Abstract
Introduction
Late morbidity and mortality in patients with diabetes mellitus (DM) are mainly caused by macrovascular and microvascular complications associated with this condition. Recent data suggest that different gene polymorphisms might be involved in the development of these complications. 1,2
Vascular endothelial growth factor (VEGF) is a glycoprotein secreted by the vascular endothelium, smooth muscle cells and other cell types. An increase in the expression of VEGF and its receptors has been described in diabetic vascular complications. 3 It plays a key role in the modification and acceleration of tissue damage caused by hyperglycaemia, which represents a risk factor for diabetic complications.
Several researchers have focused on the relationship between intra-plaque angiogenesis and formation of atherosclerotic plaque. VEGF is the major initiating factor in angiogenesis within atherosclerotic plaques. 4 It can promote proliferation of endothelial cells, induce vascularisation, enhance permeability of neovessels, and also induce inflammatory reaction within atherosclerotic plaque. VEGF levels have been analysed in patients with peripheral artery disease (PAD), and an association has been found between high VEGF levels and PAD severity. 5 These findings suggest that VEGF acts as an endogenous regulator of the endothelial integrity of the vascular wall. 6
VEGF is also a potent mediator of microvascular permeability, which might cause the development of complications such as diabetic retinopathy (DR), as shown by an increased expression of this factor in these patients. 7 Furthermore, there is wide experience in the treatment of diabetic macular oedema, DR and diabetic neovascular glaucoma with anti-VEGF agents. 8
Many single nucleotide polymorphisms (SNP) have been described in the VEGF gene. The genetic variations in the VEGF gene influence levels of VEGF protein expression. Of particular interest is an insertion/deletion polymorphism (I/D) at the −2549 position of the promoter region which has been associated with the development of DR. 9 Other polymorphisms frequently involved in the development of DR or coronary artery disease (CAD) are G→C at +405 in the 5′-untranslated region and −2578 C/A located in the VEGF promoter. 9,10
The objective of this study was to determine the potential genotype differences in the gene encoding VEGF in diabetic patients, which might explain or be associated with the difference in terms of clinical manifestation and evolution of DM in two phenotypically different groups, based upon the development of clinical complications: great vessels atherosclerosis vs. retinopathy. Secondarily, potential differences in the proteinomic expression of the VEGF gene in these two patient groups were determined.
Subjects
A cross-sectional case–control study was conducted with patients diagnosed with Type 2 DM, split into two groups according to their diabetic vascular complications in an exclusive and defining way, DR vs. PAD, with the inclusion/exclusion criteria shown in Table 1. Patients affected with diabetic nephropathy were excluded in order to avoid a potential confounding factor. Previous studies have revealed an association of several VEGF polymorphisms with this disease. 