Abstract
Objective:
To investigate the relationship between diabetic retinopathy, neuropathy and low ankle-brachial index in mild-to-moderate chronic kidney disease of type 2 diabetic patients.
Methods:
We enrolled 875 type 2 diabetic patients who were divided into two phenotypes (with or without albuminuria) and stratified into three groups (stage 1 with estimated glomerular filtration rate ⩾ 90 mL/min/1.73 m2, stage 2 with estimated glomerular filtration rate of 60–89, stage 3 with estimated glomerular filtration rate of 30–59). The prevalence of diabetic retinopathy, neuropathy and low ankle-brachial index was compared and the risk factors of renal impairment were determined.
Results:
Among chronic kidney disease stages, the prevalence of diabetic retinopathy increased from 42.5%, 56.6% to 66.7% in albuminuric subjects and from 29.4%, 33.0% to 50.0% with no significant trend in normoalbuminuric subjects (p = 0.005, 0.007 and 0.399 compared with albuminuric subjects in each stage). There was a significantly increased prevalence of low ankle-brachial index (17.5%, 22.6% and 44.4%) in normoalbuminuric subjects but no significant trend in albuminuric subjects. Diabetic retinopathy (odds ratio = 2.474, 95% confidence interval = 1.009–6.068) was an independent risk factor of declining kidney function in albuminuric patients.
Conclusion:
The prevalence of diabetic retinopathy was graded according to the estimated glomerular filtration rate declining in albuminuric patients while the prevalence of low ankle-brachial index was gradually increased in normoalbuminuric patients, indicating the diverse underlying mechanisms of mild to moderate chronic kidney disease between these two phenotypes.
Keywords
Introduction
Chronic kidney disease (CKD) has affected approximately one-third of diabetic patients and become the leading cause of end-stage renal disease worldwide. 1 However, the latest China National Survey has shown that the prevalence of severe kidney damage was relatively lower in China when compared with developed countries. For example, the prevalence of stage 3 CKD in China was 1.6% as compared with that of 7.7% in the United States and 4.2% in Norway. 2 Since mild-to-moderate CKD is also associated with an increased risk of cardiovascular disease (CVD), 3 this lag is uniquely important in terms of studying the underlying mechanisms of early-stage CKD in China, and hence the prevention.
In relation to kidney impairment, diabetic retinopathy has been demonstrated to have a close relationship with albuminuria.4,5 Albuminuria has long been recognized as the hallmark of diabetic nephropathy, preceding glomerular filtration rate decline. 6 However, a growing body of evidence has indicated that glomerular filtration rate loss may occur also in normoalbuminuric subjects, 7 especially in type 2 diabetes.8–13 While diabetic retinopathy has been described classically as a microvascular disease, ankle-brachial index (ABI) has been found to be a marker of macrovascular complications and associated with an increased risk of CVD, cardiovascular mortality, and all-cause mortality.14,15 Our previous study in Hangzhou has shown a high prevalence of low ABI (39.5%) in early-stage CKD patients with type 2 diabetes who had no albuminuria and took no medication of the renin–angiotensin–aldosterone system inhibitors. 16 Thus, it is of pivotal importance to investigate the performance of macro- and microvascular complications in albuminuric and non-albuminuric forms of CKD in type 2 diabetic subjects, which, however, is seemingly contradictory in previous studies17,18 and far from being completely established. 19
Therefore, this study aimed first to investigate the relationship between diabetic retinopathy, neuropathy and low ABI in mild to moderate CKD (stage 1–3) of type 2 diabetic patients with albuminuria and second to determine these associations among those without albuminuria.
Methods
Study population
A total of 1117 patients diagnosed with Type 2 diabetes were consecutively recruited at the Department of Endocrinology and Metabolism of the Sir Run Run Shaw Hospital (Key centre of diabetes in Zhejiang Province) between January 2010 and December 2012, among which 133 patients with clinical CVD had been excluded because CVD was considered to have a strong association with low ABI and estimated glomerular filtration rate (eGFR), 20 but a weak association with diabetic retinopathy. 21 The present study is focused on the underlying risks of early-stage CKD in two phenotypes with or without albuminuria, so the inclusion of patients with clinical CVD may confuse the results. The presence of CVD was diagnosed by the physician as a history of ischemic stroke and/or coronary heart disease. Stroke (ischemic cerebrovascular disease) included only symptomatic brain infarction, but did not include silent brain infarction, transient ischemic attack or brain haemorrhage. Coronary heart disease included a previous history of myocardial infarction, angina pectoris or the presence of coronary interventions. The resting electrocardiograms were examined for the rest of the patients and the coronary artery computed tomography was conducted in 32 of those with an abnormal resting electrocardiogram to further exclude silent coronary heart disease according to the guidelines of American Diabetes Association. 22 Because of the relatively low prevalence of severe kidney damage in China (as well as in our data) 2 and the focus of mild to moderate CKD in the present study, we excluded nine patients with eGFR lower than 30 mL/min/1.73 m2. In all, 45 patients with acute infection including pneumonia, diarrhoea and cholecystitis; 40 patients who accepted kidney operations such as nephrectomy or with other chronic renal diseases such as chronic glomerulonephritis, nephropyelitis and hydronephrosis; and 15 patients at the end stage of all kinds of cancers were also excluded. At last, 875 patients were enrolled in the study. Furthermore, the patients were divided into two phenotypes with or without albuminuria. All participants provided written informed consent, and the study was approved by the investigational review boards of Zhejiang University Affiliated Sir Run Run Shaw Hospital.
