Abstract
Hypothesis/Introduction:
The aim of this study was to assess the antihypertensive efficacy and safety of aliskiren versus ramipril or losartan in hypertensive patients with type 2 diabetes mellitus, microalbuminuria and uncontrolled hypertension, despite the use of optimal conventional antihypertensive therapy.
Materials and methods:
In this open-label active comparator study, 126 patients were randomly assigned to receive 24 weeks of additional therapy with aliskiren (Group A) or either losartan or ramipril (Group B), according to whether a patient was already treated with an angiotensin-converting enzyme inhibitor or an angiotensin receptor blocker, respectively.
Results:
After 24 weeks, both treatment groups experienced a significant reduction of systolic blood pressure (−11.37% and −8.47%, respectively; both p <0.001 vs. baseline) and diastolic blood pressure levels (−10.67% and −9.28%, respectively; both p <0.001 vs. baseline), with a greater reduction of mean systolic values in Group A compared with Group B (p <0.001). Furthermore, after six months microalbuminuria was significantly decreased in both treatment groups (−67.62% and −49.1%, respectively; both p <0.001), with a reduction rate in Group A significantly higher than in Group B (p<0.001).
Conclusions:
The addition of aliskiren to optimal conventional therapy provided a higher reduction of blood pressure and urinary albumin excretion when compared with the addition of losartan or ramipril.
Keywords
Introduction
It is well established that the renin–angiotensin–aldosterone system (RAAS) plays a central role in the development of arterial hypertension. Angiotensin II (AngII) plasma levels are clearly involved in vascular endothelium alterations and atherogenesis, and contribute to the progression of target organ damage.1–8 Drugs inhibiting the RAAS, such as angiotensin-converting enzyme inhibitors (ACE-is) and angiotensin receptor blockers (ARBs), have been shown to be effective in reducing blood pressure (BP) and hypertension-associated target organ damage, 9 particularly heart failure (HF),10–12 coronary artery disease (CAD),13,14 left ventricular hypertrophy (LVH)15,16 and chronic kidney disease (CKD). 17 A treatment that includes an ACE-i or an ARB can contribute to cardiorenal protection effects; however, these drugs act by blocking the RAAS at different levels. In addition, they are not able to completely block RAAS activity because of the reactive rise in renin release induced by the withdrawal of the feedback inhibition exerted by AngII (the so-called short feedback loop), as evidenced by a reactive increase in plasma renin activity (PRA), a well-established cardiovascular risk marker, 18 providing an incomplete cardiorenal protection. 19
The central role of the RAAS in the pathogenesis of diabetic nephropathy is accepted because several studies have shown that ACE-is and ARBs can significantly reduce diabetic nephropathy.20–25 Blockade of the RAAS with ACE-is or ARBs may be delayed; however, this delay does not avoid the progression of diabetic nephropathy towards end-stage renal disease (ESRD).24,26,27 Dual blockade with ACE-is and ARBs offers no additional benefit in patients with hypertension as well as normal renal and left ventricular function; indeed, PRA increases consistently with the dual blockade. 19 The ONTARGET study showed that the combination of telmisartan and ramipril, despite reducing BP by a few millimetres of mercury more than therapy with either ramipril or telmisartan, was associated with more adverse events.28,29 More recently, a new antihypertensive class, the direct renin inhibitors (DRIs), was introduced; 30 In fact, renin is the first and primary enzyme involved in the RAAS cascade. Since the early 1980s, several compounds have been synthesized to test the effects of a blockade of renin enzyme activity (e.g. enalkiren, ramikiren and zankiren). However, none of these reached the clinical arena, because of their low inhibiting activity, lack of oral bioavailability and short half-life. Aliskiren (Novartis) is the first DRI suitable for oral administration, and it has been available since 2008. 18 Aliskiren is a powerful renin inhibitor which is highly specific for human renin. It inhibits the enzyme activity of renin at the onset of the conversion cascade of the RAAS, thus avoiding the activation of angiotensinogen into angiotensin I and consequently into AngII. 31
