Abstract
Background:
Physician-led thoracic ultrasound (TUS)-guided biopsies provide a prompt route to tissue diagnosis in pleural and peripheral lung malignancies. This retrospective study reviews the diagnostic performance and safety of this approach in a UK District General Hospital.
Methods:
Time to biopsy, diagnostic yield and complication rate were analysed in a cohort of 49 patients undergoing ultrasound-guided tissue sampling between September 2019 and December 2022.
Results:
Fifty-one TUS-guided biopsies were attempted. Mean time from decision to biopsy was 5 days. The overall diagnostic yield was 82%. Complication rate was low; 3 minor adverse effects were reported which led to no change in routine care.
Conclusion:
This single centre retrospective study shows that physician-led TUS-guided biopsy provides a safe and timely method of obtaining a tissue diagnosis in thoracic malignancy. It offers an alternative to computer tomography (CT)-guided or thoracoscopic biopsies and should be considered in selected patients where local procedural expertise exists.
Keywords
Introduction
Thoracic Ultrasound (TUS) is an essential tool in the diagnosis and management of pleural disease. Immediate pre-procedure bedside TUS, or direct TUS guidance, is a pre-requisite for safe pleural procedure delivery.1,2 Training in TUS is incorporated into postgraduate training and supported by a respiratory-specific ultrasound training framework,3,4 which includes provision for ‘Expert’-level operators to perform costal pleural biopsies percutaneously under real-time direct ultrasound guidance. This approach may also be used to sample peripheral lung masses with adequate pleural contact.
Until recently, these techniques have been the reserve of radiologists. However, due to an increase in ultrasound-trained physicians, and increasing demand on radiology services, physician-led services are becoming more widespread. The procedure is performed under local anaesthetic and may be integrated into a comprehensive ‘one-stop’ outpatient assessment in appropriately selected cases. A physician-led TUS biopsy service has been offered at our district general hospital since August 2019.
Methods
Data were collected retrospectively for all physician-led TUS-guided biopsies performed over a 3-year period between September 2019 and August 2022 at St John’s Hospital, Livingston, UK. As a service evaluation utilising routinely collected data, informed consent and ethical approval was not required. Local Caldicott approval for data collection was obtained from NHS Lothian [Caldicott reference 2349]. Patient demographics, Eastern Cooperative Oncology Group performance status (PS, recorded in 49/51 cases), time from biopsy decision to procedure, biopsy result, final diagnosis, treatment planned in response to biopsy result, and any biopsy-related complications were recorded. Simple descriptive statistics were used to present the data.
Patient selection
Patients for investigation with TUS-guided biopsy were selected by a respiratory consultant with an interest in pleural disease (GM). Indications included:
Discrete chest wall, pleural or peripheral lung mass, or nodular pleural thickening
Pleural effusion with non-diagnostic cytology from preceding pleural fluid sampling
Initial investigation of pleural effusion with high clinical suspicion of either tuberculous pleuritis or malignant pleural mesothelioma (MPM); defined as asbestos exposure history and/or presence of calcified pleural plaques
Safe acoustic access, that is, biopsy target not obscured by ribs or scapula, and a lack of proximity to major vascular structures, was a requirement in all cases. The presence of pleural fluid was not deemed essential in cases where it was expected that the biopsy target could be accessed without lung contact.
Biopsy procedure
Biopsies were obtained in a variety of settings including inpatient and outpatient procedure rooms. They were performed by a Respiratory Physician (GM) or a trainee physician-operator under direct supervision, assisted by a Respiratory Nurse Practitioner, without radiology department support. 14- or 18-gauge Achieve® (Merit Medical, South Jordan, UT, USA) core-cutting needle and a non-throw technique, or a 16-gauge BioPince™ (Argon Medical, Plano, TX, USA) core biopsy needle with a 13–23 mm throw were used to obtain samples.
