Abstract
Background and method:
Increased plasma clot density and prolonged lysis times are associated with cardiovascular disease. In this study, we employed a functional proteomics approach to identify novel clot components which may influence clot phenotypes.
Results:
Analysis of perfused, solubilised plasma clots identified inflammatory proteins, including complement C3, as novel clot components. Analysis of paired plasma and serum samples confirmed concentration-dependent incorporation of C3 into clots. Surface plasmon resonance indicated high-affinity binding interactions between C3 and fibrinogen and fibrin. Turbidimetric clotting and lysis assays indicated C3 impaired fibrinolysis in a concentration-dependent manner, both in vitro and ex vivo.
Conclusion:
These data indicate functional interactions between complement C3 and fibrin leading to prolonged fibrinolysis. These interactions are physiologically relevant in the context of protection following injury and suggest a mechanistic link between increased plasma C3 concentration and acute cardiovascular thrombotic events.
Introduction
Cardiovascular disease (CVD) is a major cause of morbidity and mortality, accounting for over one third of all deaths in Western societies, with acute thrombosis precipitating the common clinical manifestations, myocardial infarction and ischaemic stroke. Alterations in factors influencing the delicate balance between coagulation and anticoagulation, activation/inhibition of fibrinolysis and platelet activation/inhibition may influence cardiovascular outcomes by affecting the extent of thrombosis. A variety of haemostatic abnormalities, including increased fibrinogen and plasminogen activator inhibitor-1 (PAI-1) have been associated with increased risk for acute thrombotic events.1,2 Altered fibrin clot properties have also been related to CVD, with denser ex vivo clot structures correlated with severity of coronary artery stenosis 3 and associated with premature myocardial infarction. 4 In addition, ex vivo clot density and impaired fibrinolysis are observed in healthy relatives of patients with CVD5,6 and in subjects with the metabolic syndrome, 7 suggesting that alterations in ex vivo clot structure and susceptibility to fibrinolysis pre-date the development of CVD.
Fibrinogen concentration is a major determinant of clot density and susceptibility to fibrinolysis; however, a variety of other plasma proteins are known to be incorporated into fibrin clots, via direct or indirect binding or by activated FXIII (FXIIIa)-dependent cross-linking, and influence aspects of fibrin structure and function.8–10 Identification and characterisation of novel clot proteins may further understanding of the factors which influence clot density and susceptibility to fibrinolysis and potentially identify novel risk factors for thrombosis.
In this study we applied a functional proteomics approach to identify and validate novel plasma clot components.
Materials and methods
Proteomic analysis of plasma clots
Pooled plasma preparation
Venous blood from 20 medication-free healthy volunteers (10 male, 10 female; from whom consent was obtained according to a protocol approved by the Leeds NHS Trust research ethics committee) was collected into 0.109 M trisodium citrate and centrifuged at 3000g for 30 min. Plasma was pooled, aliquoted and stored at −80°C.
Plasma clot preparation, gel electrophoresis and silver staining
Plasma clots were formed by addition of 10 µl activation mix (1 U/ml human thrombin (Calbiochem, Damstadt, Germany), 10 mM CaCl2, final concentrations) to 50 µl pooled plasma, with the polymerising mixture incubated in open tubes in a humidified chamber, based on a previously published method 5 , for 4 h at 37°C. Non-specifically incorporated proteins were removed by perfusion with Tris buffer (0.05 M Tris, 0.10 M NaCl, pH 7.4).
Proteins were solubilised in 100 mM dithiothreitol and separated by one-dimensional gel electrophoresis (1-DE; SDS-PAGE) on NuPAGE® 3–8% Tris-acetate gels (Invitrogen, New York, USA) or by 2-DE on 7 cm NuPAGE® 4–12% bis-Tris gels following separation into narrow-range pH fractions using the ZOOM®IEF Fractionator, according to manufacturer’s guidelines (Invitrogen, New York, USA). Proteins were visualised by silver staining, according to previously published methods. 11
Gel imaging, protein visualisation and MS identification
Stained gels were scanned on a Densitometer-GS800® (BioRad, Hemel Hempstead, UK). Protein profiles of three independent experiments were compared using PDQuest® (v7.1.1; Bio-Rad, Hemel Hempstead, UK) and a ‘master’ gel created to identify protein spots/bands of interest. Bands from 1-D gels were excised and cut into ~1 mm pieces. Spots from 2-DE gels were excised using a OneTouch Plus 1.5 mm spot picker (Web Scientific Ltd, Crewe, UK). Gel pieces were subjected to automated trypsin digestion using a MassPREP workstation (Waters Ltd, Elstree, UK), including destaining, reduction/alkylation, digestion with trypsin and plate spotting, according to manufacturer’s recommended guidelines. The dried plate was transferred to a matrix-assisted laser desorption ionisation mass spectrometer (MALDI-MS) (MALDI LR, Waters Ltd) and each digest analysed in reflectron mode, according to previously published methods. 11 The initial peak list for each sample was manually edited to exclude common keratin and trypsin autolysis peaks and the optimised set of sample-specific monoisotopic peptide masses used to search the SwissProt human database (v57.2, 20402 sequences) using the Mascot search engine to identify the parent protein. Searches were performed using an unrestricted protein molecular mass range, variable modifications of carbamidomethyl-Cys and oxidised-Met (and propionamido-Cys for 1-DE bands), allowing one missed tryptic cleavage site and 100 ppm error tolerance in the peptide mass. Identifications were accepted on the following criteria: (i) at least five matching peptides; (ii) a MOWSE score of more than 56 (significance threshold p < 0.05); and (iii) sequence coverage >8%.