By blocking the first and rate-limiting step in the RAAS, aliskiren reduces PRA by at least 70% and buffers the compensatory increase in PRA observed with ACE-is and ARBs. 19 The combination of a DRI and an ARB or an ACE-i is an effective approach for lowering BP and available data indicate that such combinations favourably affect proteinuria, left ventricular mass index and brain natriuretic peptide levels in patients with albuminuria, LVH and HF.15,19,24,32
Four different trials evaluated the potential cardiorenal effects of aliskiren on morbidity and mortality. 33 One of these, the ALTITUDE, was halted by the recommendation of its Data Monitoring Committee (DMC) because of a higher event rate. 34 The recently published ASTRONAUT trial reported post-discharge mortality and HF readmissions among patients with acute HF and reduced left ventricular ejection fraction treated with aliskiren or placebo in addition to standard therapy. 35
The ATMOSPHERE trial is currently evaluating the effects of an additional treatment with aliskiren in patients with chronic HF. 36 The APOLLO trial (to date at phase 3) will provide new information regarding the role of aliskiren administered with or without additional therapy with a diuretic or a calcium channel blocker (CCB) in elderly subjects (≥65 years) with systolic BP (SBP) from 130 to 159 mmHg. The trial is designed to assess the ability of DRI to prevent major cardiovascular (CV) events, as well as its impact on global measures of physical, executive and cognitive function (ClinicalTrials.gov identifier: NCT01259297). Ongoing outcome studies will clarify which subclass of patient will derive benefit from the combination therapy of aliskiren with an ACE-i or an ARB.
The aim of the present study was to evaluate the impact on BP and on urinary albumin excretion of a dual RAAS blockade strategy in hypertensive patients with type 2 diabetes mellitus (T2DM), impaired renal function (IRF), microalbuminuria (MA) and uncontrolled hypertension, despite the administration of an optimal antihypertensive therapy, already including an ACE-i or an ARB; the study design included the addition of aliskiren to a conventional therapy with an ACE-I or an ARB, compared with the addition of ramipril or losartan to a therapy already including an ARB or an ACE-I, respectively. Furthermore, the short-term safety of these dual blockade strategies was also evaluated.
We want to underline that our study was completed before Novartis announced the termination of the ALTITUDE study due to the unexpected increased incidence of renal impairment, non-fatal stroke, hyperkalaemia and hypotension. Moreover, the patients enrolled in the ALTITUDE study presented with adequate BP levels.
Materials and methods
Study population
From June 2009 to June 2010, 1107 outpatients presented at the Hypertension Care Centre of the University of Messina, Italy. Each patient underwent a complete history and physical examination, blood sampling for routine blood chemistry, 24 h urine collection (to measure creatinine clearance and albumin excretion), electrocardiography and echocardiography. Patients with secondary hypertension, acute cardiovascular (myocardial infarction (AMI) or unstable angina) or cerebrovascular (transient ischemic attack (TIA) or stroke) disease, which occurred within the previous 12 months, were excluded from the study. We also excluded patients with left ventricular dysfunction (ejection fraction ≤40%), creatinine clearance (CrCl) ≤40 ml/min, urinary albumin excretion ≤30 mg/24 h or ≥300 mg/24 h, or with serum potassium levels ≥5.5 mEq/l. Glycosylated haemoglobin was also assessed.
We included 126 hypertensive patients (65 males and 61 females, mean age 66.8 ± 8.9 years), with T2DM, IRF and MA, defined as urinary albumin excretion ≥30 mg/24h but ≤300 mg/24 h, and BP values higher than recommended by ESC-ESH guidelines (>130/80 mmHg). 37
All patients included in the study were receiving the maximum tolerated dosage of any ACE-i or any ARB for at least six months before inclusion. During the study period, the subjects were allowed to continue their previous pharmacological treatment with their antihypertensive drugs, eventually including diuretics, β-blockers, CCBs, or any other antihypertensive drug, without dose adjustment. Biometric parameters, including weight and height, were measured in the morning under fasting conditions; accordingly, the body mass index (BMI) was estimated. All patients provided written informed consent, and the study protocol was approved by local ethical review boards. The study was conducted in accordance with good clinical practice and in accordance with the Declaration of Helsinki (2002) of the World Medical Association.