A Sonosite SII ultrasound machine (FujiFilm Sonosite, Bothell, WA, USA) and low-frequency (2–5 MHz) curvilinear probe using ‘abdominal’ mode pre-settings was used to identify and characterise the area of interest. Local anaesthetic infiltration (up to 3 mg/kg 1% lidocaine) and tissue sampling were then performed under direct real-time image guidance. A strict aseptic technique utilising a sterile ultrasound probe cover was utilised.
Focussed ultrasound was performed during, and post procedure to assess for immediate complications. Observations were taken and the patients were monitored for at least 1 h. Post-procedure chest radiographs were requested at 1-h post-biopsy in all lung biopsy cases and in those pleural biopsy cases where minimal pleural fluid was present. Samples were sent for pathology assessment in formalin and for microscopy and culture in 0.9% saline solution where pleural infection was suspected. Molecular pathology analysis was performed on samples where the histology indicated a diagnosis of lung cancer, and all cases of thoracic malignancy were reviewed at the local Lung Cancer multi-disciplinary team meeting.
Follow-up arrangements
All patients received post-biopsy clinic follow-up to receive results either via local pleural or lung cancer services. To assess diagnostic performance, a final diagnosis was made by the responsible consultant after a minimum of 6 months of clinical follow-up with all available clinical, radiological and pathological data considered.
Results
Tissue was obtained via TUS-guided biopsy in 49/51 planned cases. Two planned procedures were abandoned; one due to inadequate TUS image quality, and one due to pre-syncope during pre-procedure local anaesthetic infiltration. 12/51 (24%) of biopsy subjects were male. The median age was 73 years (range 31–89) and the median PS was 1 (range 0–4).
Time to biopsy
The median time from initial consultation and sampling decision to biopsy was 5 days (IQR 0–7 days, range 0–20 days). The outlying patient that waited 20 days had a non-diagnostic aspiration of pleural fluid performed prior to TUS-biopsy.
Diagnostic yield
A definitive pathological diagnosis was made in 42/51 (82%) TUS-guided biopsy procedures (see Figure 1), rising to 42/49 (86%) where the procedure was completed. Fifteen samples were sent for molecular assessment, of which, 13/15 (87%) were sufficient for full molecular analysis. Of those that needed further sampling: five cases received a final diagnosis of MPM following further tissue sampling (thoracoscopy or repeat TUS-guided biopsy); in one case of tuberculous pleuritis, the TUS-biopsy identified mycobacterium tuberculosis following culture but sampling was insufficient for an earlier diagnosis based on microscopy alone, therefore considered non-diagnostic for the purposes of analysis; one case received a diagnosis of biphasic mesothelioma at post mortem; one case was abandoned after local anaesthetic administration due to inadequate US-images with subsequent supraclavicular node biopsy revealing metastatic lung adenocarcinoma; finally, one case of solitary fibrous tumour of the pleura was diagnosed after a repeat US-guided biopsy.
