Abstract
It is widely claimed that in well-nourished populations, very low female waist–hip ratios (WHRs) together with low body mass indices (BMIs) are judged attractive by men because these features reliably indicate superior health and fertility. However, studies show that mortality rates are higher in women with low BMIs than in women with average BMIs and are inversely related to BMI in subsistence populations. Measures of current health in women of reproductive age have not been similarly studied. We analyze large U.S. samples of reproductive-age women and show that controlling for other factors known to affect health, those with low BMIs (<20), WHRs, or waist/stature ratios did not have better health than those with values in the middle range, and there was no relationship between subsequent health outcomes and BMI in early adulthood. Lower self-reported BMIs were linked to poorer health and an increased risk of infection. However, based on recent U.S. natality data, primiparas with lower BMIs had a lower risk of an operative delivery and of gestational hypertension. Beyond these two parity-restricted effects, relevant studies and new tests fail to support the view that women with the very low BMIs and WHRs consistently judged attractive are generally healthier than women with average values; significant correlations were consistently in the opposite direction.
Introduction
Because evolution by natural selection depends on relative reproductive success, in sexual species, there should be strong selection on the preferences of both sexes for mates who will enhance the quantity and/or quality of joint offspring. This perspective suggests that predominant human mate preferences for certain body shapes and feature sizes should reflect long-enduring relationships between the preferred traits and positive fitness outcomes.
During the past few decades, many studies have sought to identify the anthropometric aspects of women’s bodies that are most strongly preferred by men. In general, the women judged most attractive in well-nourished populations have lower waist–hip ratios (WHRs), smaller waist sizes, and lower body mass indices (BMI; see Lassek & Gaulin, 2016, for review and new data supporting those preferences). In these populations, low WHRs are judged attractive only in women with low BMIs and small waist sizes (Andrews, Lukazweski, Simmons, & Bleske-Recheck, 2017; Brooks, Shelly, Jordan, & Dixson, 2015; Lassek & Gaulin, 2016; Rilling, Kaufman, Smith, Worthman, & Patel, 2008).
How then might men’s preferences for low BMIs and small waists enhance their own reproductive success? Beginning with Singh’s (1993a, 1993b) original publications on female body shape, there has been widespread acceptance of his claim that women with lower WHRs have higher fertility and better health (e.g., Grammer, Fink, Moller, & Thornhill, 2003; Marlowe, Apicella, & Reed, 2005; Pawlowski & Dunbar, 2005; Singh, 1993a, 1993b, 2002, 2006; Singh & Singh, 2011; Sugiyama, 2005; Weeden & Sabini, 2005), and similar claims have been made for lower BMIs independent of WHR, for example, “the optimal BMI for health and fertility is struck at around a value of 18-19” (Tovée, Maisey, Emery, & Cornelissen, 1999, p. 216). Because the relevant literature has grown large, in this article, we focus only on the health aspects of the “health-and-fertility” hypothesis: the idea that women whose body shapes are perceived as more attractive are healthier than average. In a companion paper, we show that fertility is lower, not higher, for women with attractive values (Lassek & Gaulin, in press).
If we assume that stabilizing selection causes the mean to approach the optimum, the claim of enhanced health for the BMIs and WHRs judged most attractive seems questionable because the most attractive values are much lower than mean or modal values. Attractiveness increases with larger negative deviations (SD) from the mean. For example, Lassek and Gaulin (2016) examined a number of samples that differed in attractiveness; as attractiveness increased, WHR dropped monotonically. In typical college women, the mean WHR of the most attractive quartile (0.72) was 1 SD lower than the mean for the whole college sample (0.74); the mean WHR of Playboy Playmates (0.68) was 2 SD below the college women’s mean, and the WHR of the most attractive imaginary female (0.39) was 5 SD below the mean of college women.
As is the case for WHR, preferred BMIs in well-nourished populations are also much lower than those of typical young women. Studies have generally found that preferred BMIs are less than 20 (Courtiol, Picq, Godelle, Raymond, & Ferdy, 2010; Crossley, Cornelissen, & Tovee, 2012; Tovee & Cornellisen, 2001; Wang et al., 2015), and the BMI with the highest attractiveness rating has been found to be just 17 in several studies (Fan, Liu, & Dai, 2004; Grillot, Simmons, Lukaszewski, & Roney, 2014; Koscinski, 2013; Lassek & Gaulin, 2016; Stephen & Perera, 2014). This BMI is 2.5 SD below the mean value of 22 in recent normative Dutch and American student samples (Fredriks, Buren, Wit, & Verloove-Vanhorick, 2000; Lassek & Gaulin, 2016) and 2.2 SD below the mode of 21 for 147 samples of 17- to 18-year-old women in 78 countries (Eveleth & Tanner, 1990). The mean value for Playboy Playmates, beauty contestants, and adult film actresses is 18.5 (see Lassek & Gaulin, 2016). For comparison, the mean BMI in a sample of women with anorexia nervosa was 17.9 (Kirchengast & Huber, 2004), and death is not uncommon in anorectic patients with BMIs below 16 (Huas et al., 2011; Okabe, 1993). Thus, men’s preferences for female body shape in well-nourished populations lie in the extreme negative tail of the actual distributions. If the most attractive female shape honestly reflects better health, why would stabilizing selection not have caused female phenotypes to converge on these low values?