History collection
Interviews were conducted by trained examiners who used a well-established questionnaire to collect demographic information of the participants studied, including date of birth, sex, smoking status and personal medical history. Waist circumference was measured to the nearest 0.1 cm at expiration along a horizontal plane through the abdomen at the level of the midpoint between the lowest rib and the iliac crest. Weight and height were measured while patients were dressed in light clothing. Body mass index was calculated as weight (kg) divided by the square of the height (m). Blood pressure was measured twice with the subjects in the sitting position after a 5-min rest. The lower value of two measurements was used for the study.
Laboratory measurement
Venous blood samples were collected in the morning following an overnight fast. Lipid profiles including triglyceride, total cholesterol, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol and very low-density lipoprotein cholesterol, fasting blood glucose, fasting insulin, fasting C peptide, serum creatinine, serum uric acid and C reactive protein levels were measured using another automatic analyser (AEROSET; Abbott Laboratories, Abbott Park, Illinois, USA). The dyslipidaemia was defined as a serum total cholesterol level of >220 mg/dL, a triglyceride level of >150 mg/dL, a high-density lipoprotein cholesterol level of <40 mg/dL or the use of lipid-lowering agents. HbA1c was examined using an automatic analyser (VARIANT II; Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Diagnosis of diabetic retinopathy and neuropathy
Diabetic retinopathy was assessed by the digital non-mydriatic fundus photography (Nonmyd; Kowa Company, Ltd., Japan) according to the protocol previously reported.23,24 Diabetic retinopathy was classified as none and diabetic retinopathy containing mild non-proliferative diabetic retinopathy, moderate non-proliferative diabetic retinopathy, severe non-proliferative diabetic retinopathy, proliferative diabetic retinopathy and diabetic macular oedema by a trained ophthalmic photographer and a retinal specialist. A total of 30 patients who were diagnosed as having cataract by ophthalmologist were excluded from the analysis. The diagnosis of diabetic neuropathy was depending on the nerve conduction study performed by a trained physiatrist. Electrophysiological tests were done in recommended standard situations 25 for all patients by Synergy electromyograph machine (Keypoint; Dantec Dynamics A/S, Denmark), which included ulnar and median nerves (sensory and motor fibres) in upper extremities; and sural (sensory), deep peroneal and tibial (motor) nerves in lower extremities. Diagnosis of diabetic neuropathy was based on at least one abnormal nerve conduction result.
Diagnosis of albuminuria and calculation of eGFR
An early morning spot urine sample was collected to calculate the urine albumin creatinine ratio. Albuminuria was defined as an albumin creatinine ratio ⩾ 30 mg/g based on the recommendation of American Diabetes Association. 26 eGFR was estimated using the CKD Epidemiology Collaboration (CKD-EPI) Study equation. 27 The enrolled patients were stratified into three groups according to the CKD-EPI eGFR level: stage 1 group with eGFR level ⩾ 90 mL/min/1.73 m2, stage 2 group with eGFR of 60–89 and stage 3 with eGFR of 30–59.
ABI measurement
The ABI measurements were performed in a supine position and blood pressure was measured in the bilateral brachial and dorsalis pedis arteries with an 8-MHz Doppler probe (Vista AVS; Summit Doppler Systems, Inc., Golden, CO USA). According to the guidelines of American Heart Association, 28 ABI was calculated as the ratio of the higher value of the systolic blood pressure of the two ankle arteries of that limb (either the anterior or the posterior tibial artery) and the higher value of the two brachial systolic blood pressures. For each patient, the lower ABI from both legs was used for further evaluation. Among the 875 enrolled patients, ABI levels ranged from 0.19 to 1.39. Therefore, we categorized ABI into three groups: normal (1.0–1.39), low–normal (0.9–0.99), and low (<0.9).
Statistical analysis
Statistical analyses were performed using the SPSS 15.0 software package (v. 15.0; SPSS Science, Chicago, IL, USA). Analysis of variance (ANOVA) and the χ2 test were used for between-group comparisons of continuous and categorical variables, respectively. An ordinal logistic regression analysis was applied to examine the independent risk factors for declining kidney function in patients with or without albuminuria, respectively. Odds ratio (OR) and respective 95% confidence interval (95% CI) were determined to examine the strength of the relationship between the prevalence of diabetic macro- and microvascular complications with the CKD stage 1–3. Differences with p < 0.05 (two-tailed) were considered statistically significant.
Results
Characteristics of the participants among the CKD stages
Characteristics of all the participants and the subgroup patients with or without albuminuria were shown in Tables 1 to 3, respectively. Among all the participants, the proportions of stage 1–3 were 74.4%, 21.4% and 4.2%, respectively. Among the patients with normoalbuminuria, the proportions were 79.8%, 18.7% and 1.5%, respectively, while the CKD stage 2 was noted in 28.9% and stage 3 in 11.9% among the patients with albuminuria. The duration of diabetes, systolic blood pressure and the prevalence of hypertension, use of insulin and renin–angiotensin–aldosterone system inhibitors were increased with eGFR declining in all the participants and in the two subgroup patients. There were no statistically significant trends with respect to lipid metabolism, diastolic blood pressure, body mass index, waist circumference, fasting glucose and fasting C peptide level.
Characteristics of the participants among CKD stages.
