Abstract
Background:
Several molecular epidemiological studies have been conducted to examine the association between angiotensin-converting enzyme (ACE) I/D polymorphism and diabetic peripheral neuropathy (DPN) risk. However, the conclusions remained controversial. We therefore performed this meta-analysis.
Methods:
PubMed and EMBASE were searched to identify case-control studies investigating the associations of ACE I/D polymorphism with risk of DPN. Odds ratios (ORs) with 95% confidence intervals (CIs) were used to assess the strength of association between ACE I/D polymorphism and risk of DPN.
Results:
A total of seven case-control studies including 1316 cases and 1617 controls were included. ACE I/D polymorphism was associated with a significantly increased risk of DPN (OR=1.46, 95%CI 1.11–1.92, p=0.006, I2=48%). This result remained statistically significant when the adjusted ORs were combined (OR=1.84, 95%CI 1.14–2.99, p=0.01, I2=53%). In the subgroup of ethnicity, a significantly elevated risk was observed in Caucasians (OR=1.32, 95%CI 1.10–1.59, p=0.003, I2=43%).
Conclusions:
Our meta-analysis confirmed that ACE I/D polymorphism was associated with the risk of DPN.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is increasing in epidemic proportions worldwide and is becoming a global health challenge. It has been estimated that the global burden of diabetes will further increase to more than 550 million by 2030. 1 Diabetic peripheral neuropathy (DPN) is the most common form of diabetic neuropathy associated with the microvascular, long-term complications of T2DM. The pathogenesis of DPN has not been understood completely. There is a common view that DPN is a multifactorial disease caused by complex interactions between a variety of genetic and environmental factors.
Angiotensin-converting enzyme (ACE) is an important component of the renin–angiotensin system (RAS) that converts angiotensin (Ang) I to Ang II. It has been suggested that high levels of Ang II may play a key role in glucose and insulin regulation, and may increase the risk of diabetes. 2 In addition, Shiuchi et al. showed that Ang II type-1 receptor blockade increased insulin sensitivity and glucose uptake in skeletal muscle of diabetic mice. 3 Furthermore, ACE inhibitor (ACEI) or angiotensin receptor blocker (ARB) may decrease patients’ odds of developing new-onset T2DM. 4 Thus, high levels of ACE and Ang II might play an important role in the pathogenesis of T2DM.
The ACE gene is localized on the band 17q23 of 17th chromosome in human. 5 Within intron 16 a polymorphism exists, consisting of the presence or absence of a 287-bp fragment. 6 The ACE genotype is classified into three types: II, DD, and ID. The serum ACE levels of DD homozygotes are approximately twice as high as those of II genotype individuals. 7 The serum ACE levels of ID type are intermediate. 7 Recently, Niu et al. found that the ACE D allele was associated with an increased risk of T2DM. 8 Wang and coworkers suggested that ACE I/D polymorphism may contribute to diabetic nephropathy development. 9 In addition, Lu and colleagues showed that I/D polymorphism in the ACE gene was associated with diabetic retinopathy. 10
However, whether ACE I/D polymorphism is associated with DPN is still unclear. Li found that the ACE D allele was significantly linked with essential hypertension susceptibility. 11 Forrest et al. suggested that hypertension was an independent predictor of the incidence of DPN. 12 These results indicated that ACE I/D polymorphism may be involved in the development of DPN. Several studies have investigated this issue to date.13-19 The results, however, were inconsistent and conflicting. This present meta-analysis was performed to evaluate the association between this polymorphism and DPN.
Materials and methods
Publication search
Online electronic databases (PubMed and EMBASE) were searched using the search terms: (angiotensin-converting enzyme or ACE) and (polymorphism or variant or variation) and (diabetes mellitus or neuropathy). Additional studies were identified by a hand search from the references of original studies or review articles on this topic. There was no language restriction.
Study selection
The following inclusion criteria were used: (1) the study should have evaluated the association between the ACE I/D polymorphism and DPN risk; (2) the study should have a case-control design; (3) sufficient data should have been provided in order to calculate odds ratios (OR) and 95% confidence interval (CI), or the studies offered ORs and 95% CIs. Studies were excluded if any of the following conditions applied: (1) only abstracts or reviews were available, without sufficient data; (2) animal studies; (3) studies were repeated or publications overlapped.
Data extraction and qualitative assessment
The following data were recorded from each article: first author, year of publication, ethnicity of participants, numbers of cases and controls, and genotype numbers in cases and controls. The data were extracted by two authors independently. Discrepancies between two authors were resolved by discussion.
Two authors completed the quality assessment independently. The Newcastle–Ottawa Scale (NOS) was used to evaluate the methodological quality, which scored studies by the selection of the study groups, the comparability of the groups, and the ascertainment of the outcome of interest. We considered a study awarded 0–3, 4–6, or 7–9 as a low-, moderate-, or high-quality study, respectively. Discrepancies were resolved by consensus and discussion.