9 Twenty-six patients with PAD were included, with no presence of DR, as well as 14 patients diagnosed with DR and without PAD. Type 2 DM was diagnosed on the basis of the WHO criteria. All patients with PAD established as a DM complication underwent a complete ophthalmological examination for a DR screening, including corrected visual acuity, anterior segment biomicroscopy, gonioscopy, intraocular pressure, fundoscopy with pharmacological mydriasis, macular ocular coherence tomography (OCT) and fluorescein angiography (AFG). DR was classified as mild, moderate or severe non-proliferative DR or proliferative DR, associated or not with diabetic macular oedema, according to Early Treatment Diabetic Retinopathy Study (ETDRS) criteria: the presence of microaneurysms, haemorrhages, cotton wool spots, intraretinal microvascular abnormalities, hard exudates, venous beading and new vessels. 11
Inclusion/exclusion criteria for both study arms
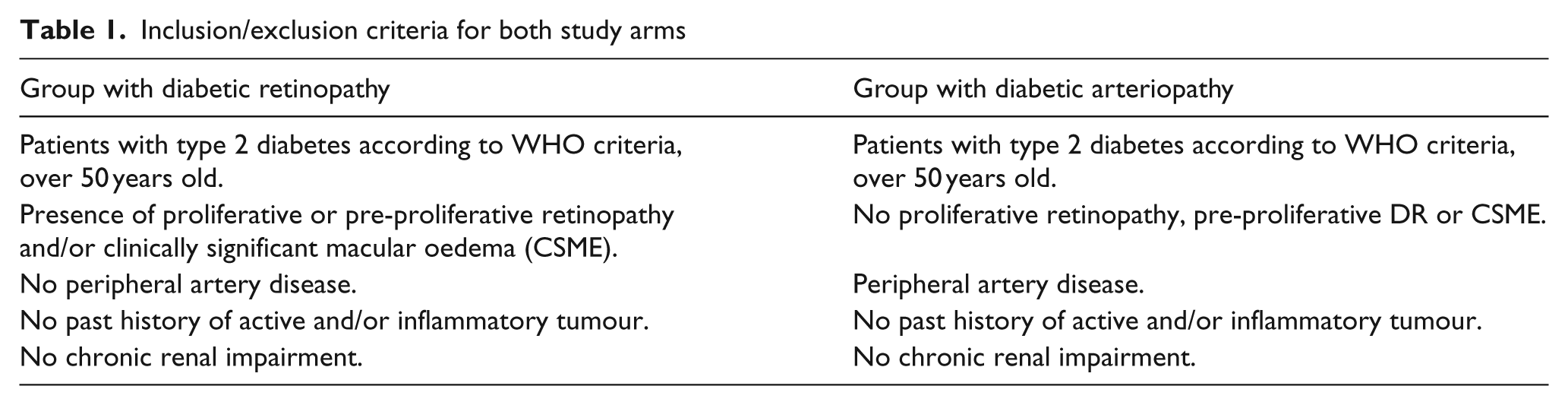
Likewise, the DR arm was assessed by a vascular surgeon, with PAD screening in physical examination and haemodynamic laboratory tests: ankle-arm index (AAI), which was considered normal when >0.9. Patients were considered as having PAD when their AAI was <0.9 or >1.3, when they presented symptoms of intermittent claudication, pain at rest and/or trophic lesions. Additional complementary tests, such as duplex, angioresonance or angiography, were carried out in those cases in which they were deemed necessary in order to establish a solid diagnosis of PAD.
Glycaemic control was evaluated by measuring glycated haemoglobin (HbA1c) levels. All individuals involved in this study were Caucasians of Spanish origin. Written informed consent was obtained from all subjects in accordance with the principles of the Declaration of Helsinki. The protocol of the study was approved by the Hospital Ethics Committee. A venous blood sample was collected from all patients after the consent form had been signed.
Materials and methods
DNA was isolated from a 3 ml blood sample using the NucleoSpin Dx Blood (Macherey-Nagel) kit according to the manufacturer’s recommendations. The I/D polymorphism was analysed using the following primers: forward 5′-GCTGAGAGTGGGGCTGACTAGGTA-3′ and reverse 5′-GTTTCTGACCTGGCTATTTCCAGG-3′. Genomic DNA (300 ng) was amplified in a final volume of 30 µl using the following conditions: denaturation at 95ºC for 6 min followed by 35 cycles at 94ºC for 1 min, 57ºC for 1.5 min and 72ºC for 2 min. A final extension was at 72ºC for 10 min. The reaction products were analysed by capillary electrophoresis (Bioanalyzer 2100, Agilent). Different genotypes were shown by the presence of different bands: 211 bp (D allele) and 229 bp (I allele).
For the VEGF G/C+405 polymorphism the forward and reverse primers were 5′ATTTATTTTTGCTTGCATT-3′ and 5′GTCTGTCTGTCTGTCCGTCA-3′, respectively. For the VEGF C/A−2578 polymorphism the forward and reverse primers were 5′- GGATGGGGCTGACTAGGTAAGC-3′ and 5′- AGCCCCCTTTTCCTCCAAC-3′, respectively. A specific TaqMan genotyping assay was used (Assay ID: C_8311614_10 for the VEGF G/C+405 polymorphism and Assay ID: 186522873_1 for the VEGF C/A−2578 polymorphism). They were analysed using real-time polymerase chain reaction (7500 Fast Real-time PCR, Applied Biosystems), according to the manufacturer’s recommendations.
Serum samples were stored at −20ºC. Serum VEGF levels were measured using the Human VEGF ELISA Kit (R&D Systems, Inc., Minneapolis, MN, USA).