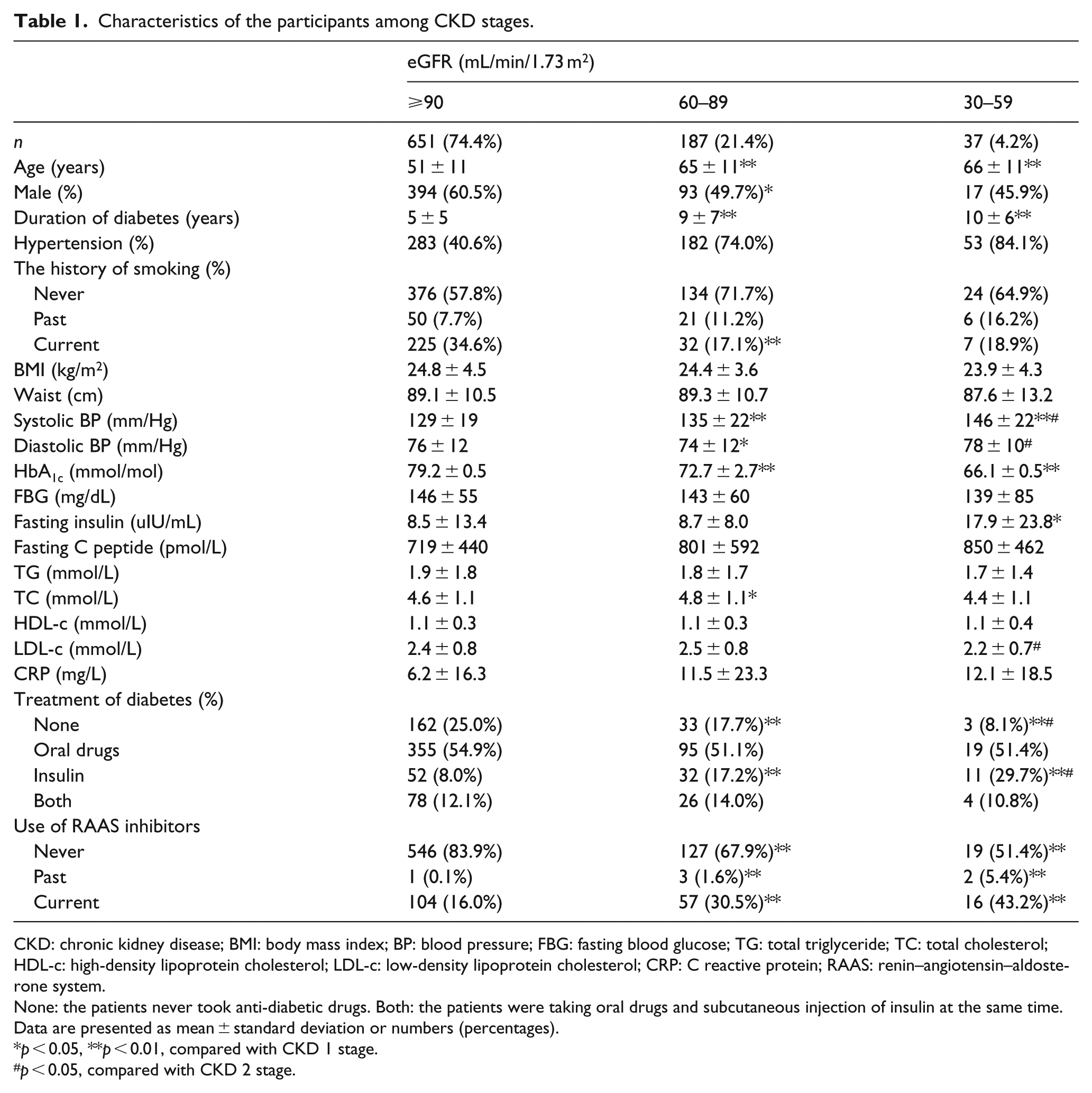
CKD: chronic kidney disease; BMI: body mass index; BP: blood pressure; FBG: fasting blood glucose; TG: total triglyceride; TC: total cholesterol; HDL-c: high-density lipoprotein cholesterol; LDL-c: low-density lipoprotein cholesterol; CRP: C reactive protein; RAAS: renin–angiotensin–aldosterone system.
None: the patients never took anti-diabetic drugs. Both: the patients were taking oral drugs and subcutaneous injection of insulin at the same time. Data are presented as mean ± standard deviation or numbers (percentages).
p < 0.05, **p < 0.01, compared with CKD 1 stage.
p < 0.05, compared with CKD 2 stage.
Characteristics of the participants without albuminuria among CKD stages (n = 615).
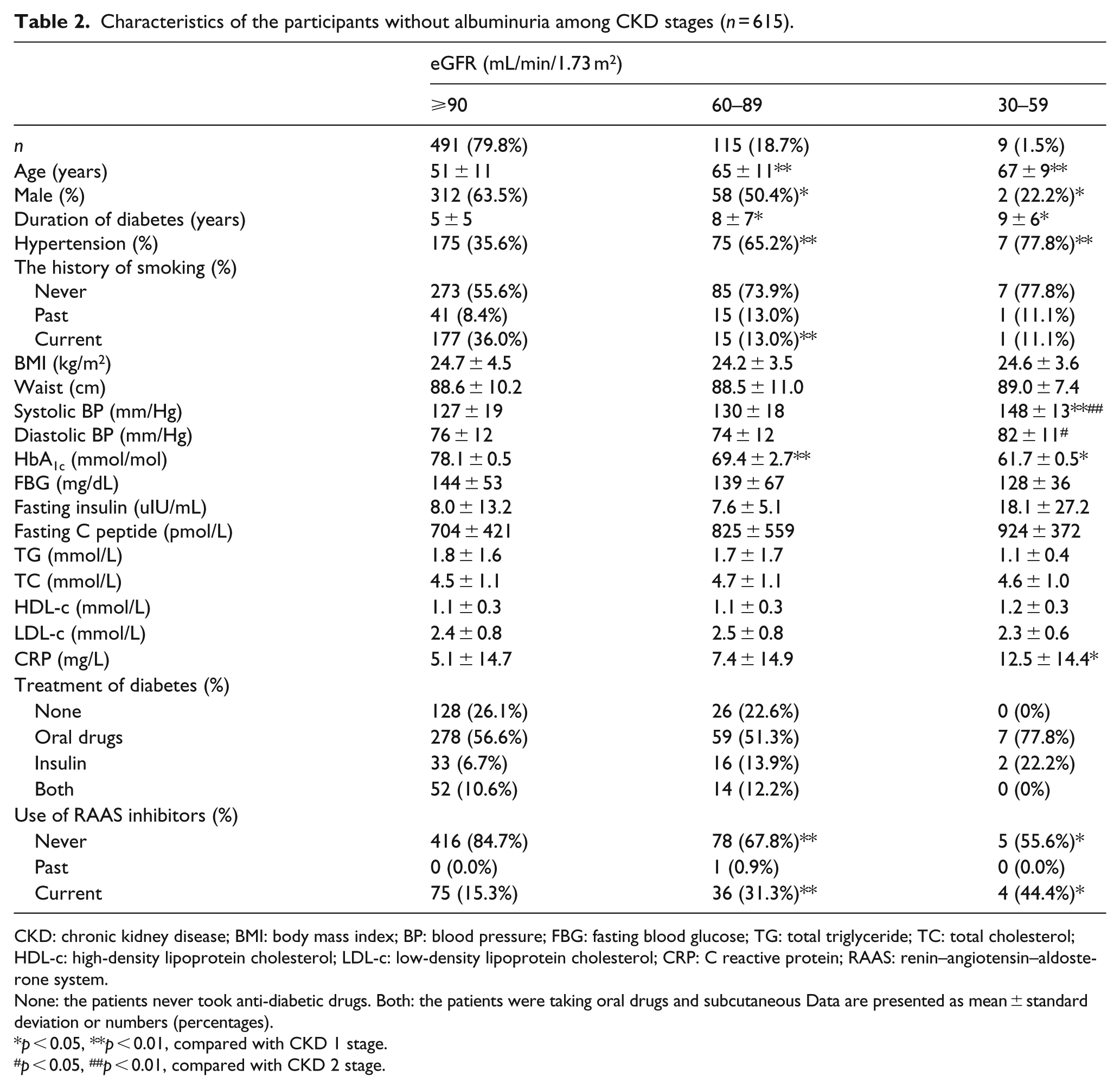
