Abstract
Introduction:
Dipeptidyl peptidase (DPP)-4 inhibitors, a novel oral anti-diabetic agents, exert a protective effect on pancreatic β-cell function in patients with type 2 diabetic mellitus (T2DM). However, their beneficial effect in hypertensive T2DM patients treated with angiotensin receptor blockers (ARBs) has not been investigated.
Methods:
In this open-label multicenter randomized study, a total of 55 hypertensive T2DM patients treated with ARBs were randomly assigned to receive the DPP-4 inhibitor sitagliptin or sulfonylurea (SU).
Results:
After 24 weeks of treatment, a significant reduction in fasting blood glucose was only observed in the sitagliptin group, while HbA1c was significantly reduced in both groups. Homeostasis model assessment of insulin resistance was not significantly improved in either group. Indicators of pancreatic β-cell function, including proinsulin to insulin ratio and homeostasis model assessment of β-cell function, were significantly improved in the sitagliptin group, but not in the SU group. The beneficial effects of sitagliptin were observed in hypoglycemic drug naïve patients, but not in patients who had received SU monotherapy prior to the study.
Conclusion:
Treatment with the DPP-4 inhibitor sitagliptin might exert beneficial effects on pancreatic β-cell function in ARB-treated T2DM patients and its efficacy might be more pronounced in hypoglycemic drug naïve patients.
Keywords
Introduction
Pancreatic β-cell dysfunction is profoundly implicated in the pathogenesis of type 2 diabetes mellitus (T2DM) and preservation of β-cell function is an important therapeutic strategy to reduce the risk of T2DM and prevent diabetic complications.1–3 Sulfonylurea (SU) is a commonly used anti-diabetic agent that stimulates insulin secretion from the β-cell; however, long-term treatment with SU has been demonstrated to exhaust β-cell function, despite its glucose-lowering effects.3,4 Recently, dipeptidyl peptidase (DPP)-4 inhibitors, a novel class of oral anti-diabetic agents, have been shown not only to exert antihyperglycemic effects, but also to preserve pancreatic β-cell function in animal experiments and clinical studies.5–9
Hypertensive patients often have concomitant T2DM, and diabetic hypertensive patients are more susceptible to the development of cardiovascular diseases.10,11 Accumulating evidence indicates that treatment with angiotensin receptor blockers (ARBs) prevents the new onset of T2DM, and that ARBs exert a protective role in the development of diabetic nephropathy.12,13 Consequently, clinical guidelines recommend that hypertensive patients with T2DM receive renin-angiotensin system inhibitors as the first line of antihypertensive agents.14,15 In addition, ARBs have recently been reported not only to improve insulin sensitivity, but also to preserve pancreatic β-cell function in both animal experiments and clinical studies.16–19 Therefore, it is clinically relevant to investigate whether the beneficial effect of DDP-4 inhibitors could be exerted in ARB-treated T2DM patients.
In this study, we investigated the protective effects of the DPP-4 inhibitor sitagliptin on pancreatic β-cell function in ARB-treated T2DM patients and compared its efficacy with that of the conventional anti-diabetic agent SU. We showed for the first time that pancreatic β-cell function in ARB-treated T2DM patients was significantly improved by sitagliptin treatment, but not by SU treatment, and that the beneficial effect of sitagliptin was likely to be exerted in hypoglycemic drug naïve patients rather than in patients receiving add-on treatment to SU monotherapy.
Methods
Study subjects
This study was conducted at 10 Kyoto Lifestyle Study Group centers (Appendix). Patients with essential hypertension and T2DM (20–84 years), who visited the attending clinics from May 2012–March 2013, were considered for screening and potential recruitment into the trial. Patients were excluded from the study if there was any evidence of type 1 diabetes mellitus, treatment with antihyperglycemic agents except for SU, renal failure (serum creatinine⩾1.5 mg/dl), or severe liver dysfunction. Patients receiving insulin therapy were also excluded. The study was conducted in accordance with the principles of the Helsinki Declaration. The investigational protocol was approved by the ethics committee for human studies at the Kyoto Prefectural University of Medicine. Informed consent was obtained from all patients before any procedures were performed.