Study design
The aim of the study was to assess and compare the efficacy and safety of add-on therapy with aliskiren versus losartan (ARB) or ramipril (ACE-i) in hypertensive patients with a very high cardiovascular risk profile, due to concomitant presence of T2DM, IRF and uncontrolled hypertension, despite the current administration of optimal antihypertensive therapy, which included ACE-is or ARBs. See Figure 1. Patients were randomly assigned to receive 24 weeks of treatment with aliskiren (Group A; 63 patients), or losartan or ramipril (Group B; 63 patients), according to whether a patient was already receiving treatment with an ACE-i or an ARB, respectively. In Group A, we added aliskiren 150 mg to the previous therapy (T0), while in Group B we added ramipril 5 mg or losartan 50 mg. Drug up-titration was mandatory for SBP or diastolic BP (DBP) values ≥130 or ≥80 mmHg. Aliskiren was increased from 150 to 300 mg, ramipril from 5 to 10 mg and losartan from 50 to 100 mg; no further dose adjustments were allowed until the end of the observation period. If the glomerular filtration rate (GFR) estimated by CrCl decreased by more than 30% from baseline within four weeks after initiation of aliskiren, we would have decreased or discontinued ACE-i or an ARB. 38 The dosage of oral antidiabetic drugs or insulin was adjusted as needed. BP values, MA, CrCl and clinical chemistry were evaluated at baseline and after four (T1), 12 (T2) and 24 (T3) weeks.

Study design.
Blood pressure measurement
Clinical BP measurement was performed at our hospital in the morning between 8:00 and 10:00; it was taken in the supine position after at least 10 min of rest. Three measurements were performed with a mercury sphygmomanometer and averaged, according to current recommendations. 38
Evaluation of CrCl and albuminuria
Each enrolled patient underwent a 24 h urine collection to evaluate CrCl and MA. CrCl was calculated according to the standard formula: (urinary creatinine × 24h urinary volume)/(serum creatinine × 1440); MA was evaluated by standard immunoturbidimetric assay.
Statistical analysis
The Kolmogorov Smirnov test verified that several variables had a non-normal distribution; consequently, given also the relatively small size of our sample, we chose a permutation test-based analysis. This subset of non-parametric statistics, widely used in biomedical research, is considered preferable to the classic non-parametric approach 39 since it is based on more realistic foundations; furthermore, it is intrinsically robust and the resulting inferences are credible because they estimate the entire data distribution and exploit all information contained in the sample. 40 Accordingly, data were expressed as mean ± standard deviation (SD). Comparisons were conducted with the Nonparametric Combination test (NPC-test), which is based on a simulation or resampling procedure that is conditionally dependent on the data; thus, it provides a simulated estimate of the permutation distribution of any statistic. 41
Moreover, in order to verify the results obtained with this approach, we integrated the statistical analysis with a traditional non-parametric approach (median and IQR); consequently, the comparisons between the variables were conducted with the Mann–Whitney U test. Comparisons between different observation times were carried out with the Wilcoxon test. The correlations among the variables were assessed with Spearman’s test. To perform the statistical analyses, we used the SPSS statistical package (SPSS V. 17.0, Chicago, IL), and the NPC test 2.0 (Statistical software for multivariate permutation tests; Methodologica srl, Treviso).
Results
Patient characteristics
Table 1 presents the characteristics of the study population at baseline. Besides age, gender and biometric parameters, in the table are reported adjunctive CV risk factors (CVRFs), such as smoking habit and low high-density lipoprotein cholesterol (HDL-C) plasma levels, and comorbidities/complications such as history of CAD, previous AMI, TIA, stroke, history of coronary revascularization procedures, and the prevalence of LVH and of IRF. No significant differences could be found in regard to the age, gender, heart rate and CVRF prevalence. Group A had a higher BMI compared with Group B (28.6 ± 4.8 vs. 26.8 ± 3.6; p < 0.05). At baseline, patients were receiving an average of 2.4 antihypertensive drugs.