Statistical calculations were performed using SPSS 15.0 for Windows (SPSS, Chicago, IL, USA). Data are presented as means ± standard deviations (SD). Genotype distribution, allele frequencies and allele positivity were assessed by a χ2 test of independence with 3 × 2 and 2 × 2 contingency tables and z-statistics. Fisher’s Exact Test was used to analyse allele frequencies and allele positivity in the various patient sub-groups. ANOVA test was used to compare different means. Values of p < 0.05 were considered statistically significant. Logistic regression analysis was used to assess the role of the VEGF genotype and other coexisting factors in retinopathy and arteriopathy. Cochran’s test was used for homogeneity of variances to assure that data were normally distributed. The data were analysed for a modification of the effect of the VEGF I/D, the VEGF G/C and the VEGF C/A genotypes on risk of retinopathy and arteriopathy by other risk factors.
The allele frequency data and the allele positive data were used for testing the null hypothesis (Ho) of association between the allele and the disease. In order to determine the sample size, the penetrance and the frequency of the alleles were estimated assuming the disease prevalence available in previous case–control data in our environment. 12,13
Power calculations were done using a power calculator available on-line (http://calculators.stat.ucla.edu). To calculate the study power, a difference in allele frequency of 0.10 was considered as relevant. The total number of subjects needed to obtain a power of 0.8 with a type I error (alpha error) rate of 0.05 in our population based on association study should be 40.
Results
Table 2 presents the clinical characteristics and biochemical variables studied in both patient groups. The gender distribution was similar in both groups. There were no statistically significant differences in terms of clinical characteristics between groups of patients, except for acute myocardial infarction (p = 0.001) and dyslipidaemia (p = 0.007), which were more frequent in the PAD group. Past smoking was also more frequent in the PAD group (p = 0.018). No statistically significant differences were observed when analysing the years of diabetes duration, either for glycated haemoglobin levels or for total cholesterol.
Clinical and biochemical characteristics of studied subjects
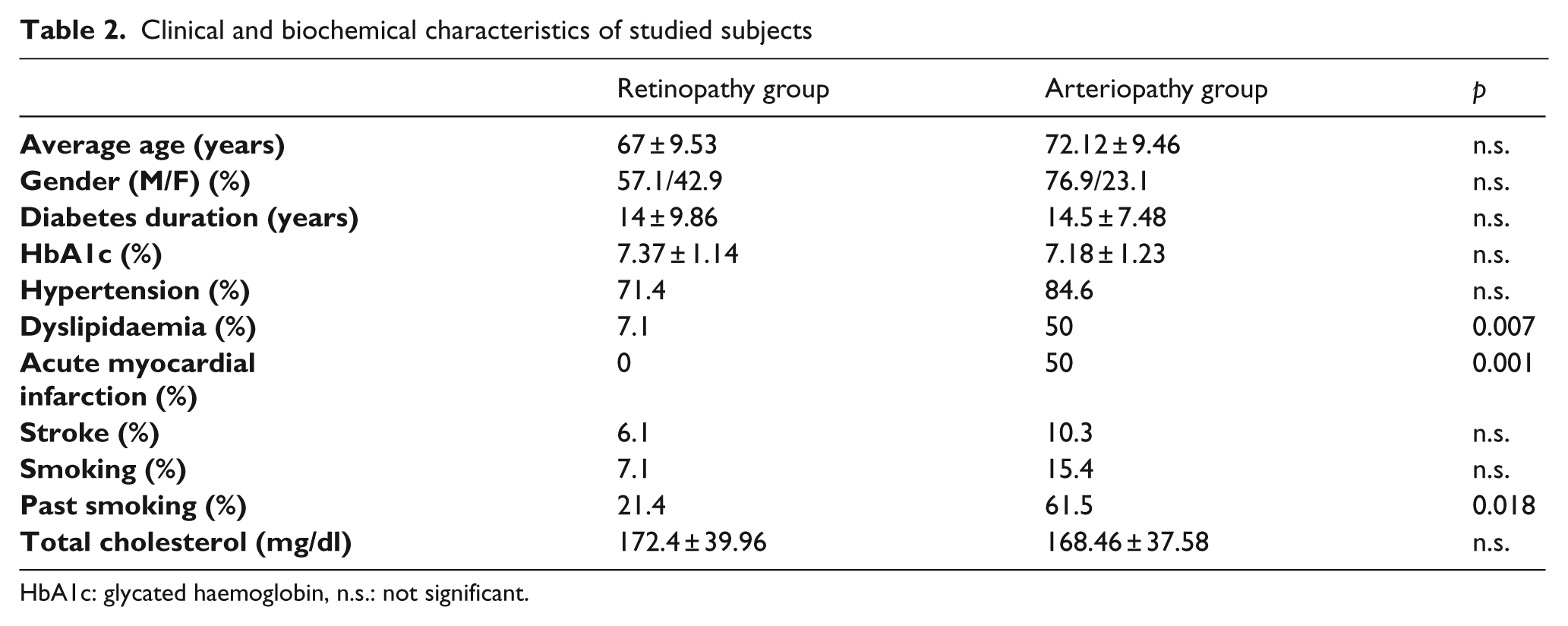
HbA1c: glycated haemoglobin, n.s.: not significant.