Study design
This study was an open-label multicenter study consisting of a two-month screening/baseline period and a six-month treatment period. We included patients with HbA1c levels ⩾7% who either had never taken a hypoglycemic agent or had taken standard fixed doses of SU for at least two months prior to beginning the study. After screening, 61 patients entered the trial and either the hypoglycemic agent sitagliptin (50 mg/day) or SU was started. When patients had received SU monotherapy prior to the study and were assigned to the sitagliptin group, prescribed fixed doses of SU was administered throughout the study, but they were not switched to sitagliptin treatment alone. Adjustments of hypoglycemic agents, if needed, were only allowed for sitagliptin or SU in each group.
Efficacy assessment
After an overnight fast, fasting blood glucose (FBG), HbA1c, insulin, proinsulin, and fasting lipids were measured at baseline and during the study. To assess pancreatic β-cell function, proinsulin to insulin ratio and homeostasis model assessment of β-cell function (HOMA-β) were evaluated at baseline and after 24 weeks of treatment. Homeostasis model assessment of insulin resistance (HOMA-IR) was also evaluated at baseline and after 24 weeks of treatment to assess the insulin sensitivity.
Statistical analysis
All data are expressed as the mean±standard deviation (SD). The significance of differences was evaluated by a two-way analysis of variance (ANOVA) with repeated measures on the time course of variables followed by Bonferroni post-hoc comparison tests. Comparisons of parameters among subgroups were made by unpaired t-test. The chi-square (χ2) test was used to compare categorical values. p<0.05 was considered statistically significant.
Results
Originally, 61 patients were enrolled in this study and six patients dropped out due to protocol violation. The remaining 55 patients were available for the safety analysis. During the entire period, four (7.2%) patients experienced one or more adverse events. One patient (1.8%) experienced a hypoglycemic attack and stopped receiving agents. One patient complained of nausea and abdominal discomfort after increasing the dose of SU. Two patients (3.6%) experienced cerebral infarction and acute coronary syndrome, respectively. In another four patients, fasting blood samples were not obtained after 24 weeks of treatment. Finally, 43 patients completed the entire trial and were used for evaluation of efficacy.
The baseline characteristics of the study population are summarized in Table 1. Patients’ mean age and gender did not differ between the two groups. The percentage of oral antidiabetic drug naïve patients was relatively, but not significantly, higher in the sitagliptin group than that in the SU group. The percent of patients receiving ARB monotherapy was equivalent between the two groups. The information of prescribed ARBs including drug name and dosage form were shown in Supplementary Material, Table 1. Prescribed fixed doses of ARBs were administered throughout the study. The prescription rates for other antihypertensive drugs did not differ between the two groups except for calcium channel blockers (CCBs), which were preferably used in the SU group. Mean systolic pressure, diastolic pressure, and lipid profiles did not differ between the two groups. Although fasting blood glucose and HbA1c were equivalent between the two groups, the serum concentration of insulin and HOMA-IR were modestly, but not significantly, higher in the SU group than in the sitagliptin group (p=0.08, p=0.07, respectively), suggesting that insulin sensitivity was moderately impaired in the SU group compared to the sitagliptin group. Proinsulin to insulin (PI) ratio and HOMA-β did not show any difference between the two groups.
Baseline characteristics of all patients.

ARB: angiotensin receptor blocker; CCBs: calcium channel blockers; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; HOMA-β: homeostasis model assessment of β-cell function; HOMA-IR: homeostasis model assessment of insulin resistance; NGSP: national glycohemoglobin standardization program; PI: proinsulin to insulin. Data are mean±standard deviation (SD).