Statistical analysis
The strength of association between the ACE I/D polymorphism and DPN risk was assessed by calculating ORs with 95% CI. The pooled ORs were performed for recessive model since most of the studies reported the results in this genetic model. Departure from Hardy–Weinberg equilibrium (HWE) in controls was tested by the Chi-square test. A statistical test for heterogeneity was performed based on the Q statistic. The p>0.10 of the Q-test indicated a lack of heterogeneity among studies. If heterogeneity was observed among the studies, the random-effects model was used to estimate the pooled OR (the DerSimonian and Laird method). Otherwise, the fixed-effects model was adopted (the Mantel–Haenszel method). Stratified analysis was performed by ethnicity. To explore the source of the heterogeneity, Galbraith plots were used. To access the stability of the meta-analysis, one-way sensitivity analysis was conducted. Cumulative meta-analysis was also performed. Because no more than 10 studies were included in this meta-analysis, we did not assess the publication bias. All statistical tests were performed with the software STATA version 11.0 (Stata Corporation, College station, TX, USA). A p value <0.05 was considered statistically significant.
Results
Eligible studies
A total of seven articles on ACE I/D polymorphism and DPN risk met the study inclusion criteria, and were included in the meta-analysis.13-19 In total, 1316 DPN cases and 1617 controls were included in this meta-analysis. Six case-control studies included Caucasian populations, while only one study included an Asian population. Four studies reported the adjusted ORs and 95% CIs. All studies were assessed by NOS. The quality scores ranged from 6 to 9, suggesting that the methodological quality was acceptable. Two studies did not provided the genotype numbers of ACE I/D polymorphism among DPN cases and controls. Other studies suggested that the distribution of genotypes in the controls was consistent with HWE. The characteristics of each case-control study and the genotype in each study are presented in Tables 1 and 2.
Characteristics of the case-control studies included in meta-analysis.

PCR: polymerase chain reaction; NA: not available.
Distribution of ACE I/D polymorphism among patients and controls.
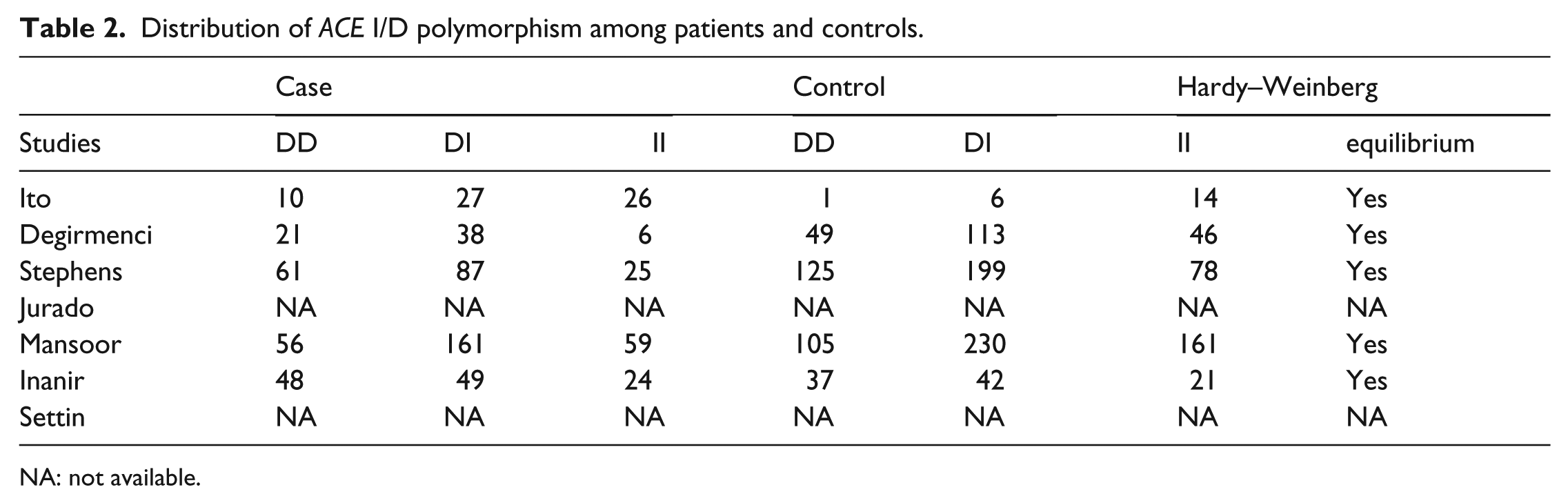
NA: not available.