All patients included in the study were genotyped for the insertion/deletion (I/D) polymorphism at −2549, the G+405C and the C−2578A polymorphisms in the VEGF gene.
VEGF gene I/D polymorphism at −2549
The distribution of the I/D genotype in both patient arms is shown in Table 3. The frequency of genotypes did not deviate significantly from the Hardy–Weinberg equilibrium. There was a significant increase in the frequency of the DD genotype in the PAD arm compared with the DR arm (34.6% vs. 0%, p = 0.016), as well as in the ID genotype in the DR arm vs. the PAD arm (85.7% vs. 38.4%, p = 0.005).
Genotype distribution of VEGF gene polymorphisms in both groups
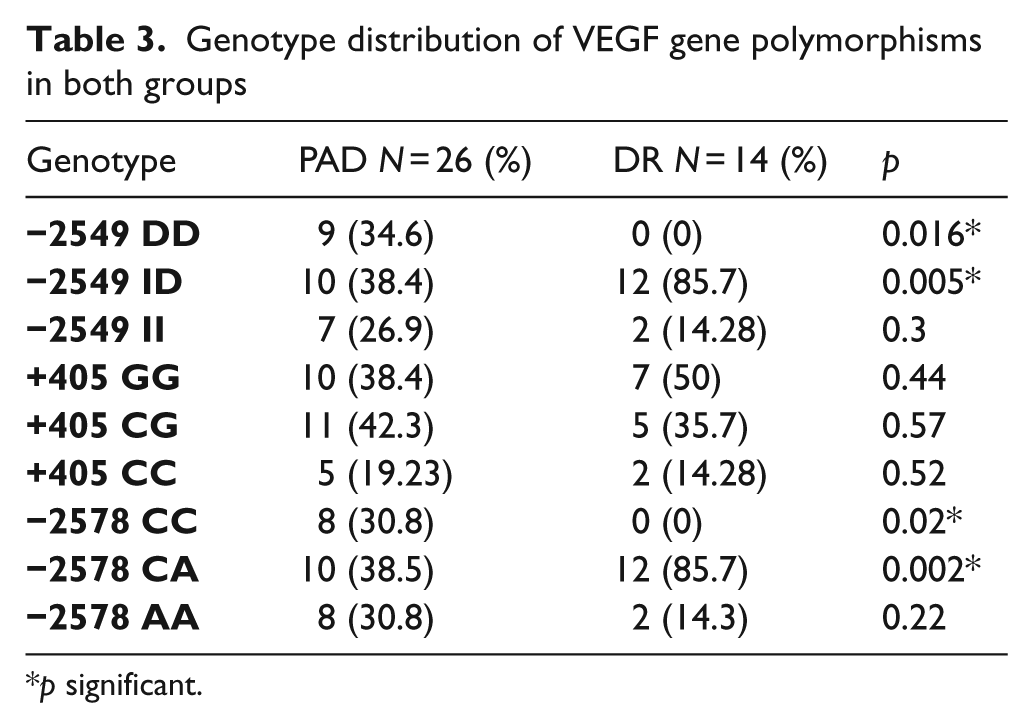
p significant.
The distribution of the allele frequencies is shown in Table 4. The allele distribution in the PAD group showed a statistically significant prevalence of D allele (p = 0.04). No differences were found in terms of a higher frequency of any allele in DR. The results of the allele positivity data of alleles with a higher frequency are shown in Table 5.
