Abstract
The present study was designed to identify the changes in microvesicle-dipeptidyl peptidase-IV (DPP IV) levels in human urine and serum, and to determine whether there were correlations with the severity of diabetic kidney disease (DKD). A total of 127 patients with type 2 diabetes mellitus (T2DM) were divided into three groups according to the urinary albumin/ creatinine ratio (UACR): microalbuminuria group (n = 50); macroalbuminuria group (n = 34) and normoalbuminuria group (n = 43), and 34 age- and sex-matched non-diabetic healthy subjects were selected as controls. Microvesicle-bound DPP IV and free urinary DPP IV were separated by a filtra-centrifugation method. The total microvesicles were captured by a specific monoclonal antibody, AD-1. DPP IV activity was determined by measuring the cleavage of chromogenic free 4-nitroaniline from Gly-Pro-p-nitroanilide at 405 nm with an ELISA plate reader. DPP IV protein levels were determined by ELISA and Western blot. Our results showed that the microvesicle-bound type was the major form of DPP IV in urine; the urinary microvesicle-DPP IV excretion of each T2DM group was significantly higher compared with controls. The urinary microvesicle-DPP IV level was positively correlated with UACR in patients with T2DM. These findings suggest that the urinary level of microvesicle-bound DPP IV is associated with the severity of DKD.
Introduction
Diabetic nephropathy (DN) is one of the major microvascular complications of diabetes and the most common cause of end-stage renal disease (ESRD).1,2 The clinical stages of DN are classified as follows: stage 1, normoalbuminuria (urinary albumin/creatinine ratio (ACR < 30 mg/g creatinine); stage 2, microalbuminuria (ACR from 30–300 mg/g creatinine); stage 3, macroalbuminuria (ACR ≥ 300 mg/g creatinine and/or persistent proteinuria with serum concentration of creatinine < 1.2 mg/dl); stage 4, chronic renal failure (serum concentration of creatinine ≥ 1.2 mg/dl with proteinuria); and stage 5, under chronic dialysis therapy. 3
Previously, diabetic kidney disease (DKD) was thought to be a unidirectional process that starts with microalbuminuria and leads to end-stage renal failure. However, recent studies have shown that a large proportion of patients diagnosed with DKD may reverse back to normoalbuminuria.4,5 Therefore, the development of more sensitive and reliable markers for early detection of DKD are needed.
Urine is one of the most useful resources for such a study, as its collection is simple and non-invasive. In addition to soluble plasma proteins, urinary microvesicles, such as exosomes and microparticles, have recently been the targets of urine proteomic analysis. Urinary exosomes are membrane vesicles, secreted by tubular cells, with a diameter of 40–100 nm, 6 while microparticles are membrane-shed vesicles with a size range between 100–1000 nm. Both the exosomes and microparticles are characterised by the same lipid bilayer, but the majority of microvesicles isolated from urine are thought to be exosomes. A previous report has shown that aquaporin-2 was only identified in urinary exosomes and not in exosomes from other sources, thereby revealing a link between these exosomes and their urogenital tract origin. These exosomes may produce novel biomarkers for renal dysfunction and structural injury. 7
Previous reports have indicated that a high level of plasma dipeptidyl peptidase-IV (DPP IV), also known as CD26, is positively correlated with diabetes mellitus (DM). DPP IV degrades active glucagon-like peptide-1 (GLP-1), an incretin released from L cells in the intestine after meal intake that enhances insulin secretion in a glucose-dependent manner. DPP IV inhibition showed blood glucose-lowering effects in both animal models of diabetes and in patients with type 2 diabetes mellitus (T2DM). Currently, some of the DPP IV inhibitors such as sitagliptin and vildagliptin have been widely used as an adjunct to diet and exercise to improve glycaemic control in adults with T2DM.8,9 DPP IV is a membrane-associated peptidase and is widely expressed in all tissues. In the kidney, where the enzyme is exceptionally concentrated, it is located primarily in the cortex and found in the brush border and microvillus fractions.
