Abstract
The functional relevance of NOS3 and ACE genetic variations to endothelial cell function is largely unstudied. Here we tested the functional relevance of the NOS3 (Glu298Asp) polymorphism and ACE (I/D) polymorphism in endothelial cells in vitro. Our hypothesis was that these genetic polymorphisms alter endothelial cell sensitivity to glucose and 3-nitrotyrosine (3NT). Genotyped HUVECs were incubated with glucose, free 3NT or a combination of these two toxicants. Significant differences in glucose-induced cell death and free 3NT-induced cell death were observed among the NOS3 genotypes. Combined glucose/3NT caused increased toxicity among the NOS3 genotypes. No differences were observed among the ACE genotypes in their responses to glucose/3NT. These data demonstrate that the NOS3 genotype may be an important predictor of, or be mechanistically involved in, endothelial vulnerability, whereas the ACE I/D genotype is apparently less important. Thus this NOS3 genetic variation may play a role in vulnerability to endothelium-dependent diabetic vascular complications.
Keywords
Introduction
Vascular endothelial dysfunction is known to precede structural remodelling during the progression of diabetes. 1 Micro- and macrovascular complications are the major causes of morbidity and mortality associated with diabetes. Patients with type I diabetes have a three- to six-fold increased risk of cardiovascular death before the age of 60 compared with non-diabetic subjects. 2 Loss of the modulatory role of the endothelium as a result of hyperglycaemia has been implicated in the pathogenesis of diabetic vascular complications, but the underlying mechanisms remain poorly understood. 3
Two key components of normal function of the vascular endothelium are the production and regulation of nitric oxide (NO) and angiotensin II (AngII). 4 NO from endothelial cells (ECs) is known to decrease vascular tone, enhance angiogenesis and prevent vascular smooth muscle cell migration and proliferation. 5 AngII stimulates NADPH oxidase both by increasing expression of NADPH oxidase subunits as well as by increasing production of reactive oxygen species in vascular smooth muscle cells, ECs, adventitial fibroblasts6,7 and in intact arteries. 8 In light of these opposing effects, several reports have illustrated important ‘cross-talk’ and interactions between the NO and AngII pathways in endothelium. 9 In addition, it has been shown that polymorphisms of the endothelial nitric oxide (NOS3) gene, and the angiotensin-converting enzyme (ACE) gene, are potential contributors to vascular disease risk.3,10-12 The NOS3 (Glu298Asp) polymorphism has been consistently associated with increased risk of vascular disease in most reports where it has been investigated. 13 We have recently demonstrated that the presence of the NOS3 polymorphism causes loss of functional responses in ECs. 14 Similarly, it appears that at least one variation in the ACE gene, an insertion/deletion genotype (I/D), is most consistently associated with increased risk of cardiovascular disease and poorer outcomes post diagnosis.15-19 However, the contributions of these two important polymorphisms in endothelial dysfunction have not been rigorously studied.
Recent studies have shown that hyperglycaemia is associated with increased formation of reactive oxygen and nitrogen species in vivo, and that these oxidants can promote related complications. 20 It has been shown that diabetic patients have elevated plasma levels of 3-nitrotyrosine (3NT), 21 and that this analyte has been statistically related to decreased vascular reactivity. 22 We have previously demonstrated that 3NT is directly toxic to ECs and that this may be an important contributor to vascular dysfunction in diabetes and other cardiovascular conditions. 23 Although glucose and 3NT have both been demonstrated to cause injury to vascular endothelium, their roles and interactions in diabetic vascular disease have not been well characterised. Furthermore, predisposing patient variables that may modify their toxicities have not been identified.
Here we investigated potential contributions of these two individual genotypes with respect to glucose toxicity, 3NT toxicity, or their combination. We used an approach of performing initial genotyping of primary human EC cultures and subsequent challenging of cells in controlled settings of endothelial stress. These studies of genotype-specific cellular responses to stress complement our recent findings of altered responses to physiological shear and growth factor sensitivities.
Methods
Materials
Human umbilical vein endothelial cell preparations (HUVECs) were used in all the experimental studies. These ECs were obtained from Biowhittaker and each cell line was obtained from individuals with no history of cardiovascular diseases or hyperglycaemia. Each primary cell culture was isolated from an individual donor and all the experiments were carried out in 3–5 individual donors in each group. For all the experiments, cells at less than passage 5 were used. D-Glucose and 3NT were purchased from Sigma. ECs were cultured in Endothelial Growth Medium-2 (EGM-2). Agarose was purchased from Fisher Biotech. All the polymerase chain reaction (PCR) reagents and precast 6% TBE gels were purchased from Invitrogen Corporation. BanII restriction enzyme was purchased from Roche.
DNA extraction
DNA was extracted from the ECs by using DNAzol Reagent (GIBCO) using the manufacturer’s protocol for extraction. In short, DNAzol was added to the cell pellets. Cells were lysed gently by pipetting. The cell pellet was then centrifuged for 10 min at 10,000×g at 4°C. The supernatant was transferred into a new tube and DNA was precipitated by adding 100% ethanol. The DNA was washed three times with 75% ethanol and solubilised in 8 mM sodium hydroxide. Optical density (OD) at a 260/280 ratio was used to determine the purity of the DNA sample.