The idea that low BMIs are preferred because they indicate better health is also called into question by preferences for higher BMI values in subsistence and forager groups including Shiwiar (Sugiyama, 2005), Hadza (Wetsman & Marlowe, 1999), Tsimane (Sorokowski, Koscinski, Sorokowska, & Huanca, 2014), Matsigenka (Yu & Shepard, 1998), Zulu (Tovée, Swami, Furnham, & Mangalparsad, 2006), and in the Gambia (Siervo, Grey, Nyan, & Prentice, 2006). A survey of 58 cultures from the Standard Cross-Cultural Sample found that men preferred women who were “plump or fat” in 81% (Brown & Konner, 1987). In a study of 54 cultures, this plumpness preference was particularly strong in low-food cultures (Anderson, Crawford, Nadeau, & Lindberg, 1992). Studies of the effects of BMI on health in such subsistence populations should be of particular interest but have not previously been discussed in relation to the health-and-fertility hypothesis.
For these reasons, we believe the claim that the most attractive WHRs and BMIs indicate better health needs to be reevaluated. Studies of either WHR or BMI and health in well-nourished populations are relevant because, as noted above, low WHRs are attractive only in women whose BMIs are also low. Our intention is not to dispute that preferences for low values of BMI and WHR predominate in well-nourished populations; instead, we evaluate the claim that such preferences have evolved because these low values reflect better health in prospective mates than mean or modal values of BMI or WHR.
BMI, WHR, and Mortality
Claims for better health in women with low BMIs and WHRs have been exclusively based on mortality studies in modern well-nourished populations. Evidence from such studies should be treated with caution for several reasons.
First, women’s BMIs are strongly related (r = 0.96) to the amount of stored body fat (Bigaard et al., 2004), and body fat is an essential source of both energy (Caro & Sellen, 1990) and essential fatty acids (Lassek & Gaulin, 2006) needed for survival and reproduction. Body fat often decreases with increasing parity in women in subsistence populations (Lassek & Gaulin, 2006). Thus, one would expect that women with lower levels of fat would be at a disadvantage, although this might be less severe in well-nourished populations with relatively more abundant food and better access to modern medicine.
Second, mortality rates in well-nourished populations are not a good measure of the health of women in their (evolutionarily relevant) reproductive years, since almost all deaths occur in postreproductive women. Based on U.S. mortality data for 1979–1998, only 5% of deaths in women occurred in the 15–44 age-group (CDC Wonder, 2018). This pattern stands in sharp contrast to the situation in non-Westernized populations. For example, among the precontact Hiwi, 56% of female deaths occurred in the 10–39 age-group (Hill, Hurtado, & Walker, 2007). A similar pattern is seen in 19th-century American mortality. Based on mortality data collected as part of the 1860 U.S. census (Edmunds, 1866), 30% of all deaths and 64% of deaths in women 15 and older occurred in the 15–49 age-group. In a meta-analysis of 129 studies that measured BMI and investigated subsequent mortality, the average age of enrollment was 55.7 and the average age at death was 81.0 (Global BMI mortality collaboration, 2016). There have been no previous studies of the relationship between waist size or BMI and current health in women in the reproductive age group.
Third, most disease-related deaths in well-nourished populations are due to chronic diseases in the elderly. Conversely, the major causes prior to the 20th century were infections that affected children and younger adults. Based on the 1860 U.S. mortality data (Edmunds, 1866), 76% of deaths in women aged 15–49 were due to diseases that we now know to be caused by infectious agents. The chronic diseases linked with high WHRs and BMIs in obese older women were quite rare until relatively recently and, moreover, have minimal impact during the reproductive years.
Finally, the very high BMIs that are associated with higher mortality in some studies in affluent groups are rare in subsistence populations (see below). If the negative correlation between BMI and health is driven mainly by this evolutionarily novel very-high-BMI group, it cannot explain an evolved preference for very low BMIs. For these reasons, the relationship between attractive body shapes and health contributions to fitness is overdue for review.
Relevant to this last point, it should be noted that Singh and coauthors (Singh, 1993a, 1993b; Singh & Singh, 2006, 2011), originators of the health-and-fertility hypothesis, did not claim that the most attractive WHRs are healthier, but rather that WHRs < 0.80—a value already 1.5 SD above the mean of 0.74—are healthier than even higher WHRs. While this claim may be true in the modern West today, it is of little evolutionary relevance since this relationship is unlikely to explain why WHRs several SD below the mean are judged to be most attractive in well-nourished populations. No evidence suggests that the very low WHRs rated as most attractive are healthier than mean or modal values.
Historically, the putative link between WHR and health dates to 1947 when the French physician Jean Vague observed that, among obese middle-aged women, those who had more lower body fat (“pears”) appeared to have a lower risk of diabetes and cardiovascular disease than those with more waist fat (“apples”; Vague, 1947). While some studies show that women over 50 with very high WHRs (e.g., >0.88) have increased mortality rates from chronic diseases (Kahn, Bullard, Barker, & Imperatore, 2012; Zhang, Rexrode, van Dam, Li, & Hu, 2008), even in this postreproductive age group, other well-designed studies have found no relationship between WHR and total female mortality (Flegal & Graubard, 2009).