On the basis of these findings, we hypothesised that DPP IV might be a protein component of urinary microvesicles, and its activity might represent the concentration of microvesicles, secreted by tubular epithelial cells, in the urine. To test this hypothesis, we have developed an enzyme-linked immunosorbent assay (ELISA) method for DPP IV on urinary microvesicles using a specific monoclonal antibody, AD-1, developed by Deng et al.10,11 After it is immobilised on the plate, the antibody can specifically capture and purify the intact microvesicles from urine samples, and the DPP IV activity in the microvesicles can be determined. With this assay, urinary microvesicle-bound DPP IV activity in patients with T2DM was tested and compared with age- and sex-matched normal individuals. We hypothesised that the variable hydration state of individuals providing urine specimens may lead to some differences in water/salt content of urine, so we pooled all 24-h urine samples from an individual, then used a portion of that for our analysis. The applicability of microvesicle-bound DPP IV as an early biomarker for determining the status of DKD was evaluated.
Methods
Subject selection
In this study, a group of 127 Chinese Han patients with T2DM were recruited upon admission the Second Hospital of Shandong University between June and December in 2009. These patients were divided into three groups by urinary albumin/creatinine ratio (UACR): microalbuminuria group (DN1, n = 50, ACR from 30–300 mg/g creatinine); macroalbuminuria group (DN2, n = 34, ACR > 300 mg/g creatinine); and normoalbuminuria group (DM, n = 43, ACR < 30 mg/g creatinine). There were no significant differences in the three groups with respect to age, sex and body mass index. All patients were treated with angiotensin-converting enzyme inhibitors and/or angiotensin-receptor blockers, human insulin and 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors to control blood pressure (or to reduce proteinuria), blood glucose and blood lipid. No additional medicines were taken within 1 week of urine collection. Exclusion criteria were as follows: tumours, urinary tract disorders, pregnancy, known renal diseases other than DKD, decompensated heart failure, chronic inflammatory diseases, prostatic diseases (in males), haematological diseases, liver diseases and recent myocardial infarction or unstable angina within past 6 months. In addition, 34 age- and sex-matched Chinese Han healthy controls were enrolled after a careful history and clinical examination. Written informed consent was obtained from all subjects, and protocols were approved by the institutional ethics committees.
Collection of urinary and serum samples
Urine samples were collected from all subjects with a 24 h period. Two 0.95 ml aliquots of the urine sample were collected and neutralised with 50 μl aliquots of neutralisation buffer (1 M TrisHCl, pH 7.4, containing 1% NaN3). The treated samples were immediately stored at −80°C until analysis. For collection of serum samples, fasting blood samples of DN patients and healthy volunteers were collected by venipuncture. Immediately after centrifugation at 400 g for 10 min, the serum samples were collected and stored at -80°C until analysis.
Microvesicle-specific antibody preparation
The specific monoclonal antibody to the microvesicle (AD-1) was originally raised against human membrane-bound liver alkaline phosphatase isolated from the serum of patients with liver cancer. 12 The hybridoma cells were cultured in RPMI 1640 medium (GIBCO Invitrogen) containing 10% foetal calf serum to produce antibody. The culture supernatant containing AD-1 antibody was collected for assays.
AD-1 plate preparation
Goat anti-mouse antibody was diluted to a concentration of 10 μg/ml in Tris/HCl buffer (10 mM Tris, 10 mM NaCl and 10 mM NaN3 pH 8.5). The diluted antibody solution was distributed to the wells of 96-well plate (Costar Co Ltd, USA, 50 μl/well) and incubated at 37°C until the wells were dry. The plate was washed once with phosphate buffered saline containing 0.05% Tween-20 (PBST) and blocked with 200 μl/well of 10% saturated casein in PBST for 1 h at 37°C. After washing the plate twice with PBST, the culture supernatant containing the AD-1 antibody was added and incubated overnight at 4°C. After washing with PBST again, the coated plate was used immediately or stored at 4°C with 200 μl/well of incubation buffer (50 mmol/l TrisHCl, pH 8.0, containing 0.15 mol/l NaCl and 0.05% NaN3) to be used later.
Determination of the microvesicle-bound DPP IV proteins in urine samples
To capture urinary microvesicles, 50 μl of urine samples were added to the wells of the AD-1 coated plate and incubated at 37°C for 3 h. After washing the plate with PBST three times, 50 μl of rabbit anti-human DPP IV antibody conjugated with horseradish peroxidase (HRP) (R&D System) was added and incubated for another 2 h at 37°C. After five washes with PBST, HRP substrate solution was added to each well and incubated for 30 min at room temperature. Finally, after adding 25 μl of stop solution to each well, the OD was read using an ELISA reader at 450 nm.
