Abstract
Offspring within families, both human and nonhuman, often differ. The obvious question is: Why? Work on psychological differences on children within human families has focused primarily on differences in the nonshared environment of contemporary siblings, though the precise location of this nonshared environment is still the subject of much debate. Here I explore the range of explanations for within-brood diversity from the perspective of nonhuman families, particularly birds that share certain key features with human families. I examine the role of social rank in creating a nonshared environment within the family, and present data from a model system (an altricial bird) to illustrate how different the effective environments experienced by offspring sitting side-by-side in the same confined physical space, tended by the same parents, and experiencing similar ecological variability, can be. These broodmates can effectively live in different worlds. I then briefly explore other sources of diversity among offspring in nonhuman families, including within brood genetic differences and non-genetic maternal (parental) effects that often covary with birth / hatching rank. Given the ubiquity and far-reaching consequences of maternal effects in nonhuman families, and some human data suggestive of similar patterns, it would seem worthwhile to explore the potential role of maternal effects in creating phenotypic diversity in psychological traits among children in human families.
Keywords
Introduction
Why do offspring within the same family so often differ? This fundamental question crosses discipline lines and has vexed social and natural scientists alike. Workers in different fields, drawing from different traditions, using different research methods, not surprisingly provide different answers to the same question. A sociologist might explain differences among children from societal, cultural and political contexts (Corsaro, 2005; Freese, Powell, and Steelman, 1999). A psychologist might explain differences among children with reference to the features of the nonshared environment among contemporary siblings, either inside or outside the family (Harris, 1995, 1998; Plomin, 2011; Plomin and Daniels, 1987; Sulloway, 1996). A behavioral ecologist might focus upon developmental plasticity in heterogeneous environments (Pfennig, 1992; Stearns, 1989) or one of the myriad maternal effects that have far reaching effects on offspring phenotypes (Groothuis, Muller, von Engelhardt, Carere, and Eising, 2005; Mousseau and Fox, 1998). Even a casual reading of these diverse literatures makes it obvious that they have much to offer each other, both in terms of explanatory framework and methodologies used. For example, the methods used by psychologists interested in the question of jealousy in human infants may be directly transferable to animal models (Hart and Legerstee, 2010). Here I have the modest goal of using studies of animal models to help potentially explain the origin of differences among children within human families.
It has been clear ever since the seminal paper of Plomin and Daniels (1987) that a chief source of such differences lurks in the nonshared environments of contemporary offspring within the same family. But exactly what and where are these nonshared environments? Are they the peer relations outside of the family as Harris (1995, 1997) suggests? Or do they rest within different niches created within the family by birth order as Sulloway and his colleagues argue (Sulloway, 1996, 2007; Zajonc and Sulloway, 2007)? Or is it the “gloomy prospect” that Plomin and Daniels originally raised, that differences among children arise from “random, idiosyncratic, or serendipitous events such as accidents, illnesses and other traumas” (Plomin and Daniels 1987, p. 8)? The exact location and nature of the nonshared environment remains uncertain, but its existence seems to be required to explain differences among children within a family: It is our environmental “dark matter.”
From the perspective of a researcher working on nonhuman families, a further question arises: Is the nonshared environment the only source of variation among offspring within the same family? Recent work on nonhuman families, and in particular birds (see below), suggests that the answer is “no.” Both heritable variation, and nonheritable maternal effects can generate substantial differences among progeny within the same family in both human (e.g., Bouchard and Loehlin, 2001) and non-human families (see below). For example, mother birds may make systematic adjustments to egg size or the quantity of hormones deposited in eggs in relation to laying sequence (Groothuis et al., 2005; Slagsvold, Sandvik, Rofstad, Lorentsen, and Husby, 1984), creating intrinsic differences among progeny at birth or hatching. I shall discuss below why parents might do this, and what the implications are for the study of human families.
I am going to make the bold and perhaps precarious assumption that certain animals provide useful models for the study of human families. Those animals are not other mammals. Humans are unlike most mammals that usually raise offspring more or less simultaneously in multi-offspring litters. Instead, humans give birth to usually single offspring at intervals, and because of the protracted period of parental care, often have families of different-aged dependent offspring. This pattern finds no strong parallel even among our closest primate relatives. Rather, a closer match is found among altricial birds that practice hatching asynchrony (Magrath, 1990). As in human families who also raise altricial offspring, parent birds are often confronted with mixed-age broods of dependent offspring at different developmental stages and with differing resource needs. Age, size and developmental asymmetries lead to unequal competitive abilities in accessing parentally delivered resources. The age differences involved may appear small at first glance − 1, 2 or 3 days in the case of the red-winged blackbirds that I shall discuss below – but the nestling period lasts only a dozen days. These age differences generate an asymmetric sibling rivalry (sensu Forbes and Glassey, 2001), and the asymmetry is very large, perhaps the rough equivalent of a 2-to-6-year inter-birth interval between human children.
