Abstract
The global prevalence of diabetes mellitus (DM) continues to climb, and is accompanied by an increase in DM associated complications, most often manifesting as coronary heart disease. Platelet dysfunction has been implicated as a central contributor to the increased risk of coronary artery disease in patients with DM, and it is not surprising that the anti-platelet agent, clopidogrel, has been shown to have efficacy in both short and long term outcomes in patients with acute coronary syndrome and those undergoing percutaneous coronary intervention. However, accumulating data suggest a clinically relevant sub-optimal clopidogrel response in some patients with DM. The exact mechanism of these observations is not yet fully understood, but appears to be related to reduced concentrations of circulating clopidogrel active metabolite, with less variability in pharmacodynamic and clinical response suggested by the evaluation of newer P2Y12 antagonists, such as prasugrel and ticagrelor. More research is needed to better understand both the pharmacology and clinical consequences of these observations.
Keywords
Diabetes mellitus and coronaryheart disease
The global prevalence of diabetes mellitus (DM) continues to climb, with a commensurate increase in the cardiovascular complications associated with DM and representing a global public health crisis.
Macrovascular atherosclerotic disease, most often manifesting as coronary heart disease (CHD) and its complications, account for the majority of morbidity and mortality associated with DM, underscoring the imperative to develop more effective and specific strategies towards mitigation of the cardiovascular disease (CVD) risk associated with DM. 1
One specific therapeutic target of interest is the prothrombotic state associated with DM, involving endothelial dysfunction, impaired proteo-fibrinolytic pathways, and platelet dysfunction. 2 As perturbations of platelet function in DM have been implicated in contributing to increased CVD risk, 3 the clinical effects of antiplatelet therapies remain a focus of intense interest, both in therapeutic decision-making and ongoing drug development. The aim of this review is to summarise and discuss the aggregate evidence supporting an increased variability of responsiveness to clopidogrel associated with DM and its potential clinical implications.
Platelet dysfunction in DM
Increased platelet activation and aggregation in DM have been attributed to a number of metabolic abnormalities, including hyperglycaemia, 4 insulin resistance, 5 and dyslipidaemia. 6 Furthermore, cellular abnormalities, primarily mediated through platelet and endothelial cell dysfunction,7,8 also contribute to the DM prothrombotic milieu. As reviewed elsewhere,3,9 platelets in DM patients have increased expression of activation markers and platelet surface receptors – such as purinergic receptor P2Y, G-protein coupled, 12 (P2Y12), glycoprotein Ib (GPIb), glycoprotein IIb/IIIa (GPIIb/IIIa), and P-selectin – and increased concentrations of intracellular calcium that lead to augmented platelet degranulation and aggregation. DM is also associated with increased platelet surface area, size, and volume, each associated with increased aggregability and activation.10,11 DM is also associated with increased production of reactive oxygen and nitrogen species and decreased platelet antioxidant production, resulting in increased platelet activation and decreased endothelial cell production of nitric oxide and prostacyclin. 7 In aggregate, these platelet perturbations associated with insulin resistance and DM represent an important pathobiological link between DM and increased CHD risk, both for acute coronary complications and for in-stent thrombosis following percutaneous coronary intervention (PCI), and remain a key therapeutic target for which more specific and/or more potent antiplatelet therapies may yield significant clinical improvements.
Clinical applications
Effective therapies for any particular disease are typically most effective in highest risk patients, especially when said therapies specifically target pathologies underpinning the clinical risk. For example, in the DM arena, GPIIb/IIIa inhibitors are potent antiplatelet medications that act by selectively binding to the GPIIb/IIIa receptors on the platelet surface, thus preventing aggregation. Given the increased expression and activation of GPIIb/IIIa receptors on the platelets of patients with DM,3,9 one would expect these inhibitors to be particularly effective in patients with DM. This hypothesis has been supported by a meta-analysis of patients with DM from six large-scale, randomised trials evaluating GPIIb/IIIa inhibitors used in the setting of acute coronary syndrome (ACS) demonstrating a significant reduction in 30-day mortality in DM patients treated with GPIIb/IIIa inhibitors compared with placebo [odds ratio (OR) 0.74; 95% confidence interval (CI) 0.59–0.92; p = 0.007], a mortality treatment effect not observed in the absence of DM (adjusted OR 1.01; p = 0.85). 12
Despite the consistency of demonstration of platelets perturbations associated with DM, there remain discordant data in the antiplatelet therapy field. In contrast to the exaggerated benefits of GPIIb/IIIa inhibitors in patients with DM, 12 aspirin efficacy is attenuated in patients with DM, as demonstrated in the Antithrombotic Trialists’ Collaboration (ATT) meta-analysis of antiplatelet therapy, with a non-significant 7% odds reduction of vascular events in patients with DM treated with aspirin compared with a 22% odds reduction observed in the overall meta-analysis cohort. 13 Similar observations derive from the primary cardiovascular risk prevention setting, with a recent meta-analysis estimating a modest and statistically non-significant 9% relative risk reduction (RRR) for major CHD events [hazard ratio (HR) 0.91; 0.79–1.05] in the DM patient population. 14 The clinical relevance of these observations remains largely uncertain and is especially relevant with the emergence of alternative antiplatelet agents.
