Abstract
Pioglitazone and metformin are insulin sensitisers used for the treatment of T2DM. The effects of pioglitazone and metformin on endothelial function, assessed by FMD, in T2DM patients treated with sulfonylureas were compared. Patients were randomised to receive pioglitazone (n = 15) 30 mg once daily or metformin (n = 16) 850 mg twice daily for six months. Pioglitazone significantly decreased fasting insulin, HbA1C and HOMA-IR (p < 0.05 for all) and increased FMD (p = 0.002). Metformin induced a significant decrease in HbA1C (p = 0.02) and only a trend for increase in FMD (p = 0.08). The greater improvement in FMD with pioglitazone, compared with metformin, did not reach significance (p = 0.11). Treatment-induced changes in FMD were not associated with the effects of the two insulin sensitisers on glycaemic control or insulin resistance. The beneficial effects of pioglitazone and metformin on endothelial function in T2DM patients did not differ greatly. Larger studies are needed to explore whether a potentially greater benefit with pioglitazone may exist.
Keywords
Introduction
Patients with T2DM present an accelerated progression of the atherosclerotic process 1 that is the major substrate of cardiovascular diseases, the most important cause of morbidity and mortality in these patients. 2 Impaired vascular endothelial function, perhaps the earliest detectable abnormality of atherosclerosis, 3 has been well recognised in patients with T2DM. 4 Insulin resistance is considered to play a central role in the pathogenesis of T2DM and has been associated with endothelial dysfunction in these patients, while reduction in insulin resistance has been shown to predict improvement of endothelial function. 4–7
Metformin and pioglitazone are two widely used insulin sensitisers with established value in glycaemic control and reduction of insulin resistance in patients with T2DM. 8 Pioglitazone has been also consistently shown to improve endothelial function 6,7,9,10 and delay the progression of subclinical atherosclerosis 11–14 while it has been associated with a lower risk of cardiovascular events 15 in patients with T2DM. On the other hand the effects of metformin on early vascular atherosclerotic changes have been little studied 5,16 and a neutral or beneficial effect in cardiovascular prognosis has been demonstrated with metformin use in T2DM. 17,18 However, there is not enough evidence as to which agent should be preferred for improved cardiovascular prognosis in patients with T2DM since the comparative effects of pioglitazone versus metformin have not been previously studied in these patients.
The aim of this study was to assess and compare the effects of pioglitazone and metformin on vascular endothelial function in patients with uncontrolled T2DM treated only with glimepiride.
Patients and methods
Study population
Patients with T2DM not optimally treated with sulfonylureas were recruited from the endocrinology outpatient clinics of the University Hospital and Hatzikosta General Hospital of Ioannina. Eligible subjects were between 40 and 80 years old with a HbA1C above 7.0% during treatment with sulfonylurea monotherapy (glimeperide) for at least six months prior to the study. Thirty-four patients were enrolled: 10 males and 24 females; all females were in menopause and did not receive hormone replacement therapy. All patients had LDL-c >100 mg/dl or were using statins. Patients reporting a history of coronary artery, cerebrovascular, or peripheral vascular disease, chronic heart failure, liver or renal disease (or abnormal liver enzymes and renal function at study entry), anaemia, thyroid dysfunction, acute or chronic metabolic acidosis, alcoholism, known intolerance to metformin or pioglitazone and new onset of any medications within the previous eight weeks were excluded from the study.
Study design
This was an open-label, randomised study evaluating endothelial function in patients with T2DM already treated with glimeperide, when metformin or pioglitazone was added to their treatment for six months. After review of their medical history, all patients underwent a complete physical examination and an electrocardiogram. Patients eligible for the study who consented to participate were then randomised based simply on the order of their presentation in the out-patient clinic in two groups: metformin (850 mg b.i.d) was added in group A (n = 17), while pioglitazone (30 mg o.d.) was added in group B (n = 17). Cardiovascular risk factors (hypertension, dyslipidaemia, smoking status, family history of coronary artery disease), medications received, and time since the diagnosis of diabetes were recorded. Clinical assessment and evaluation of endothelial function were performed in all patients, at baseline and at six months. The body mass index (BMI) of each subject and the minimum waist measurement between the pelvic brim and the costal margin were recorded. Blood samples were drawn at baseline and at six months early in the morning after overnight fasting and vascular studies were performed the next day at the Michaelidion Cardiac Centre, University of Ioannina. Patients were asked to maintain the same diet and level of physical activity and stable doses of concurrent medications used (glimeperide, antihypertensive, hypolipidaemic and antiplatelet medications) throughout the study.