Validation of complement C3 as a novel clot component
C3 quantification and activity analysis
C3 antigen was analysed by enzyme-linked immuosorbent assay (ELISA) using antibodies from DAKO (Ely, UK). 12 C3 activity was analysed by haemolytic assay according to the method of Jessen et al. 13
C3and factor H incorporation into plasma clots
The concentration of C3 and factor H in paired plasma and plasma-derived serum samples, formed according to a previously reported method, 14 was determined by C3 ELISA or factor H ELISA (antibodies from Quidel, San Diego, USA) and C3 and factor H incorporation into plasma clots was calculated.
Plasma C3 purification
C3 was purified from EDTA plasma using a method described by Tack and Prahl. 15 The C3 obtained was >95% pure as assessed by SDS-PAGE and free from C5 as assessed by western blotting with anti-C5 antibody (Quidel). C3 was quantified by ELISA and was functionally active by haemolytic assay, retaining 43.8% of the initial activity of the starting plasma.
Analysis of binding interactions by surface plasmon resonance
Surface plasmon resonance (SPR) experiments were performed using a Biacore 3000 (GE Healthcare, Little Chalfont, UK) and SPR running buffer (150 mM NaCl, 20 mM HEPES, 1.5 mM CaCl2, 0.005% v/v P20, pH 7.4). Briefly, 500 response units (RU) of affinity-purified fibrinogen 16 or 150 RU of purified C3 was directly immobilised on a CM5 sensor chip by amine coupling, according to the manufacturer’s instructions (GE Healthcare). One fibrinogen surface was converted to fibrin as previously described. 17 A two-fold serial dilution of 0.7–100 nM purified C3 or fibrinogen was injected in ascending order for 120 s (flow rate 30 µl/min) and the surface regenerated (20 s, 5 mM NaOH, 100 mM NaCl) and re-equilibrated in running buffer. Kinetic analysis was performed using BIAevaluation software 4.1 (GE Healthcare, Little Chalfont, UK) with reference-subtracted and buffer blank-subtracted data. Experiments were performed in triplicate.
Turbidimetric assays of fibrin clot density and lysis
The influence of C3 on clot density was assessed by turbidimetric clotting assays in 96-well plates using affinity-purified fibrinogen (0.5 mg/ml), FXIII (5.5 µg/ml; gel-filtration purified from Fibroammin P (CSL; Behring, Haywards Heath, UK)) and purified C3 (final concentrations 0, 0.15, 0.35, 0.55 and 0.75 mg/ml) in Tris buffer (final volume 100 µl). Clot formation was initiated by 50 µl of a thrombin/Ca2+ activation mix (final concentrations: 0.025 U/ml thrombin, 5 mM CaCl2 in Tris buffer). Plates were read every 12 s at 340 nm for 1 h (BIO-TEK ELx808 spectrophotometer; Potton, UK). Three independent experiments were carried out. The influence of C3 on fibrinolysis was assessed by turbidimetric assay. Clots formed at the end of the turbidimetric clotting assay were overlaid with 100 µl of lysis mix (100 ng/ml tPA (Pathway Diagnostics, Dorking, UK), 50 µg/ml purified plasminogen (purified by lysine–sepharose affinity chromatography during the C3 purification process) in Tris buffer), incubated for 1 h and the plate read every 108 s at 340 nm for 9 h. The influence of factor H on clot density and fibrinolysis was carried out using similar methods employing purified factor H (Quidel; final concentrations 25, 50, 75 and 100 µg/ml). Three independent experiments were carried out.
Ex vivo fibrin structure and fibrinolysis were previously analysed in stored plasma from participants in the Leeds Family study (informed consent was obtained according to a protocol approved by the Leeds Teaching Hospitals Trust research ethics committee) using high-throughput turbidimetric assays, as described. 7 Complement C3 was analysed by ELISA and the relationships between C3 and measures of clot structure and function were evaluated after accounting for significant haemostatic determinants. The following measures were evaluated: clot density (maximum absorbance, MaxAbs), 50% lysis time and area under the lysis curve (AUC).
Statistical analyses
Statistical analyses were carried out using SPSS v12.0 (SPSS Inc., New York, USA). Differences between groups were assessed by independent samples t-test or one-way ANOVA, with Bonferroni adjustment for multiple comparisons. The relationship between quintiles of C3 and measures of ex vivo plasma clot structure and fibrinolysis was assessed by linear regression analyses after accounting for pedigree, age, sex and known haemostatic determinants.
Results
Proteomic analysis of perfused solubilised ex vivo plasma clots identified inflammatory proteins including complement C3 as novel clot components
Using a 2-DE approach, solubilised clot components were subjected to pre-fractionation into narrow pI ranges prior to 2-DE (Figure 1A–D). As expected, fibrin α, β and γ chain products were the predominant protein isoforms detected in all gels (data not shown). Only 13 non-fibrin proteins were consistently observed by 2-DE (Figure 1A–D and Table 1; further details are provided in the Supplementary Information).