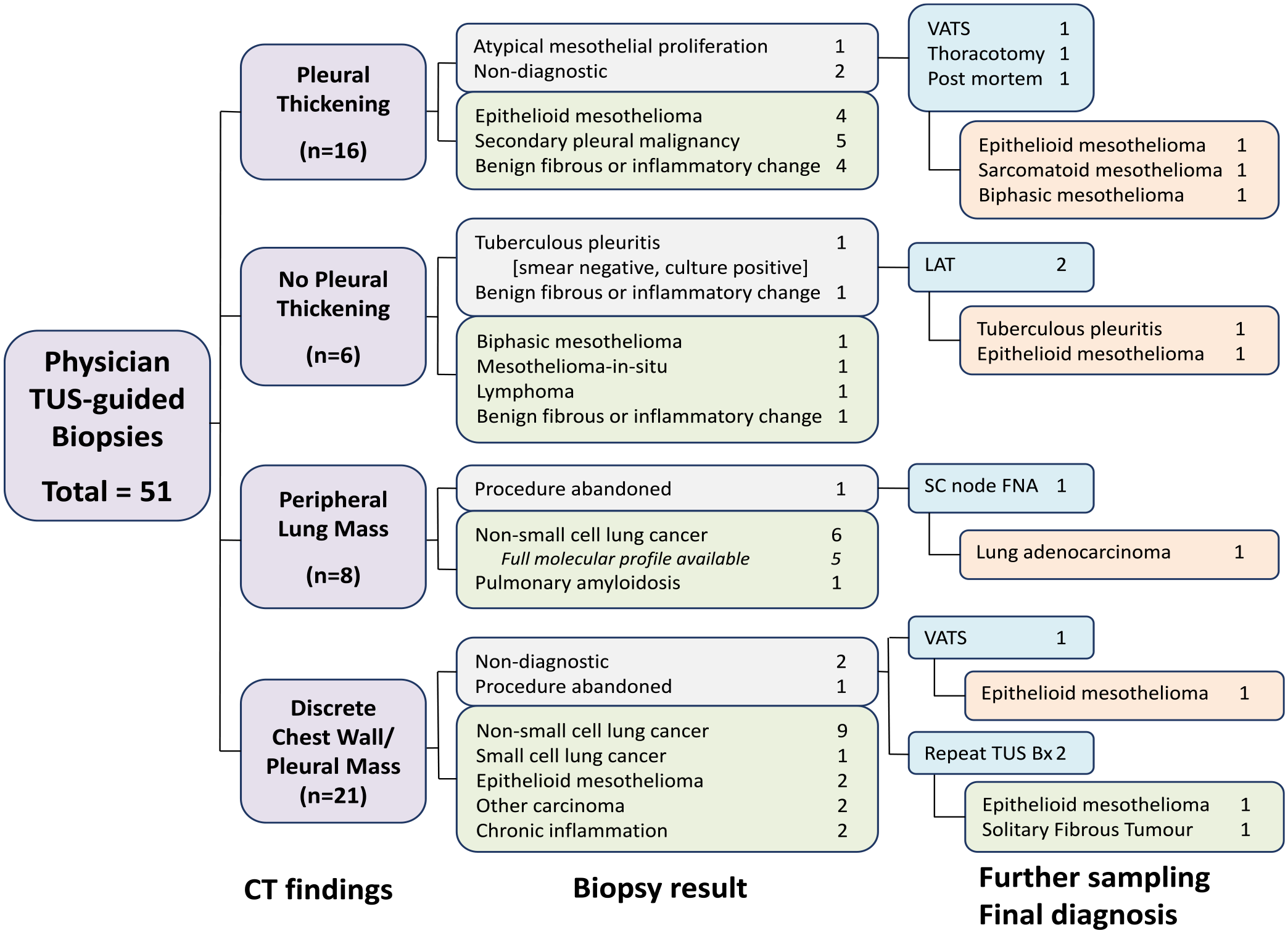
Flowchart of cross sectional imaging findings and diagnostic outcomes of patients undergoing physician-led thoracic ultrasound-guided biopsy.
Complications
There were no serious complications and no post-procedure hospital admissions in those patients attending for biopsy as ambulant outpatients. 2/51 procedures (4%) resulted in asymptomatic minor post procedural intra-pleural bleeding identified on TUS; one patient received a brief period of external thoracic compression and the other patient required no additional management. Both cases of intra-pleural bleeding had pleural thickening identified on cross-sectional imaging.
Discussion
The data presented here demonstrate that a physician-led TUS-guided biopsy service provides a safe, effective and timely means of tissue sampling in pleural and peripheral lung pathology. An overall diagnostic yield of 82% is consistent with reported experiences from tertiary pleural centres5,6 and only slightly below that which would be expected from thoracoscopic approaches (90–95%).7,8 Importantly, in those with peripheral lung cancer, full molecular pathology was available in 87%. This yield is comparable to Endobronchial ultrasound-guided tranbronchial needle aspiration (EBUS TBNA), which provides full molecular profiling in 82–96%.9,10 Furthermore, it is noteworthy that a significant minority of patients 8/49 (16%) obtained a prompt histological diagnosis of MPM without an onwards tertiary centre referral for thoracoscopy.