Baseline characteristics of study groups.
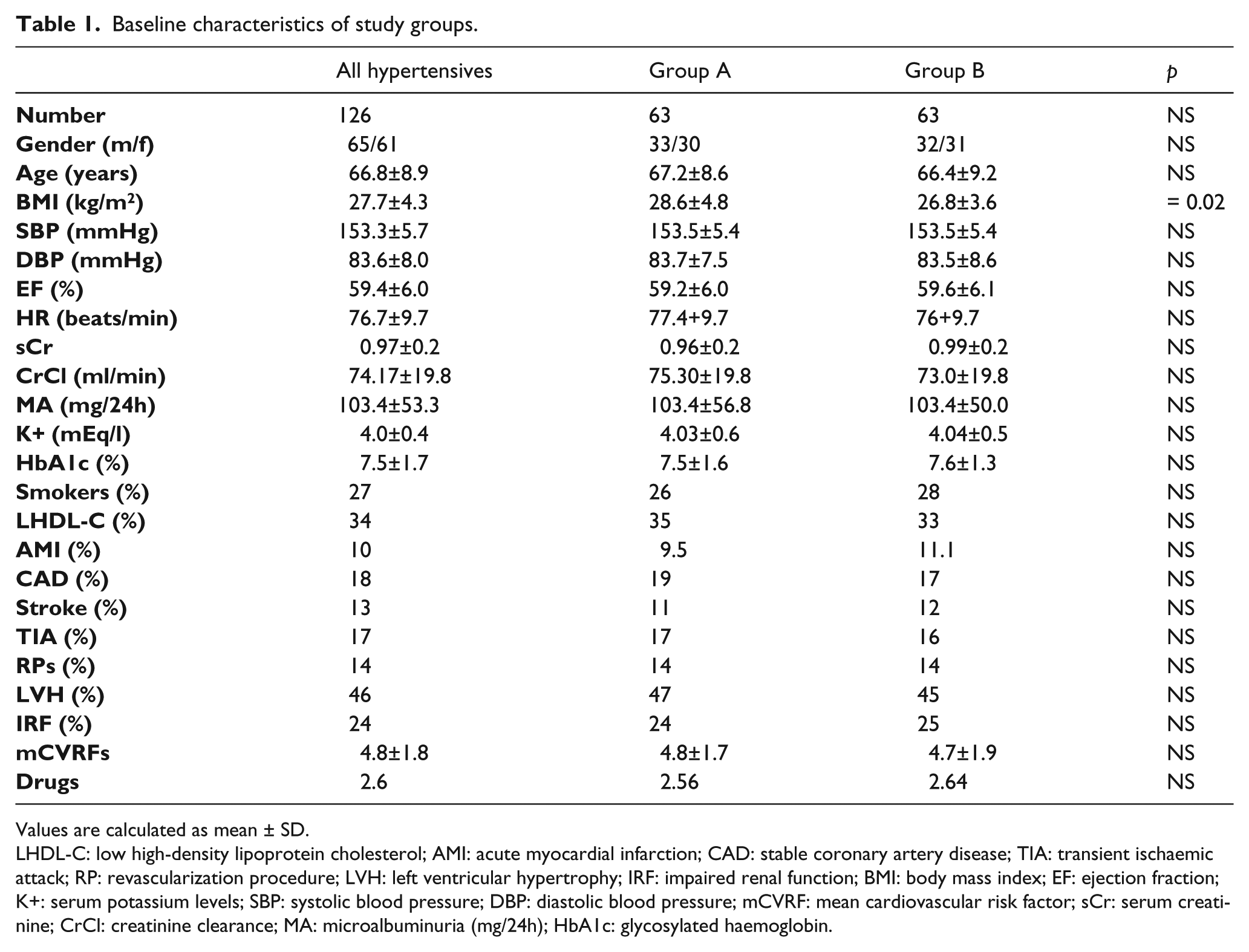
Values are calculated as mean ± SD.