CKD: chronic kidney disease; BMI: body mass index; BP: blood pressure; FBG: fasting blood glucose; TG: total triglyceride; TC: total cholesterol; HDL-c: high-density lipoprotein cholesterol; LDL-c: low-density lipoprotein cholesterol; CRP: C reactive protein; RAAS: renin–angiotensin–aldosterone system.
None: the patients never took anti-diabetic drugs. Both: the patients were taking oral drugs and subcutaneous Data are presented as mean ± standard deviation or numbers (percentages).
p < 0.05, **p < 0.01, compared with CKD 1 stage.
p < 0.05, ##p < 0.01, compared with CKD 2 stage.
Characteristics of the participants with albuminuria among CKD stages (n = 235).
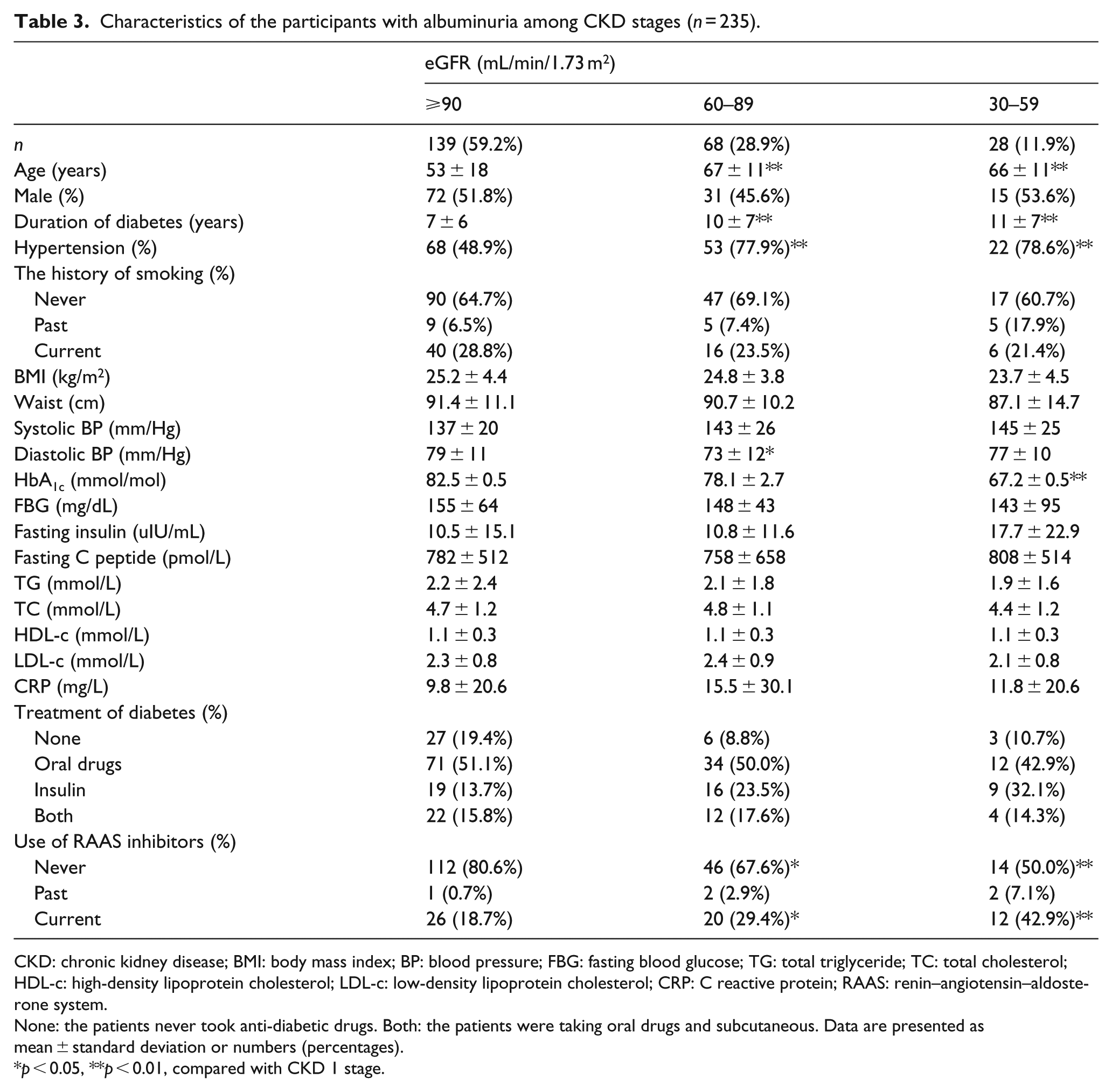
CKD: chronic kidney disease; BMI: body mass index; BP: blood pressure; FBG: fasting blood glucose; TG: total triglyceride; TC: total cholesterol; HDL-c: high-density lipoprotein cholesterol; LDL-c: low-density lipoprotein cholesterol; CRP: C reactive protein; RAAS: renin–angiotensin–aldosterone system.