Pharmacology of clopidogrel
Clopidogrel is a prodrug that is metabolised by two sequential oxidative pathways involving hepatic cytochrome P450 enzymes [cytochrome P450, family 2, subfamily C, polypeptide 19 (CYP2C19), cytochrome P450, family 3, subfamily A, polypeptide 4/5 (CYP3A4/5), cytochrome P450, family 2, subfamily B, polypeptide 6 (CYP2B6), cytochrome P450, family 2, subfamily C, polypeptide 9 (CYP2C9), and cytochrome P450, family 1, subfamily A, polypeptide 2 (CYP1A2)] (Figure 1). 15 The first pathway oxidises clopidogrel to 2-oxo-clopidogrel, followed by further metabolism to the active thiol derivative. This active metabolite selectively and irreversibly binds to the platelet ADP P2Y12 receptor and prevents ADP-induced activation of the GPIIb-IIIa complex, which is necessary for platelet aggregation. As drug binding is irreversible, the pharmacodynamic effects of clopidogrel persist for the lifespan of the affected platelet, approximately 7–10 days. In a second, competing clearance pathway, clopidogrel is hydrolysed via circulating esterases to its carboxylic acid derivative, accounting for approximately 85% of all circulating metabolites.
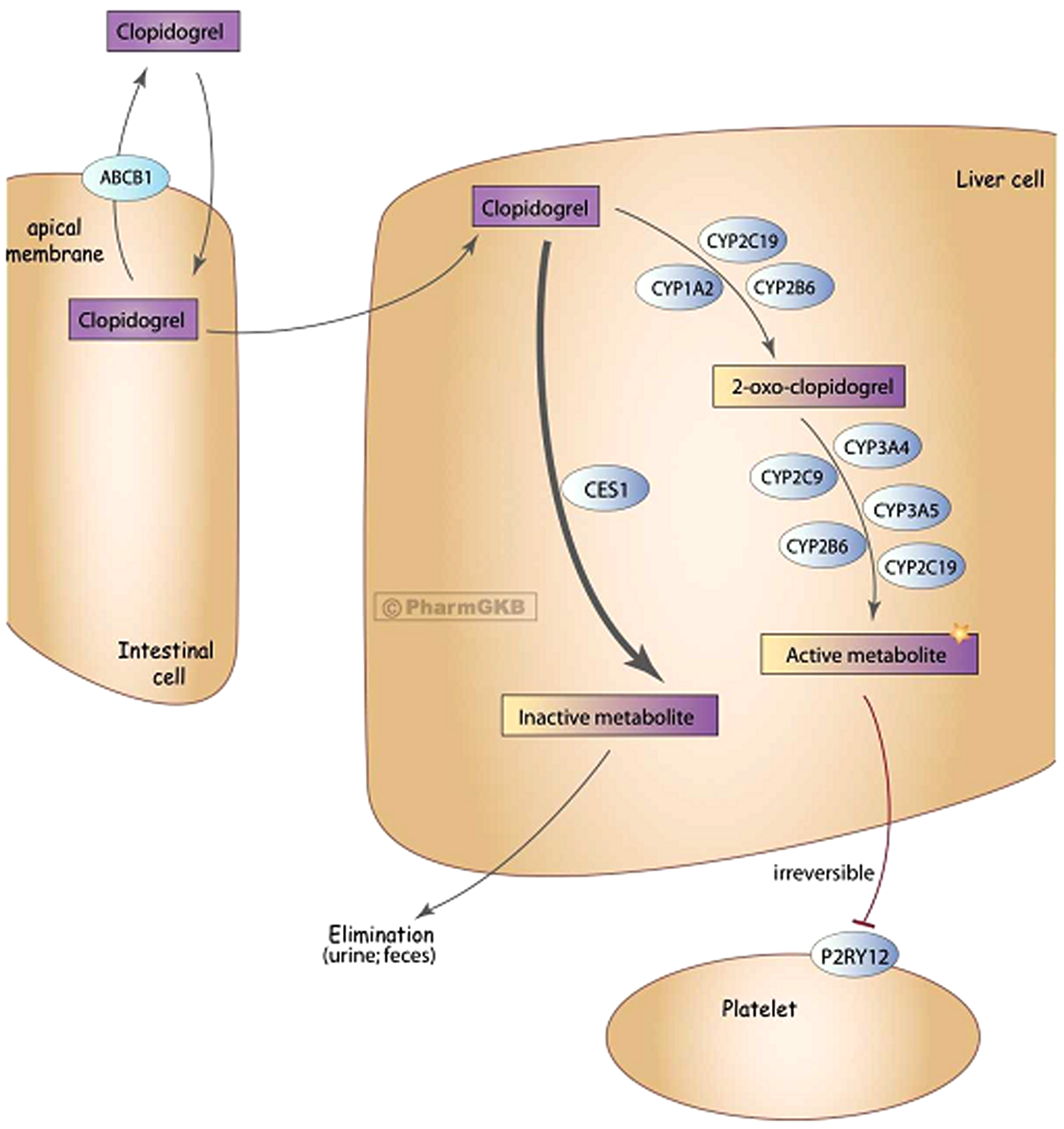
The metabolism of clopidogrel.
Impaired metabolism of clopidogrel from prodrug to active metabolite compromises pharmacodynamic efficacy, with much recent focus on the role of CYP2C19. Pharmacogenomic studies have shown that clopidogrel metabolism varies greatly based on an individual’s CYP2C19 genotype.16,17 In addition, drugs that inhibit CYP2C19, such as many of the proton pump inhibitors and omeprazole specifically, interfere with ex vivo pharmacodynamic measures of clopidogrel. 18 However, the clinical relevance of these drug interactions remains unclear, with discordant findings in observational studies.18–21 In one clinical trial, patients taking clopidogrel were randomised to omeprazole versus placebo, with no adverse cardiovascular clinical signals observed, though the trial was limited by a relatively small sample size and by premature administrative discontinuation of the trial. 22
Clopidogrel use in coronary artery disease
Clopidogrel is effective in the management of both acute and chronic coronary artery disease (CAD) and following percutaneous coronary interventions, as summarised in Table 1.23–29 Based on the observed variability of pharmacodynamic and clinical response to clopidogrel therapy among patients with DM, the concept of clopidogrel unresponsiveness has emerged. Gurbel et al. 30 were among the first to describe a variable platelet inhibitory response to clopidogrel in patients undergoing PCI, associated with increased platelet aggregation and increased expression of platelet surface receptors. Since then, the pharmacodynamic evidence of an impaired response to clopidogrel and the functional assays to detect such responses have vastly evolved to show that an impaired clopidogrel response and high residual platelet reactivity associated with DM is associated with worse short- and long-term clinical outcomes.31–34
Summary of clopidogrel trials in coronary heart disease
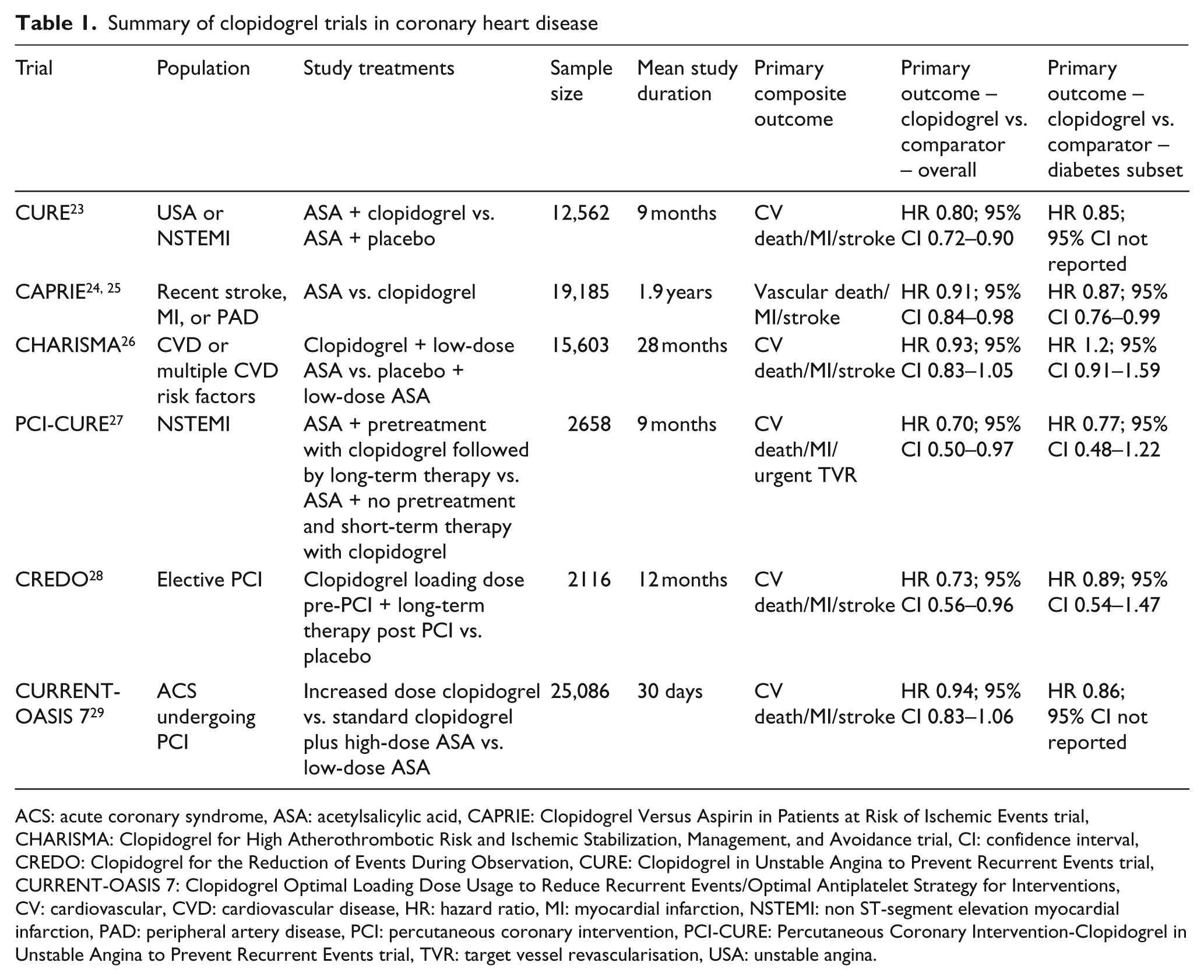
ACS: acute coronary syndrome, ASA: acetylsalicylic acid, CAPRIE: Clopidogrel Versus Aspirin in Patients at Risk of Ischemic Events trial, CHARISMA: Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management, and Avoidance trial, CI: confidence interval, CREDO: Clopidogrel for the Reduction of Events During Observation, CURE: Clopidogrel in Unstable Angina to Prevent Recurrent Events trial, CURRENT-OASIS 7: Clopidogrel Optimal Loading Dose Usage to Reduce Recurrent Events/Optimal Antiplatelet Strategy for Interventions, CV: cardiovascular, CVD: cardiovascular disease, HR: hazard ratio, MI: myocardial infarction, NSTEMI: non ST-segment elevation myocardial infarction, PAD: peripheral artery disease, PCI: percutaneous coronary intervention, PCI-CURE: Percutaneous Coronary Intervention-Clopidogrel in Unstable Angina to Prevent Recurrent Events trial, TVR: target vessel revascularisation, USA: unstable angina.
Ex vivo and function assays, and metabolite measures
The most commonly used ex vivo testing method to assess clopidogrel response is light transmittance aggregometry (LTA), 35 which assesses ADP-stimulated platelet aggregation. Unfortunately, LTA is laborious and requires a specialised laboratory and specialised specimen processing, thus precluding bedside assessment.35,36 Another method of measuring clopidogrel response uses intracellular signalling pathways activated by ADP coupling with the platelet P2Y12 receptor. This coupling triggers an intracellular cascade resulting in the phosphorylation of vasodilator-stimulated phosphoprotein (VASP). Monoclonal antibodies can then be used to quantify the phosphorylated VASP, thus giving a measure of unblocked P2Y12 receptors. 37 Although specific, this technique is also relatively expensive and labour intensive. Additionally, measurement of platelet activation-dependent receptors, most commonly P-selectin and GPIIb/IIIa, using monoclonal antibodies can be used to assess the platelet effects of clopidogrel. Two point-of-care assays to assess P2Y12 antagonism have been developed, the ‘VerifyNow’ rapid platelet function assay and the platelet function analyser 100 (PFA-100), with the former validated against LTA. 38
Variability of pharmacodynamic and clinical effects of clopidogrel in DM
Despite several large-scale clinical trials supporting the efficacy of clopidogrel as summarised in Table 1, there remains a growing evidence base regarding impaired clopidogrel responsiveness associated specifically with insulin resistance and DM.39,40 These reports are based both on ex vivo measures of on-treatment platelet reactivity and subanalyses of clinical trials evaluating the effect of clopidogrel among those participants with DM, with trends toward attenuated clinical efficacy and increased stent thrombosis.31–34,41,42
Several studies have shown a diminished response to clopidogrel in patients with DM using ex vivo assays. Angiolillo et al. investigated platelet function profiles in DM and non-DM patients taking aspirin and clopidogrel acutely after a loading dose of clopidogrel (group 1), and after chronic therapy (group 2). 43 In group 1, ADP-induced platelet aggregation was significantly increased in patients with versus without DM, both at baseline (p = 0.03) and 24 h after clopidogrel loading (p = 0.03). Group 1 was further classified as non-responders, low responders, or responders (platelet inhibition of < 10%, 10–29%, and ≥ 30%, respectively), with a significantly higher number of clopidogrel non-responders in the group with versus without DM (p = 0.04). Group 2 analyses revealed similar results, as patients with versus without DM had significantly increased platelet aggregation (p = 0.03), GPIIb/IIIa activation (p = 0.01), and P-selectin expression (p = 0.005).
The prognostic implications of high on-treatment platelet reactivity in DM have also been assessed in 173 patients with CHD on chronic aspirin and clopidogrel therapy. 41 Platelet function testing was done at study entry, with patients divided into quartiles based on maximal ADP-induced platelet aggregation, with the highest quartile defining high platelet reactivity (HPR). After continuation of dual therapy for a further 3–6 months, clopidogrel therapy was discontinued and patients were followed for an additional 2 years. The average maximum platelet aggregation at study entry was 52%, with the upper quartile defined by HPR > 62% with a mean value in the upper quartile of 68.6 ± 6%. After 2 years, a total of 41 major adverse cardiovascular events (MACEs) occurred, primarily unstable angina and/or non-ST elevation myocardial infarction (NSTEMI), 37.7% of which were in patients in the upper quartile with HPR (p = 0.005). Multivariable Cox regression analysis demonstrated that HPR independently predicted MACE (HR 3.4, 95% CI 1.7–6.7).
The Optimizing Antiplatelet Therapy in Diabetes Mellitus (OPTIMUS) trial assessed the pharmacodynamic effect of high maintenance dosing of clopidogrel (150 mg) in patients who had DM, CHD, and suboptimal clopidogrel-induced platelet effects. 44 This randomised, double-blind trial found that those patients taking higher-dose clopidogrel experienced a significant reduction in maximum platelet aggregation when compared with baseline values and when compared with those taking standard-dose clopidogrel (p = 0.002). Interestingly, patients on standard dosing all continued to have maximal platelet aggregation values > 50%, a value that has been associated with a higher risk of ischaemic events following PCI. 32
Erlinge et al. also investigated the prevalence and mechanisms of clopidogrel unresponsiveness in patients with and without DM. 40 This double-blind trial randomised 110 patients (19 with DM) on aspirin therapy to receive clopidogrel (600 mg loading dose, followed by 75 mg maintenance dose) or prasugrel – a newer thienopyridine drug that requires less extensive hepatic metabolism and that is not impacted by CYP2C19 activity or inhibitors – (60 mg loading dose, followed by 10 mg maintenance dose) for 28 days. Results showed a significantly increased proportion of poor responders in the clopidogrel group when compared with the prasugrel treatment group. Patients with DM were also noted to be significantly over-represented in the poor responders at all time points, and there were very few poor-responder DM patients in the prasugrel group, suggesting that DM poor responsiveness was limited to clopidogrel-treated patients. When compared with patients without DM, those with DM were also noted to have significantly lower levels of clopidogrel active metabolites at the 2-h post-loading dose (p = 0.0064) and at day 29 of the maintenance dose (p = 0.0047). Importantly, platelets from patients considered to be poor responders and from patients with DM responded fully to the clopidogrel active metabolite added ex vivo, excluding drug resistance at the level of the P2Y12 receptor as a contributing mechanism.
Evidence for impaired clopidogrel clinical efficacy in the setting of DM
Despite significant evidence showing the short- and long-term cardiovascular clinical efficacy associated with clopidogrel (Table 1), patients with DM enrolled in these trials continue to experience increased risk for subsequent adverse cardiovascular events when compared to those without DM. In addition, subanalyses of the efficacy of clopidogrel within the subsets of patients with DM across the published trials have consistently yielded point estimates of relative treatment effects numerically no greater than, and most often smaller than, those observed in the overall trial and in the subsets without DM, indirectly suggesting an incomplete response to clopidogrel.
Subanalysis of the Clopidogrel Versus Aspirin in Patients at Risk of Ischemic Events (CAPRIE) trial,24,25 which included 3866 patients with DM, showed that clopidogrel was superior to aspirin among patients with DM for the primary composite of vascular death, MI, and stroke (15.6 versus 17.7%, p = 0.042). This treatment effect was similar in magnitude to that observed in the overall trial (9% risk reduction overall; 13% reduction in the DM subset), with no statistical evidence for a differential treatment effect by DM status (p interaction = 0.36). The Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management and Avoidance (CHARISMA) trial, 26 which assessed the cardiovascular efficacy of clopidogrel versus placebo among 15,603 patients with stable CHD or with multiple CHD risk factors, showed no significant benefit on the primary outcome of time to first event of the composite end point of cardiovascular death, MI, or stroke in the overall study cohort (RR 0.93; 95% CI 0.83–1.05; p = 0.22), with a statistically non-significant point estimate in the opposite direction in the subset with DM (RR 1.2; 95% CI 0.91–1.59; p = 0.20).
In the Clopidogrel in Unstable Angina to Prevent Recurrent Events (CURE) trial, 23 a randomised trial of patients with either unstable angina or recent MI, including 2840 patients with prevalent DM, the efficacy associated with aspirin alone versus aspirin plus clopidogrel was numerically smaller in the subset with DM (primary event rates 16.7% versus 14.2%; p > 0.05) compared with the non-diabetic group (9.9% versus 7.9%; p < 0.05); i.e. a 15% non-significant relative reduction in the DM group and 20% RRR in those without DM, though no statistical comparison or interaction testing was reported. In the Clopidogrel for the Reduction of Events During Observation (CREDO) trial, 28 which randomised over 2000 patients with CAD undergoing elective PCI, the point estimate at 1 year of combined death, MI, and stroke for clopidogrel versus placebo showed only a non-significant 11.2% RRR (95% CI 46.2 to -46.8) in patients with DM, compared with 26.9% RRR (95% CI 3.9–44.4) observed in the overall study cohort. Although the DM by treatment interaction was not statistically significant, this is a numerically smaller treatment effect than observed in other higher-risk subsets such as those with ACS (27.6% RRR, 95% CI 47.8 to −0.40), and those who underwent a stent procedure (28.8% RRR, 95% CI 47.4–3.6).
The Clopidogrel and Aspirin Optimal Dose Usage to Reduce Recurrent Events – Seventh Organization to Assess Strategies in Ischemic Symptoms (CURRENT-OASIS 7) trial was a randomised trial that sought to evaluate the efficacy of different clopidogrel and aspirin regimens using a 2 × 2 factorial design in the prevention of MACE and stent thrombosis in patients with ACS events. 29 Patients were randomised to increased-dose clopidogrel (600 mg on day 1, 150 mg on days 2–7, then 75 mg daily), versus standard-dose (300 mg on day 1, then 75 mg daily), and factorially to ‘high-dose’ aspirin (300–325 mg daily) versus low-dose (75–100 mg daily). Results showed that increased-dose clopidogrel, when compared with standard dosing, did not statistically reduce the rate of cardiovascular death, MI, or stroke at 30 days (HR 0.94; 95% CI 0.83–1.06; p = 0.30), and was associated with an increased bleeding risk (HR 1.41; 95% CI 1.09–1.83; p = 0.009). Subgroup analysis of the 5880 patients with DM included in the trial showed consistent and similarly non-significant treatment effects of increased- versus standard-dose clopidogrel in patients with DM (HR 0.86; 95% CI not reported; p = 0.16).
Increased risk for stent thrombosis in patients with DM while on clopidogrel therapy has also been reported, representing a form of clinical clopidogrel failure.30,33,42,45,46 Although there is some controversy in the accuracy of adjudication of stent thrombosis versus ‘usual’ ACS, the diabetic cohort consistently represents those at highest risk of definite or probable stent thrombosis in the contemporary era of prevalent clopidogrel use.47,48 When analysing data from a prospective observational study, Iakovou et al. have reported that DM independently predicts stent thrombosis (HR 3.71; 95% CI 1.74–7.89; p = 0.01) despite dual antiplatelet therapy in patients who underwent successful implantation of drug-eluting stents. 42
Clopidogrel compared with other P2Y12 antagonists
In the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel-Thrombolysis in Myocardial Infarction 38 (TRITON-TIMI 38), 49 13,608 patients with ACS who were scheduled to undergo PCI were randomised to therapy with prasugrel versus clopidogrel. The primary study results demonstrated superiority of prasugrel with regard to the primary composite outcome of death from cardiovascular causes, non-fatal MI, or non-fatal stroke when compared with patients treated with clopidogrel (HR 0.81; 95% CI 0.73–0.90; p < 0.001). An important subanalysis of the TRITON-TIMI 38 trial evaluated the effect of prasugrel versus clopidogrel among 3146 study participants with DM. 50 In this subset, the effect of prasugrel versus clopidogrel was statistically superior and greater in magnitude than observed in the overall study cohort (HR 0.74; p = 0.001), with the treatment-by-diabetes interaction test just missing statistical significance (p interaction = 0.05). Rates of MI were also significantly reduced in patients with DM when compared to those without (40% versus 18%, respectively; p < 0.001, p interaction = 0.02), underscoring the importance of enhanced platelet inhibition in this high-risk population. Furthermore, this incremental efficacy of prasugrel over clopidogrel among the diabetic participants was observed in the absence of significantly increased bleeding risks associated with prasugrel as was observed in the overall trial and in the non-diabetic subset.
The Platelet Inhibition and Patient Outcomes (PLATO) trial was a randomised, multicentre, double-blind trial that compared ticagrelor, a reversible, non-thienopyridine, ADP P2Y12 antagonist, to clopidogrel for the prevention of cardiovascular events in patients with ACS. 51 Results for the primary outcome showed that treatment with ticagrelor as compared with clopidogrel resulted in significantly reduced rate of cardiovascular death, MI, or stroke (HR 0.84; 95% CI 0.77–0.92; p < 0.001), without a significant increase in major bleeding (p = 0.43). Similar (but not greater) efficacy was observed in the subanalysis evaluation of the 4662 patients with DM, 52 which showed significant reductions with ticagrelor compared with clopidogrel in the primary composite end point (HR 0.88; 95% CI 0.76–1.03), all-cause mortality (HR:0.82; 95% CI 0.66–1.01), and stent thrombosis (HR 0.65; 95% CI 0.36–1.17).
Mechanisms of clopidogrel unresponsiveness
Though the specific aetiological underpinning of clopidogrel hyporesponsiveness associated with DM remains unclear, there are several plausible mechanisms that may be contributing. In patients with DM, decreased circulating active metabolite while maintaining normal ex vivo clopidogrel responsiveness has been observed in patients described as having clopidogrel unresponsiveness, 40 whether as a result of impaired absorption, impaired metabolism from prodrug to active metabolite, increased rate of clearance, increased platelet turnover, or some combination thereof. Additional mechanisms postulated to influence clopidogrel unresponsiveness include genetic differences in P2Y12 receptors, leading to decreased effectiveness of clopidogrel,16,53–56 increased expression of P2Y12 receptors, increased circulating ADP, increased circulating esterases, and upregulation of other platelet activation pathways.7–9 In patients both with and without DM, interactions between clopidogrel and other drugs have been largely inconsistent, but with the majority of evidence supporting no clinically significant interaction. 22 Similarly, evidence regarding the impaired absorption of clopidogrel has been inconclusive.