Allele distribution of VEGF gene polymorphisms
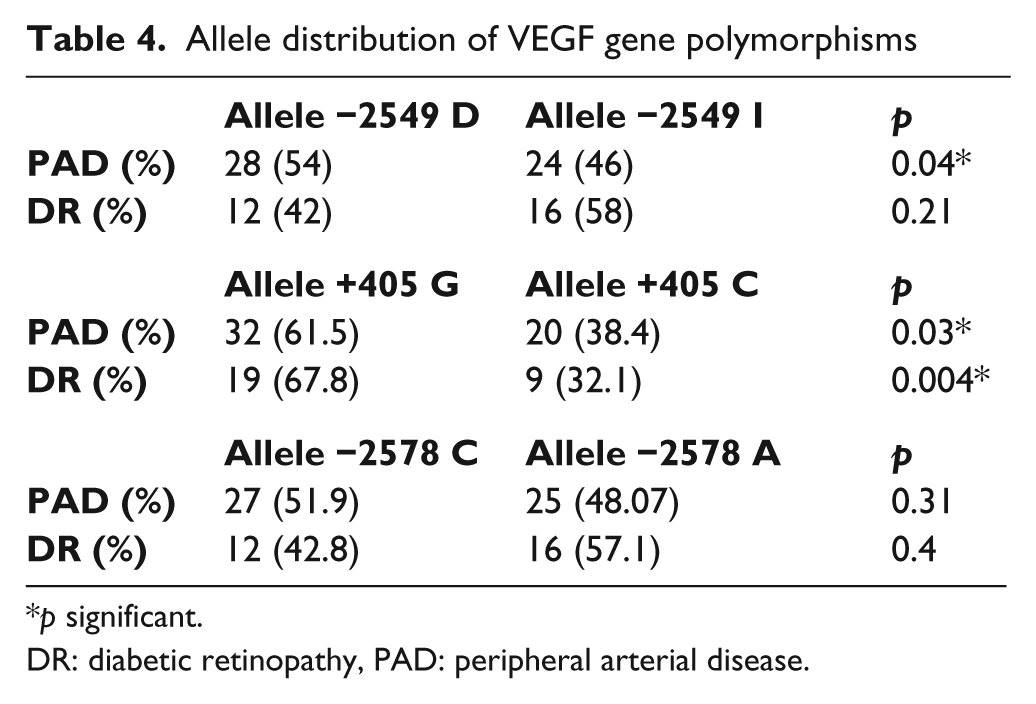
p significant.
DR: diabetic retinopathy, PAD: peripheral arterial disease.
Allele positivity table
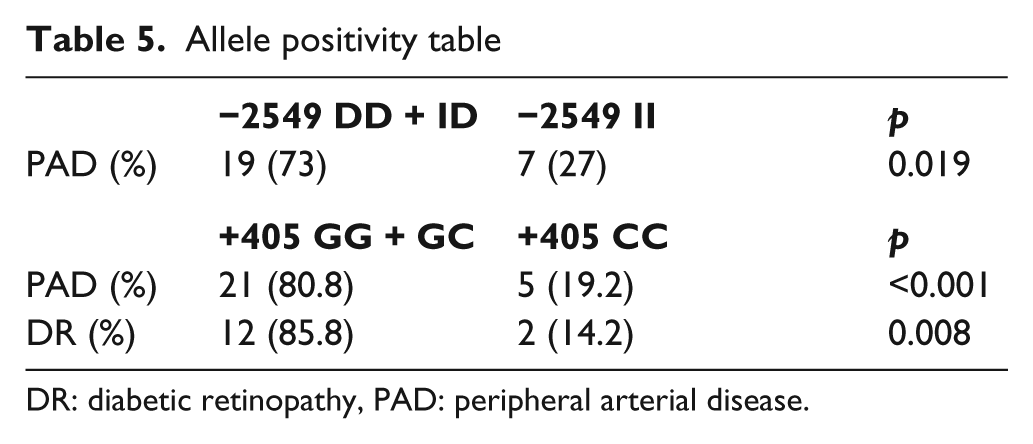
DR: diabetic retinopathy, PAD: peripheral arterial disease.
VEGF gene +405 polymorphism
There were no significant differences in genotype distribution when compared between DR and PAD groups (Table 3). However, the frequency of the G allele was increased in PAD patients, as well as in DR patients (Table 4). The results of the allele positivity data of alleles with a higher frequency are shown in Table 5.
VEGF gene −2578 polymorphism
There was a significant difference in the frequency of the CC genotype in the PAD arm, compared with the DR arm (34.6 vs. 0, p = 0.016), as well as in the CA genotype in the DR arm vs. the PAD arm (85.7 vs. 34.6, p = 0.002) (Table 3).