Verification of the specificity of ELISA method
To verify the specificity of the ELISA method, AD-1 antibody was replaced by mouse IgG1 in the preparation of the ELISA plate. Eleven human urine samples were added to the plates coated with either AD-1 or mouse IgG1 antibodies. The amount of microvesicle-bound DPP IV was measured as described above. The intra-assay precision was tested on different plates in the same assay run by parallel measurements of six different serum or urine samples. The inter-assay precision was evaluated using the same samples on six different days. The intra-assay and inter-assay coefficients of variation in our laboratory were 2.6% and 7.8%, respectively.
Serum DPP IV activity determination
Serum DPP IV activity was measured using a 96-well plate colorimetric assay. 13 Briefly, 5 μl of plasma in a total volume of 100 μl of 71 mmol/l glycine/NaOH (pH 8.3) buffer was incubated with 0.5 mg/ml substrate Gly-Pro-p-nitroanilide (Sigma) for 60 min at 37°C. The substrate is cleaved by serum DPP IV to release the chromogenic free 4-nitroaniline and monitored at 405 nm with an ELISA plate (Labsystems, Finland) reader. All measurements were performed in duplicate and corrected for spontaneous hydrolysis of the substrate using blank wells without plasma. The DPP IV activity was calculated using the p-nitroaniline standard (Immuno Kontact) included on each plate.
Determination of microvesicle-bound DPP IV and free DPP IV in urine samples
Microvesicle-bound DPP IV was separated from its free form by a filtra-centrifugation method as described by Miranda et al., 14 with some modifications. Briefly, urine samples (500 μl) were centrifuged at 17,000 g for 10 min at 4°C to remove urinary sediment. The supernatants were transferred into filtration concentrators (100 kDa MWCO, Millipore) and centrifuged at 4000 g for 5–10 min at room temperature to let almost all solution go through the filters. After washing the filters with PBS, PBS containing 0.1% Triton-X100 was added in the concentrators and incubated on ice for 5 min with agitation to dissolve the microvesicle-bound DPP IV proteins. This step was repeated with 50 μl of PBS-Triton buffer and, finally, a total of 150 μl of solution containing microvesicle-bound proteins was collected. The DPP IV activities in both fractions (filtration flow-through fraction and PBS-Triton solubilised fraction) were measured as described above.
Immunoprecipitation and immunoblotting assays
Microvesicles in urine samples were immunoprecipitated with the AD-1 antibody while DPP IV proteins in urine or serum were pulled down with mouse anti-human DPP IV antibody (AbD serotec, MCA 1317) as below. After pre-clearing human serum or urine of healthy controls with protein G-sepharose CL-4B (Pharmacia Biotech), aliquots were immunoprecipitated with either AD-1 or mouse anti-human DPP IV AB-conjugated protein G-sepharose CL-4B in PBS overnight at 4°C. A mouse IgG (AbD Serotec, MCA 928) was also included in the assay as negative control. In some urine AD1-precipitated samples, PBS containing 0.1% Triton X-100 was added to dissolve the membrane-bound proteins. After washing five times with PBST, immunoprecipitates were denatured with NuPAGE sample buffer (Invitrogen), containing 5% beta-mercaptoethanol, at 95°C for 5 min and separated on NuPAGE 4-12% Bis-Tris gels (Invitrogen). Gels were blotted onto nitrocellulose using the XCell II blot module (Invitrogen) according to the manufacturer’s instructions. Membranes were stained with Ponceau S to check the transferring efficiency, briefly washed with PBST, blocked in PBST/5% low-fat milk for 30 min, and probed with a goat anti-human DPP IV antibody (R&D systems, AF1180) overnight in PBST/1% low-fat milk. The blot was then washed extensively in PBST, and probed for 1 h with HRP-conjugated secondary antibodies (Abcam) at a dilution of 1:10,000 in PBST/1% low-fat milk. After extensive washing in PBST, blots were developed with Supersignal West Dura Extended Duration Substrate (Perbio, Cheshire, UK). Images were captured using a Chemigenius imaging system (Syngene, Cambridge, UK).