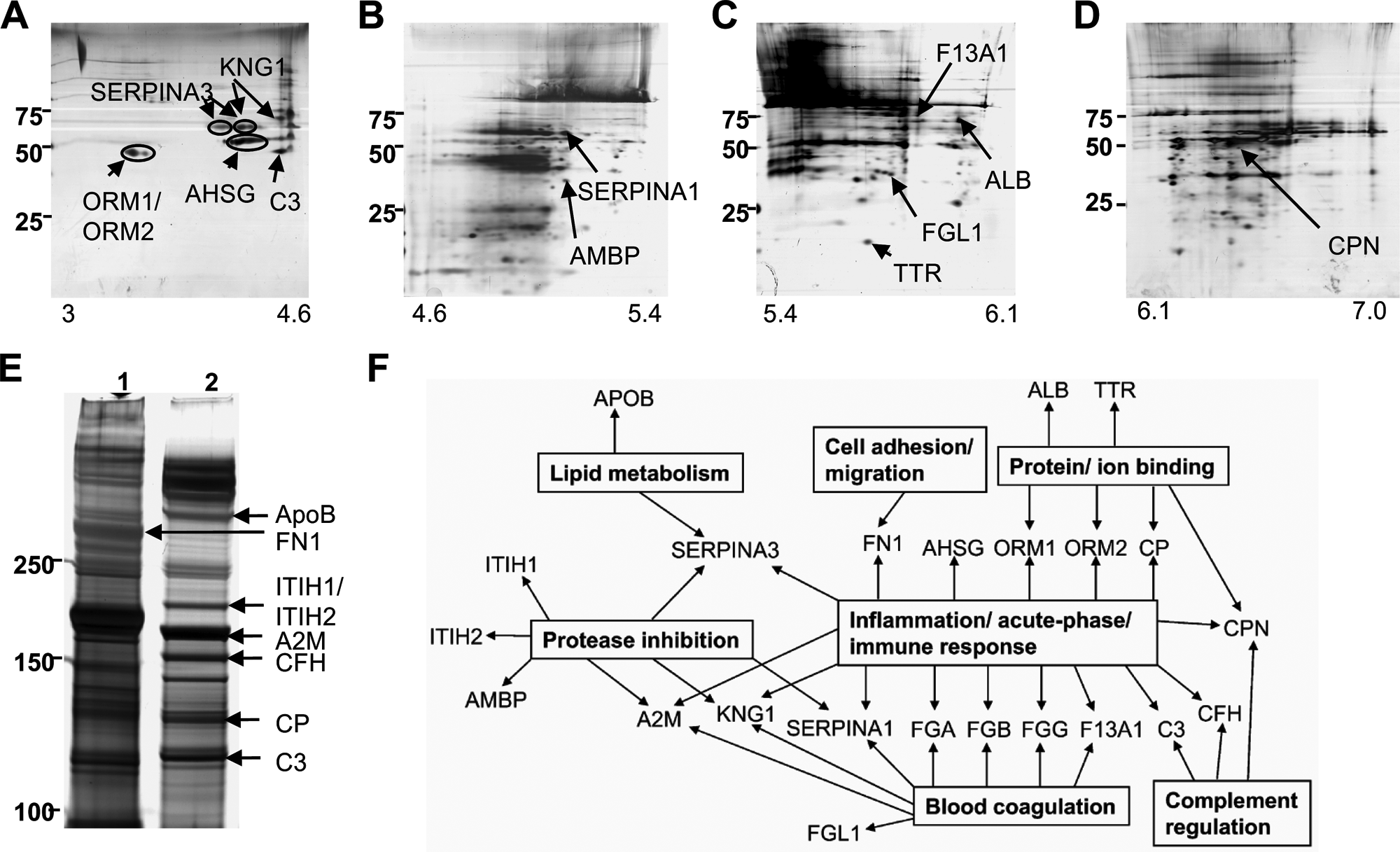
Proteomic analysis of perfused, solubilised plasma clots. (A–D) Two-dimensional polyacrylamide gel electrophoresis of solubilised plasma clot components (NuPAGE® 4–12% Bis-Tris gels): (A) pH 3.0–4.6, (B) pH 4.6–5.4, (C) pH 5.4–6.1, (D) pH 6.1–7.0. (E) SDS-PAGE of high molecular weight proteins (NuPAGE® 3–8% Tris-acetate gels). Lane 1: solubilised clot formed from pooled normal plasma; lane 2: solubilised clot formed from factor XIII-deficient plasma. Gels are representative of a minimum of three independent experiments. Positions of MW markers are shown. Bands and spots were identified using MALDI-MS peptide mass fingerprinting (see Tables 1 and 2 for protein identification). (F) Gene ontology analysis of plasma clot proteins (using DAVID18,19).
Protein identifications from two-dimensional gel electrophoresis spots using MALDI/TOF-MS peptide mass fingerprinting.