Claims for better health in women with BMIs in the attractive range of 17–19 (Courtiol et al., 2010; Crossley et al., 2012; Fan et al., 2004; Grillot et al., 2014; Koscinski, 2013; Stephen & Perera, 2014; Tovee & Cornellisen, 2001; Wang et al., 2015) are even more problematic, especially in subsistence populations. Female mortality exhibits a negative linear relationship to BMI in Polynesians, Pima, and Australian aboriginals (Hanson, McCance, Jacobsson, Narayan, & Nelson, 1995; Hodge, Dowse, Collins, & Zimmet, 1996; Wang & Hoy, 2002). A study in rural Gambia also found a strong inverse relationship between mortality and BMI, with mortality reaching a minimum for (the rare) BMIs over 25 (Sear, 2006). In these populations, women with low BMIs—in the range consistently preferred by men in well-nourished populations—have significantly elevated mortality rates. Most adult women in forager and subsistence populations tend to have low BMIs in the range of 17–20 (Howell, 2000, 2010; Kramer & Greaves, 2010; Leonard, 2008; Nyak, 2014; Remis & Jost Robinson, 2014; Sherry & Marlowe, 2007, Sugiyama, 2005) and, as mentioned above, the obese BMIs associated with higher postreproductive mortality in well-nourished populations are rare.
Despite the fact that they might be expected to be better buffered from nutritional and disease stresses than their counterparts in subsistence populations, low-BMI women in better nourished populations also have higher mortality than women with BMIs closer to the mean, as shown in several meta-analyses. A recent meta-analysis with data from 139 studies (Global BMI Mortality Collaboration, 2016) with an average age of enrollment of 55.3 found that women with a BMI of 22–25 had the lowest subsequent mortality, whereas those with a BMI of 18.5–20 had a 15% greater mortality risk and those with BMIs <18.5 had a 55% increase in mortality risk. A second recent meta-analysis with data from 230 studies (Aune et al., 2016) found the lowest mortality for women with a BMI of 23.6–26.4, with a 7% increase for BMIs of 21.0–23.4, a 30% increase for BMIs of 18.5–20.9, and a 103% increase for BMIs <18.5. Two other meta-analyses found the lowest mortality for a BMI of 25–29 (Flegal, Kit, Orpana, & Graubard, 2013; McGee & Diverse Populations Collaboration, 2005).
A study of particular relevance followed 12,000 pregnant women in Finland (most under 30 when recruited) for 29 years and found that the women with a prepregnancy BMI of 25–29 had the lowest subsequent mortality (Laara & Rantakallio, 1996). Also, in a longitudinal study of women enrolled at ages 38–60 in Sweden which looked at the joint effects of BMI and WHR, the tertile with lowest WHRs and BMIs had the highest mortality rates (Lapidus et al., 1984).
It should be noted that, although BMI is strongly correlated (.96) with the fat mass index (Bigaard et al., 2004), women with the same BMI can differ in their percentage of body fat. However, this does not change the relationship of low BMIs with mortality. At the high end of the BMI distribution, more body fat is associated with higher mortality, whereas more fat-free mass is not. At the low end of the distribution, less body fat and less fat-free mass are both associated with higher mortality, so that lower BMIs are reliably linked to higher mortality (Bigaard et al., 2004).
The BMIs associated with the lowest mortality are those typical of adult women in well-nourished populations (Finucane et al., 2011; Komlos & Brabec, 2010). Thus, the pattern of lowest mortality at adult women’s modal BMI values seems very well established, as would be expected assuming stabilizing selection had established the current mode.
A study of nurses aged 30–55 when recruited and followed for 16 years (Manson et al., 1995) has been cited in support of better health for lower BMIs (Tovée et al., 1999), but it does not support that claim. Participants reported their BMIs at age 18 and also at the time of recruitment (ages 30–55). Mortality rates did not differ for women whose self-reported BMIs at age 18 fell between 19 and 27. Thus, there was no evidence that lower BMIs in young women were associated with better subsequent health. The relationship between self-reported BMI at the time of recruitment and mortality over the following 16 years was U-shaped with the lowest mortality at a recruitment BMI of 25–26, the same pattern commonly found in other studies of female BMI and mortality.
A recent study of women in Bangladesh, a population with very low mean BMIs, found the lowest mortality for a BMI in the third quartile, which in this population represented a BMI of 18.4–19.6 (Hosegood & Campbell, 2003). However, body fat levels have been rising sharply in Bangladesh (Mohsena, Goto, & Mascie-Taylor, 2016), and there is more adipose in Bangladeshi women with low BMIs than in BMI-matched women in European populations (Shaikh et al., 2016), which makes this result difficult to interpret.
To summarize the mortality data in well-nourished populations, the evidence shows lowest mortality rates in for BMIs and WHRs near the population mean, as would be expected from stabilizing selection. In subsistence populations, mortality decreases monotonically with increasing BMI. In neither context are BMIs or WHRs in the extreme negative tail of the distribution (where attractiveness peaks) associated with lower mortality rates.
BMI and Infection
Until quite recently, infectious diseases accounted for most deaths in women of childbearing age, including many maternal deaths (Charles & Larsen, 1986; Finch, 2010; Fogel, 2004; Hogberg & Brostrom, 1985), and, as noted above, accounted for three quarters of deaths in American women in the reproductive age group in 1860. As recently as 1900, infectious diseases were responsible for more than half of all American deaths under age 45 (Armstrong, Conn, & Pinner, 1999), and a 1908 insurance-company study found substantially higher death rates in adults under 30 with lower-than-average weights (Symonds, 1908).
