetes exacerbates mesenteric endothelial dysfunction. These disorders are associated with a systemic inflammatory process and may contribute to inadequate tissue perfusion, mucosal ischaemia and lead to non-occlusive mesenteric ischaemia. Mesenteric endothelial injury may play a key role in the development of multiple organ failure after CPB and may trigger higher postoperative mortality and perioperative complications that occur in diabetic patients after CPB.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
None declared.