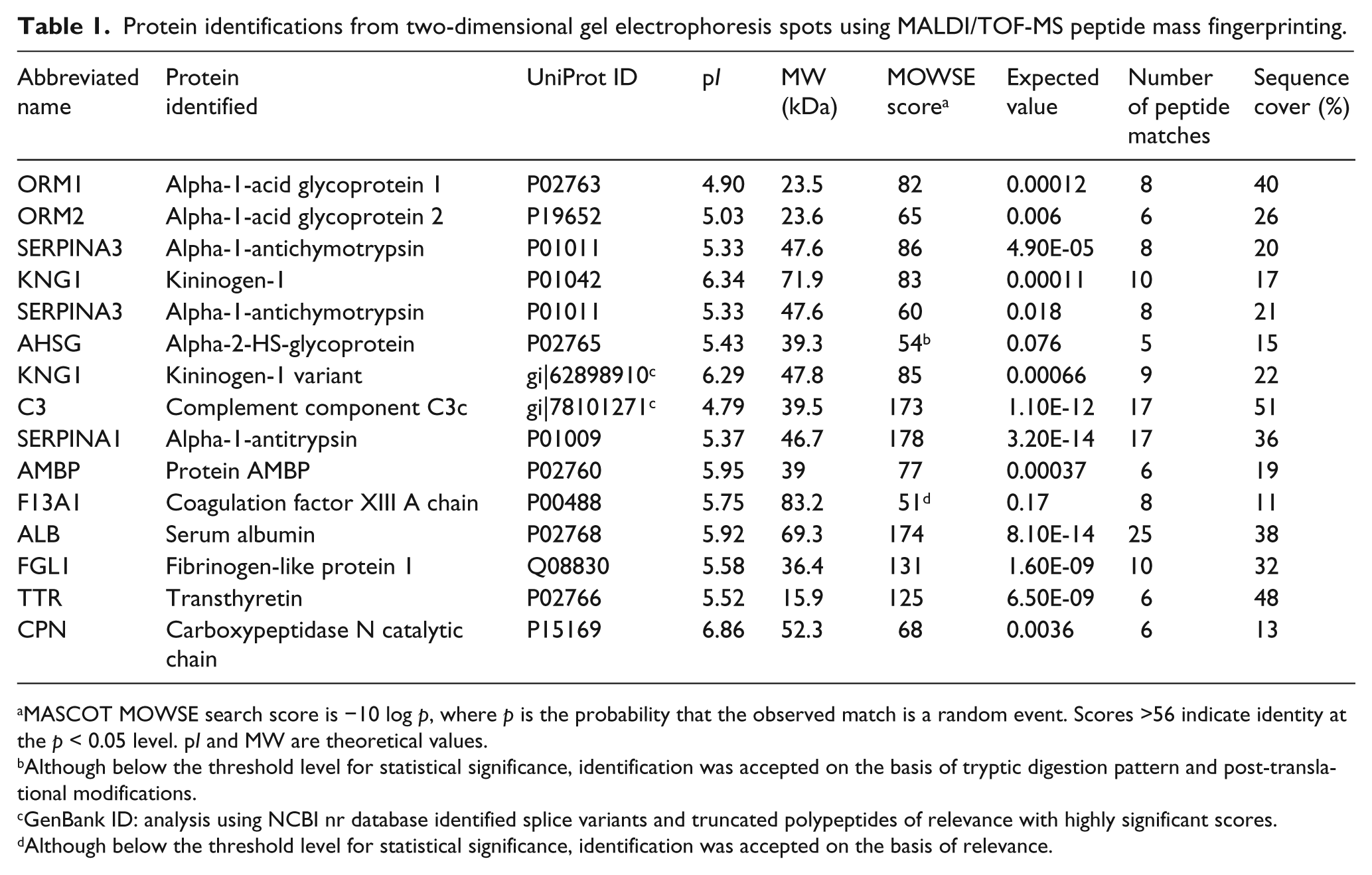
MASCOT MOWSE search score is −10 log p, where p is the probability that the observed match is a random event. Scores >56 indicate identity at the p < 0.05 level. pI and MW are theoretical values.
Although below the threshold level for statistical significance, identification was accepted on the basis of tryptic digestion pattern and post-translational modifications.
GenBank ID: analysis using NCBI nr database identified splice variants and truncated polypeptides of relevance with highly significant scores.
Although below the threshold level for statistical significance, identification was accepted on the basis of relevance.
Protein identifications from one-dimensional gel electrophoresis bands using MALDI/TOF-MS peptide mass fingerprinting.
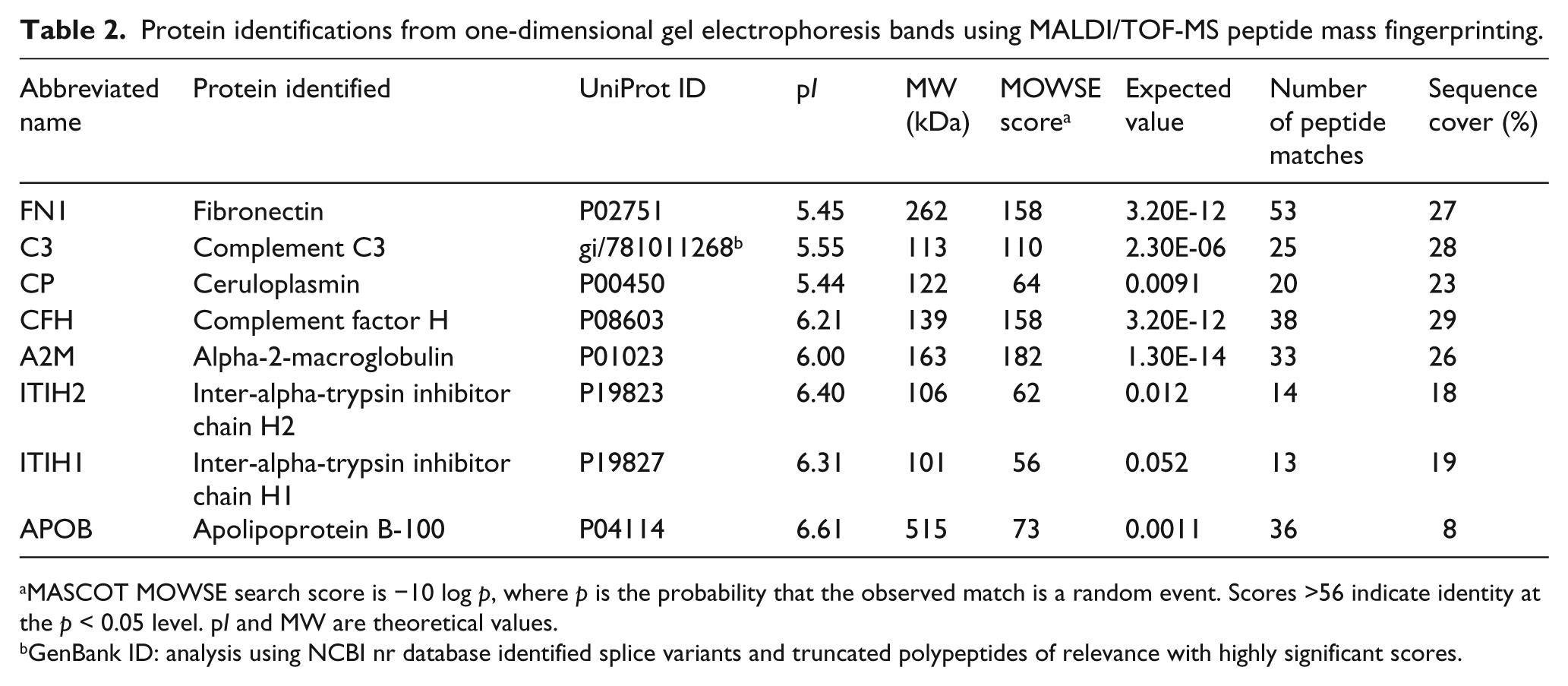
MASCOT MOWSE search score is −10 log p, where p is the probability that the observed match is a random event. Scores >56 indicate identity at the p < 0.05 level. pI and MW are theoretical values.
