Abstract
Objective:
This study was undertaken to determine whether use of the renin–angiotensin system (RAS) inhibitors would increase colorectal cancer morbidity and mortality.
Methods:
Databases were electronically searched to collect data of RAS use and colorectal cancer morbidity and mortality from inception to October 2018. Stata 12.0 software was used to perform a meta-analysis.
Results:
A total of 16 publications involving 2,847,597 participants were included. RAS inhibitor use was related to colorectal cancer risk (relative risk (RR): 0.86; 95% confidence interval (CI): 0.78–0.93) and mortality (RR: 0.80; 95% CI: 0.66–0.98) decrement. Subgroup analysis showed angiotensin-converting enzyme inhibitor (ACEI)/angiotensin II receptor blocker (ARB) (RR: 0.82; 95% CI: 0.69-0.96) or ARB (RR: 0.86; 95% CI: 0.73–0.98) or ACEI (RR: 0.81; 95% CI: 0.70–0.92) were related to colorectal cancer risk decrement. Furthermore, RAS inhibitor use was related to colorectal cancer risk decrement in Caucasians (RR: 0.88; 95% CI: 0.80–0.96) and Asians (RR: 0.72; 95% CI: 0.61–0.85). Additionally, dose–response showed that per one year duration of RAS inhibitor use incremental increase was related to 6% colorectal cancer risk decrement (RR: 0.94; 95% CI: 0.90–0.97).
Conclusion:
According to the evidence, RAS inhibitor use was associated with colorectal cancer risk and mortality decrement.
Introduction
Colorectal cancer is the third most common cancer worldwide, and fourth in cancer-related deaths. 1 At present, the effective treatment methods of colorectal cancer include surgery, radiotherapy, chemotherapy and targeted therapy. Comprehensive treatment based on surgery is the only method that can treat colorectal liver metastases (CRLMs). Currently, the five-year survival rate of CRLM patients after surgical resection is up to 50%, but only 20% of patients have the opportunity to undergo surgical resection. 2 Neoadjuvant chemotherapy can reduce the primary tumor or metastasis focus, reduce the tumor stage, change the unresectable tumor into a resectable tumor, increase the rate of radical resection, reduce the recurrence rate and control the microcarcinoma that exists before operation. But the timing of surgery after neoadjuvant chemotherapy did not reach a consensus. 3 Therefore, there are enormous challenges in treatment, and there is no satisfactory universal treatment for all.
Numerous renin–angiotensin system (RAS) agents have been shown to prevent risk of cancer. Angiotensin-converting enzyme inhibitors (ACEIs)/angiotensin II receptor blockers (ARBs) act as RAS inhibitors and play a fundamental role in the treatment of hypertension. 4 However, with the further exploration of RAS inhibitors, more and more studies have found that RAS inhibitors play a role not only in the treatment of hypertension, but also in colorectal cancer.5–7
Considering controversial evidence on the relation between RAS inhibitor use and colorectal cancer risk and mortality, we performed a meta-analysis to summarize the relative risk of RAS inhibitor use and colorectal cancer risk and mortality, in order to provide guidance for the clinic.
Methods
Search strategy
Medline, Embase, Web of Science and Cochrane Database were electronically searched to collect RAS use and colorectal cancer morbidity and mortality data from inception to October 2018, with keywords including ‘Angiotensin-converting enzyme inhibitor’ OR ‘Angiotensin I-converting enzyme inhibitor’ OR ‘ACE inhibitor’ OR ‘ACEI’ OR ‘Angiotensin receptor blockade’ OR ‘Angiotensin II receptor blocker’ OR ‘Angiotensin II antagonist’ OR ‘AT1 receptor antagonist’ OR ‘ARB’ OR ‘Renin–angiotensin system inhibitor’ OR ‘Renin–angiotensin–aldosterone system inhibitor’ OR ‘RAS inhibitor’ AND ‘Colorectal neoplasms’ OR ‘Colorectal tumors’ OR ‘Colorectal cancer’ OR ‘CRC’.