The changes in glycemic parameters after 24 weeks of treatment are shown in Table 2. Sitagliptin treatment significantly reduced HbA1c from 7.4±0.7% to 6.7±0.6% (p<0.01), whereas a significant reduction in FBG was not achieved from 148±33 to 133±35 mg/dl (p=0.08). Likewise, SU treatment significantly reduced HbA1c from 7.3±1.0% to 7.0±1.0% (p=0.02), without affecting FBG (p=0.53). HOMA-IR was not significantly improved in either group. We next examined the effect of hypoglycemic treatment on pancreatic β-cell function. PI ratio and HOMA-β after sitagliptin treatment were substantially improved from 3.81±1.56 to 2.79±1.29 (p<0.01) and from 48.8±29.6 to 87.4±72.7 (p=0.01), respectively. In contrast, SU treatment did not significantly improve PI ratio (3.18±1.46 to 2.81±1.10, p=0.14) or HOMA-β (72.5±67.7 to 83.4±46.9, p=0.43). We further compared the mean changes in PI ratio and HOMA-β between the sitagliptin and SU groups; however, there were no significant difference between the two groups (p=0.12, p=0.16, respectively).
Changes in glycemic parameters in all patients.

HOMA-β: homeostasis model assessment of β-cell function; HOMA-IR: homeostasis model assessment of insulin resistance; NGSP: national glycohemoglobin standardization program; PI: proinsulin to insulin. Data are mean±standard deviation (SD).
We also analyzed the effect of sitagliptin on blood pressure and compared with that of SU. Twelve weeks of sitaglyptin treatment significantly reduced systolic blood pressure from 138.7±14.3 mm Hg to 132.4±14.8 mm Hg (p<0.05) and diastolic blood pressure from 80.6±9.3 mm Hg to 76.9±9.7 mm Hg (p<0.05); however the blood pressure at 24 weeks was not significantly reduced (Supplementary Material, Table 2). In contrast, SU treatment significantly decreased only systolic blood pressure at 24 weeks from 134.8±11.6 mm Hg to 127.2±14.2 mm Hg (p<0.05); however, there was no significant difference in antihypertensive effect between sitagliptin and SU groups.
The percentage of antihyperglycemic agent naïve patients in sitagliptin group was relatively, but not significantly, higher than in the SU group. We therefore performed a two-way ANOVA and found that the beneficial effect of sitagliptin on PI ratio was significantly affected by the prescribed antihyperglycemic agent SU (p<0.01). Thus, the efficacy of sitagliptin was separately analyzed in patients with or without history of SU treatment prior to the study. Baseline characteristics in hypoglycemic drug naïve patients did not show any significant difference between the 2 treatment groups (Table 3). Sitagliptin treatment reduced HbA1c to the same extent as SU treatment, while FBG was not significantly decreased in either group (Table 4). Marked improvements in PI ratio (3.87±1.54 to 2.68±1.05, p=0.01) and HOMA-β (48.1±27.4 to 83.4±66.3, p=0.03) were observed after treatment with sitagliptin. In contrast, SU treatment did not significantly improve PI ratio or HOMA-β (p=0.08, p=0.07, respectively). The mean changes in PI ratio and HOMA-β did not differ between the two groups.
Baseline characteristics of antihyperglycemic drug naïve patients.

ARB: angiotensin receptor blocker; CCBs: calcium channel blockers; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; HOMA-β: homeostasis model assessment of β-cell function; HOMA-IR: homeostasis model assessment of insulin resistance; NGSP: national glycohemoglobin standardization program; PI: proinsulin to insulin. Data are mean±standard deviation (SD).
Changes in glycemic parameters in antihyperglycemic drug naïve patients.

HOMA-β: homeostasis model assessment of β-cell function; HOMA-IR: homeostasis model assessment of insulin resistance; NGSP: national glycohemoglobin standardization program; PI: proinsulin to insulin. Data are mean±standard deviation (SD).