As also noted above, adult women in forager groups tend to have very low BMIs, and a study of 19 hunter-gatherer groups found that 70% of deaths were due to illnesses caused by infections (Gurven & Kaplan, 2007). In a study of female Hiwi hunter-gatherers, 43% of deaths and 69% of noninjury deaths were due to infection (Hill et al., 2007). In a study of the Iban in rural Sarawak, 69% of women with BMIs less than 18.5 had symptomatic illness versus 44% with higher BMIs (Strickland & Ulijaszek, 1993).
Even in the modern West, despite the availability of modern antibiotics, women with lower BMIs are more likely to die from infection, whereas higher BMIs are protective (Flegal, Graubard, Williamson, & Gail, 2007). Some studies have found that obese women may be at higher risk of hospital infections than nonobese, but those with low BMIs have the highest risk (Falagas, Athanasoulia, Peppas, & Karageorgopoulos, 2009; Huttunen & Syrjanen, 2013; Milner & Beck, 2012).
The relationship of BMI to mortality from tuberculosis, the leading cause of death in the United States in 1900, has been especially well studied, and mortality from this disease is inversely related to BMI (Edwards, Livesay, Acquaviva, & Palmer, 1971; Leung et al., 2007; Marks, 1960; Tverdal, 1986). This inverse relationship of BMI to infection may account for its inverse relationship to total mortality in subsistence populations.
The likely reason that women with lower BMIs have higher mortality from infections is BMI’s relationship to immune function (Wensveen, Valentic, Sestan, Wensveen, & Polic, 2015). As BMI and waist size decrease, fewer pro-inflammatory immune products are made and fewer infection-fighting white blood cells circulate (Alam, Larbi, & Pawelec, 2012; Alvehus, Buren, Sjostrom, Goedecke, & Olsson, 2010; Ilavska et al., 2012; Pou et al., 2007; Thewissen et al., 2011), and women with lower WHRs have increased colonization with pathogenic bacteria (Pawlowski, Nowak, Borkowska, & Drulis-Kawa, 2014).
Health Effects of Low BMIs During Pregnancy
In well-nourished populations, pregnant women with low BMIs are at increased risk of complications which adversely affect the fetus and newborn, such as stillbirths, prematurity, and low birth weight (Han, Mulla, Beyene, Liao, & McDonald, 2011), and their lower BMIs may also put them at greater risk of infections. Nevertheless, low-BMI women themselves may be at lower risk of two complications which can affect maternal health: gestational hypertension and problems during labor requiring an operative delivery, both of which occur more frequently in first pregnancies (Barau et al., 2006; Bhattacharya, Campbell, Liston, & Bhattacharya, 2007; Bodnar, Ness, Harger, & Roberts, 2005; Bodnar, Ness, Markovic, & Roberts, 2005; Nuthalapaty, Rouse, & Owen, 2004; O’Brien, Ray, & Chan, 2003).
Study Design and Hypotheses
The studies cited in support of the purported health benefits of low WHR and BMI in women have used mortality as the outcome measure; but in the contemporary data sets used in those studies, almost all deaths are due to chronic diseases in the postmenopausal age-group. Although postmenopausal women may increase their inclusive fitness by provisioning their daughters’ children (Sear, Mace, & Macgregor, 2000), it seems unlikely that such distal effects could significantly influence the evolution of male mating preferences. Thus, we focus here on relationships in the reproductive age group and predict that, contrary to the prevailing view, the very small waist sizes, WHRs, and BMIs associated with higher female attractiveness will not be associated with better health measures in women in this more evolutionarily relevant segment of the population.
We draw on large American samples to construct new empirical tests of the relationships of WHR and BMI to measures of health in women in the reproductive age group. Based on the foregoing literature review, we make the following predictions.
(1a) Current self-rated health status, limited activity, and health care versus measured BMI, WHR, and waist size. (1b) Disability days, inability to perform a major activity, and significant infections versus current BMIs based on self-report. (1c) Current health status versus lowest BMI after age 18 based on self-report.
Descriptive Statistics for National Health and Nutrition Examination Survey (NHANES), National Health Interview Survey (NHIS), and U.S. Natality Samples With Mean Values and Standard Deviations for Variables.