No differences were found between groups in terms of a higher frequency of any allele (Table 4).
VEGF serum levels
VEGF serum levels were measured in all patients. In the group of diabetic patients with PAD as a complication, the average level of VEGF in serum was 67.35 ± 74.9 pg/ml. When VEGF levels were analysed in the group of patients with DR as a complication, the average was 235.23 ± 184.21 pg/ml.
When median levels of VEGF in serum were compared between both types of diabetic complications, a statistically significant increase was observed in the DR group (p = 0.005).
Lastly, we compared VEGF levels with the different polymorphisms to assess possible association between one polymorphism and a higher production of VEGF. Increased serum VEGF levels were observed in −2578 CA patients compared with those with other genotypes of that polymorphism. Also, a trend toward higher VEGF levels was observed in patients with −2549 ID genotype, although it was not significant (Table 6).
Comparison between different genotypes of different polymorphisms and serum VEGF levels

p significant.
VEGF: vascular endothelial growth factor.
A multivariable logistic regression analysis was performed with the VEGF genotype and several clinical features adjusting by age, gender and lipid disorders as independent variables, and the presence of PAD or DR as dependent variables. This analysis showed that the presence of a DD genotype and the D allele of the VEGF I/D polymorphism and CC genotype of C−2578A polymorphism increased the risk of macroangiopathy, suggesting that these are independent risk markers of PAD. On the other hand, the presence of ID genotype of the VEGF I/D polymorphism and CA genotype of C−2578A polymorphism increased the risk of DR. We did not find any statistically significant differences for the remaining variables (Table 7).
Multivariable logistic regression analysis results
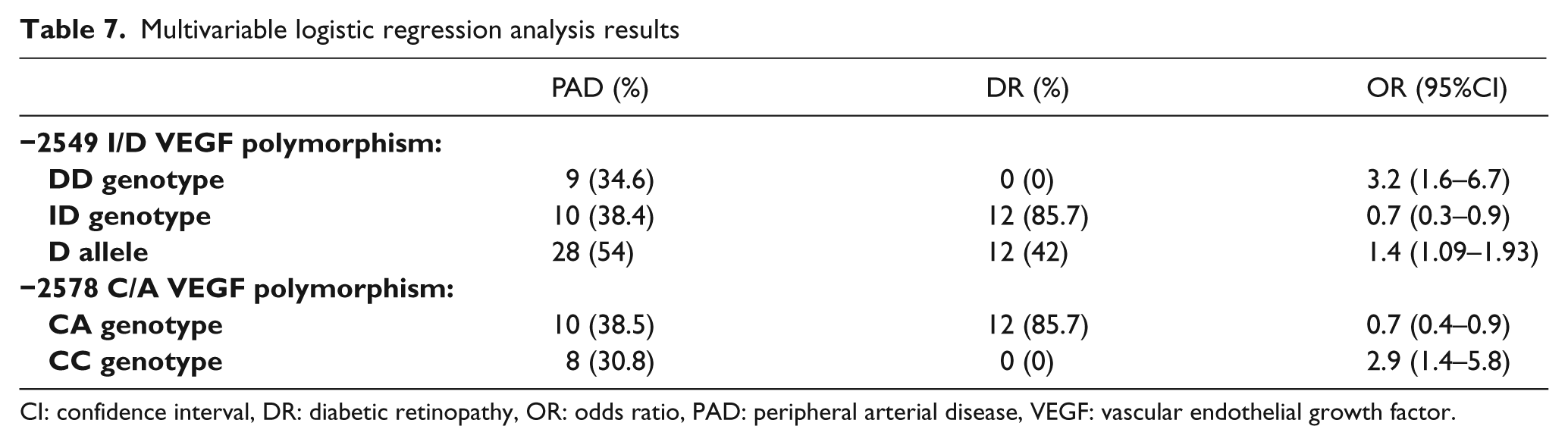
CI: confidence interval, DR: diabetic retinopathy, OR: odds ratio, PAD: peripheral arterial disease, VEGF: vascular endothelial growth factor.