GenBank ID: analysis using NCBI nr database identified splice variants and truncated polypeptides of relevance with highly significant scores.
The cross-linking of fibrin α chains and other plasma proteins during clot formation resulted in the formation of numerous high molecular weight (HMW) protein isoforms, which were poorly resolved by 2-DE (Figure 1A–D). Therefore, 1-DE was also performed to maximise the separation of cross-linked and HMW clot components, with increased resolution of discrete bands in the HMW range (Figure 1E, lane 1). Once again fibrin α and γ chains were the predominant protein isoforms detected; the only non-fibrin protein that could be reliably identified was fibronectin (Figure 1E, lane 1, and Table 2; further details are provided in the Supplementary Information), a known clot component and substrate for FXIIIa. To facilitate the identification of HMW proteins capable of binding to fibrin clots, clots formed from FXIII-deficient plasma were also analysed by 1-DE (Figure 1E, lane 2) and eight HMW non-fibrin protein components were identified (Figure 1E and Table 2; further details are provided in the Supplementary Information).
Gene ontology analysis18,19 indicated that of the 20 non-fibrin proteins identified in plasma clots, eight proteins are known to play a role in haemostasis and 16 proteins are involved in inflammatory processes, in particular response to injury including the acute phase and immune responses (Figure 1F). Other protein clusters identified included protease inhibitors and binding proteins and several proteins belonged to more than one functional class (Figure 1F).
C3 is incorporated into plasma clots
To validate C3 as a clot component, we quantified the incorporation of C3 into plasma clots, by comparing C3 concentrations in paired plasma and plasma-derived serum samples (n = 82). The concentration of C3 within clots increased with increasing plasma C3 concentration (Figure 2A), although the proportion of plasma C3 incorporated into the clot was independent of concentration (R2 = 0.065) with a mean C3 incorporation into fibrin clots of 25.7% (SD 7.5).
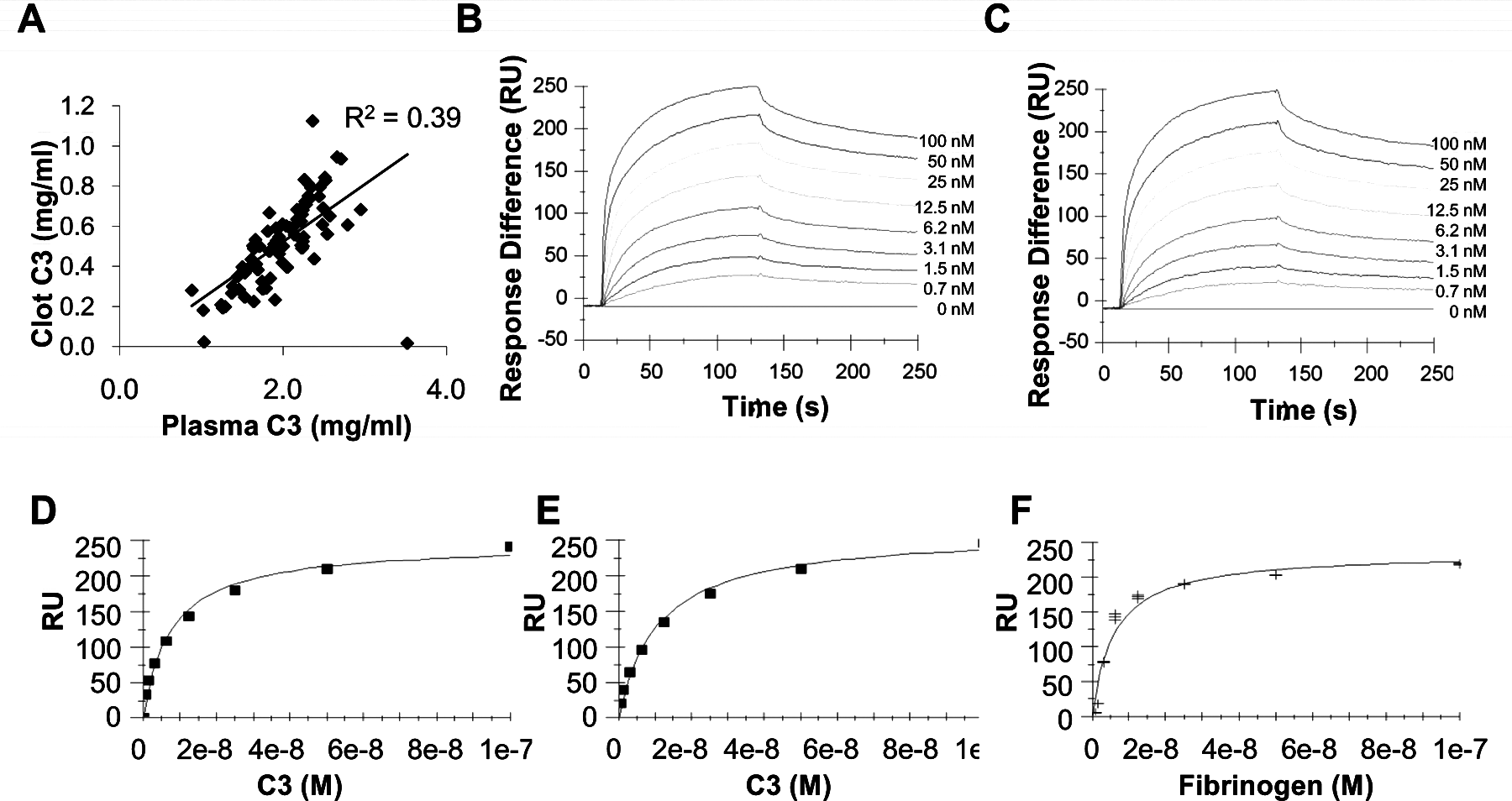
Complement C3 interactions with fibrin. (A) The concentration of C3 in plasma and plasma-derived fibrin clots. (B–F) SPR analysis of C3 interactions with fibrin(ogen). (B and D) C3 (0.7–200 nM) interactions with immobilised fibrinogen; (C and E) C3 (0.7–200 nM) interactions with immobilised fibrin; (F) fibrinogen (0.7–200 nM) interactions with immobilised C3. Data are fitted to a heterogeneous ligand (B and C) and steady state affinity (D–F) models. Three independent experiments.