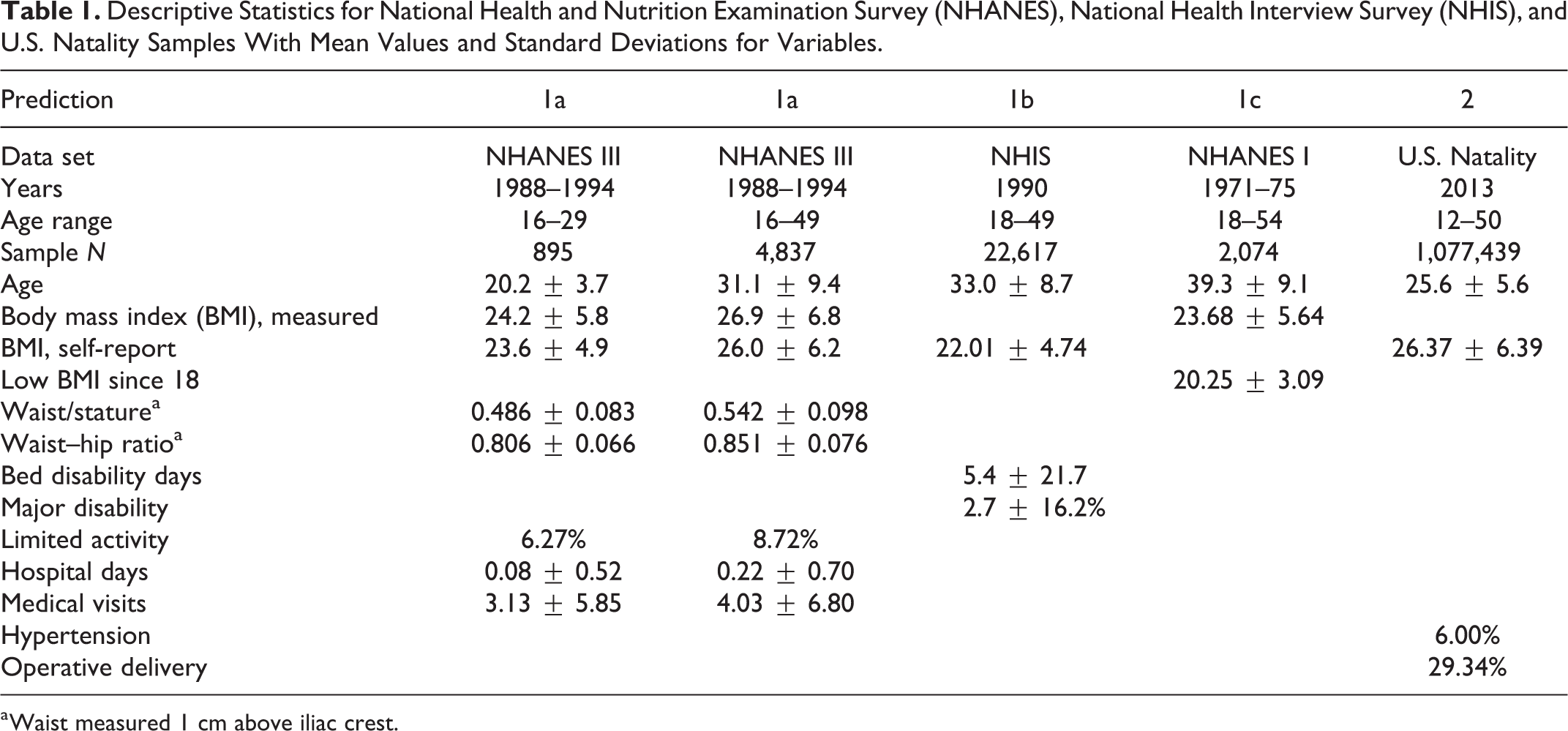
a Waist measured 1 cm above iliac crest.
Method
BMIs Based on Self-Report Versus Measured BMIs
In three of the data sets used in this analysis (see below), BMIs were based on self-reported heights and weights. Because women often report lower weights than those measured (Stommel 2009), the relationship between self-reported and actual BMIs is relevant to interpretation of analyses using self-reported values. We use data for 5,076 women aged 18–49 from the third U.S. National Health and Nutrition Examination Survey (NHANES III) conducted from 1988 to 1994; the methodology of that study allows us to contrast BMIs based on contemporaneous self-reported weights versus measured weights in the same women.
Waist Size, Measured BMI, and Health Measures (Prediction 1a)
Data from the NHANES III (the only NHANES to measure hip circumference) were also used to investigate the relationship of measured BMI, WHR, and waist/stature to four measures of self-reported health in 895 nulliparous women aged 16–29 and in 4,837 women (of various parities) aged 16–49. The four health measures were self-rated health status (coded 1 = poor, 2 = fair, 3 = good, 4 = very good, and 5 = excellent), numbers of medical visits, numbers of hospitalization days in the previous year, and whether or not activity was limited due to health problems. Ordinal regression was used to test for effects on health status, linear regression for hospital days and physician visits, and logistic regression for limited activity. For each measure, we compared BMI <20 with BMIs of 20–29 and the low quartile of WHR and waist/stature with the second and third quartiles.
To examine whether any association between poor health and high BMI (or WHR) might be driving the conclusion that low BMI (and WHR) is healthy, we also compared these same four health outcomes among those with BMIs over 30 and in the fourth quartiles of WHR and waist/stature to health outcomes among those with lower values of these same body-shape measures.
In all analyses, age, family income, years of education, parity (ages 16–49), and serum cotinine (a biomarker for cigarette smoking) were used as continuous covariates and race/ethnicity and current smoking and 100+ lifetime cigarettes as categorical factors.