I do note that the comparison between altricial birds and altricial humans is not perfect. Multi-offspring human families are built over a number of years and a common pattern is for older parents to be wealthier (see, e.g., Steelman and Powell, 1989). Another key difference is the origin of hatch vs. inter-birth intervals. Birds can potentially control hatching asynchrony simply and precisely by varying the onset of incubation, and maternal effects are easy to envision. Whether a similar modulation of competitive asymmetries among progeny occurs in humans via birth spacing is less clear. If inter-birth interval is adaptively modulated in humans, it seems likely to be controlled with less precision than in birds. While there are important similarities between birds and humans in their manner of parenting, their differences should not be understated.
Developmental psychologists, behavioral geneticists, and sociologists are all interested in the origins of differences among children within families in personality, behaviour, intelligence, growth trajectory or health status, and differentiate between the shared and nonshared environment of siblings (Frampton, Jenkins, and Dunn, 2010; Harris, 1995; Kristensen and Bjerkedal, 2007; Plomin and Daniels, 1987; Steelman, Powell, Werum, and Carter, 2002; Sulloway, 1996). The nonshared environment is those phenomena unique to each child, such as birth rank or peer relations, and not general to the entire family (Plomin and Daniels, 1987). The shared environment includes those environmental phenomena shared among all members of the same family such as resource availability, family size and the number of parents (Harris, 1998; Plomin and Daniels, 1987) and is expected to generate similarities among siblings, as would shared hereditary effects (Plomin and Daniels, 1987). Recent work, however, shows that shared environments can exert differential effects on individual offspring conditional on the properties of the offspring. For example, human children with a negative outlook on life may be more vulnerable to the adverse effects of maternal depression (Frampton et al., 2010; Jenkins, Simpson, Dunn, Rasbash, and O'Connor, 2005). Here I borrow this framework to examine differences in the performance of offspring in a non-human animal model – an altricial songbird – to study the environmental origins of within-family differences among siblings. I use this approach to address two questions. First, how different are the effective environments of contemporary offspring in the same family? Second, how does an environmental stressor – year to year ecological variability – affect offspring performance in relation to the properties of the shared and unshared environment?
For both questions I use a simple metric: whether an offspring lives or dies during the nestling period in relation to the features of the within-family environment that affect sib-sib competition, the size and structure of the family in which a nestling lives (shared environments), and the social rank of the offspring (a mixture of shared and nonshared environments). At the outset I will emphasize one important point. I have noted above in my introduction the different potential locations of the unshared environment. In the system that I am about to describe – an altricial bird – all offspring are confined to the same physical space – the nest – for the duration of the nestling period. This simplifies matters greatly. Any differences that arise among individual offspring must arise from within the family.
Materials and Methods
I examined within-family variation in offspring performance in the red-winged blackbird (Agelaius phoeniceus) – that serves as a model system for the study of sibling competition and parental care (Forbes and Glassey, 2001). Like humans, these birds exhibit developmental asynchrony created by asynchronous hatching of nestlings; this results in the age differences among progeny (up to 3 days, or 25% of the nestling period) that underpin an asymmetric sibling competition between older, larger and stronger core siblings, and weaker “marginal” counterparts (Forbes and Glassey, 2001). Core progeny as a result enjoy growth and survival advantages over their marginal nestmates (Forbes, 2009, 2010a, 2011; Forbes, Thornton, Glassey, Forbes, and Buckley, 1997; Mock and Forbes, 1995; Mock and Parker, 1997).
I studied red-winged blackbirds nesting in near Winnipeg, Manitoba over 18 field seasons from May to August 1993 to 2010. Daily censuses were conducted at an average of 278 nests each year with the assistance of a field crew. Detailed methods have been described elsewhere (Forbes, 2009, 2010a; Forbes and Wiebe, 2010) and I present only a précis here. Nests were visited from the onset of egg-laying until the youngest nestlings reached at least 8-d of age (hatching = day 1). For ethical reasons, nestlings were generally not handled after day 10 to prevent premature fledging. Eggs and nestlings were marked for individual identification, and nest contents were monitored.
I divide the brood into core and marginal elements (Mock and Forbes, 1995) based upon the hatching pattern of individual offspring (Forbes et al., 1997). All nestmates hatching together on the first day of the nestling period are the core brood; nestlings hatching 1 or more days later are the marginal brood (see Forbes, 2010a for detailed methods of classifying core vs. marginal progeny). I further subdivide the marginal brood into three levels: m1, m2 and m3 (Forbes, 2011). Because of the incubation pattern of red-winged blackbirds, these subscript designations correspond both to the number of days that a marginal offspring hatches after its core counterparts, and its rank within the marginal brood. If, for example, three marginal offspring are present alongside one or more core offspring, the m1, m2, and m3 progeny will usually have hatched 1, 2 and 3 days after the core offspring.