The study was approved by the Ethics Committee of the Michaelidion Cardiac Centre, University of Ioannina, Greece and informed consent was obtained from all patients. The study complies with the Declaration of Helsinki.
Laboratory investigations
Serum glucose was determined by the hexokinase method and HbA1 C by a latex agglutination inhibition assay (Randox, UK) while serum total cholesterol and HDL-c and triglycerides were measured by an enzymatic colorimetric assay. All assays were performed using an Olympus 640 analyser (Olympus Diagnostica GmbH, Hamburg, Germany). LDL-c was calculated using the Friedewald formula: LDL-c = total cholesterol - HDL-c - (triglycerides/5). Insulin was measured by microparticle enzyme immunoassay on an AXSYM immunoanalyser (Abbott Laboratories, Abbott Park, IL, USA). Serum hs-CRP was measured using rate turbidimetry (IMMAGE Immunochemistry Systems and Calibrator 5 Plus, Beckman Coulter Inc., Fullerton, CA, USA).
The fasting glucose-to-insulin ratio was calculated as fasting glucose (mg/dl)/fasting insulin (µIU/ml). 19 Whole body insulin resistance was assessed using the homeostasis model approximation (HOMA-IR), calculated as (fasting glucose, mmol/L × fasting insulin, µIU/ml)/22.5. 20
Assessment of endothelial function
Endothelial function was evaluated early in the morning with subjects fasted and refrained from smoking for at least 14 hours before the study. All measurements were taken in the supine position in a quiet, temperature controlled room (~22°C) after a 30-min period of rest. Supine blood pressure and heart rate were measured twice using an automated brachial sphygmomanometer (Omron M7, Omron Healthcare Co., Kyoto, Japan). All studies were done by the same operator, who was unaware of the treatment of patients.
Endothelial function was assessed in all patients by measurement of endothelium-dependent shear-induced vasodilation in the right brachial artery in response to hand hyperaemia (FMD). FMD was measured as previously described 7,21 according to published guidelines. 22 Optimal imaging of the right brachial artery was obtained using an Echo-Doppler ultrasound (Ultrasound ATL, HDI 5000, Bophell, WA, USA) and a 5–12 MHz transducer. Images were recorded on super-VHS videotape (VCR Panasonic AG-MD 835, Osaka, Japan) for off-line analysis. Brachial artery diameter was measured by another blinded operator, at end-diastole coincident with the R-wave on the electrocardiogram, using electronic callipers from the anterior to the posterior m-line at a fixed distance from an anatomic marker.
Images of the brachial artery were acquired at baseline, during hand hyperaemia, i.e. 90 s after deflation of a wrist cuff inflated to suprasystolic pressure for 5 min for measurement of FMD, and at 4 min after 400 µg of sublingual glyceryl trinitrate for measurement of endothelium- independent, nitrate-mediated vasodilatation (NMD). FMD and NMD were calculated as the per cent increase in diameter during hyperaemia and after nitrate administration respectively compared with the diameter at rest. Brachial artery blood flow was measured by continuous wave Doppler at rest and at 15 s after cuff release. Internal repeatability data of FMD and NMD measurements in our laboratory have been previously published. 7
Statistical analysis
Continuous variables are presented as mean ± SD. Kolmogorov–Smirnov Z-test was used to determine the normal distribution of continuous variables; all variables were found to be normally distributed. The χ2-test and the unpaired Student’s t-test were used to compare categorical and continuous variables respectively between the two groups at baseline. To assess changes from baseline at follow-up within each group, the paired Student’s t test was used. Comparison of treatment-induced changes in studied parameters between the two groups at six months was performed using the unpaired Student’s t-test. Based on our previous data in patients with T2DM 7 we anticipated a ≥ 1.5% absolute improvement in FMD (ca. one SD or 70–75% compared with baseline) following treatment. We estimated that a sample size of 17 patients per group would provide a power of approximately 80% to detect an increase in FMD > 1.5% following medications within each group as well as a difference of > 1.5% in treatment-induced changes in FMD between groups (α = 0.05).