Relationship of Self-Reported BMI and Health Outcomes (Prediction 1b)
Data from 22,617 women aged 18–49 from the 1990 National Health Interview Survey (NHIS) were used to investigate the relationship of (self-reported) BMI to three disability measures (bed days, hospital days, and inability to perform a major activity) and to three infection measures (presence, associated bed days, and associated restricted activity). This annual interview survey has a larger sample size and more health-related variables than NHANES. Diagnostic codes assigned to persons reporting an acute condition associated with disability days, doctor visits, or hospitalizations in the 2 weeks preceding the interview were used to identify those with infections. Age, family income, race, and years of education were controlled using logistic or multiple regression, as appropriate. In the logistic regressions, those with a BMI less than 20 were compared with those with a BMI of 20–24 because many women with self-reported BMIs of 25–29 are likely to be obese (see Results section).
Minimum BMI After Age 18 (Youth BMI) and Current Health Status in the Reproductive Years (Prediction 1c)
The NHANES I (1971–1975) included a question on the self-reported lowest weight since age 18. For most women, this occurred before age 25. A “youth BMI” was calculated from this weight and measured current stature for 2,074 women aged 18–54. Ordinal regression was used to determine the relationship between youth BMI and self-rated health status (coded as for Predication 1a), with current age, age at minimum weight, family income, years of education, and parity as continuous covariates and race/ethnicity, current smoking, and 100+ cigarettes as categorical factors. In a separate analysis, BMI was treated as a categorical variable with four groups (<20, 20–24, 25–29, and 30+).
Risk of Pregnancy Complications in Relation to Prepregnancy BMI (Prediction 2)
Natality data from U.S. birth certificates for 1,077,439 singleton births to primiparous women in 2013 were used to determine the relationship of prepregnancy BMI to the occurrence of operative delivery by caesarian section and to gestational hypertension (which includes mothers with preeclampsia). Prepregnancy BMI was calculated from maternal stature and self-reported prepregnancy weight as recorded on the birth certificate. Control variables included maternal age, race (White, Black, American Indian, and Asian), self-reported weight gain per month of gestation, cigarette smoking, and years of education. Logistic regression was used for analysis with age as a continuous variable and BMI treated as a categorical variable with grouping <15, 15–19, 20-24, and 25–29 chosen as the reference level.
Results
Mean values for selected variables for the four U.S. data sets are shown in Table 1.
Self-Reported Versus Measured BMIs
Table 2 shows the BMI groups for measured BMIs in relation to those based on self-reports for women aged 18–49 in NHANES III. Of those with BMIs less than 20 based on self-report, 23% actually have higher BMIs; for self-reported BMIs of 20–24, 6.3% are lower and 17.7% are higher. Importantly, for those with self-reported BMIs of 25–29, 7.1% are lower but nearly one quarter (24.2%) are actually obese, a group that may have significantly different health outcomes. To minimize the influence of obese cases on our analysis, we restrict our comparison groups to BMI <20 and BMI 20–24 when using self-reported BMIs.
Comparison of Body Mass Indices (BMIs) Calculated From Self-Reported and Measured Values With Percentage Distribution for BMI Ranges in 5,076 Women Aged 18–49, National Health and Nutrition Examination Survey III.

Prediction 1a: Relationship of Current BMI, WHR, and Waist Size to Four Health Measures
Controlling for age, family income, years of education, race/ethnicity, and cigarette smoking, in nulliparas aged 16–29, and in all women aged 16–49, there was no significant difference between BMIs of <20 and BMIs of 20–29, nor between the first quartiles of WHR and waist/stature ratio (WSR) and the second and third quartiles of those variables for any of the four health measures tested, including self-rated health status, limited activity, hospital days, and medical visits (Table 3). Lower values of BMI, WHR, or waist size were not correlated with better health. As found in some mortality studies, obese women and those in the fourth quartile of WHR and WSR did have significantly poorer health, more limited activity, and more hospital days and medical visits (except for nulliparous obese women). Also, age was negatively related to health status and positively related to disability in women 16–49. Education and family income were positively related to beneficial health outcomes, while cigarette smoking was negatively related. BMI was positively and significantly correlated with age in both nulliparas and in all women aged 16–49 (r = .067, p = .026; r = .244, p < .001,).
Effect of Low Body Mass Index (BMI <20 vs. 20–29) and Waist–Hip Ratio (WHR) and Waist/Stature Ratio (WSR) in the First Quartile Versus Quartiles 2 and 3 on Health Status, Hospital Days, Physician Visits, and Limited Activity in Women Aged 16–29 (n = 895) and 16–49 (n = 4,837) in National Health and Nutrition Examination Survey III, Controlling for Age, Income, Education, Smoking, Parity (16–49), and Race/Ethnicity.

Prediction 1b: Relationship of Current Self-Reported BMI to Three Disability and Three Infection Measures
Controlling for age, family income, race, and years of education in 22,617 females aged 18–49 with a self-reported BMI <25 in the 1990 NHIS, BMI was negatively related to both bed disability and hospital days (Table 4). Those with lower BMIs had more disability and hospital days. In addition, those with a BMI <20 were significantly more likely to be unable to perform a major activity due to medical conditions and to have an infection diagnosis than those with BMI of 20–24. Overall, 7.4% of women had one or more infection diagnoses. In women with infections, those with lower BMIs had more restricted activity and bed disability days due to infection (Table 4). Age was a significant positive predictor of disability, hospital, and bed days, while education and family income were significant negative predictors.
Relationship of Body Mass Index (BMI) With Bed Disability Days, Hospital Days, Major Disability, Infection Diagnosis, and Infection Related Outcomes in 22,617 Women Aged 18–49 With Self-Reported BMI <25 in National Health Interview Survey 1990.