Electron microscopy analysis of urinary microvesicles
First, urine samples were concentrated with filtra-centrifugation columns, and the concentrated solution was incubated overnight at 4°C with 80 μg/ml of AD-1 in 100 nmol/l Tris-HCL buffer, pH 8.0. Antibody-bound microvesicles were immunoprecipitated by rabbit anti-mouse immunoglobulin, and the precipitate was processed for electron microscopy as described previously. 10 Briefly, the precipitate was fixed for 2 h at room temperature with 2% glutaraldehyde in 0.1 mol/l Na-cacodylate-HCL buffer, pH 7.4, containing 1% sucrose. After rinsing with 0.1 mol/l Na-cacodylate buffer, pH 7.4, containing 4% sucrose, the pellet was fixed at room temperature, first for 2 h with 2% osmium tetroxide in 0.1 mol/l Na-cacodylate buffer and then treated for 1 h with 2% uranyl acetate in distilled water. Finally, the fixed pellet was embedded in EPON, cut into 70 nm ultra-thin sections, and the morphology of the vesicles was analysed by electron microscopy.
Statistical analyses
All data were analysed by SPSS software 16.0 (SPSS, Chicago, IL, USA). Values are presented as means ± SD. The difference among groups was compared by single-factor ANOVA. Pearson correlation, and stepwise multiple regression analyses were used to explore the relationship between urinary microvesicle-DPP IV and the clinical characteristics of patients with T2DM. All tests were two-tailed, and p < 0.05 was considered statistically significant.
Results
Analysis of clinical characteristics
Baseline anthropometric and biochemical characteristics for all examined groups are given in Table 1. In total, 34 age- and sex- matched healthy volunteers were enrolled as controls after a careful history and clinical examination. As shown in Table 1, there were no significant differences in age, sex and body mass index among these four groups. Patients with T2DM with microalbuminuria and macroalbuminuria had increased UACR, glycated haemoglobin, serum creatinine, systolic blood pressure and total cholesterol (p < 0.01) compared with those of the control group. In addition, T2DM patients with macroalbuminuria had significantly elevated serum creatinine and UACR compared with other groups, while the levels of glycated haemoglobin, serum urea nitrogen and total cholesterol were not significantly different in three diabetic groups (Table 1).
Baseline anthropometric and biochemical characteristics.
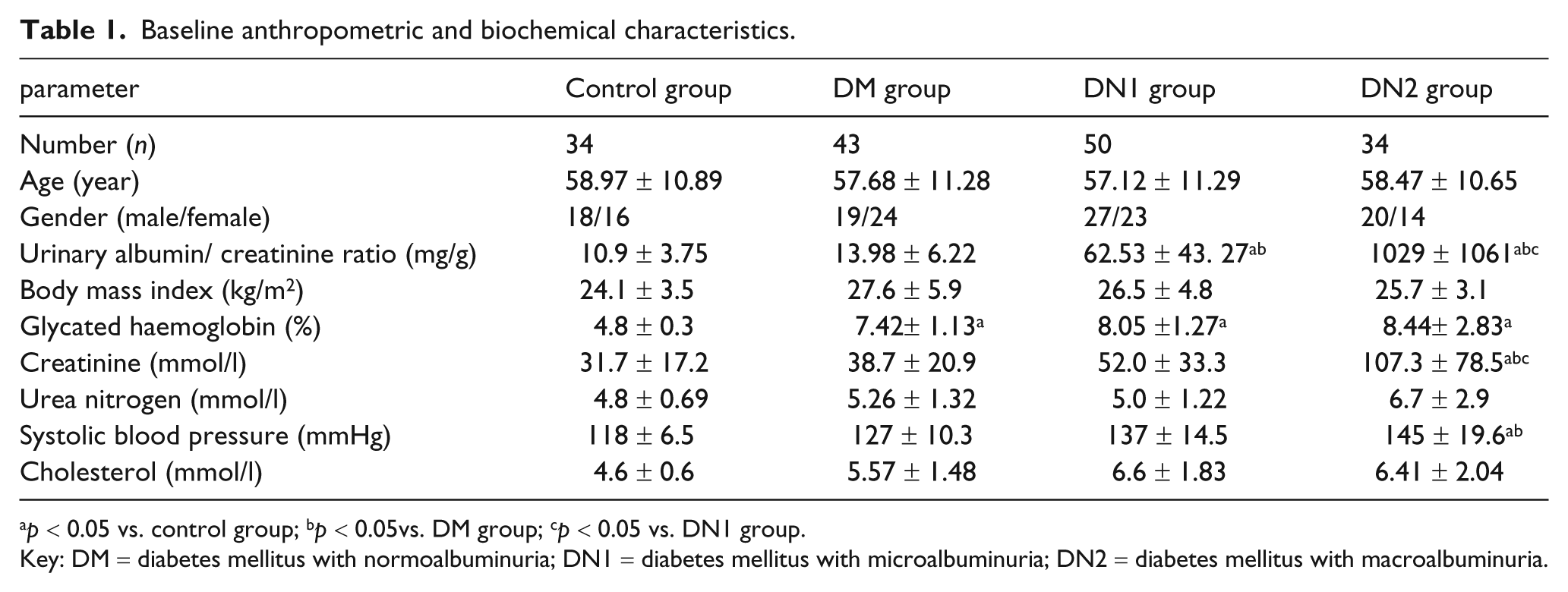
p < 0.05 vs. control group; bp < 0.05vs. DM group; cp < 0.05 vs. DN1 group.