NOS3 genotyping
ECs were screened for NOS3 genotype using PCR-based DNA amplification followed by restriction enzyme digestion. 24 For detection of the Glu298Asp polymorphism of the NOS3 gene, the following primer pairs were used: sense – 5’ TCC CTG AGG AGG GCA TGA GGC T 3’ and antisense – 5’ TGA GGG TCA CAC AGG TTC CT 3’. Samples were amplified for 30 cycles, consisting of denaturation at 94°C for 1 min, annealing at 61°C for 1 min, and extension at 72°C for 1 min. The resulting 457-bp amplification product was incubated at 37°C for 3 h with 10 U of the restriction enzyme BanII (Roche). The amplified fragments were digested by BanII into smaller fragments (137 and 320 bp). In the case of a G to T substitution, a BanII restriction site is lost. The restriction digest fragments were separated on 6% TBE gels with ethidium bromide staining. ECs were categorised as Homozygous Wild Type (Glu/Glu), Heterozygous variant (Glu/Asp) and Homozygous variant (Asp/Asp).
ACE genotyping
DNA was extracted from cultured HUVECs. For detection of the I/D polymorphism of the ACE gene we amplified genomic DNA with primer pairs: sense – 5’ CTG GAG ACC ACT CCC ATC CTT TCT 3’ and antisense – 5’ TCG AGA CCA TCC CGG CTA AAA C 3’. An insertion- specific primer was also used to reduce the chances of a false negative for the I/D genotype: 5’ GAT GTG GCC ATC ACA TTC GTC AGA T 3’. Samples were amplified for 30 cycles, consisting of denaturation at 94°C for 5 min, annealing at 64°C for 1 min, and extension at 72°C for 1 min, and then final extension at 72°C for 10 min. The PCR products were subjected to 2% agarose gel electrophoresis. The I allele (an insertion of a 287-bp repeat sequence in region 16) can be identified by the presence of bands at 479 and 277 bp originating from the two reverse primers, one in exon 17 and one, the insertion-specific sequence, in intron 16. A band at 199 bp represents the D allele.
Cell viability assay
ECs (1×104 cells/well) were seeded in UV-transparent 96-well plates, grown to confluence for 24 h, then treated for 24 h in medium alone or in medium fortified with 10 µM and 100 µM free tyrosine or toxicants (10 nM–5 mM free 3NT, 10 mM, 20 mM and 30 mM glucose, and a combination of 10 nM 3NT and 20 mM glucose). Cells were then washed and fixed in 5% buffered formalin, and stained with crystal violet as a marker of cell viability. Crystal violet signal was assayed spectrophotometrically at 590 nm.
Nitric oxide estimation
The concentration of nitrite in cell culture supernatant, which reflects cumulative NO production, was measured by using an enzymatic nitric oxide assay kit (Oxford Biomed). Cultured supernatant (50 µl) was sampled. The assay was performed as per the manufacturer’s instructions. Briefly, nitrite reductase was added to the samples in the presence of NADH. This was followed by addition of Sulfanilamide (p-Aminobenzenesulfonamide) dissolved in 3 N HCl and N- (1-Naphthyl) ethylenediamine dihydrochloride dissolved in deionised H2O. Absorbance was read at 540 nm. KNO3 at 500 pmol/ml (equivalent to 500 mM NO), was used as a standard to generate the standard curve.
Cell lysis and immunoprecipitation
After washing three times with ice-cold PBS, cells were lysed in NP-40 Lysis Buffer containing 1% NP-40, 50 mM Tris, pH 7.5, 150 mM NaCl, 5 mM EDTA, 0.5 mM Na3VO4, 50 mM NaF, 10 µg/ml aprotinin, 10 µg/ml leupeptin, 10 µg/ml pepstatin-A, and 1 mM PMSF. The cell extracts were used in an immunoprecipitation reaction, using the Protein G Immunoprecipitation Kit from Sigma. The cell extracts were incubated overnight at 4°C with NOS3 polyclonal antibody on protein G agarose beads. The beads were washed thoroughly after incubation and the sample was prepared for SDS-PAGE.
Western blotting
Samples prepared as described above were separated on 4–20% Tris-glycine gel. After electrophoresis, the proteins were transferred onto nitrocellulose membranes. The membranes were blocked with 5% non-fat milk in Tween 20 (0.1%) and Tris-buffered saline (10 mM Tris, pH 7.5; 150 mM NaCl), and incubated with primary antibody for 1 h at room temperature. An HRP-conjugated secondary antibody (Vector Laboratories), an enhanced chemiluminescence reaction (PerkinElmer Life Sciences), image capture using UVP EpiChemi dark room system and image analysis (ImagePro, Media cybernetics) were used to assess the relative amount of target at the appropriate molecular weight.
Statistical analysis
All data are expressed as Mean±SEM of three replicates from 3–5 individuals for each genotype. Each cell line was treated as an individual sample. GraphPad Prism software was used to fit the data and carry out the statistical analysis. A simultaneous comparison of groups was handled by one-way analyses of variance (ANOVA), with Student Newman–Keuls post-hoc analysis. For analysing the combination effects, two-way ANOVA was used to test differences between the genotypes and various treatments. In all cases, significance was defined as p<0.05.