We next analyzed the patients who had received SU monotherapy prior to the start of study. The baseline characteristics are shown in Table 5. Except for systolic blood pressure, which was significantly higher in the sitagliptin group than the SU group (144.1±11.3 vs 134.6±10.8 mm Hg, respectively, p=0.03), there was no difference between the two groups. Although FBG and HbA1c were significantly reduced in the sitagliptin group, PI ratio and HOMA-β were not improved (Table 6). SU treatment did not improve PI ratio or HOMA-β as in the hypoglycemic drug naïve patients. These findings suggest that the protective effect of sitagliptin on pancreatic β-cell function is likely to be exerted in hypoglycemic drug naïve patients rather than patients who had received SU monotherapy. To examine the underlying mechanism, we compared the baseline glycemic parameters in the sitagliptin group between hypoglycemic drug naïve patients and patients receiving SU monotherapy prior to the study (Supplementary Material, Table 3). However, PI ratio, HOMA-β, as well as other parameters did not show any significant differences between the two groups, suggesting the possibility that SU treatment could counteract the beneficial effect of sitagliptin on pancreatic β-cell function.
Baseline characteristics of patients receiving sulfonylurea monotherapy prior to the study.
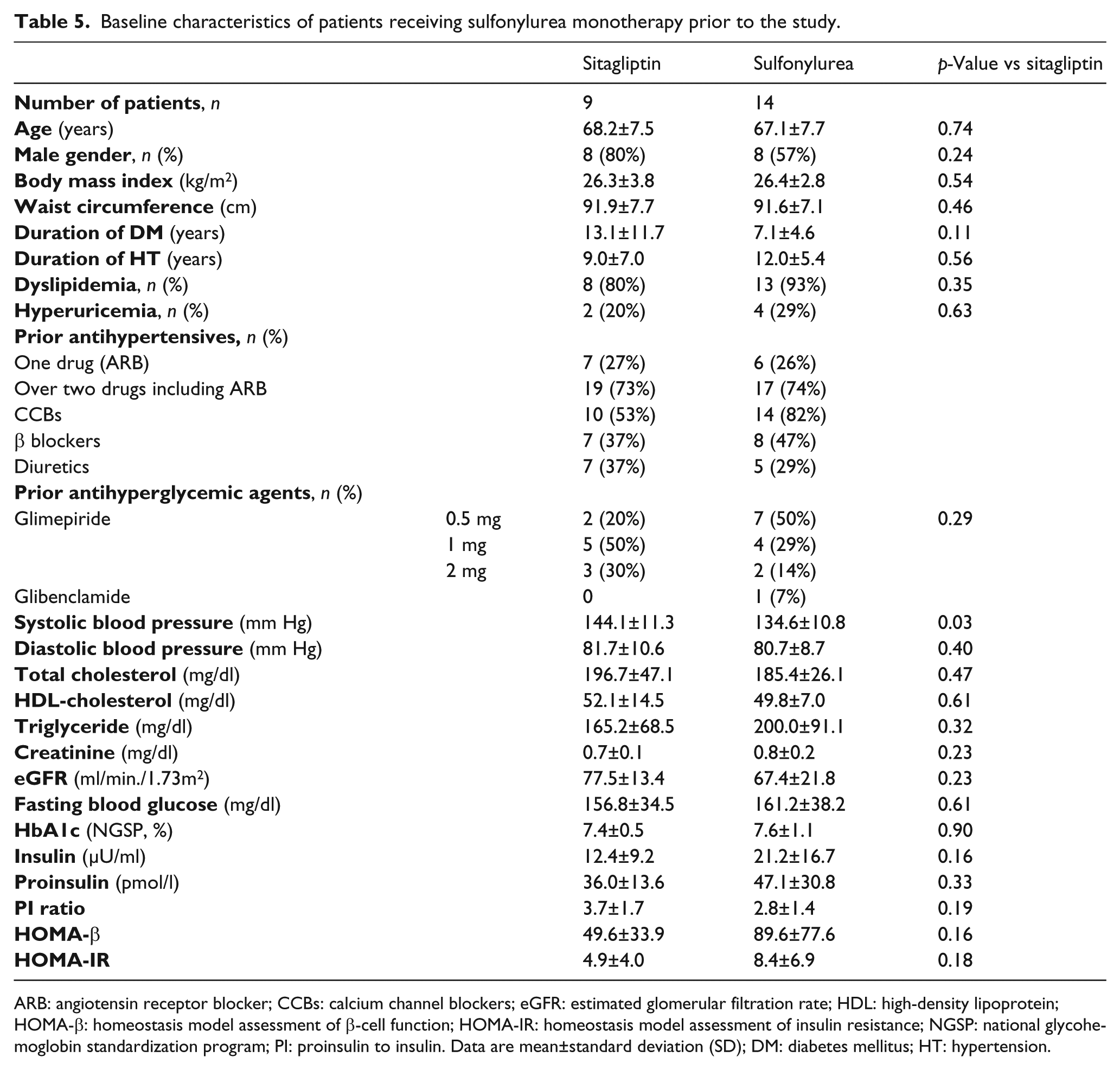
ARB: angiotensin receptor blocker; CCBs: calcium channel blockers; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; HOMA-β: homeostasis model assessment of β-cell function; HOMA-IR: homeostasis model assessment of insulin resistance; NGSP: national glycohemoglobin standardization program; PI: proinsulin to insulin. Data are mean±standard deviation (SD); DM: diabetes mellitus; HT: hypertension.
Changes in glycemic parameters in patients receiving sulfonylurea monotherapy prior to the study.
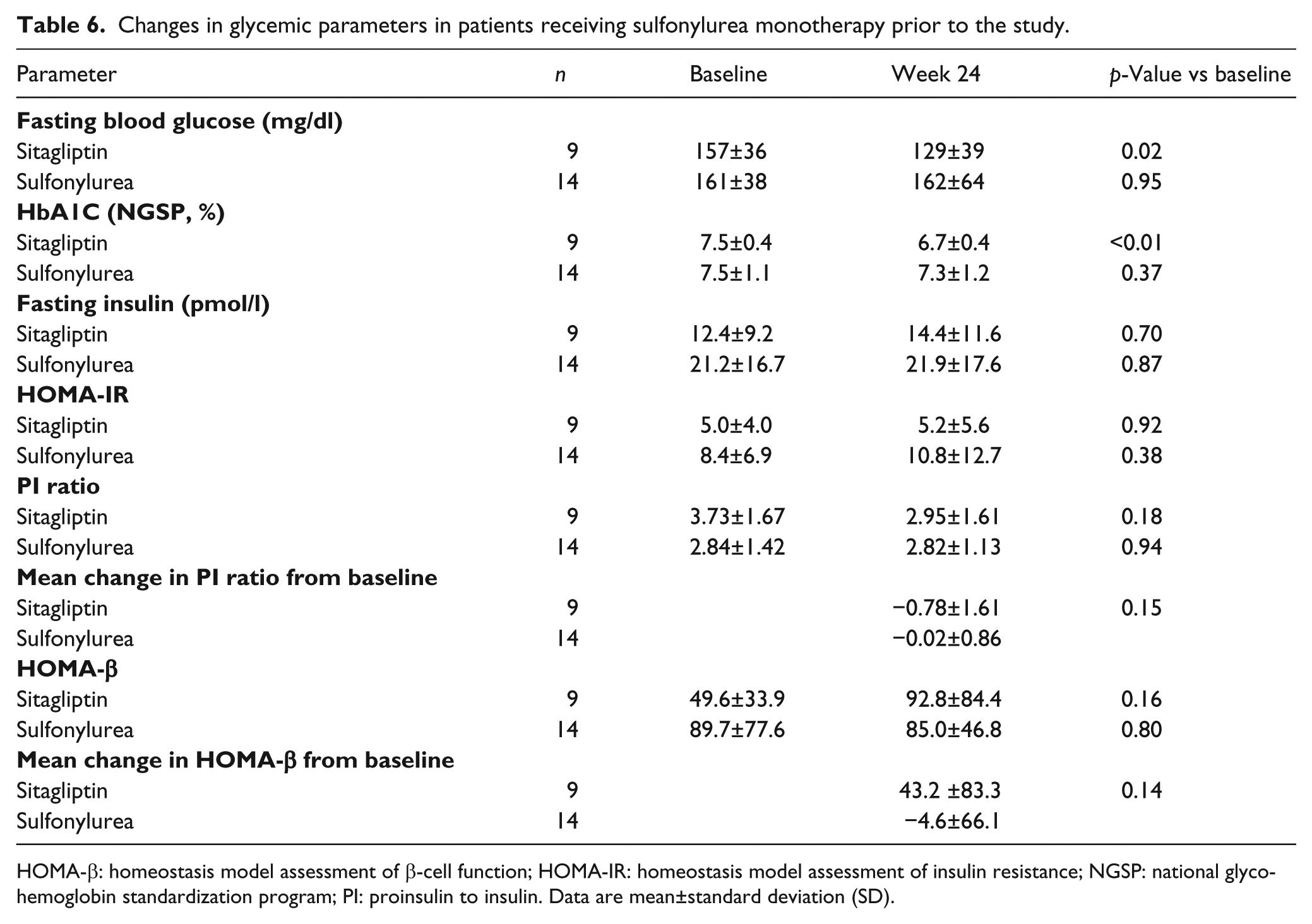
HOMA-β: homeostasis model assessment of β-cell function; HOMA-IR: homeostasis model assessment of insulin resistance; NGSP: national glycohemoglobin standardization program; PI: proinsulin to insulin. Data are mean±standard deviation (SD).
Discussion
The present study showed that sitagliptin treatment could exert a protective effect on pancreatic β-cell function as well as an antihyperglycemic effect in ARB-treated hypertensive patients with T2DM. Furthermore, the beneficial effect of sitagliptin might be more pronounced in hypoglycemic drug naïve patients than in patients who had received SU monotherapy prior to the study. These findings suggest that the protective effect of sitagliptin could be independent of the ARB-associated beneficial effects on glucose homeostasis and that the ability of sitagliptin to preserve pancreatic β-cell function is reduced in combination with SU therapy.
Impaired insulin sensitivity is initially compensated by hyperinsulinemia, which exhausts pancreatic β-cell function, leading to the development of T2DM.1–3 Preservation of pancreatic β-cell function is a potential therapeutic strategy to prevent the transition from insulin resistance to T2DM. Novel oral antidiabetic DPP-4 inhibitors have recently been shown not only to exert hypoglycemic effects, but also to preserve pancreatic β-cell function in animal experiments and clinical studies.5–9 On the other hand, ARBs, the first line antihypertensive agents in T2DM patients, have recently been shown to exert beneficial effects on pancreatic β-cell function as well as on the insulin signaling cascades in skeletal muscle cells and hepatocytes.16–19 It is therefore clinically relevant to determine whether the beneficial effects of DPP-4 inhibitor on pancreatic β-cell function could be observed in T2DM patients treated with ARBs. Our findings demonstrated for the first time that the DPP-4 inhibitor sitagliptin improved pancreatic β-cell function in ARB-treated T2DM patients. These findings suggest that the protective effect of sitagliptin on pancreatic β-cell function is independent of ARB treatment and that DPP-4 inhibitors may have an additive effect to that of ARBs.