Inclusion
Then, the study was screened for retrieval based on the following criteria: (a) the study design of the selected literature must be an observational study; (b) the content of selected articles must be related to the effect of RAS use on the risk or mortality of colorectal cancer; (c) the relative risks with 95% confidence intervals. The selection of articles was carried out by two authors strictly according to the above inclusion criteria, and the discordant articles were discussed before deciding whether to include them.
Exclusion criteria
We excluded duplicate published studies, literature that was not available on outcomes or outcomes and non-English literature.
Data extraction and methodological quality evaluation
Two researchers independently screened the literature, extracted the data and cross-checked. If there was any disagreement, a third party was consulted to assist in the judgment. When reading the literature, the questions and abstracts were read first. After excluding the clearly unrelated documents, the full text was read to determine whether the item was appropriate for final inclusion. The data extraction content included: (a) basic information for inclusion in the study, including first author, publication time; (b) the basic characteristics of the subjects, including the number of samples in each group, the average age of the patient, and the disease; (c) key elements of bias risk assessment; (d) outcome indicators and outcome measurement data of interest. There was a lack of information to enable contacting the author for supplementary information. Quality assessment was performed according to the Newcastle–Ottawa Scale. 8
Statistical analysis
The count data uses the relative risk (RR) and the interval estimate uses 95% confidence interval (CI) as the effect size indicator with p<0.05 as a statistically significant standard. The heterogeneity between the included studies was analyzed by χ2 test (test level is α=0.10), and the size of heterogeneity was quantified by combining I2. If the heterogeneity was small, the meta analysis was carried out directly; if the heterogeneity was large, the source of heterogeneity was analyzed first, the obvious clinical heterogeneity excluded, and then the meta analysis carried out. When heterogeneity could not be explained, only descriptive analysis was carried out.
Results
Literature search results
A total of 931 related articles were obtained in the initial inspection, which were screened by layer and 16 were finally included, comprising 16 randomized controlled trials involving 2,847,597 participants.9–24 The flow chart of the literature search is presented in Figure 1. The characteristics of the included studies are shown in Tables 1 and 2.
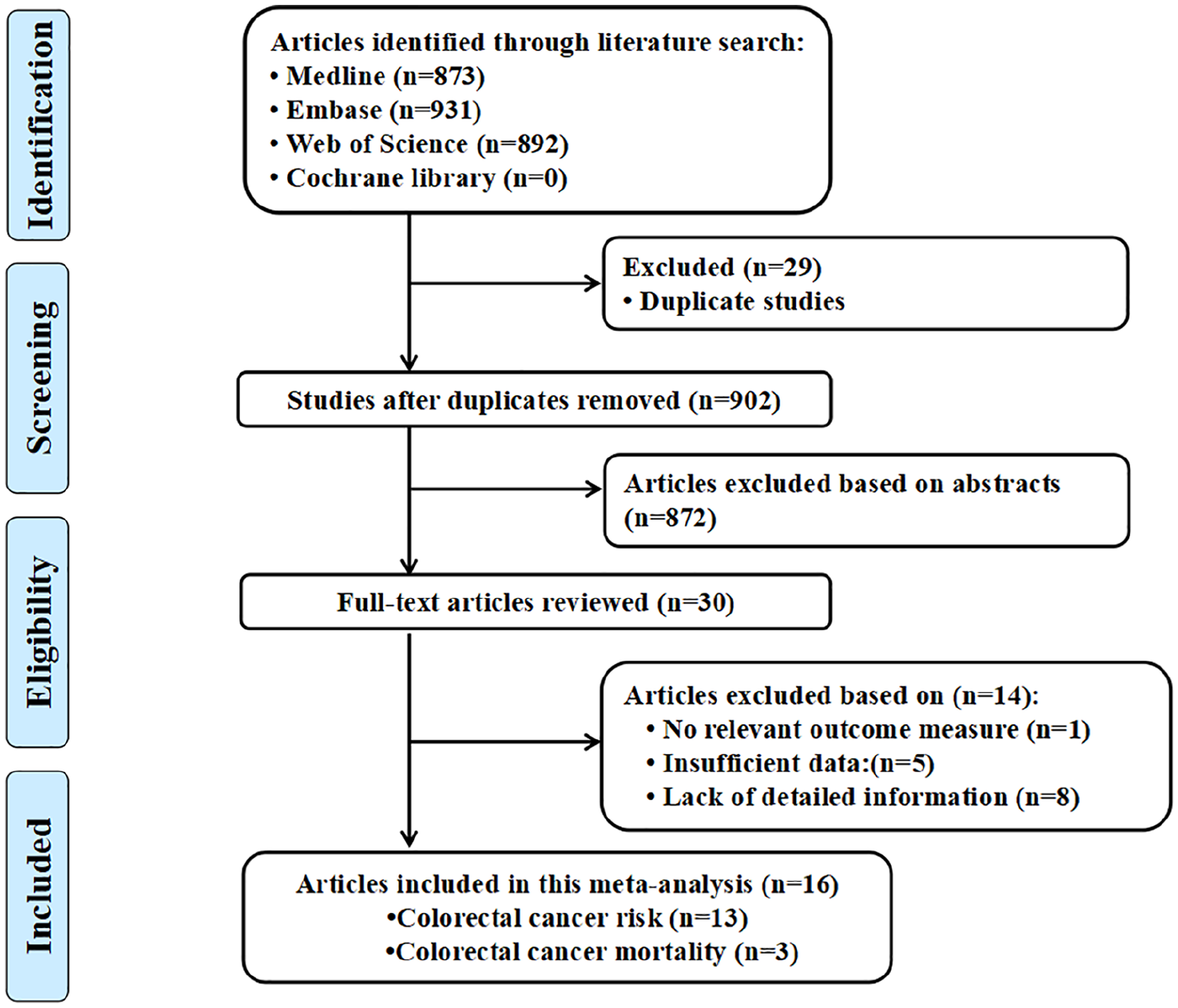
Flow diagram of the study selection process.
Characteristics of participants in included studies.

CRC: colorectal cancer; ARB: angiotensin receptor blocker; ACEI: angiotensin-converting enzyme inhibitor.
Outcomes and covariates of included studies.

CI: confidence interval; CRC: colorectal cancer; NSAID: non-steroidal anti-inflammatory drug; BMI: body mass index; ARB: angiotensin receptor blocker; ACEI: angiotensin-converting enzyme inhibitor.
RAS inhibitors and colorectal cancer risk
Figure 2 displays the results of RAS inhibitors and colorectal cancer risk. Thirteen studies (six cohort studies and seven case–control) were included in the meta-analysis to evaluate the association between RAS inhibitors and colorectal cancer risk. RAS inhibitors use was related to colorectal cancer risk decrement (relative risk (RR): 0.86; 95% confidence interval (CI): 0.78–0.93). Subgroup analysis showed ACEI/ARB (RR: 0.82; 95% CI: 0.69–0.96) or ARB (RR: 0.86; 95% CI: 0.73–0.98) or ACEI (RR: 0.81; 95% CI: 0.70–0.92) were related to colorectal cancer risk decrement. Furthermore, RAS inhibitor use was related to colorectal cancer risk decrement in Caucasians (RR: 0.88; 95% CI: 0.80–0.96) and Asians (RR: 0.72; 95% CI: 0.61–0.85; Table 3).

Forest plot showing the pooled effects of renin–angiotensin system use on the risk of colorectal cancer.
Main results of eligible studies evaluating angiotensin system inhibitor use and risk of colorectal cancer.

CI: confidence interval; ACEI: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker.
RAS inhibitors and colorectal cancer mortality
Figure 3 displays the results of RAS inhibitors and colorectal cancer mortality. Three cohort studies were included in the meta-analysis to evaluate the association between RAS inhibitors and colorectal cancer mortality. RAS inhibitor use was related to colorectal cancer mortality decrement (RR: 0.80; 95% CI: 0.66–0.98).

Forest plot showing the pooled effects of renin–angiotensin system use on colorectal cancer mortality.
Dose–response meta-analyses between RAS inhibitors and colorectal cancer risk
Six studies were included in the dose–response meta-analyses to evaluate the association between RAS inhibitors and colorectal cancer risk. A dose–response analysis showed that per one year duration of RAS inhibitor use the incremental increase was related to 6% colorectal cancer risk decrement (RR: 0.94; 95% CI: 0.90–0.97; Figure 4).

Dose–response analysis between renin–angiotensin system use and colorectal cancer risk.
Publication bias
The distribution of the included study is roughly symmetrical on both sides of the funnel diagram, and it can be considered that there is little possibility of publication bias (Figure 5). Meta-regression analysis found that the type of study (0.994), the year of publication (p=0.972) and different drugs (p=0.980) had no effect on heterogeneity. On the contrary, the number of participants (p=0.002) had a greater impact on heterogeneity. The size is the source of heterogeneity (Figure 6).
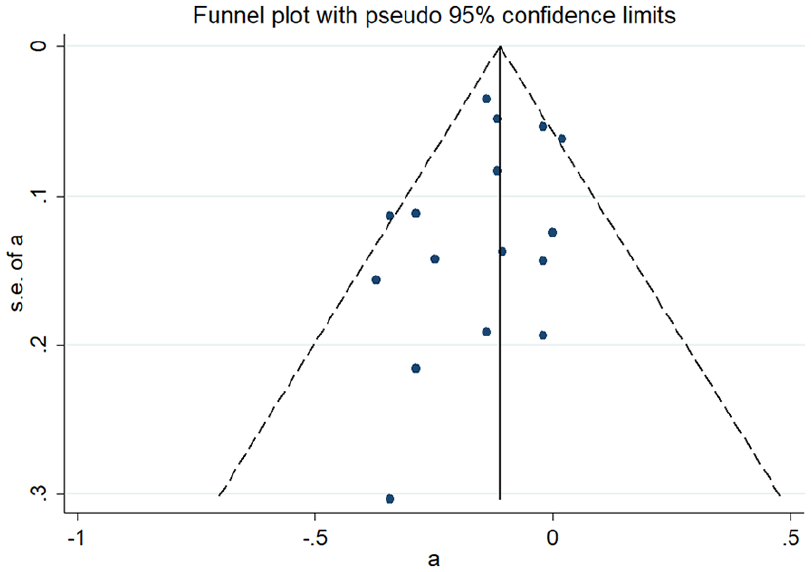
A funnel plot for the meta-analysis between renin–angiotensin system use and colorectal cancer risk.

The association between renin–angiotensin system inhibitor use and colorectal cancer risk based on the type of study (a), the year of publication (b), different drugs (c) and the number of participants (d), using meta regression.
Discussion
There is already solid evidence that some drugs that act on the cardiovascular system (such as statins, aspirin) can reduce the risk of cancer.25,26 As an inhibitor of RAS, ACEI/ARB plays an important role not only in the treatment of cardiovascular diseases, but also in cancer. 27 In recent years, a number of in vitro simulation experiments have suggested that RAS inhibitors can inhibit angiogenesis, cancer proliferation and metastasis.28–30 In contrast, some animal experiments have shown that RAS inhibitors increase the expression of vascular endothelial growth factor (VEGF) and decrease the level of platelet reactive protein 1 in tissues, thereby promoting tumor growth.31–33
What really caught the attention of the medical community regarding the safety of these drugs was a meta-analysis by Sipahi et al. in 2010. The results show that the use of ARB can increase the risk of cancer. Sipahi et al. included five randomized controlled trials with a follow-up period of at least one year and showed that the incidence of cancer in the experimental group (ARB use group) was 7.2% and the cancer risk in the control group was 6.0% (RR: 1.08; 95% CI: 1.01–1.15; p= 0.016). 31 However, the latest meta-analysis, in 2015, showed different conclusions. Yang et al. included 10 observational studies and showed that the use of ARB was not associated with cancer risk. 34 To further clarify the relationship between ACEI/ARB and colorectal cancer risk and mortality, we included 16 studies in this meta-analysis. The results showed that RAS inhibitor use was associated with colorectal cancer risk and mortality decrement. Subgroup analysis showed ACEIs/ARBs or ARBs or ACEIs were associated with colorectal cancer risk decrement.
VEGF is a heparin binding growth factor specific to vascular endothelial cells and has a strong role in inducing new angiogenesis in the human body. The expression of VEGF and its receptor has been proved to be involved in the growth and metastasis of many kinds of malignant tumors, and it is the main target of anti-angiogenesis of ACEI. 35 In many animal experiments, ACEI blocks angiotensin-converting enzyme (ACE) and the formation of angiotensin II is reduced.36,37 Since the angiotensin II can stimulate the expression of VEGF, the expression of VEGF is reduced correspondingly, so that the growth and invasion of the tumor are limited, and the purpose of anti-tumor is achieved. 38 In addition, ACEI can inhibit the formation of new blood vessels by stimulating the production of angiostatin (the hydrolysate of plasminogen) and synergism with other drugs (such as statins, vitamin K, interferon). 39 Shorning et al. used microarray analysis to detect the angiotensin converting enzyme gene. They found that the ACE gene was overexpressed during the formation of colorectal cancer. However, ACEI inhibits the activity of ACE, so ACEI has a certain preventive effect on upper colorectal cancer. 40
The expression of angiotensin receptor I was up-regulated in pancreatic carcinoma, bladder cancer and cell renal cell carcinoma.41–43 It was also found that the expression intensity of angiotensin receptor I could be used to evaluate the stage and prognosis of some malignant tumors. 44 Through selective blocking of angiotensin receptor I, ARB’s possible anticancer mechanism is as follows: (a) it has been suggested that epidermal growth factor expression may be associated with the proliferation and invasion of some malignant tumors such as skin cancer, gastric cancer and so on. Angiotensin receptor I activates some enzyme pathways, such as epidermal growth factor receptors. The selective blocking of angiotensin receptor I by inducing epidermal growth factor aggregation in tissue may lead to tumorigenesis. ARB may have an inhibitory effect on epidermal growth factor. 45 (b) Angiogenesis of malignant tumor tissue is a necessary condition for tumor growth and invasion. 46 Ino et al. point out that angiotensin receptor I can up-regulate the expression of vascular endothelial growth factor and its receptors, thus promoting angiogenesis. 47 Imai et al. found that angiotensin receptor I can be continuously synthesized in local tissues of tumor, which upregulated the expression of VEGF and promoted tumor invasion. 48 This shows that ARB may exert its anticancer effect by inhibiting the production and expression of vascular endothelial growth factor.
Meta-analysis is a descriptive quadratic analysis, which has some defects. First of all, different treatment courses and doses of each study may have greater clinical heterogeneity. Second, although systematic literature retrieval has been carried out, only 16 studies have been included in the meta analysis, so the size of the sample may have an impact. Third, there are differences in the inclusion conditions of each study, resulting in incomplete or inaccurate collection of some data, and it is not possible to clearly explain when RAS should be used, and the relationship between the dosage and the incidence of colorectal cancer. Moreover, the language of the retrieval was limited to articles published in English, which may ignore unpublished articles and cause the deviation of the language.
In summary, based on this study, the results of meta-analysis showed that RAS inhibitor use was associated with colorectal cancer risk and mortality decrement. ACEI/ARB or ARB or ACEI use was related to colorectal cancer risk decrement. But a large sample of high-quality randomized controlled studies is still needed in order to provide further confirmation.
Footnotes
Acknowledgements
XC and CY contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