I denote family size and structure as cj, where c is the number of core hatchlings (1 ≤ c ≤ 5) and j is the number of marginal offspring at hatching (≤0 j ≤ 4) (Forbes, 2009, 2011). Rank within the brood is denoted as either c (= core) or mi, where m denotes marginal status and the subscript i denotes rank within the marginal brood based on age); m1 progeny are found in all broods with ≥1 marginal offspring; m3 progeny are only found in broods with ≥3 marginal progeny. Table 1 shows the mean survival to day 8 of core, m1, m2 and m3 progeny in the 13 most common brood structures; five additional brood structures were vanishingly rare. Though survival to leave the nest is an obviously incomplete measure of fitness, it is a useful proxy as survival among specific classes of offspring showed a near maximal range of variation (see Table 1 and Figure 1) and fledging success is a robust predictor of recruitment to the breeding population in redwings (Weatherhead and Dufour, 2000). I note that I do not expect that all parents surveyed here to be of equal quality: Parents laying larger clutches quite probably have access to more resources (e.g., Högstedt, 1980). Given that the lowest survival of progeny is found in the largest broods, this represents a conservative assumption for my analysis.
Estimate of the effective core brood size ĉ in relation to social rank, family structure, and year type in red-winged blackbirds studied near Winnipeg, Manitoba from 1993 to 2010
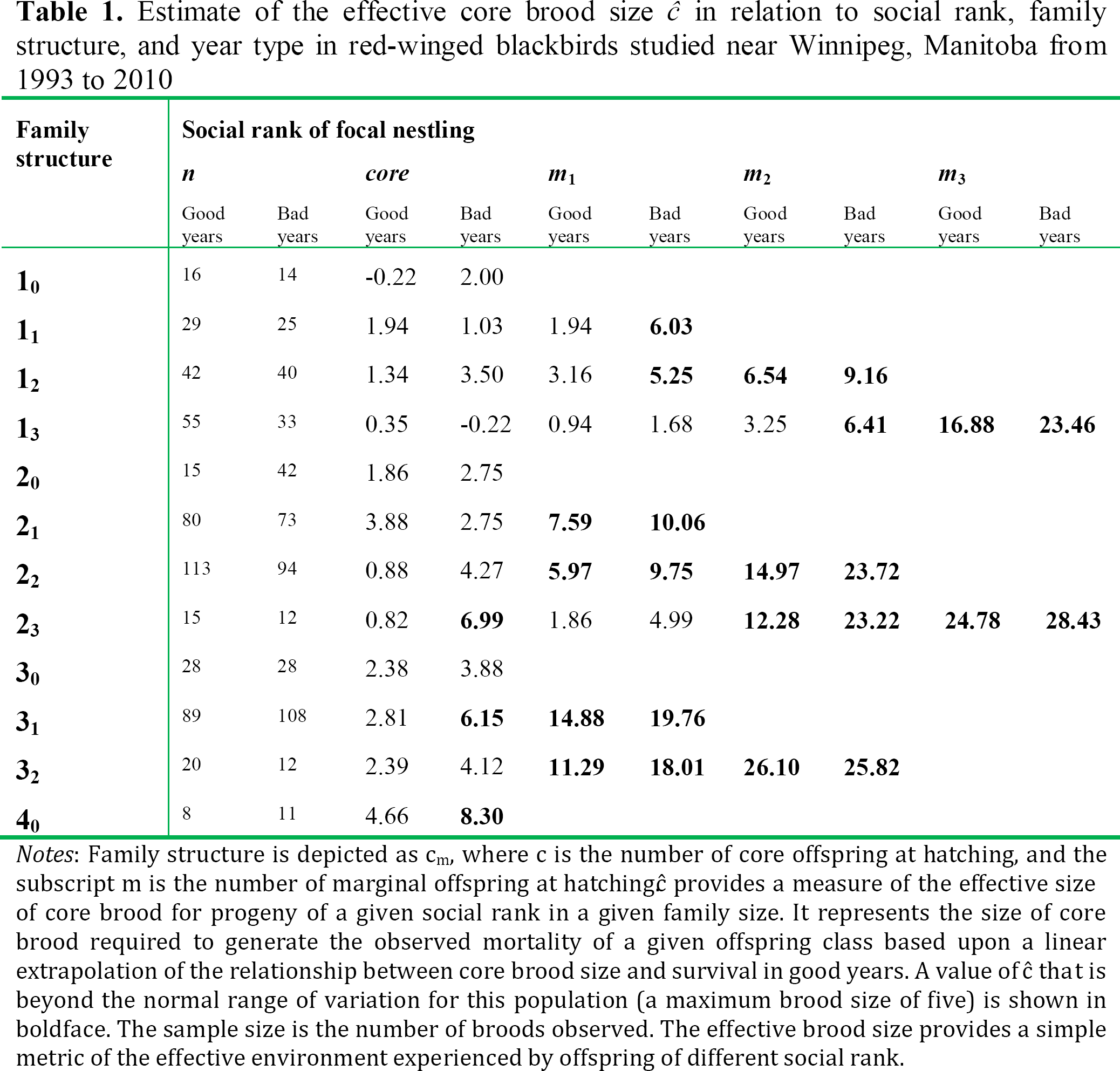
Notes: Family structure is depicted as cm, where c is the number of core offspring at hatching, and the subscript m is the number of marginal offspring at hatchingĉ provides a measure of the effective size of core brood for progeny of a given social rank in a given family size. It represents the size of core brood required to generate the observed mortality of a given offspring class based upon a linear extrapolation of the relationship between core brood size and survival in good years. A value of ĉ that is beyond the normal range of variation for this population (a maximum brood size of five) is shown in boldface. The sample size is the number of broods observed. The effective brood size provides a simple metric of the effective environment experienced by offspring of different social rank.

Survival of nestling red-winged blackbirds in good (open bars) and bad years (shaded bars) in relation to social rank and family structure
Results
I measured the survival of nestlings to fledging in relation to social rank and family structure in years of above and below average reproductive success (see Forbes, 2009, 2010a for details about how year type was categorized): I shall call these “good” and “bad” years as a shorthand description, and the data are shown in Figure 1. To estimate a “bad” year's effect, I first analyzed the relationship between the size of the core brood (c) and the survival of core nestlings (s), in years, of above reproductive success using simple linear regression:

I used the regression coefficients to interpolate the observed mortality of marginal offspring of a given social rank (m1, m2, or m3) in a given family structure into an estimate of the effective core brood size, ceff − i.e., what size of core brood would have yielded the equivalent mortality rate observed in a focal marginal offspring in a good year (see Table 1).
Inspection of the data showed, as expected, a reasonably good fit between the observed and estimated core brood sizes (r = 0.802, p = 0.002). The question of direct interest, however, is what would the equivalent core brood size (ĉ) have to be to generate the observed mortality (see Figure 1) for marginal offspring in those brood structures? To estimate this, I simply rearranged the regression equation above:
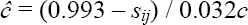
Where sij is the survival of a nestling of social rank i in brood structure j, and ĉ is the effective size of the core brood at hatching in a good year.
I acknowledge that extrapolation outside the observed range of variation is dangerous practice and violates the norms of statistical etiquette. That caveat notwithstanding, the analysis provides a rough measure of the effective environments experienced by offspring of different social rank. And sometimes the effective environments were very different.
How vulnerable are offspring to bad years?
I examined the relationship between social rank, family size, and vulnerability to below average ecological conditions (bad years) by computing the difference in mean survival for offspring between years of above and below average reproductive success (data shown in Figure 1). The difference (d) shows the extent to which offspring were affected by adverse conditions (−1 ≤ d ≤ 1) – e.g., a value of zero indicates that a focal offspring was not affected by bad years; a positive value of d indicates that offspring exhibited lower survival in bad years than in good years, a negative value of d the opposite.
Multiple regression analysis indicated separate and significant effects of both social rank (core = 0, m1 = 1, m2 = 2, m3 = 3) on the difference in survival across years (core brood size at hatching: β= 0.040, p = 0.022; social rank: β= 0.065, p = 0.001; overall regression: R2adj = 0.328, F2,24 = 7.337, p = 0.003); the size of the marginal brood was not strongly related to the magnitude of d (marginal brood size at hatching: β= 0.009, p = 0.616) and this variable was deleted from the best-fit model. In short, low ranking marginal offspring in families with more core offspring were more strongly affected by bad years than either core offspring or high-ranking marginals (see Figure 1).
Discussion
Offspring within both human and nonhuman families differ, often dramatically. The key question is: Why? As Plomin and Daniels (1987) noted more than a quarter century ago, hereditary effects and the shared environment are expected to produce similarities among progeny within the same family. Differences among progeny, conversely, are expected to stem primarily from the nonshared environment of individual offspring. But where does the nonshared environment lie? Here I report the results of work on a model system for nonhuman families – an altricial bird – that examines the role of social rank in creating phenotypic differences among progeny. Differences in social rank were created by hatching asynchrony, a maternal effect that resulted in broods of mixed-age offspring. And although offspring within the same family shared the same confined physical space, were tended by the same parents, and experienced similar extrinsic ecological variation, they exhibited dramatic variations in overall survival and their sensitivity to year-to-year ecological variation. Both survival and the degree of sensitivity were conditional upon offspring social rank (nonshared environment), and its interaction with the size and structure of the family created by their mothers (shared environment). Here I use a simple metric, the effective brood size, to compare the effective environments experienced by offspring within the same family. And the observed differences were striking.
Although there was overlap in the effective core brood size for core and marginal offspring, particularly for high-ranking (m1) marginals and especially in small families, there were also broad regions of non-overlap. The effective brood size ranged as high as 11 in bad years and 26 in good years for lower ranking (m2 and m3) marginal offspring. As the normal maximum brood size at hatching in this population is five, with broods of six and seven occurring but being vanishingly rare (Forbes, 2010a), this places these marginal progeny far outside the normal range of brood size for this population. Overall, 16 of the 28 estimates of marginal offspring survival (bold-faced entries in Table 1) exceeded the normal brood size.
Offspring social rank also explained in large part the differential effect of a shared environmental factor for all siblings: year-to-year ecological variability. How resource shortfalls are experienced by individual offspring was conditional upon both family size and where that individual sat in the brood hierarchy. Core offspring in different brood structures were affected relatively little by year-to-year ecological variability, as too were high ranking (m1) marginal offspring in small broods. However, m1 progeny in large broods and m2 progeny were strongly affected by year-to-year variability, showing marked declines in survival in bad years. Intriguingly, low-ranking marginals in the largest broods (m2 in 32 broods and m3 in 23 broods; see Figure 1) were not strongly affected by year-to-year ecological variation: Their survival prospects were already so grim even in good years that there was little room for further decline.
This set of results is important for four reasons: 1) they show how different the effective environment for contemporary siblings can be; 2) they show in a nonhuman animal model how the response to a shared environment can differ according to intrinsic differences among offspring; 3) they show how the unshared environment that exists for contemporary siblings can exist within the family unit; and 4) they have implications for how maternal effects are allocated within families.
How different are the effective environments of contemporary siblings?
The results above show that differences in the effective environment experienced by progeny within a brood can be striking. A high ranking member of the brood hierarchy, for example, can be living in an effective brood of less than three, while a junior brood member can be living in an effective brood of more than 26 (see Table 1). A brood size of 26, in fact, does not exist in nature: It is a “meta” brood size, which shows that offspring of lower social rank are living in quite different worlds than their counterparts sitting in close physical contact beside them.
These different family niches should favor different offspring traits. For example, Mainwaring, Dickens, and Hartley (2010) show that social rank, and not maternal effects, are the chief determinants of phenotypic variation in nestling blue tits (Cyanistes caeruleus) in a suite of traits linked to sibling competition; nestlings prioritized growth of tarsi over feather development to maintain standing within the nest. More recently, Mainwaring and Hartley (2012) have found that later-hatched nestling zebra finches (Taeniopygia guttata) exhibited greater exploratory behaviour as adults. Variation in early nutritional conditions (later hatched nestlings grow slower) appears to influence behavioral development and personality. These appear to be examples of phenotypic plasticity during development (Stearns, 1989) that serve as adaptations to different social environments. They are a polyphenism that emerges from the interaction of a hereditary program with different environmental cues.
It would not be surprising if such phenotypic plasticity in response to offspring social environment was widespread in altricial species, including humans. Sulloway (1996) argues cogently that behavioral adaptations to different niches within human families yield differences in personality in relation to birth rank. Though this work is still much discussed, the results of work on non-human models and altricial birds in particular provide strong support for this argument.
The phenotypic response to a shared environment is conditional upon social rank
How nestling blackbirds responded to features of the shared environment was conditional upon the properties of individual offspring. This follows from an asymmetric sibling rivalry that parents initially established via hatching asynchrony, conferring advantages to progeny of high social rank and disadvantages to others. As such, offspring of low social rank were more vulnerable to resource shortfalls.
This result parallels work on human children, where siblings exposed to maternal depression (an adverse and shared environmental effect) differed according to the perspective of the children in the mother-child relationship, a nonshared environmental factor (Frampton et al., 2010; Jenkins et al., 2005). Similarly in blackbirds, the offspring response to the challenge of adverse ecological conditions was conditional upon social rank and the interaction with family size. In the objective environment of these altricial birds, ecological stress is a shared family-level risk factor, but in bad years, some siblings are profoundly affected, others little at all. A shared family-level risk had a nonshared effect conditional on offspring social rank.