Key: DM = diabetes mellitus with normoalbuminuria; DN1 = diabetes mellitus with microalbuminuria; DN2 = diabetes mellitus with macroalbuminuria.
Specificity of the ELISA method
The specificity of our developed ELISA method was evaluated by replacing AD-1 with the mouse antibody of the same immunoglobulin class but of irrelevant specificity. As shown in Figure 1, the microvesicle-bound DPP IV captured by the control antibody was almost undetectable (0.373 ± 0.052 pg/ml vs. 0.004 ± 0.000 pg/ml, p < 0.001), indicating the high specificity of the assay.
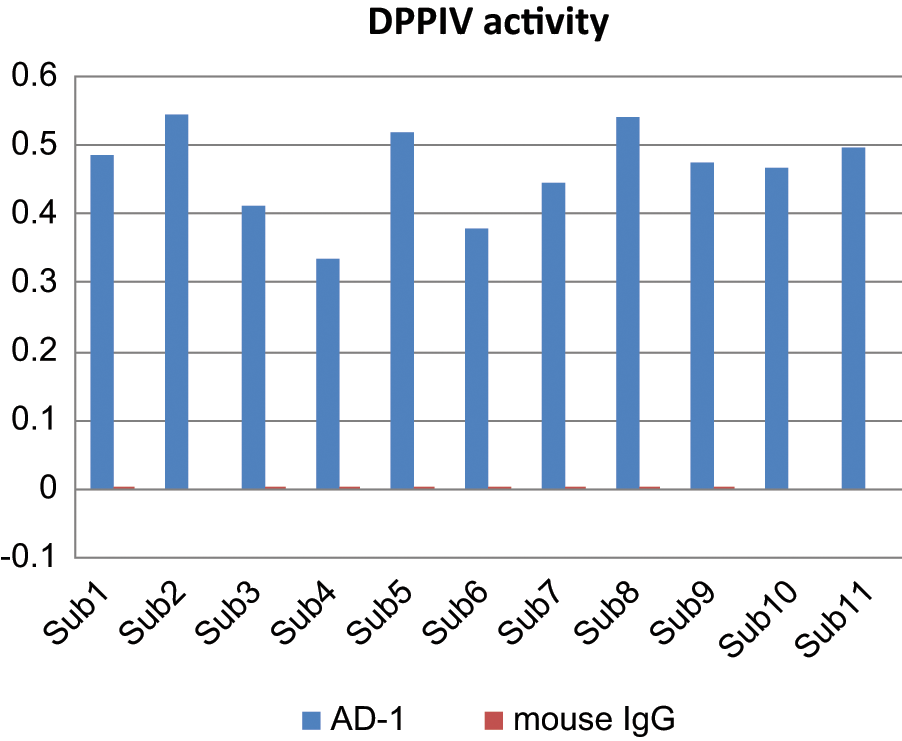
The dipeptidyl peptidase-IV (DPP IV) activity level of the specific monoclonal antibody to the microvesicle (AD-1) and the mouse antibody of the same immunoglobulin class.
Excretion of urinary microvesicles-bound DPP IV in patients with T2DM and healthy controls
Excretion of urinary microvesicles-bound DPP IV in the healthy controls and in T2DM patients with normoalbuminuria, microalbuminuria and macroalbuminuria is summarised in Figure 2. The urinary microvesicle-DPP IV in DM (4.7766 ± 2.1036 pg/ml), DN1 (4.7000 ± 1.8122 pg/ml) and DN2 (7.6278 ± 3.8487 pg/ml) groups were significantly higher (p < 0.01) than in the control group (3.5857 ± 0.6959 pg/ml). The urinary microvesicle-bound DPP IV showed a stepwise increase in the patients of DM, DN1 and DN2 groups, especially in the DN2 group which has a higher amount of DPP IV than in the other two groups (p < 0.01; see Figure 2). These results clearly indicate that the urinary microvesicle-bound DPP IV excretion was at a low level in the control group, but was obviously increased in DM, DN1 and DN2 groups, especially in the DN2 group.