C3 binds to fibrinogen and fibrin with high affinity
High affinity binding interactions were identified by SPR, with concentration-dependent, saturable binding of C3 to both immobilised fibrinogen and fibrin (Figure 2B and C). Binding data did not fit a 1:1 interaction model whereas heterogeneous ligand model analysis indicated the presence of two high-affinity binding sites for C3 on both fibrinogen and fibrin (fibrinogen: site 1 KD 5.0 ± 6.2 nM, site 2 KD 0.7 ± 0.19 nM; fibrin site 1 KD 4.9 ± 3.6 nM, site 2 KD 0.7 ± 0.3 nM; shown in Supplementary Table S1). Binding analyses performed in the reverse orientation confirmed concentration-dependent, saturable, high-affinity binding between fibrinogen and immobilised C3 although interactions were not consistent with predefined kinetic models (data not shown). To obtain approximate K D values for comparison of interactions in both orientations, data were fitted to steady state models (Figure 2D–F) which indicated K D values for the interactions between C3 and immobilised fibrinogen and fibrin of 9.5 (± 1.9) nM and 11.0 (± 1.8) nM respectively and between fibrinogen and immobilised C3 of 6.0 (± 0.2) nM (Supplementary Table S1).
C3 prolongs fibrinolysis
To determine whether incorporation of C3 into fibrin clots influenced fibrin structure/function, turbidimetric clotting and fibrinolysis assays were performed. Increasing C3 concentration did not significantly influence the kinetics of clot formation or clot density (MaxAbs), as shown in Figure 3A and B. In contrast, fibrinolysis times increased significantly with increasing concentrations of C3 (Figure 3C and D). A similar relationship was identified between plasma C3 and ex vivo fibrin phenotypes in the Leeds Family Study, after adjustment for age, sex, pedigree and previously identified 7 haemostatic determinants (fibrinogen, FXIII, PAI-1 and tPA); there was no significant association between increasing quintiles of plasma C3 and plasma clot density (MaxAbs; Figure 3E), whereas 50% lysis time and AUC were independently associated with increasing C3 concentration (Figure 3F and G, p = 0.02 and p = 0.01 respectively). To further validate our in vitro analyses we evaluated the effect of spiking normal pooled plasma with purified C3 to give concentrations within the normal range and confirmed that fibrinolysis times are prolonged with increasing concentrations of exogenous C3 (see Supplementary Figure S1, panel A).