Results
High glucose toxicity
The cells were exposed to high glucose concentrations. Upon exposure to glucose (10 mM, 20 mM and 30 mM) about 40% cells were killed at the highest concentration. At 10 mM glucose the percentages of cells left alive were 93.45±1.81% for Glu/Glu; 73.72±3.07% for Glu/Asp (p<0.05); and 82.51±6.11% for Asp/Asp. The Glu/Asp heterozygote demonstrated more vulnerability as compared with the Glu/Glu wild type. At 20 mM both variants demonstrated a higher susceptibility as compared with the wild type (90.73±2.54% for Glu/Glu; 69.65±2.79% for Glu/Asp; and 71.31±2.30% for Asp/Asp). At the highest glucose concentration there was no difference in the amount of cells killed. Interestingly, ACE genotypes demonstrated no differences in their susceptibility to high glucose. At 10 mM glucose, 89.86±2.30% I/I, 92.92±0.75% I/D and 90.86±1.85% D/D cells were alive. The percentage of cells alive after 20 mM glucose treatment was 69.76±1.42% for I/I, 74.27±1.62% for I/D, and 66.40±1.33% for D/D (all NS) (Figure 1).
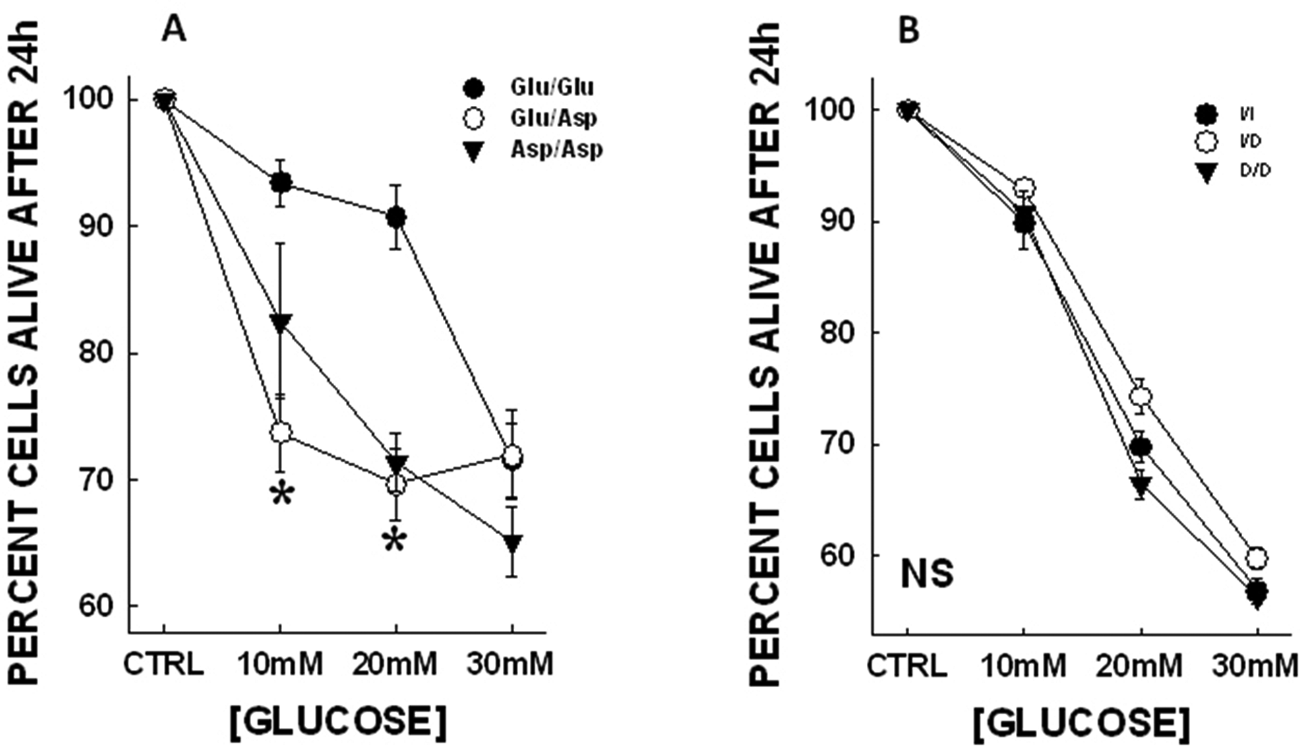
Glucose toxicity in genotyped endothelial cells. Data are represented as mean±SEM. Significant differences were observed in the NOS3 (Glu298Asp) genotypes in response to high glucose (* p < 0.05 when compared to Glu/Glu). No apparent differences were observed in the ACE (I/D) genotypes. In the NOS3 genotypes at the lowest concentration of glucose (10 mM) the amount of cells alive in Glu/Asp group is significantly lower than the Glu/Glu group (93.45±1.81% for Glu/Glu; 73.72±3.07% for Glu/Asp; and 82.51±6.11% for Asp/Asp). At 20 mM glucose both the variant genotypes have enhanced cell death as compared with the wild type cells (90.73±2.54% for Glu/Glu; 69.65±2.79% for Glu/Asp; and 71.31±2.30% for Asp/Asp; p<0.05).