LHDL-C: low high-density lipoprotein cholesterol; AMI: acute myocardial infarction; CAD: stable coronary artery disease; TIA: transient ischaemic attack; RP: revascularization procedure; LVH: left ventricular hypertrophy; IRF: impaired renal function; BMI: body mass index; EF: ejection fraction; K+: serum potassium levels; SBP: systolic blood pressure; DBP: diastolic blood pressure; mCVRF: mean cardiovascular risk factor; sCr: serum creatinine; CrCl: creatinine clearance; MA: microalbuminuria (mg/24h); HbA1c: glycosylated haemoglobin.
Four patients dropped out the study before its conclusion because of adverse events: one in Group A (diarrhoea) and three in Group B (two for cough, and one for angina pectoris). The direct causal relationship of adverse events with drugs in use and/or with pre-existing conditions was not further evaluated.
Changes in BP, MA, CrCl and serum potassium
Tables 2 and 3 present the mean changes in BP, MA, CrCl and serum potassium levels. At T1 (four weeks) in both groups there was a significant reduction of SBP (−9.08 mmHg and −5.33 mmHg, respectively; both p < 0.001) and DBP (−4.36 mmHg, p < 0.001; and −2.55 mmHg, p < 0.003, respectively).
Percentage of changes of BP, creatinine clearance, microalbuminuria and serum potassium levels at the end of observation in Group A and Group B.
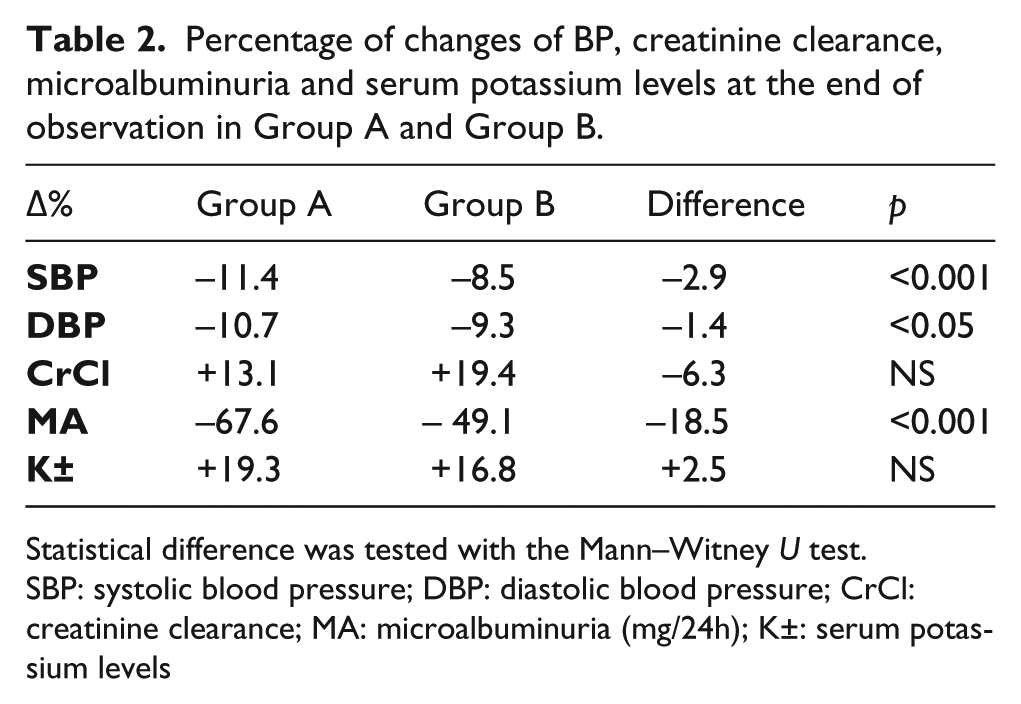
Statistical difference was tested with the Mann–Witney U test.