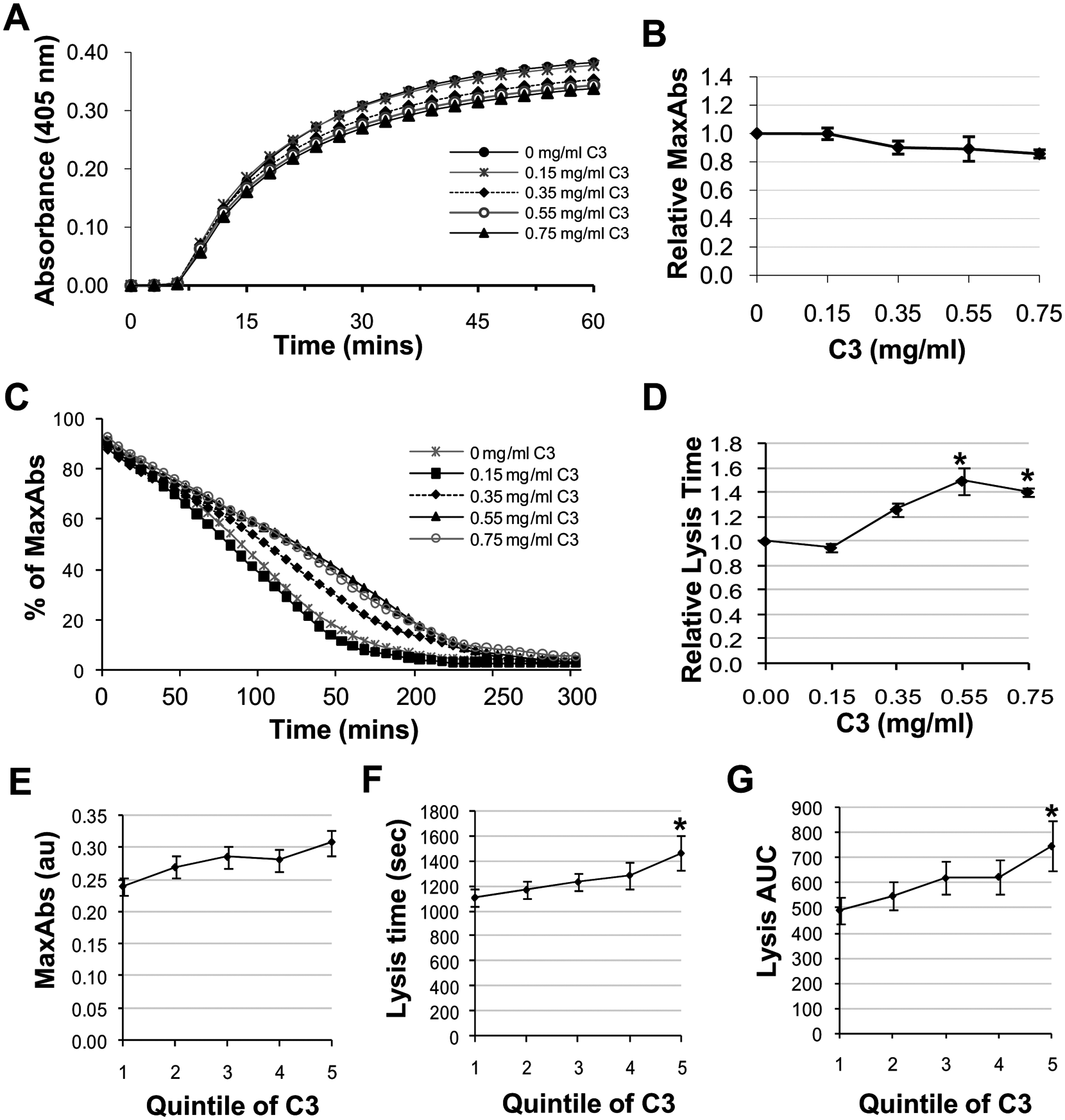
Effects of complement C3 on fibrin structure and fibrinolysis. (A and B) Turbidimetric analysis of clot density (MaxAbs). (C and D) turbidimetric analysis of fibrinolysis. Data are presented as the mean (SEM) of three independent experiments. *p = 0.01 comparing C3+ to C3− after Bonferroni adjustment. (E–G) The relationship between quintiles of C3 and measures of ex vivo fibrin structure/fibrinolysis in the Leeds Family Study (adjusting for pedigree structure, age, sex and known haemostatic determinants). *p = 0.01 after Bonferroni adjustment compared with quintile 1. C3 concentrations quintiles 1–5: 0.55–0.89, 0.90–1.02, 1.03–1.13, 1.14–1.28, 1.29–2.20 mg/ml, respectively. MaxAbs: maximum absorbance. AUC: area under lysis curve.
Factor H is incorporated into plasma clots but does not prolong fibrinolysis
To validate factor H as a clot component, we quantified the incorporation of factor H into plasma clots (n = 82). The concentration of factor H within clots increased with increasing plasma factor H concentration (Supplementary Figure S1, panel B), with a mean factor H incorporation into fibrin clots of 11.4% (SD 8.0). To determine whether incorporation of factor H into fibrin clots influenced fibrin structure/function, turbidimetric clotting and fibrinolysis assays were performed. Increasing factor H concentration did not significantly influence clot density (MaxAbs) or fibrinolysis times, as shown in Supplementary Figure S1, panels C and D.
Discussion
By employing a functional proteomics approach we have identified 20 non-fibrin proteins as clot components, predominantly related to coagulation and inflammation. As expected, fibrin components predominated in all mass spectrometry analyses. Other haemostasis related proteins included the catalytically active FXIII A subunit, α2-macroglobulin, α1-antitrypsin, kininogen-1 and fibrinogen-like protein-1, the latter protein having been previously identified as a clot component by Rijken et al. using a proteomics approach. 20 The majority of novel proteins identified in this study were related to inflammatory processes, supporting the concept that inflammation and thrombosis are fundamentally linked. The inflammatory proteins included three components of the complement cascade, C3, factor H and CPN, which have not previously been identified as clot components, although CPN has been reported to cleave fibrin under certain circumstances. 21
The identification of complement proteins as clot components is consistent with the close ancestral and physiological links between the coagulation and complement cascades. 22 which represent rapid effector systems for prevention of blood loss, protection against infection following injury and, by necessity, are required to co-localise to the same site, suggesting interactions between complement and fibrin may be physiologically relevant. There is a growing body of evidence reporting interactions between complement activation and a variety of haemostatic processes and supported by previous proteomic studies which have identified differential concentrations of C3 and other complement factors in patients with CVD23,24 and activated complement components at the site of thrombus formation in patients with acute MI. 25 These studies are further supported by clinical studies demonstrating elevated C3 predicts the development of CVD26,27 and may be specifically related to the inflammatory processes involved in CVD rather than a non-specific inflammatory marker.12,28,29 Taken together these and the present study support interactions between complement components and processes fundamentally involved in the development and progression of CVD and acute thrombosis, and on this basis complement C3 was selected for validation as a novel clot component.