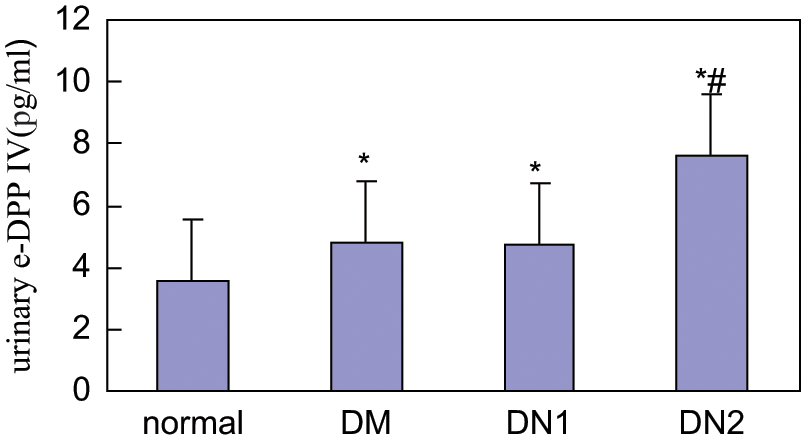
The urinary excretion of microvesicle-dipeptidyl peptidase-IV (m-DPP IV) in T2DM patients with normoalbuminuria (DM), microalbuminuria (DN1), macroalbuminuria (DN2) and healthy controls. *p < 0.01 vs. control group *# p < 0.01 vs. DM or DN1 group.
Serum DPP IV activity assay
Serum activities of DPP IV in patients with T2DM and the normal control group are summarised in Figure 3. Serum DPP IV activity (249.7 ± 73.6 pg/ml) in T2DM patients with normoalbuminuria was slightly higher compared with the controls (238.3 ± 66.5 pg/ml), but the difference was not statistically significant (p = 0.535).
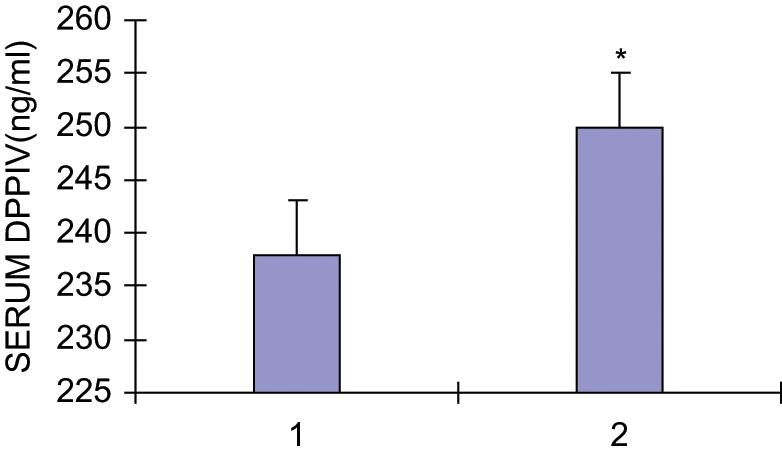
The serum activities of dipeptidyl peptidase-IV (DPP IV) in patients with T2DM with normoalbuminuria (DM) and healthy controls. * p > 0.05 vs. control group.
Pearson correlation and stepwise multiple linear regression analyses
Correlation analyses between urinary microvesicle DPP IV and biochemical parameters in patients with T2DM are summarised in Table 2. Significant correlations (p < 0.01) of urinary microvesicle-DPP IV excretion with glycated haemoglobin (p = 0.002), UACR (p < 0.001), serum creatinine (CR) (p < 0.001) and serum urea nitrogen (BUN) (p = 0.001) were obtained.
Correlation between urinary microvesicle-dipeptidyl peptidase-IV (m-DPP IV) and biochemical parameters in T2DM patients (p value).
Stepwise multiple linear regression analyses were summarised in Table 3. UACR (β = 0.538) and glycated haemoglobin (β = 0.489) are significant determinants for microvesicle-DPP IV levels in urine, p < 0.001, respectively.