SBP: systolic blood pressure; DBP: diastolic blood pressure; CrCl: creatinine clearance; MA: microalbuminuria (mg/24h); K±: serum potassium levels
Percentage of changes of blood pressure, creatinine clearance, albuminuria and serum potassium levels between baseline and T3.

Data are mean ± standard deviation. Δ between group after 24 weeks. P: two-tailed Z-test for significance (Wilcoxon).
T3: 24 weeks after treatment; SBP: systolic blood pressure; DBP: diastolic blood pressure; CrCl: creatinine clearance; MA: microalbuminuria (mg/24h); K±: serum potassium levels
At T1, 35 patients from Group A (~55%) and 41 patients from Group B (~65%) still exhibited inadequate control of BP; therefore, and according to the study design, drug up-titration was prescribed. In detail, from the fifth week of treatment until the end of the observation, 35 of 63 patients in group A took aliskiren 300 mg daily, whereas 27 continued with 150 mg daily; in Group B, 22 out of 30 patients who were taking ramipril 5 mg switched to 10 mg per day, and 19 out of 33 patients who were taking losartan 50 mg switched to 100 mg per day.
Figure 2 presents SBP, DBP, MA, CrCl and serum potassium level variation during the observation period.

Variation of systolic blood pressure (SBP) (a), diastolic blood pressure (DBP) (b), creatinine clearance (CrCl) (c), microalbuminuria (MA) (d), serum potassium (K+) (e) during observation period (four (T1), 12 (T2) and 24 (T3) weeks).
At the conclusion of the study (T3) both treatments provided a significant reduction of SBP (−11.4 mmHg and −8.5 mmHg, respectively; both p < 0.001) and DBP levels (−10.7 mmHg and −9.3 mmHg, respectively; both p < 0.001) versus baseline. The reduction rate of SBP in Group A was significantly higher than in Group B (Δ = −2.9%; p < 0.01); furthermore, the DBP reduction rate reached statistical significance (Δ = −1.4%; p = 0.05) (Figure 2(a) and (b)).
At T3 both treatments provided a significant increase of CrCl (+13.1 ml/min and +19.4 ml/min, respectively; both p < 0.001). There was no significant difference between the groups (p: NS) (Figure 2(c))
MA levels remained substantially unchanged after the first four weeks in both groups; a significant urinary albumin excretion reduction was observed after 12 weeks of treatment (T2) in both the treatment groups, and it decreased further after 24 weeks (T3) (p < 0.001 for both groups compared with baseline). The MA reduction rate was significantly higher in Group A compared with Group B (Δ = −18.5%; p < 0.001) (Figure 2(d)).
During follow-up, we observed a slight, progressive increase of serum potassium levels when compared with baseline (+ 0.78 mEq/l and + 0.68 mEq/l, respectively; both p < 0.001) (Figure 2(e)) without a significant difference between the groups (Δ = + 0.1 mEq/l; 2.5%; p = 0.51, NS). Five patients experienced a mild increase of serum potassium levels (K+ > 5.5 but < 6.0 mEq/l) and they were managed with dietary potassium restriction. None of these patients were discontinued from the trial because of increase of serum potassium levels, or increase in baseline plasma creatinine > 30%.
Discussion
Patients enrolled in our study are those that are termed ‘complicated’. We usually define ‘complicated’ patients as hypertensive subjects with diabetes, MA and previous CV events. In fact, these conditions increase the risk of new CV events and accelerate the progression of target organ damage, such as end-stage renal disease; moreover, low numbers of patients at very high CV risk achieve the BP target levels. 37
At the conclusion of the study, we observed a reduction of BP and MA in all patients. This result is likely due to the addition of a RAAS antagonist to the previous standard treatment. Notably, in this study, both treatments showed an adequate safety and tolerability profile. In fact, we actually had a low dropout incidence due to an increase of serum potassium levels: we had five cases of mild hyperkalaemia, which were managed with dietary potassium restriction. None of these patients were discontinued from the trial because of hyperkalaemia, or an increase in baseline plasma creatinine > 30%. Furthermore, our study showed that the addition of aliskiren to a previous conventional antihypertensive treatment that included a RAAS antagonist, either an ACE-i or an ARB, had a greater effect in reducing SBP than the addition of either ACE-i or ARB; further reduction of SBP by approximately 4 mmHg was found with the former regimen.