Comparison of paired plasma and serum samples indicated approximately 25% of plasma C3 becomes incorporated into clots, which compares favourably with ~40% of plasma α2-antiplasmin incorporation we previously reported using a similar approach. 14 C3 bound to immobilised fibrinogen and fibrin in a concentration-dependent manner and binding data indicated the presence of two high affinity binding sites on fibrin(ogen). In the reverse orientation, complex high-affinity binding interactions were observed consistent with the potential for fibrinogen to interact with either one or two molecules of immobilised C3 dependent upon the conformation of individual molecules of immobilised C3. Such high affinity binding may suggest plasma fibrinogen and C3 circulate in complex; however, we did not detect fibrinogen/C3 interactions in the fluid phase (data not shown). These data therefore suggest that immobilisation-dependent conformational changes facilitate fibrinogen/C3 interactions, consistent with the known binding interactions between unstimulated platelets or neutrophils and surface immobilised fibrinogen.30,31
The physiological relevance of C3 incorporation into clots was demonstrated by C3 concentration-dependent prolongation of fibrinolysis. C3 did not significantly influence the kinetics of clot formation or clot density carried out in the absence of plasminogen and tPA, suggesting that prolongation of fibrinolysis by C3 is a direct effect rather than an indirect effect as a consequence of structural differences in fibrin. The physiological relevance of interactions between C3 and fibrin is supported by our recent study indicating C3 is associated with ex vivo fibrinolysis times in relatives of patients with type 2 diabetes independent of differences in fibrinogen and PAI-1 32 and in patients with diabetes. 33 This is confirmed in the present study by the significant associations between increased C3, prolonged lysis times and increased AUC of ex vivo plasma clots in the Leeds Family Study, further validating our in vitro observations. Furthermore, spiking of normal pooled plasma with exogenous C3 within the range observed in patients with CVD also leads to prolonged fibrinolysis, confirming C3 prolongs fibrinolysis in the presence of the key regulators of fibrinolysis and complement to support a physiologically relevant interaction. In contrast, whilst we demonstrated concentration-dependent incorporation of factor H into plasma clots, factor H at physiologically relevant concentrations did not influence clot structure or fibrinolysis, supporting a specific effect of C3.
Interactions between activation of coagulation and complement have previously been described, including activation of the alternative complement cascade resulting in prolonged clot lysis, although the complement component(s) mediating these effects were not identified. 34 More recently mannan-binding lectin-associated serine protease-2, which cleaves C4 and C2, has been shown to cleave prothrombin and initiate fibrin formation. 35 Complement activation has previously been shown to influence thrombin-induced platelet-dependent clot formation, clot retraction and subsequent platelet-dependent fibrinolysis of dilute whole blood in a manner dependent upon binding of IgM to platelets at 4°C and subsequent complement interactions involving factors C1–C5 resulting in enhanced clot retraction and fibrinolysis; 36 the inconsistency in results with the present study require further analysis but are likely due to the specific interactions of IgM with platelets occurring at 4°C. Complement and coagulation cascades also share common inhibitors including C1 inhibitor, 37 antithrombin III 38 and thrombin-activatable fibrinolysis inhibitor, 39 supporting co-activation and co-regulation of these systems. The results of the present study are also consistent with our recent study evaluating the influence of C3 on fibrin structure/function in diabetes, which demonstrated enhanced incorporation of C3 into fibrin and enhanced prolongation of fibrinolysis in patients with diabetes. 33 The interactions between C3 and fibrin described herein are physiologically relevant in the context of protection against pathogen invasion upon injury, whereby incorporation of C3 into a clot would localise C3 to sites of injury and consequently sites of potential pathogen invasion. Impaired fibrinolysis would also impair bacterial migration through the fibrin mesh; the importance of this is supported by exploitation of the fibrinolytic system by certain bacteria to facilitate invasion.40–42
Limitations of the study
The predominance of highly cross-linked fibrin multimers resulted in difficulty in resolving high molecular weight complexes by both 1-DE and 2-DE approaches. The predominance of fibrin isoforms across the molecular weight and pI range by 2-DE approaches due to the very nature of the plasma clot led to a relatively small number of non-fibrin components being detected with significant MOWSE scores. A comprehensive catalogue of the fibrin clot proteome is therefore yet to be achieved.
Conclusion
Using a functional proteomics approach we have identified several inflammatory factors including complement C3 as novel clot components. C3 binds to fibrin(ogen) with high affinity and incorporation into clots results in a concentration-dependent prolongation of fibrinolysis. In the context of injury, localisation of complement components to the fibrin clot and prolongation of fibrinolysis may be beneficial in impairing pathogen migration. The mechanistic links between C3 and fibrinolysis warrant further investigation given the growing body of evidence indicating complex interactions between inflammation and haemostasis. Interactions between C3 and fibrinolysis may have important implications in the context of CVD, with the potential for C3-mediated prolongation of fibrinolysis to lead to more extensive thrombus formation upon vascular injury.
Footnotes
Funding
VR is supported by a Medical Research Council DTA PhD studentship; RS was supported by a British Heart Foundation Clinical PhD studentship (FS/03/056); VS was supported by fellowships from the Swiss National Science Foundation and the Novartis Foundation; KH is supported by a grant from the German ADUMED foundation; the Leeds Family Study was supported by the Special Trustees of the Leeds Teaching Hospitals NHS Trust (UK) and an EU 6th Framework Programme Specific Targeted Research Project (grant no. LSHM-CT-2004-005268).
Conflict of interest statement
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