An obvious question is whether parallels exist within human families. Do children of lower social rank experience resource shortfalls in a similar manner to more senior progeny? Statistically this might emerge, for example, as an interaction between socioeconomic status and birth rank. Work on altricial birds suggests strongly that resource shortfalls are not shared equally among the sibship, but disproportionately fall upon offspring of lower social rank (Forbes, 2011; Forbes and Glassey, 2001; Forbes and Mock, 1996; Forbes et al., 1997; Lack, 1947; Mock and Forbes, 1995). Asymmetric sibling rivalry effectively buffers offspring of high social rank (the core brood) from resource shortfalls, while leaving offspring of lower social rank vulnerable. A statistical signature of asymmetric sibling rivalry is increased variation in performance (in birds, offspring growth and survival) among lower ranking brood members (Forbes, 2009; Forbes and Glassey, 2000). This seems worth exploring in human families.
The nonshared environment exists within the family
A key insight derived from the study of altricial birds concerns the shared and nonshared environments experienced by contemporary offspring. Even though different individuals share the same confined physical space at the same time, are tended by the same parents, and are influenced by the same extrinsic ecological conditions, they can effectively live in different worlds (Forbes, 2011). This has potentially deep implications for the study of human families and the influence of birth order and social rank. We assume that offspring share a common family environment and that this should induce similarities among contemporary offspring. But is this assumption valid? Work on altricial birds suggests not necessarily. From below, competitive asymmetries among contemporary offspring mean that offspring often do not enjoy equal access to resources with concomitant effects on growth and survival. From above, parents may play favorites and treat offspring unequally (Lessels, 2002; Lyon, Eadie, and Hamilton, 1994). Both may contribute to offspring in the same place at the same time experiencing the world very differently.
Differences in social rank and parental favoritism may generate wide differences in the effective environment experienced by contemporary offspring. This is not a straightforward question to address. The effective environment cannot be measured directly, but rather is inferred from offspring performance. Instead, we must focus on properties of the objective environment – family size, birth rank, family income, number of parents – that can be measured directly to establish the sources of variation that exist among progeny within the same family.
It is clear that the strongest determinant of differences in offspring fitness in this avian system comes not from extrinsic environmental variation (year-to-year variation in brood-rearing conditions), but from the within-family inequality among progeny. That is, the unshared environment largely exists within the family unit living in the same confined physical space. There are no differences in peer relations outside the family (nestlings do not leave the nest), only differences in competitive ability established at birth and that unfold during the period of parental care.
Altricial birds are obviously very different from humans in many key respects, but there are sufficient parallels to at least entertain the argument that the missing unshared environment that generates differences among human children within the same family might indeed lurk within the family.
Maternal effects are both a cause and consequence of asymmetric sibling rivalry
The competitive asymmetries among nestling blackbirds were established by the maternal effect of hatching asynchrony (Glassey and Forbes, 2002). Much recent work has focused on the role of maternal effects in relation to nestling social rank in birds, and the potential to compensate for the inimical effects of hatching asynchrony (reviewed in Saino et al., 2011). This work has focused primarily on how parents might manage sibling competitions for parentally provided resources – e.g., extra maternal testosterone may enhance begging performance of last-hatched nestlings (Groothuis et al., 2005; Schwabl, Mock, and Gieg, 1997; Sockmann, Sharp, and Schwabl, 2006).
But maternal effects may also play a compensatory role. Instead of managing sibling competitions directly, maternal effects such as boosts in maternally provided hormone levels, antioxidants, immune system complements, or egg size (Groothuis et al., 2005; Müller, Boonen, Groothuis, and Eens, 2010; Royle, Surai, and Hartley, 2001; Schwabl, 1996; Slagsvold et al., 1984; Sockmann et al., 2006) may instead reflect the different worlds in which these offspring live. Life in an effective brood of 11 to 26 is very different than in a brood of two or three. Thus, these maternal effects may not serve to manage sibling competition per se, but reflect the different challenges faced by offspring of different social rank.
Slagsvold et al. (1984) long ago suggested that parent birds might vary egg size in relation to hatching asynchrony to modulate the effects of sibling competition. A larger last laid egg would provide additional resources for an offspring initially handicapped in sibling competition, promoting its survival; they called this the brood survival hypothesis. Conversely, a small final egg would exaggerate the competition and accelerate the process of brood reduction. Forbes and Wiebe (2010) provided empirical support for a brood survival function, finding that early mortality of marginal, but not core, offspring was affected by egg size.
More generally, maternal effects that establish competitive asymmetries (cardinal maternal effects) may trigger secondary maternal effects. Much recent work on altricial birds has focused on the role of maternal androgens deposited in the yolk of eggs that differ with laying and hatching order, and alter nestling phenotype. Maternal testosterone, for example, has a wide array of effects, including inducing greater begging intensity, which may in turn promote the survival of these offspring (Eising and Groothuis, 2003; Groothuis et al., 2005; Lipar and Ketterson, 2000; Schwabl, 1996). But testosterone may also bear a cost of depressed immune system function that may induce further maternal effects to compensate for the inimical pleiotropic effects of extra androgens. Royle et al. (2001) suggested that mothers may deposit extra anti-oxidants in eggs with higher titres of maternal testosterone to offset the immunological costs.
The work on nonhuman families shows quite clearly that some offspring within the same family start their postnatal existence very different than brood or littermates. I shall now turn to the role of such intrinsic differences in generating within-brood diversity among families.
Sources of intrinsic differences among progeny at birth or hatching
Work on differences among offspring within human families has focused chiefly upon differences in the nonshared environment of offspring. Less attention has been given to considering the possible role of intrinsic differences that exist within the same family among progeny – be they genetic or otherwise – in generating phenotypic differences among human children. The sources of intrinsic differences among progeny can be divided into genetic effects, those with an environmental origin, and combinations of these. Plomin and Daniels (1987) noted that heritable effects are expected to generate similarities among progeny within the same family, not differences. But work on nonhuman families reveals a growing number of mechanisms that stand as exceptions to this general rule.
The sources of intrinsic differences that stem from environmental causes are manifold. They include non-genetic maternal effects that are ubiquitous in nonhuman families and often covary with birth or hatching order. With respect to psychological differences among children in human families, these seem to have been little discussed except insofar as they stand counterexamples to estimates of heritability for psychological traits – i.e., differences in the prenatal environment experienced by monozygotic and dizygotic twins and/or singletons in classical twin studies of heritability. Here the line of argument is that these maternal effects result in estimates of heritability that are biased high (Conley, 2011; Patterson, 2007). But work on nonhuman families, particularly birds, shows that maternal effects may be used as tools to modify offspring phenotype to match the postnatal environment.
Benefits of offspring diversity within the family
There is growing interest in the role of phenotypic and genotypic diversity among siblings and the fitness consequences for both parents and offspring. The potential benefits of diversity are manifold. Developmental asymmetries among progeny, for example, may reduce peak demands for critical resources such as food, space, or oxygen. The ornithologist David Hussell long ago suggested that hatching asynchrony in altricial birds staggered the maximum food demands of nestlings, reducing peak demands on parents (Hussell, 1972; see also Mock and Schwagmeyer, 1990). McLeod and Marshall (2009) similarly suggest that genetic differences among progeny resulting in developmental asynchrony may spread out the peak oxygen demand in genetically diverse clutches of marine polychaetes. Age-differences among progeny do not necessarily create phenotypic diversity, but they do create the asymmetric sibling competitions that can lead to phenotypic diversity.
Genetic diversity among contemporary progeny may yield fitness benefits by reducing competition among sibs, creating diversity among the offspring workforce (Ergonomic Hypothesis), and creating immunological diversity within the brood (McLeod and Marshall, 2009). Spreading out resource demands and reducing peak loads, as in the marine polychaetes noted above, is one mechanism for more efficient resource partitioning, akin to encouraging electricity users to shift usage patterns to off-peak hours. In effect, the resource base available for offspring is expanded.
There is growing empirical evidence for the Ergonomic Hypothesis of offspring diversity derived chiefly from studies of social insects. Offspring from different patrilines create workers in a colony with different propensities to engage in specific tasks, and more diverse colonies respond better to changing task needs (Mattila and Seeley, 2007; Oldroyd and Fewell, 2007). Polyandry is an obvious mechanism to create genetic diversity within a brood (Birkhead and Møller, 1992). Blended families of adopted and biological children might represent a human parallel. Differences in personality that arise from differences in family niche, as Sulloway suggests, might also generate greater offspring diversity.
A further potential benefit of offspring diversity arises from bet-hedging. Bet-hedging strategies involve sacrificing arithmetic mean fitness to reduce variance in fitness (Cohen, 1966; Donaldson-Matasci, Lachmann, and Bergstrom, 2008; Seger and Brockmann, 1987). By creating a diversified offspring portfolio, parents may reduce variance in fitness and increase geometric mean fitness (Crean and Marshall, 2009; Forbes, 2009, 2010b). Laaksonen (2004), for example, suggests that hatching asynchrony in birds – a maternal effect – may represent an offspring diversification strategy that yields bet-hedging benefits by inducing phenotypic variation among progeny. Miller (1997) made the provocative and parallel suggestion concerning nonshared environments in human behavioral genetics: Quite the opposite of Plomin and Daniels' gloomy prospect of random idiosyncrasies generating differences among progeny, Miller suggests that these may be manifestations of a strategy of evolved randomness. Such randomness would serve to create phenotypic diversity within a family for a variety of reasons that include, but are not restricted to, bet-hedging.
To date there has been little if any work on the benefits of diversity within human families. Hrdy and Judge (1993) examined the social and ecological conditions that would underpin a mandatory diversity among progeny arising from primogeniture. Consolidating the wealth transfer from one generation to the next in a single offspring, usually the eldest son, appears under some circumstances to maximize the continuation of the family line. Such a wealth transfer automatically results in different life paths for daughters and younger sons. Diversity here is adaptive compared to an egalitarian distribution of resources and arises as an epiphenomenon of the rules of wealth transfer.
Sulloway (1996, 2010) cites an example that would, however, fall under the ergonomic hypothesis. Ralph Nader and his three elder sibs divided the world into four parts with each specializing on their region's culture, history, and language. Sulloway notes that the four siblings understood intuitively that they were better off specializing in different areas of study and then pooling their collective resources (Sulloway, 1996). As with genetically diverse hymenoptera families, human families might benefit from the potentially symbiotic interactions that can arise from phenotypic diversity.
Another potential example of the ergonomic hypothesis applied to humans may be sexual orientation and birth order. Androphilic males are more common later in the birth order, especially when preceded by brothers (see below). If the presence of a larger number of older brothers in the family reduced the success of later-born males, then perhaps an androphilic male not in direct competition with his brothers might benefit the family by assisting collateral relatives (Ruse, 1982; Wilson, 1975). If this arose as a maternal effect, then it need not be adaptive for the affected individual – i.e., the indirect fitness benefit of raising nieces and nephews need not compensate entirely for the loss of direct fitness, but if it did not, would represent a selfish maternal effect (sensu Marshall and Uller, 2007). The fa'afafine of Samoa, an androphilic caste of males, may provide such an example. The fa'afafine show greater avuncular tendencies than gynophilic males (Vasey and VanderLaan, 2010; Vasey, Pocock, and VanderLaan, 2007) and the benefit accrues primarily to nieces and nephews. Though conjectural, one potential evolutionary interpretation is that a male with low expected direct success might yield lesser reproductive returns for the extended family than an additional helper, especially when viewed from the perspective of the mother.
Maternal effects in humans?
There is growing evidence that maternal effects play a role in the development of an array of traits in humans and that these are mediated by birth order, and in particular fraternal birth order. The presence of older brothers, but not sisters, in the sibship is, for example, associated with a lower birth weight of the subsequent sibs, particularly males (Blanchard and Ellis, 2001; Côté, Blanchard, and Lalumière, 2003; Magnus, Berg, and Bjérkedal, 1985), an effect that carries over to adulthood as lower height (Rickard, 2008); there is a parallel fraternal birth order effect for the incidence of male homosexuality (reviewed in Blanchard, 2004). There is strong evidence that the presence of male offspring in the womb and their cells in maternal blood primes the maternal immune system against minor (HY) antigens (Verdijk et al. 2004). This has been linked to recurrent spontaneous abortion following a first-born son (Nielsen, 2011), and reduced birth weight of later-born sons (Nielsen et al., 2008). Such birth order effects could serve to modulate the extent of sibling competition via maternal effects, though they could be manifestations of a cryptic sibling rivalry (Forbes, 2010b) that uses the mother as a conduit for “sibling effects.” The effect of birth order on traits such as sexual orientation, handedness and birth weight likely represent the tip of a phenotypic iceberg, and the current evidence suggests that they are mediated by the early prenatal environment, when synaptic connections are being established and brain circuits are highly plastic (Champagne and Curely, 2005).
Evidence of the use of maternal effects to tailor offspring phenotype to their postnatal environment is burgeoning in nonhuman systems. There is also growing evidence of the importance of the early prenatal environment on a suite of behavioral and health related traits in humans (de Weerth, Buitelaar, and Mulder, 2005; Vallée et al., 1997). Some of these effects, such as the increased prevalence of hypertension and coronary heart disease in the offspring of mothers pregnant during famine (Roseboom et al., 2003; Stein, Zybert, van der Pal-de Bruin, and Lumey, 2006), are clearly pathological. But it does not follow that all responses to maternally-mediated stress early in development are pathologies. Rather, some of these may be adaptive maternal effects that match offspring phenotype to current environmental conditions (Sachser, Hennessy, and Kaiser, 2011). It would seem worthwhile to extend this view more broadly to the development of psychological traits generally. Do mothers know best? They seem to know what is best for them, but that may not always be what is best for their offspring.