The introduction of prasugrel, as evaluated in the TRITON-TIMI 38 trial, has been instrumental in elucidating the aetiology of impaired clopidogrel responses. Similar to clopidogrel, prasugrel is an oral thienopyridine that exerts its antiplatelet effects by irreversibly antagonising the P2Y12 receptors on platelets. Both drugs are prodrugs, and must be converted to their active form before they can influence platelet reactivity. However, there are key differences in their metabolism and subsequent conversion to their active metabolites. Compared with the two-step cytochrome P450 metabolic process to convert clopidogrel to active metabolite, prasugrel is rapidly hydrolysed by esterases to a thiolactone, which is subsequently metabolised to the active metabolite in a single step by several cytochrome P450 enzymes, mainly CYP3A4/5 and CYP2B6. Studies have suggested that although the mechanism of action of prasugrel is similar to that of clopidogrel, treatment with prasugrel results in a higher and more consistent level of platelet inhibition.57,58 Another possible contributing factor to the pharmacodynamic differences observed between prasugrel and clopidogrel is related to the increased activity of circulating esterases found in patients with DM. Increased esterase activity would, in theory, increase the formation of the active metabolite of prasugrel, whereas increased esterase activity would increase the proportion of inactive metabolite of clopidogrel, by participating in a competing pathway. A similar finding has been observed in aspirin resistance in patients with DM. 59
Another alternative is brought about by recent data demonstrating that patients with clopidogrel hyporesponsiveness have decreased circulating active metabolite while maintaining normal ex vivo response after exposure to clopidogrel active metabolite.40,51 In addition, an in vitro study performed by Sugidachi et al. showed that the active metabolites of prasugrel and clopidogrel were equipotent. 60 Studies evaluating polymorphisms of the P2Y12 receptor gene have also been unrevealing with regard to a genetic underpinning of clopidogrel hyporesponsiveness.61,62 Together, these observations largely exclude receptor-based resistance, and support impaired absorption and/or impaired metabolism of clopidogrel to active metabolite, and/or accelerated clearance potentially underpinning impaired responsiveness. Ultimately, clopidogrel unresponsiveness in patients with diabetes is likely multifactorial in origin, related to both the prothrombotic conditions associated with DM and decreased circulating active metabolites in patients treated with clopidogrel.
Conclusions
CHD continues to be rampant across all boundaries of society, comprising an ever-increasing subset of patients with DM. Clopidogrel has proven clinical efficacy in both short- and long-term outcomes in patients with ACS and those undergoing PCI.45,47–49,62 However, accumulating data from pharmacodynamic studies, from epidemiological analyses, and from DM subanalyses from clinical outcomes trials suggest a clinically relevant suboptimal clopidogrel response in at least some patients with DM. The exact mechanism underpinning the variability of clopidogrel response among patients with DM has yet to be fully elucidated, but the majority of evidence suggests that the underlying mechanism is likely attributable at least in part to decreased circulating active metabolite, whether by impaired absorption and/or impaired conversion of prodrug to active metabolite and/or increased rate of hydrolysis of the active metabolite. More recent P2Y12 antagonists, specifically prasugrel and ticagrelor, appear to overcome the decreased response apparent with clopidogrel in those with DM. More research is needed to better understand the pharmacology and clinical consequences of these observations.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
SB provided consultancy for the following pharmaceutical companies: Sanofi-Aventis; Boheinger Ingelheim; Medtronic; Gilead; St Jude Medical, Inc.; Angioslide, Inc.; Cordis. SB was funded by the following institutional research grants: Boston Scientific; The Medicines Company. DKM provided consultancy for the following pharmaceutical companies: Sanofi-Aventis; Novo Nordisk; Tethys Bioscience; Daiichi Sankyo; F. Hoffmann-LaRoche Ltd; Genentech. HMH: none declared.
DKM is a Steering Committee member of the TRILOGY trial, coordinated by the Duke Clinical Research Institute and sponsored by Eli Lilly/Daiichi Sankyo; and the SAVOR trial, coordinated by the TIMI Study Group and sponsored by Bristol-Myers Squibb/AstraZeneca.