Free 3NT toxicity
Upon addition of 3NT to the cell culture medium, there was no change in the pH of the medium even at the highest 3NT concentration. Various NOS3 genotypes demonstrated differing responses to free 3NT exposure. The free 3NT LC50 values for the NOS3 genotypes were 12.96±0.58 µM for Glu/Glu; 4.37±0.25 µM for Glu/Asp (p<0.05); and 10.83±0.62 µM for Asp/Asp. At the highest concentration of 3NT (5 mM) almost 95% of cells were killed in all the NOS3 genotypes. The heterozygous variant was more susceptible to 3NT toxicity. On the other hand, the ACE genotypes demonstrated no differences in their susceptibility to free 3NT concentrations. The LC50 values for the three ACE genotypes were 8.32±3.45 µM for I/I; 11.69±3.98 µM for I/D; and 8.28±1.88 µM D/D (see Figure 2).
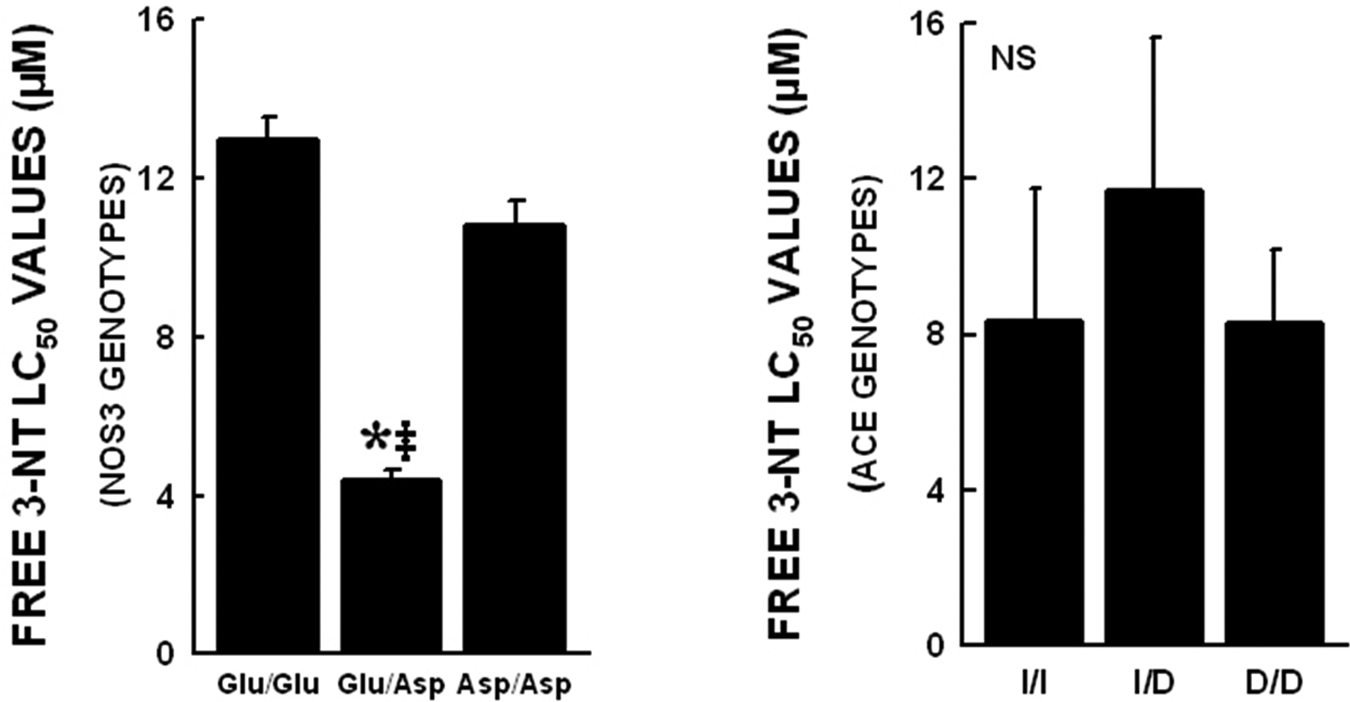
LC50 values obtained for various NOS3 and ACE genotypes are shown. Data represented as mean±SEM (*p<0.05 when compared with Glu/Glu and ‡p<0.05 when compared with Asp/Asp). No significant differences were observed among the ACE genotypes. The NOS3 Glu/Asp variant cells have enhanced vulnerability to free 3NT treatment. The LC50 values were significantly lower in this group as compared with the Glu/Glu and Asp/Asp group (12.96±0.58 µM Glu/Glu, 4.37±0.25 µM Glu/Asp and 10.83±0.62 µM Asp/Asp; p<0.05).
Combination toxicity
The combined effects of 3NT and glucose on cell survival in the NOS3 genotypes were also investigated. The concentrations chosen for both the toxicants are similar to those seen in the plasma of patients with diabetes. In all the genotypes, combination treatment resulted in increased cell death. Comparison of the combination treatment effects in the genotypes showed that the Asp variant cells were killed to a greater extent as compared with the wild type cells (Figure 3).