The stepwise multiple linear regression analysis between urinary microvesicle-dipeptidyl peptidase-IV (m-DPP IV) and biochemical parameters in T2DM patients (p value).
The presence of urinary microvesicle-bound DPP IV protein
The presence of DPP IV protein in urinary microvesicles was analysed by a combination of immunoprecipitation and Western blotting assays (Figure 4). Microvesicles in urine samples were immunoprecipitated with the AD-1 antibody, and DPP IV proteins in urine or serum were immunoprecipitated with the mouse anti-human DPP IV antibody. As shown in Figure 4, the DPP IV protein band (105 kDa) could be clearly detected in all samples precipitated with either the AD-1 or the mouse anti-DPP IV monoclonal antibody. However, the band disappeared completely after addition of 0.1% Triton X-100 to the AD-1 immunoprecipitate prior to electrophoresis, indicating the release of DPP IV from urinary microvesicles after the dissociation of the plasma membrane structure by the detergent. Indeed, DPP IV was associated with immunoprecipitation-isolated vesicles. Comparison of the urinary and serum immunoprecipitates in the blot revealed that DPP IV protein from human urine was consistent with its serum origin (Figure 4).

Western blotting with anti-dipeptidyl peptidase-IV (DPP IV) antibody. 1. Urine immunoprecipitated with anti-DPP IV antibody. 2. Urine immunoprecipitated with AD-1 antibody treated with Triton X-100. 3. Urine immunoprecipitated with AD-1 antibody. 4. Serum immunoprecipitated with anti-DPP IV antibody.
The morphology of urinary microvesicles analysed by electron microscopy
As shown in Figure 5, the electron microscopy analysis revealed that the size of the urinary microvesicles varied between 50–300 nm, compatible with the sizes of both exosomes and microvesicles. The morphology of the vesicles displayed a bilayer structure typical of cellular plasma membranes.
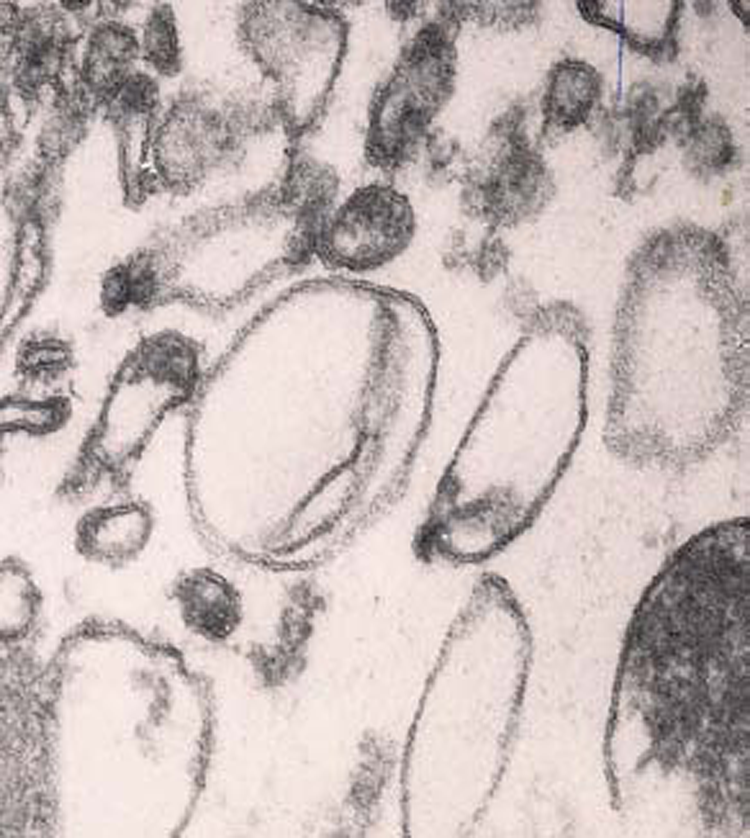
Electron micrograph of urinary microvesicles.
The relative distribution of microvesicle-bound and soluble DPP IV in human urine
The distribution of soluble and vesicle-bound DPP IV forms was analysed in urine samples (six from DN2 patients and six from normal individuals) by a two-step procedure. First, vesicle-bound enzyme was separated from its soluble form by filtra-centrifugation, and then the enzyme activities in the two fractions were measured. The results indicated that DPP IV was present mainly in the microvesicle-bound form in urine: 85.16% ± 9.68% of total activity in the samples of DN2 patients was microvesicle-bound, compared with 92.22% ± 10.66% in normal control samples, but no statistically significant difference was found between two groups.