No difference was observed in DBP reduction between the two groups.
However, our results are in agreement with the findings of other clinical trials.42–44 An eight-week, double-blind, multicentre trial 45 assessed whether the combination of aliskiren and ramipril in patients with diabetes and hypertension was safe and effective in lowering BP when compared with the respective monotherapies. In this study aliskiren showed higher SBP reduction with ramipril. Moreover, when used in combination with ramipril, aliskiren provided a significant additional reduction in both SBP and DBP. Another eight-week, randomized, controlled trial 42 compared valsartan, aliskiren and their combination at the maximum dose (320/300 mg) in patients with mild to moderate hypertension. This study showed that the combination of aliskiren and valsartan at maximum recommended dosages provides greater reduction in BP values compared with monotherapy with valsartan or aliskiren alone.
We must stress that these results were obtained in a clinical setting of mild to moderate residual hypertension in T2DM patients, where a significant degree of pressure reduction is hard to achieve, especially in a limited timeframe. A recent 24-week, open-label, single-arm study showed that the combination therapy of aliskiren and a RAAS blocker in CKD patients who were already being treated with ACE-is or ARBs for more than six months had a favorable effect on reducing residual proteinuria and BP reduction. 44 Another recent study 43 confirmed the efficacy and safety of aliskiren in a real-life setting. Aliskiren is approved for treatment of hypertension, but has also shown renoprotective potential in normotensive patients with T2DM and albuminuria.24,25,46 All patients included in the study were receiving the maximum dosage of an ACE-i or ARB for at least six months before inclusion. To date, this is the optimal treatment to reduce albuminuria and delay the progression of CKD. 38 After 24 weeks we observed a significant reduction of urinary albumin levels in both groups. In Group A, we observed a higher reduction of MA compared with group B (−67.6% vs. −49.1%, respectively; versus basal, additional reduction −18.5%; p< 0.001); furthermore, in both study groups we observed, after an initial decrease at four weeks, a significant improvement of GFR estimated with CrCl at 24 weeks with respect to baseline (+13.1 ml/min and +19.4 ml/min, respectively; both p < 0.001); however, no difference was detected in CrCl variation between the two groups. Adjunctive therapy with aliskiren appeared to provide a more beneficial improvement of diabetic nephropathy in hypertensive patients, as marked by urinary albumin excretion, compared with other treatment protocols that are considered to be the gold standard. 38 The renal protective effects of aliskiren could be related to its organ-specific mechanism of action. 18 A direct renin inhibition with aliskiren provides a greater intrarenal protective effect than other RAAS antagonists. 47 The results of our study show that the multi-level blockade of the RAAS represents a good therapeutic strategy, and confirms the efficacy and safety of aliskiren. These results are consistent with those of other and larger randomized clinical trials. The AVOID study recruited 599 patients with hypertension, T2DM and proteinuria who were already receiving the maximum recommended renoprotective treatment with losartan (100 mg daily) and exhibited an optimal management of hypertension; enrolled patients were randomized to receive adjunctive treatment with aliskiren (300 mg daily) or placebo. In the group treated with aliskiren, a significant reduction of albuminuria, compared with placebo, was observed. This benefit appeared to be independent of systemic BP reduction. 24 Adjunctive therapy with aliskiren appears to be a unique opportunity for patients at high cardiovascular risk. These patients usually show a poor response to conventional antihypertensive treatment. It is likely that the double blockade of RAAS with DRI and an ACE-i or an ARB can help us to better understand the pathogenesis of hypertension. If so, it would provide a more effective therapeutic strategy in preventing hypertension-related diseases; consequently, it would delay the progression towards the end-organ damage. More long-term data are needed to confirm the efficacy and safety of this regimen in these patient populations. The ASPIRE HIGHER programme was undertaken to evaluate potential cardiorenal effects of aliskiren over a spectrum of conditions in 14 different studies involving more than 35,000 patients. 