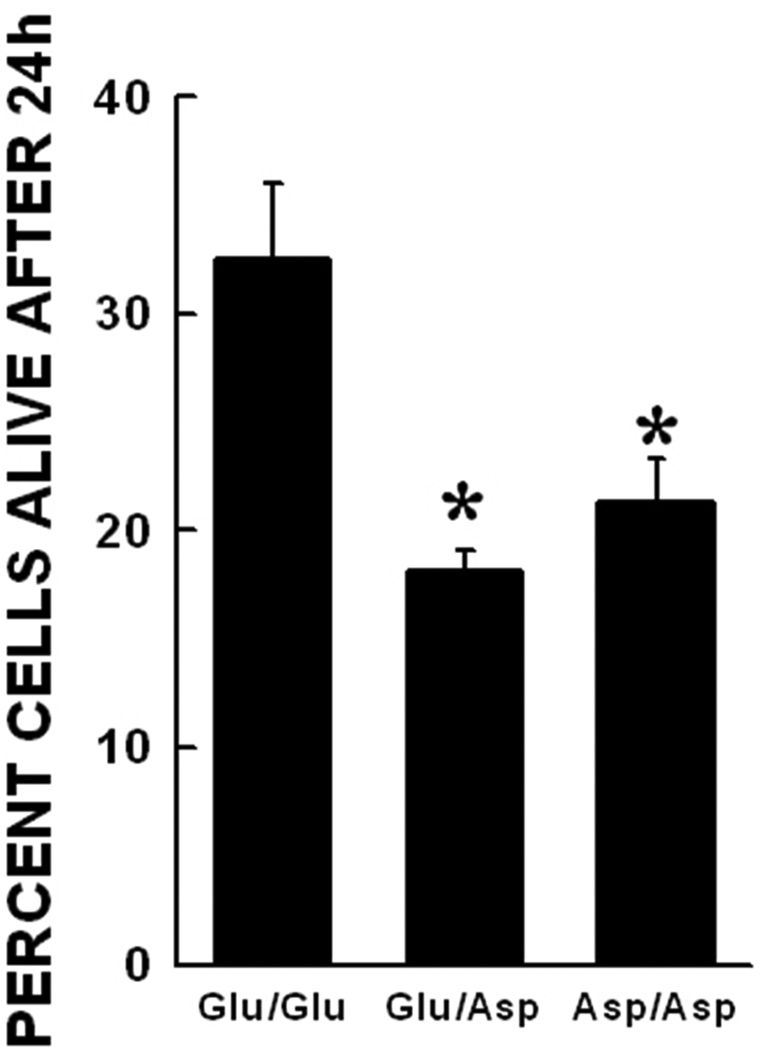
Combined glucose and free 3NT toxicity in NOS3 Glu298Asp genotypes. Data represented as mean±SEM. The combination of high glucose and free 3NT was significantly more toxic in all the genotypes. (*p<0.05 when compared with Glu/Glu genotype with similar treatment).
pAkt levels in ECs post-glucose treatment
The proteins from glucose-treated (10 mM) cells were separated on a nitrocellulose membrane. Densitometric analysis was used to calculate the ratio of the phosphorylated Akt (pAkt) band OD to the corresponding band OD after Fastblot staining. This is an indirect measure of the pAkt/Akt ratio in the cells. No significant differences in basal pAkt levels were observed across NOS3 Glu298Asp genotypes. After glucose treatment there was a significant reduction in cellular pAkt levels (p<0.05). There were no differences across the NOS3 genotypes (Figure 4).
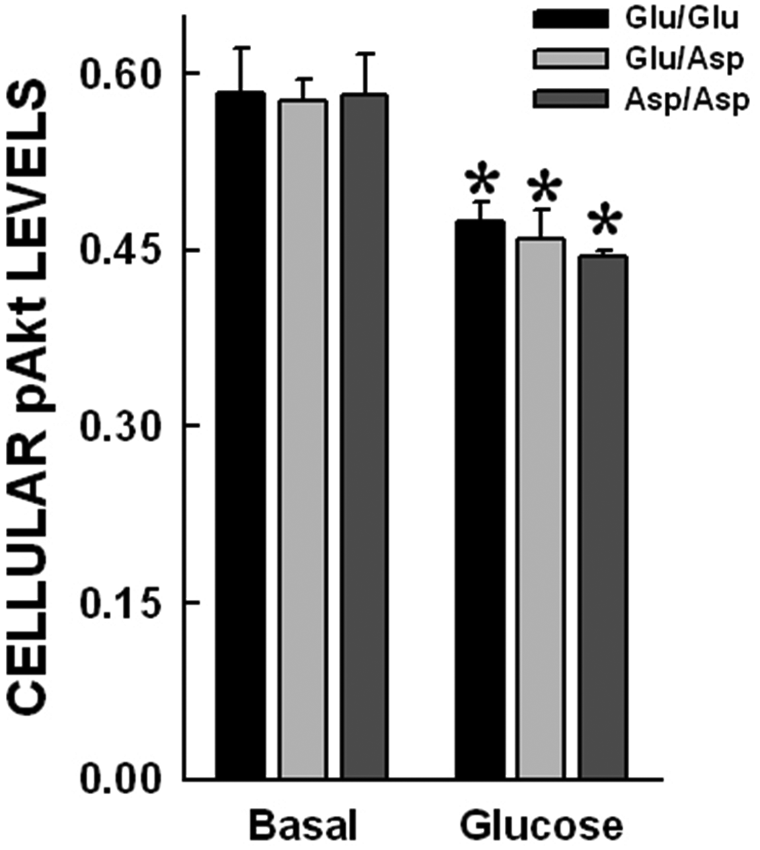
pAkt levels in ECs post-glucose treatment. The proteins from glucose (10 mM)-treated cells were separated on nitrocellulose membrane. The pAkt band OD to corresponding band OD after Fastblot staining was used as a quantitative measure of pAkt levels in the cells. No significant differences in basal pAkt levels were observed across NOS3 Glu298Asp genotypes. After glucose treatment there was a significant reduction in cellular pAkt levels (p<0.05). There were no differences across the NOS3 genotypes.
Nitrite production in ECs post-glucose treatment
Nitrite levels in the media were estimated after 24 h of glucose treatment (10 mM) in the NOS3 genotypes. The data shown in Figure 5 demonstrate that there were basal differences in media nitrite levels in the NOS3 genotypes (8.9±0.20 nM/µg for Glu/Glu; 5.3±0.1 nM/µg for Glu/Asp; and 6.4±0.11 nM/µg for Asp/Asp; p<0.05) (*p<0.05 when compared with Glu/Glu after similar treatment). No significant differences were observed after glucose treatment in all the NOS3 Glu298Asp genotypes. The Glu/Asp and Asp/Asp genotypes demonstrated lower nitrite levels in media before as well as after glucose treatment.
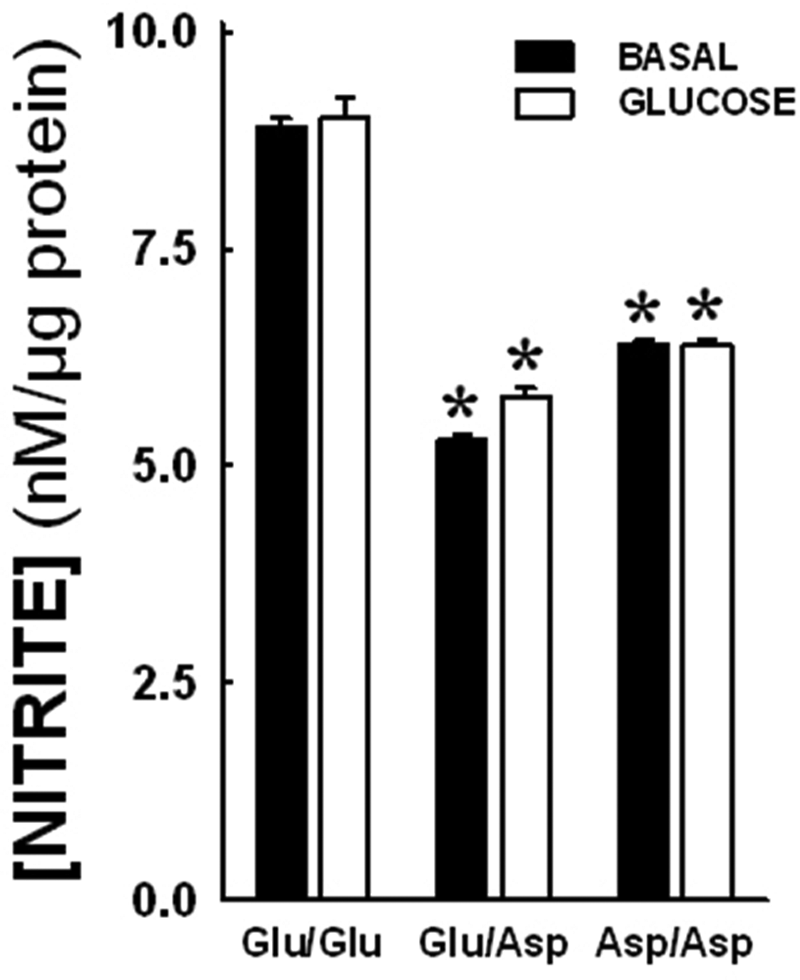
Nitrite production in endothelial cells post-glucose treatment. Nitrite levels in the media were estimated after 24 h glucose treatment. There were basal differences in media nitrite levels in the NOS3 genotypes (8.9±0.20 nM/µg for Glu/Glu; 5.3±0.1 nM/µg for Glu/Asp; and 6.4±0.11 nM/µg for Asp/Asp; p<0.05). (*p<0.05 when compared with Glu/Glu after similar treatment). No significant differences were observed after glucose treatment. The Glu/Asp and Asp/Asp genotypes consistently demonstrated lower nitrite levels in media.
NOS–Cav-1 association post challenge
The results of the immunoprecipitation studies are shown in Figure 6. These studies revealed an impaired association of NOS3 protein with caveolin-1 protein in this setting. We have previously demonstrated that NOS3 protein levels are not significantly different across the various genotypes. 14 However, basal NOS3 associated with Cav-1 in ECs is different among the NOS3 exon 7 genotypes. Immunoprecipitation of cell lysates after high-glucose treatment demonstrated that the Glu/Asp and Asp/Asp variants of the NOS3 protein have a significantly reduced (p<0.05) ability to dissociate from Cav-1.