Shao et al. showed that treatment with the ARB candesartan in db/db mice, a model of T2DM with obesity, improved glucose tolerance and increased β-cell mass. 18 They also demonstrated that oxidative stress markers as well as expression of NADPH oxidase subunit p22phox and gp91phox were significantly reduced in pancreatic β-cells, suggesting that ARB treatment preserves β-cell function by reducing oxidative stress. In addition, a recent in vitro experiment by Madec et al. showed that high glucose exposure in human islets significantly increased endoplasmic reticulum stress markers along with augmented expression of angiotensin type 1 receptor and angiotensinogen. 19 They also showed that these deleterious effects of high glucose exposure were attenuated by treatment with the ARB losartan, which also improved insulin secretion. Likewise, the underlying mechanism for DPP-4 inhibitor-mediated protective effects on pancreatic β-cells has recently been extensively investigated. Shimizu et al. developed pancreatic β-cell-specific C/EBPB transgenic mice and showed that GLP-1, the circulating level of which was increased by DPP-4 inhibitor through inhibiting degradation, induced C/EBPB degradation, leading to the attenuation of endoplasmic reticulum stress in β-cells. 20 However, an additive effect of ARBs and DPP-4 inhibitors on β-cell function has not been fully investigated. Wang et al. demonstrated that combination treatment of GLP-1 and the ARB candesartan exerted an additive anti-apoptotic effect in isolated mouse pancreatic islets. 21 Although the efficacy of DPP-4 inhibitors has been studied widely in humans, the prevalence of hypertension and concurrent administration of antihypertensive agents have not yet been addressed.22–26 In this study, we showed for the first time that the DPP-4 inhibitor sitagliptin could exert protective effects on pancreatic β-cells in T2DM patients treated with ARBs. Underlying mechanisms of the additive effect between ARBs and DPP-4 inhibitors should be investigated.
The effects of sitagliptin on β-cell function were more pronounced in hypoglycemic drug naïve patients than in patients who had received SU monotherapy prior to the study. When DPP-4 inhibitors have been used as add-on therapy to other blood glucose-lowering agents, additional beneficial effects have been reported in previous human studies; however, most of the concomitant glucose-lowering agents were metformin or thiazolidinedione, but not SU,5,22,23 which did not slow down β-cell function deterioration. 3 Considering that the baseline glycemic parameters in the sitagliptin group did not differ between patients naïve to hypoglycemic drugs and those who had received SU monotherapy, it is likely that combination therapy of sitagliptin with SU may attenuate the protective effect of sitagliptin on β-cell function, despite their glucose-lowering effects.
The limitation of the study is the lack of blinding between groups. Moreover, this trial is a small exploratory trial and is designed as a preliminary study to evaluate the beneficial effect of sitagliptin on pancreatic β-cell function in hypertensive T2DM patients treated with ARBs. To confirm the validity of these observations, a large-scale, prospective, and randomized double-blind trial is needed.
In conclusion, sitagliptin treatment could exert a protective effect on pancreatic β-cell function as well as an antihyperglycemic effect, without serious adverse effects in ARB-treated T2DM patients. The efficacy of sitagliptin might be more pronounced in hypoglycemic drug naïve patients than in patients receiving SU monotherapy prior to the study. The efficacy of combination therapy of sitagliptin with SU should be investigated in future studies, with respect to the preservation of β-cell function.
Footnotes
Acknowledgements
Members of Kyoto Lifestyle Study Group: Hajime Shirai (Aiseikai Yamashina Hospital); Yusuke Nakagawa, Kenzo Sakamoto, Masaki Tsukamoto (Akashi City Hospital); Sinsaku Matsunaga (Kyoto City Hospital); Satoshi Gojo, Naohiko Nakanishi (Kyoto Prefectural University of Medicine); Yoshihiro Matsui, Hirokazu Yokoi (Japanese Red Cross Kyoto Daiichi Hospital); Sumio Komatsu (Komatsu Heart Clinic); Morihiko Kondo, Toshiyuki Otomo, Tetsuya Tanaka, Kazuto Yamamoto (Kobe Central Hospital); Yoshifumi Nakahara (Saiseikai Shigaken Hospital); Takeshi Takenaka (Takenaka Clinic); Shigeki Kusuoka, Keizo Furukawa (Tanabe Central Hospital).
Conflict of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Funding
This study was supported by a research grant from The Waksman Foundation of Japan, Inc.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