33 Three of these studies (AVOID, ALLAY and ALOFT) evaluated surrogate end-points and confirmed the favourable effects of adding aliskiren to standard treatment. The ASPIRE HIGHER programme also included four morbidity and mortality trials. 33 One of these, the ALTITUDE, was halted by a recommendation from its DMC. 34 The basis of the DMC recommendation was futility (i.e. no prospect of demonstrating the treatment benefit anticipated in the protocol), as well as safety concerns. These concerns included renal dysfunction and hyperkalaemia. Doubling of serum creatinine occurred in 4.8% of the aliskiren group vs. 5% of the placebo group (p= NS). ESRD or renal death occurred in 2.8% of the aliskiren group vs. 2.5% of the placebo group (p= NS). Hyperkalaemia occurred in 39% of the aliskiren group vs. 29% of the placebo group, and severe hyperkalaemia was observed in 21% vs. 16%. It was assessed that there were no cases where the increase in potassium needed dialysis, but there was one case where raised potassium levels were specified as the cause of death. Other main concerns were hypotension and an excess of non-fatal strokes. In response to these findings, it has been recommended that dual aliskiren and ACE-i/ARB therapy not be used in patients with both hypertension (the current indication for aliskiren) and diabetes or moderate to severe renal dysfunction. The recently-published ASTRONAUT trial 35 reported that the addition of aliskiren to standard therapy in patients with acute HF and reduced left ventricular ejection fraction (LVEF) appeared to improve post-discharge outcomes and biomarker profiles; it was generally well-tolerated in non-diabetic patients. In contrast, diabetic patients receiving aliskiren appeared to have poorer post-discharge outcomes.
The ATMOSPHERE trial is currently being conducted on patients with chronic HF.36,48 We want to stress that, in the population enrolled in ALTITUDE, BP was well-controlled at baseline, and patients were randomized to receive aliskiren 150 mg or placebo in addition to their conventional treatment (including a maximum dose of ACE-i or ARB). After four weeks of treatment, the patients were force-titrated to receive aliskiren 300 mg. In contrast, all of the patients enrolled in our study had poor BP control at baseline, and aliskiren was up-titrated to 300 mg only if the BP target was not achieved at the fourth week. This study design allowed a low dropout rate and a lower incidence of side effects compared with other studies.
Further larger clinical studies are ongoing, and they may add further insight regarding aliskiren safety and efficacy; furthermore, they may increase understanding regarding the mechanisms underlying its cardiorenal protective potential. The APOLLO Trial should provide new information regarding the role of aliskiren (with or without additional therapy with a diuretic or a CCB) in elderly subjects (≥ 65 years; with SBP 130 to 159 mmHg) in preventing major CV events and on global measures of physical, executive and cognitive function (ClinicalTrials.gov identifier: NCT01259297). Moreover, we hope that a sub-analysis of ALTITUDE, and the outcomes of the other long-term studies (ATMOSPHERE, ASTRONAUT and APOLLO) will help us to identify the subgroup of patients that could obtain an advantage from dual RAAS blockade with DRI and ACE-I or ARB.
Study limitations
The major limitation of our study is the small sample size that limits our ability to determine statistical significance.
Conclusions
The addition of aliskiren to the standard therapy provides greater reduction of BP and urinary albumin excretion than adding an ACE-i (ramipril) to an ARB and vice versa (losartan to ACE-i). The dual blockade of RAAS can be associated with an increased risk of hyperkalaemia compared with monotherapy; therefore, we recommend that this therapeutic strategy requires closer monitoring of renal function and serum potassium levels.
Footnotes
Conflict of interest
The authors have no conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors