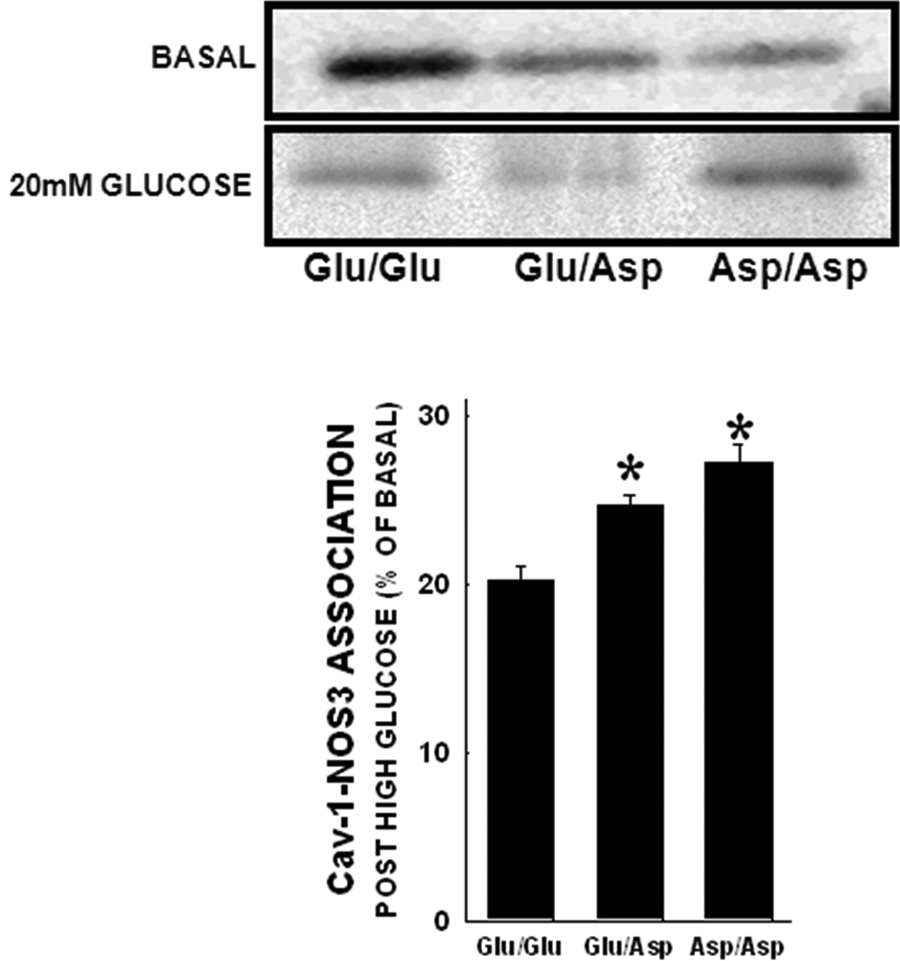
Cav-1 association in endothelial cells post high-glucose treatment. NOS3 immunoprecipitation was carried out and membranes were probed for Cav-1 levels. Under basal conditions Cav-1 associated with NOS3 is significantly lower in the Asp variant genotypes (14, 35). After high glucose treatment, the amount of cav-1 bound to NOS3 is significantly higher in the Glu/Asp and Asp/Asp variant genotypes (p<0.05).
Discussion
The vascular endothelium is an important participant in normal cardiovascular physiology, and dysfunction in this important cell type has been implicated in most forms of cardiovascular disease.25,26 Several variations in the human genome have been linked to increased risk of cardiovascular disease, and some of these are known to relate specifically to endothelium-relevant processes.27-32 Here, we focused on two genetic variations (in NOS3 and ACE) known to occur with high frequency and known to be statistically associated with various forms of vascular disease, including diabetes-related cardiovascular complications. Using this approach we were able to test the hypothesis that the presence of the NOS3 Glu298Asp genotype, or the ACE insertion/deletion genotype, affected the endothelial response to glycaemic stress or 3NT exposure. We recognise that one of the major limitations of human genetic studies in vitro is the lack of control over the genetic background of the individual donors. Variations in the genetic backgrounds may affect our observed results with regards to the gene polymorphisms studied. However, currently there are no other approaches to investigate the direct functional outcomes of genetic variations. We have used cell lines from 3–5 individual donors in each of our studies, and even with this seemingly low number of individual samples we were able to demonstrate significant differences in cellular responses.