Prediction 1c: Current Health Status Versus Lowest BMI After Age 18 Based on Self-Report
We used ordinal regression to control for age, parity, family income, years of education, cigarette smoking (current or 100+ cigarettes), age at minimum weight, and race/ethnicity. With these controls, there was no significant association between youth BMI and current health (Table 5, “all BMIs”); in contrast, age was negatively related, and education and family income were positive predictors of current health (all p < .001). When youth BMI was treated as a categorical variable with four groups (<20, 20–24, 25–29, and 30+), there was no difference between the groups with BMIs <30 (Table 5). Current BMI was a significant negative predictor of current health status (p < .001) if used in place of youth BMI, but not when the regression was restricted to BMIs less than 30.
Estimates (With 95% Confidence Intervals) From Ordinal Regression Predicting Health Status From Youth Body Mass Index (BMI) and Significant Predictors in 2074 Females Aged 18–54, National Health and Nutrition Examination Survey I.

Prediction 2: Pregnant Women With Lower Prepregnancy BMIs Will Have a Lower Risk of Pregnancy-Induced Hypertension and Operative Delivery
In American primiparas giving birth to singleton infants in 2013, risks of delivery by caesarian section and of gestational hypertension were much lower in those with prepregnancy BMIs less than 20 compared with BMIs of 25–29 and significantly lower than for BMIs of 20–24 (Table 6), controlling for age, race, gestational weight gain, cigarette smoking, and education. The risk of operative delivery was lowest for BMIs of 15–19 and was also positively related to age, with mothers under 20 having the lowest risk.
Odds Ratios for Caesarian Section, and Gestational Hypertension in Relation to a Prepregnancy Body Mass Index (BMI) of 25–29, Controlling for Age, Race, Weight Gain, Cigarette Smoking, and Education in Primiparas With Singleton Newborns, United States, 2013.

Note. n = 1,077,439. OR = odds ratio; CI = confidence interval.
Discussion
Contrary to the health-and-fertility hypothesis, studies cited in the Introduction show that female mortality in subsistence populations is inversely related to BMI. Numerous studies also show a similar pattern in well-nourished populations with higher mortality for women with low BMIs, but almost all deaths are due to chronic disease in women over 50. Health during the reproductive years is arguably more relevant to assessing the health consequences of the low BMIs and waist sizes judged most attractive. However, the results of new studies in this reproductive age group were similar: Claims of better health in women with low BMIs or smaller waist sizes were not supported, and some indicators showed the reverse relationship, with the body shapes judged most attractive having worse health outcomes. The only exception was for two specific pregnancy-related outcomes.
In young nulligravidas and women of reproductive age, those with BMIs below 20 or WHRs or WSRs in the first quartile did not have better health than those with BMIs of 20–29 or waist measures in the second and third quartiles, controlling for other factors related to health. In addition, there was no relationship between the minimum BMI in early adulthood, when many mateships would be formed, and self-rated health status in women in the reproductive age group. As in some mortality studies showing worse outcomes for very high BMIs, obese women reported poorer health.
In a larger sample of reproductive-age women with self-reported BMIs less than 25, women with lower BMIs had more bed disability days and hospital days and more major disability. They were also more likely to have had a recent significant infection and to have more hospital days and restricted activity related to an infection.
Thus, the results of tests of the relationship between WHR, BMI, and health in nonobese women of reproductive age show either no relationship to health or, with larger samples, diminished health in women with BMIs below 20—the range associated with maximum attractiveness.
There was evidence of a potential health benefit of lower BMIs in women pregnant for the first time that has not been previously discussed in relation to the attractiveness literature. As has been found in other well-nourished populations (Barau et al., 2006; Bhattacharya et al., 2007; Bodnar, Ness, Harger, et al., 2005; Bodnar, Ness, Markovic, et al., 2005; Han et al., 2011; Nuthalapaty et al., 2004; O’Brien et al., 2003), American primiparas with prepregnancy BMIs less than 20 were at lower risk of developing gestational hypertension and of needing an operative delivery (typically due to cephalopelvic disproportion).
Complications of labor cause most maternal deaths in countries where maternal mortality is high (Frost, 1984; Nkata, 1997; Rosenfield, 1989; Vork, Kyanamina, & van Roosmalen, 1997) and often result in stillbirths (Hossain, Kahn, & Kahn, 2009; Olusanya & Solanke, 2009) or infant deaths due to birth injuries (Kandasamy et al., 2009; van Roosmalen, 1988). Maternal mortality due to obstructed labor is likely to have been a major cause of death in women in the evolutionary past, although data are limited and there is some debate about its frequency (C. Wells, 1975; J. C. K. Wells, DeSilva, & Stock, 2012; Wittman & Wall, 2007). In the 19th-century Sweden, an estimated 7% of women died in childbirth (Hogberg & Brostrom, 1985), but in a preColumbian sample from Chile, the estimate is 14% (Arriaza, Allison, & Gerszten, 1988). In the study of female mortality in contemporary Hiwi hunter-gatherers, 21% of deaths in females aged 10–39 were related to childbirth, and this accounted for 4% of all female deaths and 13% of nontraumatic deaths (Hill et al., 2007).
When a first-time mother dies during pregnancy, labor, or postpartum, any child born alive is also very likely to die (Hogberg & Brostrom, 1985), in which case none of her genes will survive. Thus, there should be a strong selection pressure to enhance any characteristic which reduces this risk, in this case, a lower BMI. At the same time, the increased risk of death from infection in women with lower BMIs would be a countervailing selection pressure. Thus, the optimum BMI may be higher than the value associated with the lowest risk of obstructed labor.
Other than these two possible benefits to first-time mothers, the very small waist sizes and low BMIs associated with female attractiveness in well-nourished populations are not associated with obvious health advantages and, conversely, may increase the risk of disability and infection. In subsistence populations, these low levels of body fat are clearly detrimental.
Discussions of the relationship between attractiveness and health have been based on measures of actual health, since selection generally favors individuals attending to reliable indicators of fitness-relevant parameters. Nevertheless, it has been suggested that perceived rather than actual health might be driving male judgments of female attractiveness (Boothroyd, Tovée, & Pollet, 2012). Three studies which included ratings of perceived health and attractiveness (Furnham, Petrides, & Constantinides, 2005; Furnham, Swami, & Shah, 2006; Singh, 1993b) are difficult to interpret because the figures representing “underweight” “normal weight” and “overweight” are based on BMIs of 15, 20, and 25, because manipulations of WHR change the BMI, and because there was no systematic analysis of the relationship between health and attractiveness ratings. However, tabular data in two of the studies (Furnham et al., 2005; Singh, 1993) suggest that the attractiveness ratings of “underweight” figures were higher than their perceived health ratings, while the opposite was true for heavier figures.
A cultural-evolution explanation for male mating preferences should not only demonstrate a correlation between body shape and health but should also explain and test a model of how a false (maladaptive) belief about the relationship between health and body shape might arise and be retained in the population. Our goal is different and simpler: to show that, whatever the general populace might believe, scientists should reject the hypothesis that low BMI and low WHR are generally healthy phenotypes that are preferred for that reason.
Although low BMIs did not indicate better health, better health outcomes were consistently associated with younger age. This suggests that males seeking mates with optimal current health should prefer younger women, which is consistent with the hypothesis that males are attracted to younger women with maximal reproductive potential (Andrews et al., 2017; Symons, 1979).
As a way to better understand the relevant selection pressures, future tests of the health-and-fertility hypothesis should attempt to identify the specific benefits accruing to women with the most attractive body shapes.
Limitations
The new analyses of the effects of waist size, WHR, and BMI on health and fertility, based on recent representative samples of American women, may not reflect conditions in the environment of evolutionary adaptiveness. However, our new analyses do derive from the same kind of Westernized population that has provided most of the evidence previously advanced to support the health-and-fertility hypothesis, and they are relevant precisely for that reason. It should also be noted that there is a general concordance between the results in these samples and data from subsistence populations.
Except for analyses related to minimum BMI after age 18, the data from the NHANES samples are cross-sectional, but they investigate current relationships between variables in the same individuals. Data relating youth BMI to subsequent health and fertility are longitudinal.
The BMIs in the NHIS sample, the lowest BMIs after age 18 in NHANES I, and prepregnancy BMIs in the U.S. natality sample are all based on self-reported weights and, as noted, there is a well-known bias for heavier individuals to report lower than actual current weights; we were able to quantify this bias in our analysis of data from NHANES III. In so far as obesity is associated with a decrement in health, this would make midrange self-reported BMIs (which include some obese individuals) appear less healthy when compared to the lowest BMIs. Despite this bias, we still find that reported midrange BMIs are optimal in terms of health and, by some measures, superior to health outcomes for individuals with BMIs less than 20.
The self-reported lowest weight after age 18 occurred at different ages, although most were within 5 years of age 18; to deal with this issue, the age of minimal weight was used as a covariate in the analyses (and was never significant). Also, youth BMIs were computed using current heights, so that a loss of height could make youth BMIs appear higher, although this source of error should be minimal in the reproductive age group. However, there does not seem to be any reason why such factors should systematically skew the analysis of current health or the number of live births in ways that would systematically disadvantage the health-and-fertility hypothesis.
Conclusions
Our analysis of the relationship between BMI (and WHR or waist size) and health in women of reproductive age is consistent with others—and notably with those in subsistence populations—suggesting that the relationship between health and female body shape has not undergone significant recent change. When these anthropometric variables are predictive, women with smaller waists and lower BMIs have poorer health. This large body of results thus stands in strong contradiction to one pillar of the prevailing health-and-fertility hypothesis regarding men’s preferences for women’s body shapes. Our study did uncover one possible health advantage of a low BMIs in a well-nourished American population: Primigravid women with lower BMIs may be at lower risk of two pregnancy complications, operative deliveries and gestational hypertension, although risks of other complications could be higher.
Claims of a negative association between body fat (BMI, WHR, or waist size) and health could only find a basis in the often worse outcomes of women in the heaviest (e.g., obese) part of the distribution. But such BMIs are rare in contemporary forager and subsistence populations and were likely rare when men’s mating preferences were shaped and, in any case, provide no explanation for preferences at the opposite extreme of the distribution when, as demonstrated here—and as expected from stabilizing selection—optimal health is achieved near the population mean.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