The association between treatment-induced absolute changes in FMD and the studied clinical characteristics and laboratory measurements was assessed using Pearson correlation coefficients. A p value of < 0.05 was considered significant. The SPSS statistical software package (version 15.0 for Windows, SPSS Inc., Chicago, IL, USA) was used.
Results
One patient from group A and two patients from group B withdrew from the study because a statin was added to their treatment by their physicians during the study period. All remaining 31 patients completed the study. Adherence to medication use was assessed by tablet count at the end of the study. No serious adverse events were recorded; mild gastrointestinal complaints were reported from four patients receiving metformin, while mild leg oedema that did not result in discontinuation of treatment was reported in two patients taking pioglitazone. None of the group B patients presented severe hypoglycaemia.
Demographic, clinical, metabolic and vascular parameters of the 31 patients who completed the study are shown in Tables 1 and 2. The two groups did not differ in any studied parameter at baseline and consisted predominantly of females (75% and 67% in groups A and B respectively, p = NS) and overweight or obese T2DM patients (BMI 30.4 ± 5.5 and 31.9 ± 4.2 kg/m2 in groups A and B respectively, p = NS). In group A, treatment with metformin resulted in a significant decrease in HbA1C (p = 0.016) and a non-significant trend for reduction in diastolic blood pressure and increase in FMD (p = 0.077 for both) (Table 2). In group B, treatment with pioglitazone was associated with a significant improvement of fasting insulin (p = 0.01), HbA1C (p = 0.01), HOMA-IR (p = 0.003), and FMD (p = 0.002) and a non-significant trend for increase in HDL-c (p = 0.054) (Table 2). There was no significant difference in changes induced by metformin and pioglitazone on endothelial function and other metabolic parameters at six months (p > 0.1 for all) (Table 3).
Baseline demographic and clinical characteristics in the two groups
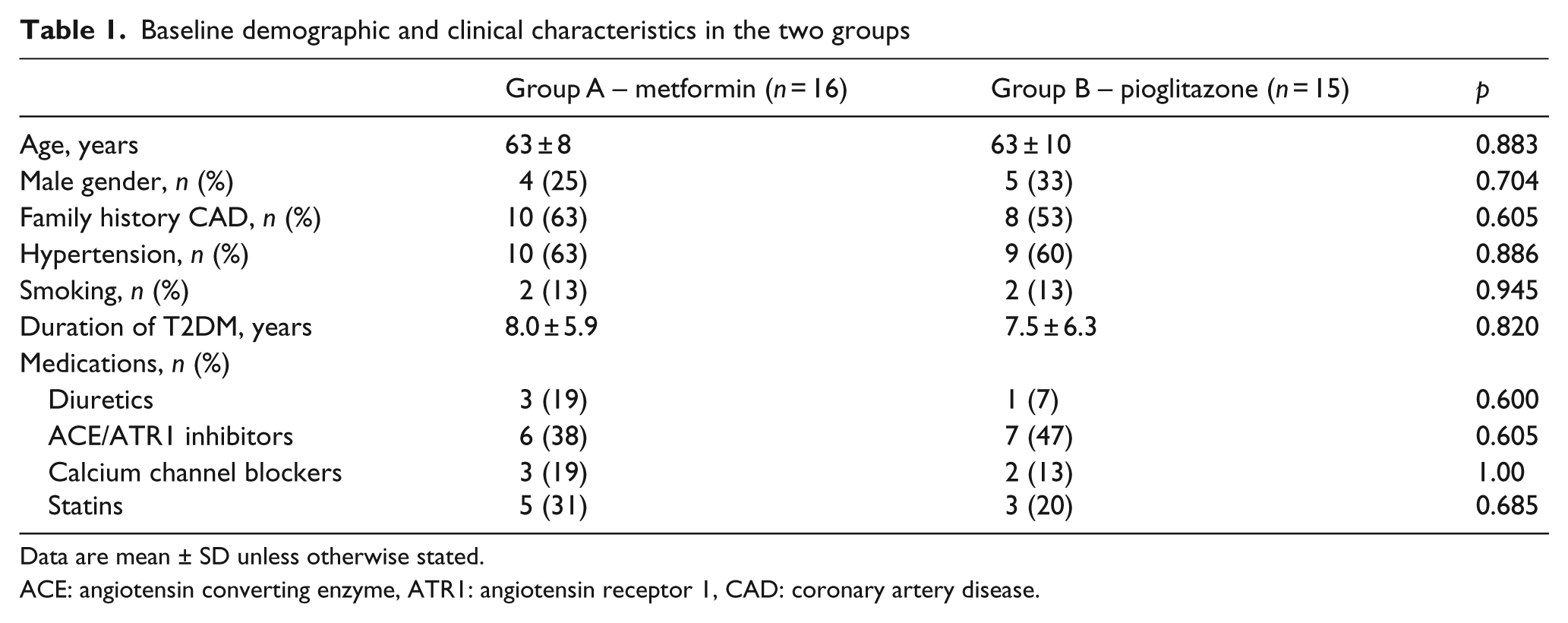
Data are mean ± SD unless otherwise stated.
ACE: angiotensin converting enzyme, ATR1: angiotensin receptor 1, CAD: coronary artery disease.
Clinical, laboratory and vascular measurements in the two groups at baseline and at follow-up
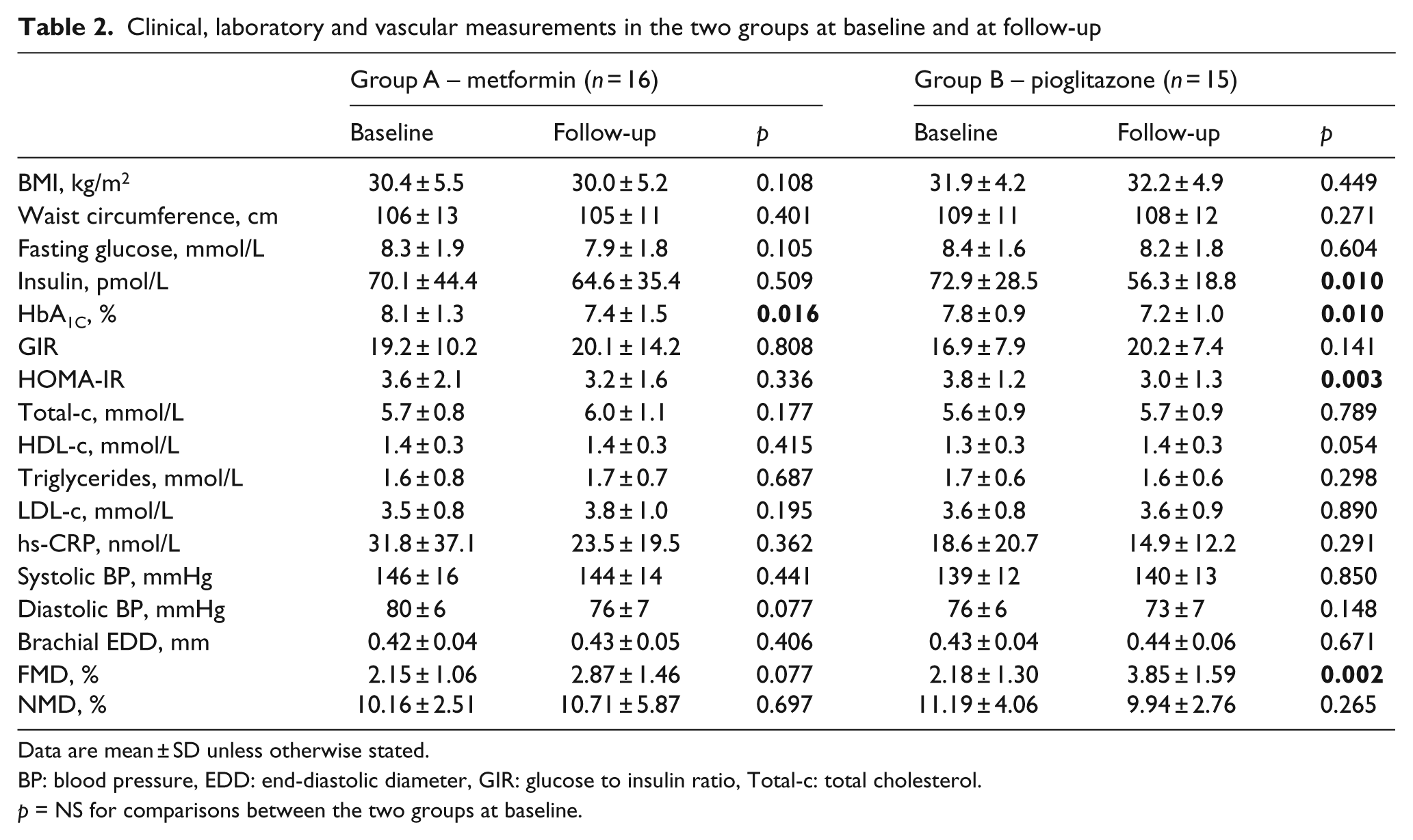
Data are mean ± SD unless otherwise stated.
BP: blood pressure, EDD: end-diastolic diameter, GIR: glucose to insulin ratio, Total-c: total cholesterol.
p = NS for comparisons between the two groups at baseline.
Treatment-induced changes in clinical, laboratory, and vascular parameters in the two groups at follow-up
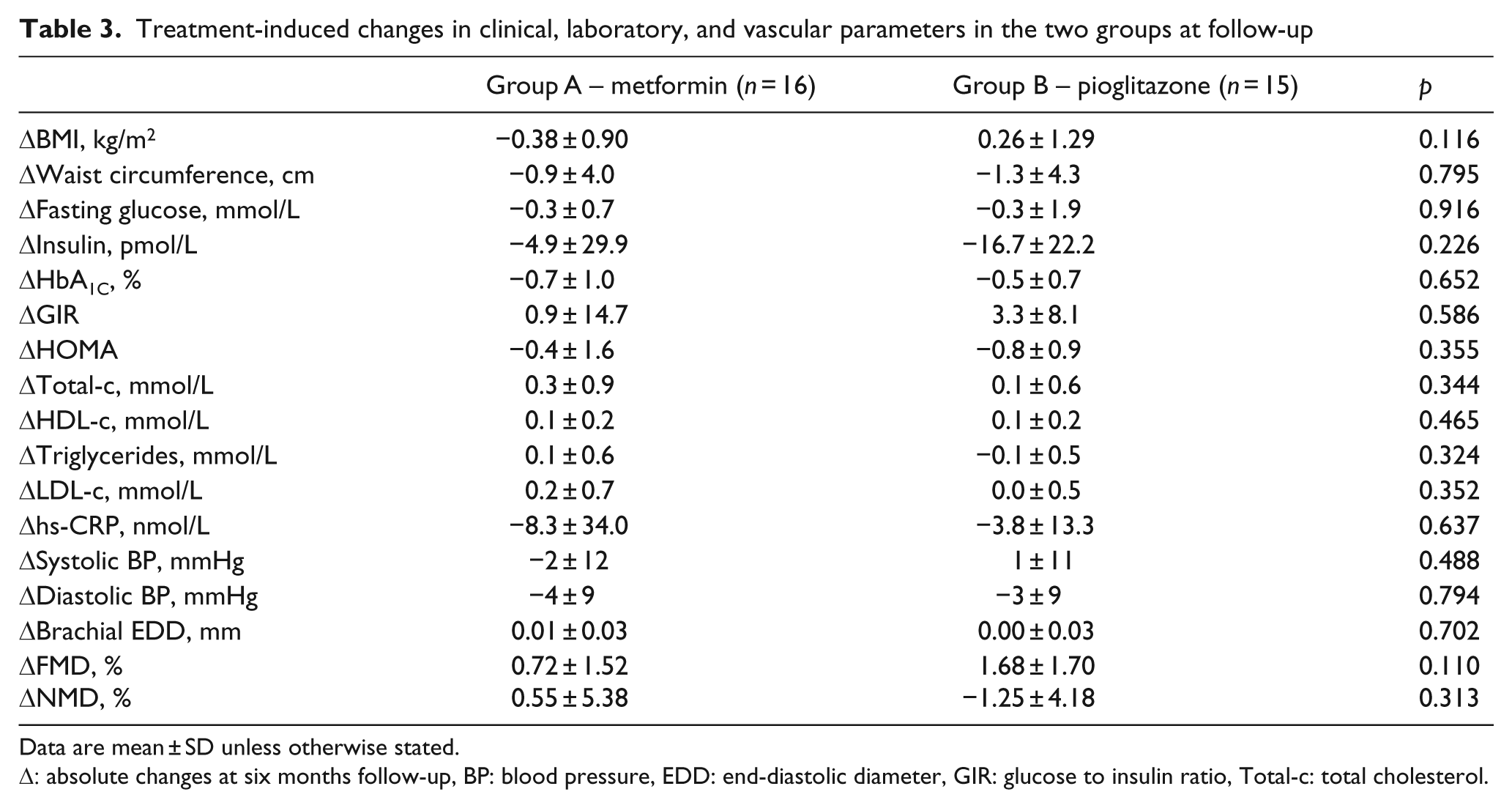
Data are mean ± SD unless otherwise stated.
Δ: absolute changes at six months follow-up, BP: blood pressure, EDD: end-diastolic diameter, GIR: glucose to insulin ratio, Total-c: total cholesterol.
Absolute change in FMD at the end of the six-month period in the 31 studied patients was inversely correlated with the absolute change in total cholesterol (r −0.424, p = 0.018) and LDL-c at follow-up (r = −0.386, p = 0.032) (Table 4).
Correlations between treatment-induced absolute changes in FMD and other studied parameters
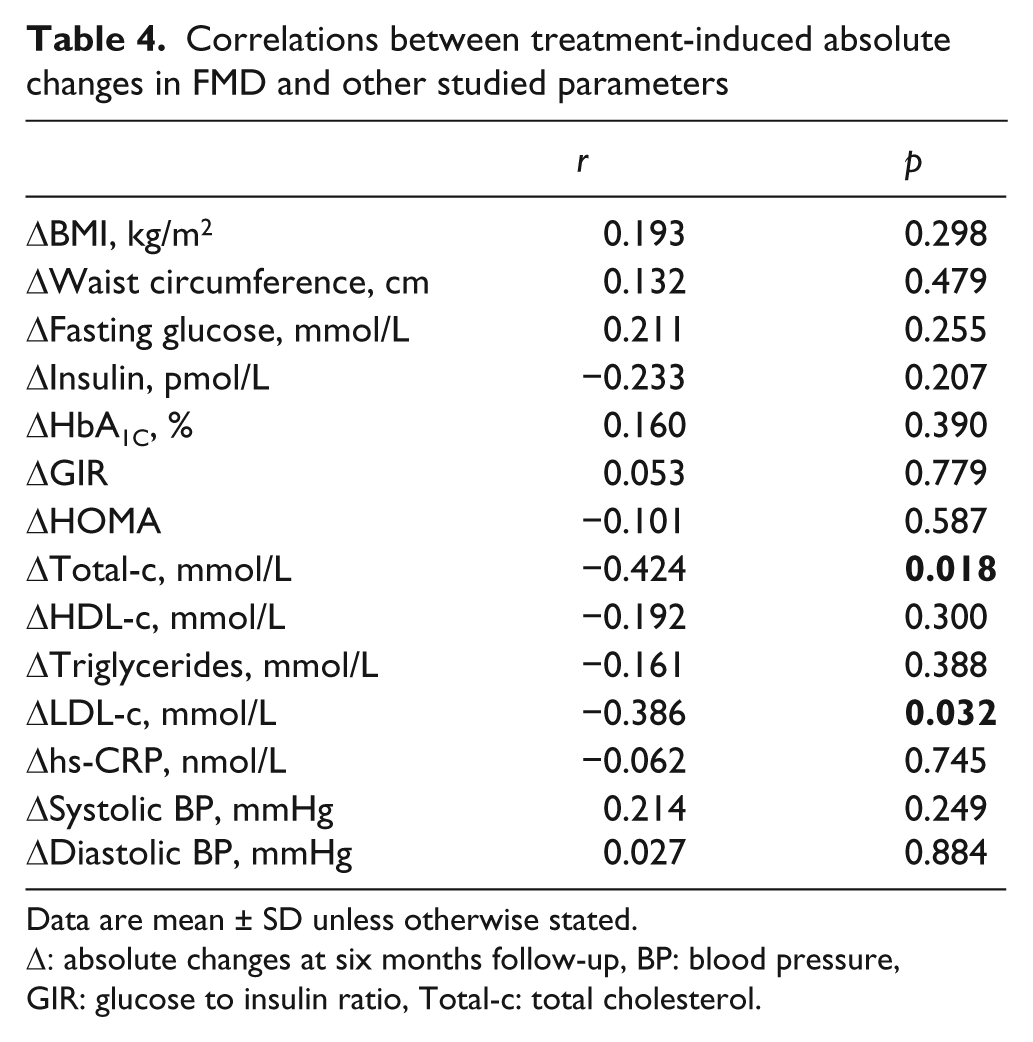
Data are mean ± SD unless otherwise stated.
Δ: absolute changes at six months follow-up, BP: blood pressure, GIR: glucose to insulin ratio, Total-c: total cholesterol.
Discussion
The results of the current study suggest that adding pioglitazone to glimepiride in T2DM patients significantly improved endothelial function, glycaemic control, and insulin resistance. On the other hand, addition of metformin to glimepiride was associated with a significant decrease only in HbA1C and a non-significant improvement in insulin resistance or endothelial function. Nevertheless, the difference in the effects of pioglitazone versus metformin did not reach statistical significance. Improvement in FMD with the use of these two insulin sensitisers was not related to glycaemic control or improved insulin sensitivity.
Endothelial dysfunction, demonstrated as reduced FMD, has been associated with various cardiovascular risk factors 23 and has been reported to predict long-term cardiovascular events in healthy and high risk populations. 24 Decreased FMD has been shown to reverse with treatment 3,22 but limited data so far suggest that improvement of FMD with treatment may translate into improved cardiovascular prognosis. 24
In our study, the administration of pioglitazone in addition to glimepiride in T2DM patients decreased HbA1C with a concomitant decrease in HOMA-IR and FMD; a nearly significant increase in HDL-c was also shown. The metabolic effects of pioglitazone in improving glycaemic control, insulin resistance and lipid profile are well established. 7,11,13,21,25 Pioglitazone has been also previously shown to improve endothelial function in T2DM 6,7,9,10 and non-diabetic patients, 21,25,26 suggesting a beneficial effect on cardiovascular prognosis. Indeed, administration of pioglitazone in T2DM patients has been associated with a delayed progression of subclinical or established atherosclerosis 11–14 as well as improved cardiovascular prognosis. 15