None: the patients never took anti-diabetic drugs. Both: the patients were taking oral drugs and subcutaneous. Data are presented as mean ± standard deviation or numbers (percentages).
p < 0.05, **p < 0.01, compared with CKD 1 stage.
Prevalence of diabetic retinopathy and neuropathy in stage 1–3 CKD with or without albuminuria
From stage 1 to 3, the prevalence of diabetic retinopathy in albuminuric subjects (Figure 1) increased gradually from 42.5%, 56.6% to 66.7% (p < 0.05 compared with stage 1). In normoalbuminuric subjects (Figure 2), the prevalence of diabetic retinopathy was relatively lower (29.4%, 33.0% and 50.0% in stage 1–3) compared with that in albuminuric subjects (p = 0.005 in stage 1, p = 0.007 in stage 2 and p = 0.399 in stage 3, respectively), and there was no statistically significant trend between different CKD stages. Compared with the normoalbuminuric subjects (Figure 2), those with albuminuria (Figure 1) had higher prevalence of diabetic neuropathy in each CKD stage. The prevalence of diabetic neuropathy increased with declining eGFR (48.9%, 51.5% and 57.1%), but there was no statistically significant trend.
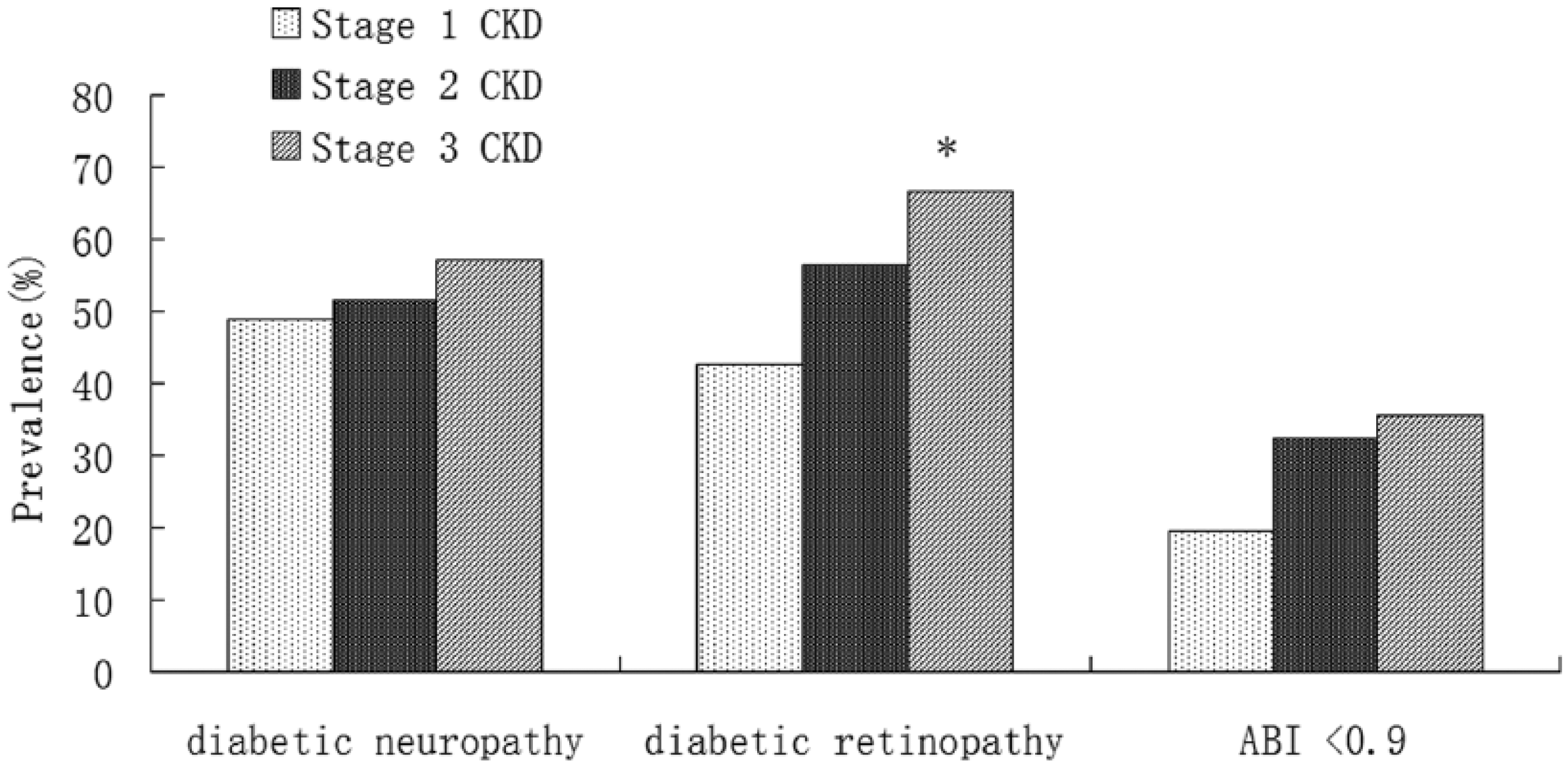
Prevalence of diabetic neuropathy, retinopathy and low ABI in patients with albuminuria among CKD stages.