Discussion
The whole proteome of urinary vesicles has already been reported, indicating that over 40% of the identified proteins are associated with several diseases.6,15 Therefore, further characterisations of these exosome-detected proteins under normal and pathological conditions will help to define them as biomarkers of diseases. 16 The present study was to evaluate the effectiveness of using analysis of urinary microvesicles to identify potential diagnostic markers for DKD.
DPP IV is an intrinsic membrane glycoprotein, localised on glomerular visceral epithelial cells, endothelial cells, and the proximal tubule brush border. 17 DPP IV has been shown to participate in T-cell biology as a co-stimulatory molecule able to regulate signalling transduction pathways.18–20 In addition to its role in T-cell biology, DPP IV also plays an important role in the pathogenesis of kidney diseases such as IgA nephropathy. 21
We here studied the excretion of DPP IV in urinary microvesicles in a small cohort of patients with T2DM by categorising them into three groups according to the severity of kidney damage. In this study, we first assessed the feasibility of using urine as a microvesicle source in the context of a clinical trial, and then investigated the changes in urinary microvesicle-DPP IV levels during different stages of DKD.
As reported, ultracentrifugation and filtration are the major methods for purifying microvesicles. The filtration method has been proposed to replace ultracentrifugation, as the latter requires expensive instrumentation and lengthy processing time. 12 However, filtration cannot isolate vesicles less than 300 nm in diameter. 22 In the present study, we have successfully developed an ELISA method for measurement of DPP IV in urinary microvesicles using our specific monoclonal antibody, AD-1. The intact microvesicle-bound DPP IV was first captured and purified from urine samples, and the DPP IV protein in the microvesicles was subsequently determined. This method is rapid and highly reproducible. In particular, it has high sensitivity and specificity. 12
Our data showed that the microvesicle-bound DPP IV in the urine of three groups of patients with T2DM was significantly increased compared with the control group. A significant correlation between the proteinuria and the increased urinary excretion of microvesicle-bound DPP IV was observed. In our previous study, in normal human kidney, AD1 staining is only present on the brush border of proximal tubular cells in the cortex (S1, S2 segments) and in the outer strip of the outer medulla and in the medullary rays (S3 segments). These results suggested that the microvesicle-bound DPP IV in urine may be secreted from tubular epithelial cells and be related to early tubular impairment, and thus it may be an early marker of renal damage before onset of albuminuria. However, the mechanism of increased urinary DPP IV excretion in patients with DKD remains unclear. DPP IV may be secreted from tubular epithelial cells following renal damage, which is known to cause the leakage of microvascular endothelial DPP IV in the kidney, or there may be some increased cleavage of the DPP IV pro-peptide.7,23–25
The morphological analysis of the AD-1 antibody-immunoprecipitated urinary microvesicles by electron microscopy indicated that antibody binding during the isolation process did not perturb the morphology and structure of these vesicles. Based on the ratio of microvesicle-bound DPP IV to total urinary DPP IV, we propose that the microvesicle-bound type is the major form of DPP IV in urine. Furthermore, we showed that the ratio of the urinary microvesicle-bound DPP IV to the free form of DPP IV is not significantly different in patients with DKD as compared with controls.
In published clinical reports over the last decade, circulating DPP IV activity has been reported to be either increased or decreased in diabetic patients.26–29 So far, the significance of changes in serum DPP IV activity is uncertain. 30 In this present study, we showed that the serum DPP IV activity in patients with T2DM with normoalbuminuria was not significantly different from that of controls, whereas we demonstrated that the urine microvesicle-bound DPP IV activity in T2DM patients with normoalbuminuria was significantly increased compared with controls, and increased alone with the aggravation of urine albuminuria. These findings indicate that urinary microvesicle-bound DPP IV is a specific marker for DKD. The measurement of urinary microvesicle-bound DPP IV might be a useful method for DKD screening and diagnosis in patients with T2DM. Whether it can be used as a marker for other kind of kidney diseases needs to be further investigated.
Footnotes
Funding
This study was supported by the research grant from the Natural Science Foundation of Shandong Province (ZR2009CM061).
Conflict of interest statement
All the authors declare that there is no conflict of interest.