On the other hand the addition of metformin to glimepiride in T2DM patients resulted in a decrease in HbA1C and a non-significant increase in FMD. The beneficial effects of metformin on glycaemic control have been previously reported. 27 Although metformin has been shown to improve endothelial function in several non-diabetic populations with insulin resistance, 21,28,29 its effects on endothelium and progression of subclinical atherosclerosis in T2DM patients have been little studied. 5,16 In this study administration of metformin caused a non-significant increase in FMD (absolute change 0.7%), an effect that was below the a priori anticipated improvement (1.5%). Our study was not powered to show such a small increase in FMD and larger studies are needed to clarify the effects of metformin on endothelium. Furthermore, an attenuated effect of metformin on endothelial function may be seen specifically with the combination of metformin plus sulfonylureas and may be related to the adverse cardiovascular prognosis previously reported with this combination in T2DM patients. 18
In this study, the difference in the effects of pioglitazone versus metformin in addition to glimepiride in any of the studied parameters did not reach statistical significance. There is no previous study comparing the effects of these two insulin sensitisers in T2DM patients on either endothelial function or metabolic parameters. Although pioglitazone appeared to induce a greater improvement in contrast to metformin in insulin resistance as well as FMD (1.7% vs. 0.7%), this difference did not prove to be significant in the present study, maybe because of the small number of patients studied. Larger studies are needed to clarify whether pioglitazone may exert a more potent effect on both insulin resistance and endothelial function. Besides, it is not known whether such a small difference (≈ 1.0%) in the effects of the two insulin sensitisers on FMD could result in a significant difference in cardiovascular prognosis between metformin and pioglitazone. We have previously shown 21 that in overweight and obese females with polycystic ovary syndrome pioglitazone and metformin resulted in an equal improvement of endothelial function despite the fact that pioglitazone was superior in reducing insulin resistance; other distinct metabolic actions of the two agents were also demonstrated. On the other hand a differential effect of these agents cannot be ruled out in the setting of T2DM and in the case of co-administration of metformin and pioglitazone with glimepiride. Although glimepiride has probably a null effect on endothelium, 7 the combinations of glimepiride with metformin or pioglitazone may have distinct cardiovascular actions. It has been previously mentioned that the combination of metformin with sulfonylureas may be associated with adverse cardiovascular prognosis 18 while the cardiovascular effects of the combination of pioglitazone plus sulfonylureas have not been previously studied.
In accordance with previous studies, 5,7,30 improvement in endothelial function was not associated to the improvement in glycaemic control achieved. Several studies have reported a significant relation between a reduction in insulin resistance and beneficial effects of metformin or pioglitazone in vascular function. 5–7,11,12,21 Interestingly, this was not demonstrated in the present study. However, the reduction in insulin resistance indices in the present study was small compared with previous studies, indicating probably that the reduction in insulin resistance achieved might have not been sufficient to lead to a subsequent improvement in endothelial function. Finally, our study showed that FMD improvement following either metformin or pioglitazone was related to the decrease in total and LDL