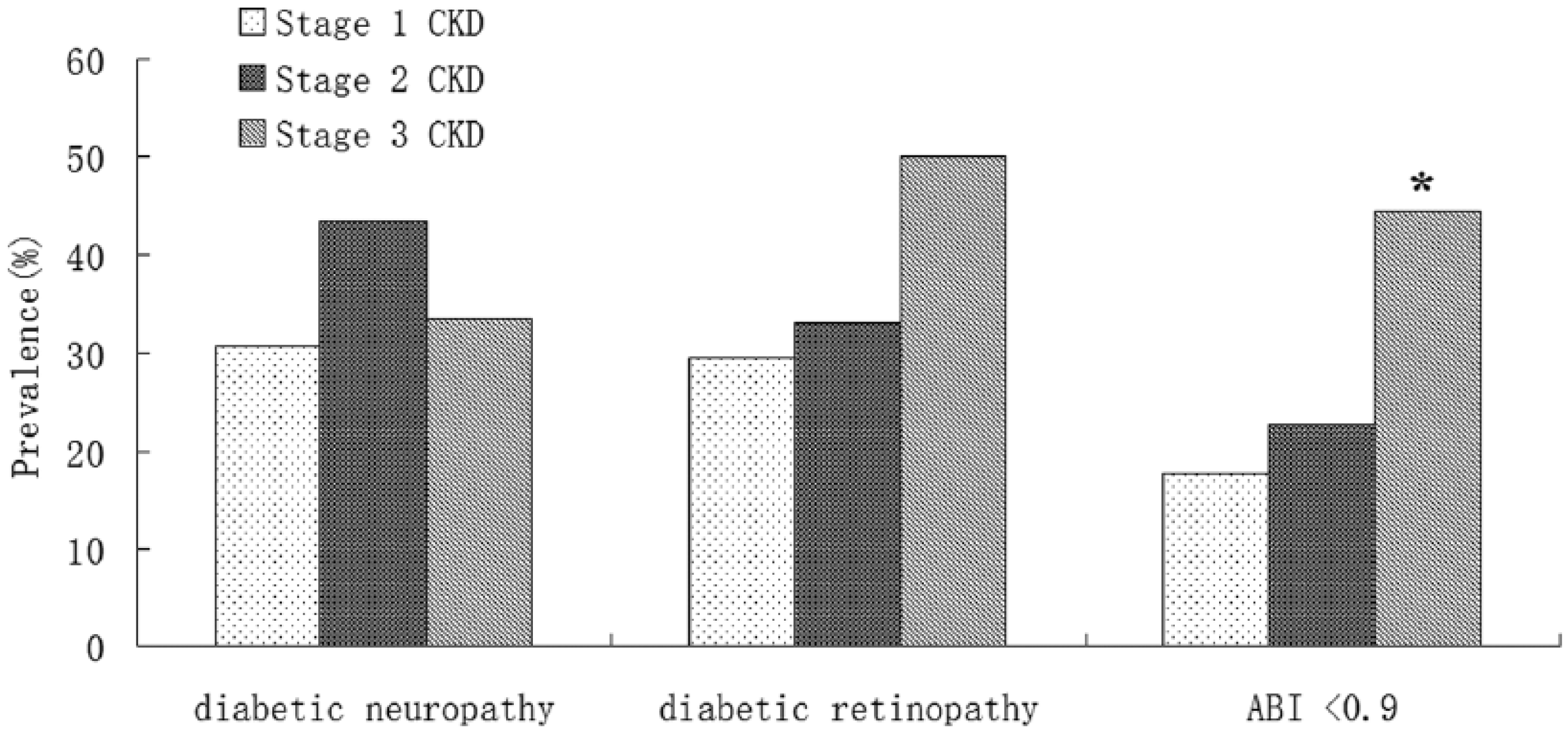
Prevalence of diabetic neuropathy, retinopathy and low ABI in patients without albuminuria among CKD stages.
Prevalence of Low ABI in stage 1–3 CKD with or without albuminuria
There was a significantly increased prevalence of low ABI from stage 1 to 3 (17.5%, 22.6% and 44.4%) in normoalbuminuric subjects (Figure 2), while there was a similar but no significant trend of low ABI in each CKD stage (19.4%, 32.4% and 35.7%) in those with albuminuria (Figure 1). Low–normal ABI (0.9–0.99) prevalence was higher in each stage of normoalbuminuric subjects than in those with albuminuria (data not shown), especially in patients with eGFR 60~89 (30.4% vs 16.2%).
Risk factors of declining kidney function in patients with or without albuminuria
In patients with albuminuria, logistic regression analysis showed that age (OR = 1.099, 95% CI = 1.049–1.149, p = 0.000) and hypertension (OR = 3.124, 95% CI = 1.075–9.079, p = 0.036) were independent risk factors of declining kidney function, while age (OR = 1.151, 95% CI = 1.112–1.194, p = 0.000) was found to be the only risk factor of CKD worsening in those without albuminuria (Table 4). Importantly, after controlling for age, sex, smoking status, diabetes duration, body mass index, HbA1c, use of renin–angiotensin–aldosterone system inhibitors, hypertension and dyslipidaemia history and other complications, diabetic retinopathy (adjusted OR = 2.474, 95% CI = 1.009–6.068, p = 0.048) still showed close relation of declining kidney function in patients with albuminuria. On the other hand, low ABI failed to show significant relation with declining kidney function in those without albuminuria. HbA1c was not significantly associated with CKD worsening after adjustment by the haemoglobin level and use of insulin in non-albuminuric patients (p = 0.087).