Our findings highlight the benefit of a US-guided biopsy service in reducing demand on thoracoscopy and CT-guided biopsy services, and providing an acceptable route to a tissue diagnosis in frailer patients who may not have sufficient fitness for thoracoscopy. 11 In our patient cohort, six patients with a PS greater than one underwent biopsies which confirmed MPM. This is of particular relevance, as a robust tissue diagnosis avoids the requirement for postmortem examination and may facilitate an earlier financial compensation claim, even if a pathological diagnosis does not lead to systemic anti-cancer therapy due to lack of fitness. The greater infrastructure required for thoracoscopy means it is likely to remain the preserve of a relatively small number of regional centres and is therefore also associated with an inherently longer diagnostic pathway for patients seen in referring peripheral hospitals. In contrast, US-guided biopsy services may be more widely deployable with evolving physician skills as most respiratory departments already have access to the modest equipment and facilities required. 12
Comparative data between the diagnostic performance of US-guided biopsy and CT-guided biopsy, or thoracoscopic biopsy, is limited. This is likely a reflection that the techniques occupy differing roles within the diagnostic pathway. A retrospective study of 711 thoracic biopsies demonstrated no difference in diagnostic yield for pleural or peripheral lung lesions between CT- and TUS-guided approaches and showed a reduced procedure time and complication rate in the TUS arm. 13 Lee et al. retrospectively analysed a cohort of 1269 patients and demonstrated superiority of TUS guidance over CT for lesions larger than 10 mm. 14 This may be in part due to the ability to biopsy in real time using TUS. Real-time guidance allows compensation for respiration which is an important consideration for biopsy targets affected by diaphragmatic excursion and is not practical using CT due to an unacceptable level of operator ionising radiation exposure. Conversely, some lesions may not be adequately visualised on TUS due to overlying scapula or rib, highlighting that both modalities have their benefits and careful patient selection is key.
Within our District General Hospital there is only one of eight respiratory physicians trained in pleural and peripheral lung biopsy. Clinician training is currently a limiting factor in the development of a flexible and consistent TUS service. The recent British Thoracic Society (BTS) respiratory-specific TUS training framework supports US-guided biopsy at the ‘Expert’ operator level although a specific and prescriptive training syllabus for US-guided biopsy is not provided. 3 Clinicians with ‘Advanced’-level skills in TUS (defined in BTS framework) wishing to develop biopsy skills should identify a local mentor with suitable expertise and use a progressive competency-based approach to training. In the authors’ experience, this would typically involve simulated practice to acquire the core motor skills required to safely handle the biopsy needle and concurrently maintain adequate US probe control, followed by directly supervised clinical practice in at least 10 cases and a summative assessment before independent practice could be considered. This advice is not intended to be prescriptive as it is well recognised that individual practitioners acquire procedural skills at different rates.
In conclusion, a physician-led US-guided biopsy service provides a useful addition to diagnostic pathways with a good safety profile and robust diagnostic performance. This technique may be a particularly advantageous tool for units without prompt access to thoracoscopy services. It is anticipated that with increasing recognition of pleural medicine as a discrete sub-specialty, and a growing number of trained operators, the use of physician-led US-guided biopsy will become more widespread.
Supplemental Material
sj-pdf-1-rcp-10.1177_14782715241231331 – Supplemental material for Physician-led thoracic ultrasound-guided biopsy; a district general hospital perspective
Supplemental material, sj-pdf-1-rcp-10.1177_14782715241231331 for Physician-led thoracic ultrasound-guided biopsy; a district general hospital perspective by M.J. Cafferkey and G.A. Martin in Journal of the Royal College of Physicians of Edinburgh
Footnotes
Data availability statement
The datasets generated and analysed during the current study are not publicly available as individual written consent was not obtained from patients. Caldicott approval obtained and included in supplemental information 1. Any further enquires relating to the dataset can be made to the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