cholesterol despite the fact that neither agent had a significant effect on cholesterol. This finding has been previously reported in non-diabetic patients who received pioglitazone, 25 but the exact mechanisms have not been clarified and are beyond the scope of the present study.
Although this was an open label study, FMD studies were performed by an operator blinded to the treatment of patients, while measurement of brachial diameters and assessment of FMD was done by another operator blinded to both the patients and the study sequence. This study did not have statistical power to detect small differences in the vascular or metabolic effects of the two medications due to the relatively low number of patients enrolled. Patients received other medications that may have affected FMD (e.g. statins, angiotensin converting enzyme inhibitors), but their use was similar between the groups at baseline and patients were requested not to change their medications during the study. Smoking was not excluded in the current study, but smoking habits did not differ between the groups and patients were requested to refrain from smoking many hours prior to the FMD studies.
In conclusion, in T2DM patients treated with sulfonylureas, the addition of pioglitazone results in a significant improvement in FMD that does not appear to differ greatly compared with metformin. Improvement in endothelial function with the two insulin sensitisers does not seem to depend upon improved glycaemic control or insulin resistance in these T2DM patients. Larger studies are needed to investigate whether a potentially greater benefit with pioglitazone compared with metformin may exist and whether this may also translate into an improved cardiovascular prognosis.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
None declared.