In our previously published data, we have demonstrated that neither genotype was associated with a change in basal cellular characteristics. 14 In contrast, striking differences in glucose-induced cytotoxicity were observed among the NOS3 Glu298Asp genotypes, wherein the variant genotypes were apparently five-fold more sensitive to glucose-induced cell death (Figure 1). We have already found evidence that this NOS3 mutation at position 298 is associated with decreased basal NO production as well as diminished shear-dependent cellular response and altered NOS3 caveolar localisation. 14 In general, the observation that glucose toxicity is enhanced is consistent with other reports showing that NO can be cytoprotective and that NOS3 activity is diminished during diabetes-related vascular disease. 33 In parallel studies we found no evidence of altered cell vulnerability to glucose with respect to ACE I/D genotype, suggesting that the NOS3 genotype may actually be a contributor to the mechanism of glucose-induced endothelial toxicity during diabetes. Although this and other NOS3 genotypes have been suggested to be associated with diabetic cardiovascular complications, this is the first report to suggest a specific mechanism involved. We further investigated the mechanisms of NOS3 genotype-dependent alterations in the EC responses.
In addition to hyperglycaemia, increases in plasma 3NT have been documented in various forms of vascular disease, including diabetes.21,23 Here we tested the hypothesis that 3NT cytotoxicity was NOS3 Glu298Asp-genotype dependent. Slightly enhanced toxicity was observed in the NOS3 Glu/Asp genotype relative to wild type (Glu/Glu), whereas no statistically significant differences were observed for cells sorted based on ACE I/D genotype. The mechanism involved in 3NT-induced cytotoxicity is not clearly defined, but others have implicated disruption of mitochondrial electron transport. 34 Further studies of the interactions of NOS3 activity, the Glu298Asp polymorphism, and 3NT cytotoxicity are warranted, especially those involving the contribution of NOS3 to mitochondrial function in this cell type. Responses to 3NT were significantly different between the Glu/Asp and Asp/Asp variants. It is difficult to explain the differences between the two variant genotypes, given the fact that both variant genotypes had lower association with Cav-1 under baseline. This seemingly enhanced toxicity in the heterozygotes could be due to asymmetry at the protein level. We have previously demonstrated that substitution at amino acid 298 causes a significant reduction in amino acid distances, and these may affect protein geometry. 35 Alterations in protein geometry are known to affect various protein–protein interactions, and it is possible that this asymmetry in the Glu/Asp variants may significantly affect NOS3 enzyme function and cell survival in response to stress.
Since we observed an enhancement of 3NT EC toxicity in the NOS3 Glu/Asp genotype, we further investigated potential interactions of glucose and 3NT. We tested whether their combination would be synergistically toxic to ECs in vitro. As we previously observed, glucose (20 mM), or 3NT (10 µM) both caused cell death at the concentrations investigated, and the extent of cell death was enhanced in the NOS3 variant genotypes (Glu/Asp, Asp/Asp) when compared with wild type (Glu/Glu). When these treatments were combined the extent of cytotoxicity was found to be non-synergistic; the toxicities were either additive or less than additive, depending on NOS3 genotype (Figure 3). These data suggest that although each of these substances may contribute to vascular endothelial injury, there is apparently no synergistic interaction with respect to signalling events involved. Thus, glucose and free 3NT may have discrete pathways of cellular toxicity.
Given the observation of enhanced glucose toxicity in the presence of the NOS3 Asp variant allele, we investigated the potential mechanisms of altered Akt activation during glucose toxicity in the variants. Hyperglycaemia has been shown to diminish activation of Akt/protein kinase in rat skeletal muscle. 36 The Akt phosphorylation-dependent survival pathways are essential for normal NOS3 activation and function; 37 high glucose is also involved in pro- apoptotic pathways, and this may alter the pAkt-dependent survival signalling mechanisms.38,39 Furthermore, this signalling pathway also regulates NOS3 phosphorylation, NO release and vasomotor tone. There was no difference in basal (5.5 mM glucose) levels of the phosphorylated form of Akt (pAkt) among NOS3 genotypes; hence we did not probe for changes in NOS3 phosphorylation levels in this setting. pAkt was reduced after glucose treatment at 10 mM. However, no NOS3 genotype-dependent differences were observed in this setting. In contrast, levels of NO were lower in the Asp variant cells under basal control glucose exposures (5.5 mM) and after 24 h at 10 mM glucose. Since basal NO production in ECs has been shown to be anti-apoptotic it is possible that the lower levels of basal NOS activity, measured indirectly via NO estimation, in the variant genotypes may predispose these cells to glucose toxicity. Similarly, we found that 10 mM glucose caused an approximate 80% dissociation of NOS3 from caveolin-1 in the wild-type cells, whereas the extent of dissociation was reduced in the Asp genotype groups (Figure 6).
Here we focused on two already known and endothelium-related genetic variations in human cells. Using an approach of detailed studies of isolated primary cell lines from humans we were able to characterise and compare the functional responses of ECs based on their known genotype. We believe that this is a valuable approach to better characterise endothelial consequences of individual genetic variations. This unique approach may serve as an example of a cell-based functional genomics strategy, and could lead to better definition of functions and mechanisms of human genetic variations.
These findings are consistent with our recent observation that the variant genotypes also have diminished responses to shear-induced NOS3 activation and translocation from caveolar structures. 14 Overall, these data suggest that the mechanism of enhanced vulnerability to glucose toxicity is likely to be related to altered NOS3 activity and/or mobilisation under stress conditions.
Footnotes
This work was supported by the National Institutes of Health [Grant numbers HL63067 and DK55053; PI: JAB] and the Victorian Government’s Operational Infrastructure Support Program.
The authors declare that they have no conflicts of interest.