The relative risk factors of mild to moderate CKD with or without albuminuria.
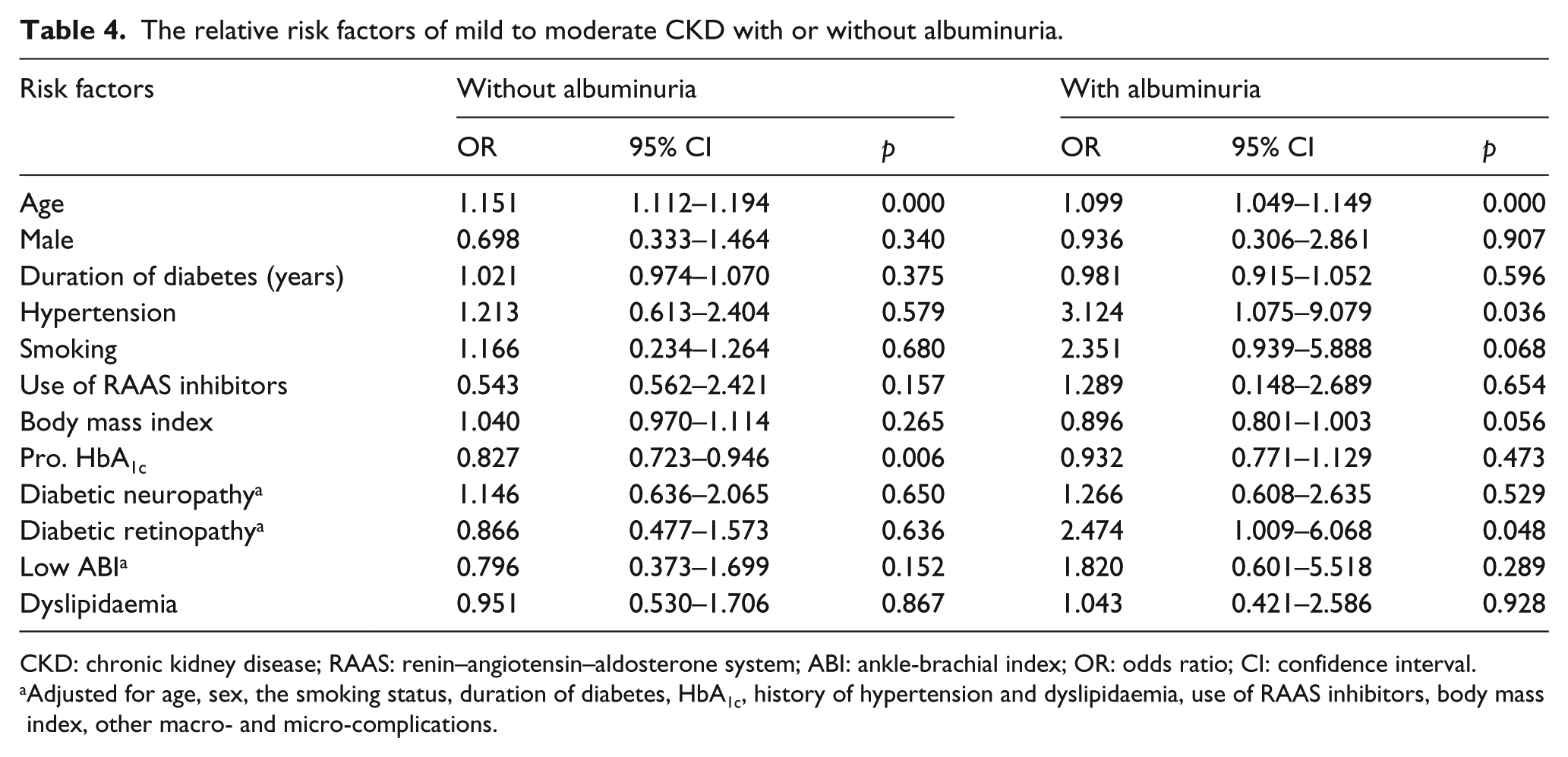
CKD: chronic kidney disease; RAAS: renin–angiotensin–aldosterone system; ABI: ankle-brachial index; OR: odds ratio; CI: confidence interval.
Adjusted for age, sex, the smoking status, duration of diabetes, HbA1c, history of hypertension and dyslipidaemia, use of RAAS inhibitors, body mass index, other macro- and micro-complications.
Discussion
In this study, we investigated the entire spectrum of patients with or without albuminuria among the CKD stage 1–3 and showed that the prevalence of diabetic retinopathy increased as eGFR decreased in albuminuric patients, while the prevalence of low ABI increased as eGFR decreased in normoalbuminuric patients, indicating the diverse underlying mechanisms of CKD between these two phenotypes. To the best of our knowledge, this is the first study to demonstrate the different performance of macro- and microvascular complications in mild to moderate CKD between type 2 diabetic patients with or without albuminuria.
There have been a limited number of studies on the risk factors of type 2 diabetic patients with reduced eGFR and normoalbuminuria. Yokoyama et al. 17 showed that age, prevalence of women, duration of diabetes and frequencies of hypertension and hyperlipidaemia were significantly higher in CKD stage 3+4 than CKD stage 1+2 among the normoalbuminuric patients. They also examined the frequencies of diabetic complications and reported that CVD were more frequent in CKD stage 3+4 compared with CKD stage 1+2; however, the frequencies of diabetic retinopathy and neuropathy were not different between the two groups. Conversely, another cross-sectional study in Japan 18 reported diabetic neuropathy and macroangiopathies were significantly more frequent in the normoalbuminuric patients with CKD stage 3+4 than in those with CKD stage 1+2. The different results of these two researches in Japan were considered to be due to the different ages of the study subjects (62 vs 70.8 years old, respectively). 18 The present study showed the prevalence of low ABI, but not the prevalence of diabetic retinopathy and neuropathy, gradually increased in normoalbuminuric patients and significantly higher in Chinese patients with CKD stage 3 than in those with CKD stage 1. Our findings further support the hypothesis that macroangiopathy may play a crucial role in the non-albuminuric phenotype. However, low ABI failed to be an independent risk factor after controlling for age, sex, smoking status, diabetes duration, body mass index, HbA1c, use of renin–angiotensin–aldosterone system inhibitors, hypertension and dyslipidaemia history and other complications. Age was the only risk of declining kidney function in type 2 diabetic patients without albuminuria. It should be noted that the present study was performed in type 2 diabetic patients with mild to moderate CKD because of the relatively low prevalence of severe kidney damage in China, especially in hospitalized diabetic patients. The difference of our results from the previous studies may have been due to the fact that 74.4% of the subjects were younger (average age: 51 years) and had a shorter average diabetes duration (5 years) and a higher average eGFR level. Moreover, CVD was considered to have a strong association with low ABI and eGFR, 20 but a lack of association with diabetic retinopathy, 21 which may have confused the results in previous studies. The different results could also be explained by the exclusion of CVD from the present study.
To date, few studies have compared macro- and microvascular complications in subjects with type 2 diabetes and early reduced eGFR with or without albuminuria, and as a result, the underlying mechanisms of CKD remain poorly understood. The relation of diabetic retinopathy and development of albuminuria has been well established by UK Prospective Diabetes Study. 8 However, there have been no sufficient data to describe the role of microvascular complications in declining kidney function. Our present study showed the prevalence of diabetic retinopathy, but not diabetic neuropathy and low ABI, was graded among the CKD stages in albuminuric patients. What’s more, diabetic retinopathy had a close relation of declining kidney function in patients with albuminuria besides age and hypertension, even after adjustment by the other risk factors of kidney function impairment. Together, our findings indicate that low eGFR could be due to age-associated declining of kidney function in both albuminuric and non-albuminuric patients, and more importantly, microvascular involvement prevails in the albuminuric phenotype, while macroangiopathy seems to play a crucial role in the non-albuminuric phenotype. This finding was in concordance with a previous biopsy study in diabetic patients with eGFR of <60 (mean eGFR of 31 mL/min/1.73 m2). The study demonstrated typical glomerular changes in patients with micro- or macroalbuminuria compared with varying degrees of arteriosclerosis in those with normoalbuminuria. 29 Further biopsy studies in early-stage CKD with or without albuminuria will be needed to clarify the initial renal structural changes.
Lower HbA1c values were observed in those with low eGFR than in those with preserved eGFR in our study, which was consistent with other studies. 17 This could be due to a reduced erythropoietin production caused by reduced renal function 30 and the intensive treatment. After adjustment by the haemoglobin level and use of insulin, the relation of lower HbA1c and declining kidney function disappeared.
The main limitation of the present study is the cross-sectional design and the single-centre data with relatively small sample size, especially the small proportion of participants with eGFR 30–59, which may explain why low ABI failed to become an independent risk factor of decreased eGFR in non-albuminuric patients. Thus, prospective studies of a larger sample should be conducted and the primary outcome of end-stage renal disease or CVD incidence should be used. Other possible limitations include the hospital-based study cohort making selection bias a potential confounding factor and the included patients with medications of the renin–angiotensin–aldosterone system inhibitors which may confuse the results. Multiple trials have demonstrated that the renin–angiotensin–aldosterone system inhibitors have a renoprotective effect.31–33 Unfortunately, less than 50% of the patients with albuminuria as well as hypertension received renin–angiotensin–aldosterone system inhibitors in our study. This was consistent with the latest multicentre study in China, which had shown more than half of diabetic patients coexisting with hypertension and/or albuminuria in China were not treated with this kind of drugs, suggesting that more educational efforts should be made to the physicians. 34 However, patients treated with renin–angiotensin–aldosterone system inhibitors in our data still assumed a definite proportion and seemed impossible to be excluded or ignored. An important methodological strength of the current study is that we determine the entire spectrum of patients in mild to moderate CKD with or without albuminuria using the reliable evaluations of macro- and microvascular disease, thereby indicating the discordancy of macro- and microvascular complications in different phenotypes of CKD. The other strength of the study, as opposed to previous population-based studies, is concerning and therefore excluding the patients with CVD, hence avoiding the confounding factors.
In conclusion, the prevalence and the important roles of macro- and microvascular complications in mild to moderate CKD patients of type 2 diabetes with or without albuminuria have been well defined in our study, implying the diverse underlying mechanisms of these two phenotypes and hence the targeted treatments in clinical work.
Footnotes
Acknowledgements
Study designed and supervised by X. D., D. W. and Y. X. ABI assessment performed by C. J. and J. L. Data analysed by X. D., Y. Z., D. W. and Y. X. Data researched by Y. X., Y. R., X. F. and J. L. Statistical analysis performed by and paper written by D. W., Y. Z., L. L. and X. D. Paper revised and reviewed by X. D., L. L. and H. L. D. W. and Y. X. contributed equally.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by grants from the National Natural Science Foundation of China for young scientists [30900703], the Zhejiang Natural Science Foundation [Y15H070010] and the National Natural Science Foundation of China [81170759].