Discussion
VEGF is an angiogenic factor which plays an important role in the pathogenesis of diabetic complications. VEGF gene polymorphisms have been involved in the development of complications such as DR and atherosclerosis progression in diabetic patients. 2,9
The hypothesis on which we based our study implies the existence of a genetic component which could be responsible for the different evolution of DM towards two opposite types of complications, in terms of type of affected blood vessels: microvascular complications (DR) or macrovascular complications (PAD). We took into account previous reports that show a potential involvement of the VEGF gene in these complications. Angiogenic factors seem to play different roles in these conditions; these observations have led to opposite therapeutic approaches, using pro-VEGF genetic therapy for treatment of critical lower limb ischaemia and anti-VEGF agents for treatment of proliferative DR and diabetic macular oedema. 14,15
We investigated the potential association of three VEGF gene polymorphisms with two long-term complications of Type 2 DM, DR and PAD.
Our data showed that the DD genotype and D allele of the SNP −2549 could be associated with a higher susceptibility to PAD among diabetic patients. Likewise, the ID genotype was also identified as a potential risk marker of DR. These results differ from those obtained in other studies, in which an association was found between the DD genotype of the VEGF gene I/D polymorphism and an increase in the risk of DR. 9,16 This might be due to different ethnicity of the patients, and to the fact that these studies analyse the above mentioned polymorphism between groups with DR vs. diabetic nephropathy, among other factors.
Of particular interest are the results obtained for the VEGF −2578 polymorphism. The presented results show that the CC genotype may be associated with susceptibility to diabetic PAD while the CA genotype may be associated with susceptibility to DR. The results are contradictory to other studies, in which the AA genotype was associated with DR 17 and the CC genotype was associated with a protective effect on atherosclerosis development. 2 Again, this might be due to different ethnicity of the patients. In general, the frequency of the susceptibility allele differs among populations. Thus, it should be noted that contradictory reports may be due to the difference in allele frequency between populations studied. 12
We also analysed the VEGF gene +405 polymorphism. There was no association found between +405 genotype and DR or PAD in our group of patients. This is in agreement with the results of Ray et al., 18 in which no significant association was found between diabetic retinopathy and the +405 polymorphism, and with the results of Howell et al., 2 who found no association between the +405 polymorphism and the number of coronary vessels affected by arteriopathy. While analysing the allele frequency, it was observed that the presence of the G allele is a risk marker for developing retinopathy as well as PAD, suggesting a potential association between this allele and an increase in activity in the promoter region of the gene. These data differ from those described by Szaflik et al., 19 in which the C allele of +405 gene was associated with an increased risk of DR.
Lastly, an increase in serum VEGF levels in DR patients was observed. This increase was also observed in −2578 CA genotype patients independently of the diabetic complications that they presented.
A limitation of our study is the small number of samples. However, we have found statistically significant differences with a small sample.
Diabetes is on its way to becoming the main cardiovascular risk factor in the next decades. Therefore, diagnosis and treatment of its complications will be very important.
The identification of genotype differences in the VEGF gene could provide a genetic marker during the first stages of DM, after diagnosis, that would allow us to predict the natural evolution and the type of specific clinical complications for each patient and to establish early preventive measures against them.
The exact association between VEGF gene polymorphisms in diabetic patients and the development of complications such as DR and PAD is possibly very complex. Also, the precise role of VEGF in diabetic vascular complications is still unclear. It is not known whether expression of VEGF is, at least in part, the cause of pathological changes in DR and diabetic PAD or, rather, represents a reparative response as a consequence of pre-existing tissue and functional alterations. The association between higher VEGF levels and DR is interesting; in fact, we found a relationship between higher VEGF levels and −2578 CA genotype. That genotype may be associated with susceptibility to DR, so that all may be interconnected. This study opens the way for future research that might analyse the efficacy of strategies aimed at treatment and early prevention of different complications associated with DM.
To summarise, our data suggest that the DD genotype and the ID genotype of the VEGF gene I/D polymorphism might possibly be associated with a higher susceptibility to PAD or DR, respectively, as a complication of Type 2 DM. Allele D has been found as an independent risk marker of PAD. On the other hand, the CC genotype and the CA genotype of the C−2578A polymorphism might possibly be associated with a higher susceptibility to PAD or DR, respectively, as a complication of Type 2 DM. When analysing the VEGF gene +405 polymorphism, we found that allele G was a risk marker of DR as well as of PAD. The role of these polymorphisms is still unclear. The presence of these alleles might be possibly associated with increased transcriptional activity. Future studies in larger diabetic patient populations are needed in order to increase knowledge about the effect of VEGF gene polymorphisms on diabetic vascular complications.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
None declared.