Meta-analysis
The DD + DI genotype of ACE I/D polymorphism was associated with a significantly increased risk of DPN when compared with the II genotype (OR=1.46, 95%CI 1.11–1.92, p=0.006, I2=48%; Figure 1). When stratified by ethnicity, the significantly elevated risk was observed in Caucasians (OR=1.32, 95%CI 1.10–1.59, p=0.003, I2=43%). Four studies reported adjusted ORs. The combination of adjusted ORs for DPN was 1.84 (95%CI 1.14–2.99, p=0.01, I2=53%).
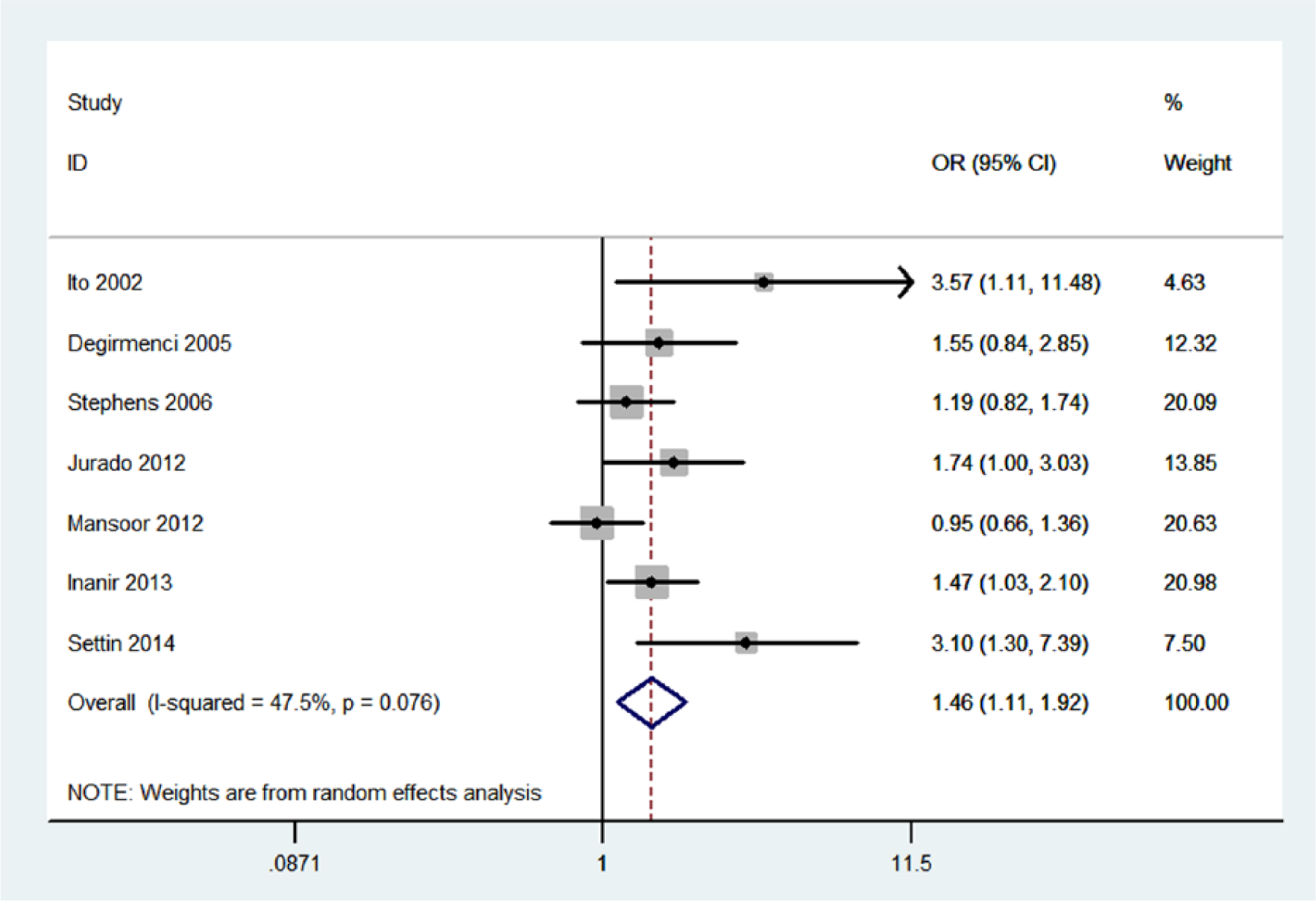
Meta-analysis for the association between DPN risk and the ACE I/D polymorphism.
As shown in Figure 2, significant associations were evident with each addition of more data over time. The results showed that the pooled ORs tended to be stable. A single study involved in the meta-analysis was deleted each time to reflect the influence of the individual data set to the pooled ORs, and the corresponding pooled ORs were not materially altered (Figure 3).

Cumulative meta-analysis for the association between DPN risk and the ACE I/D polymorphism.

Sensitivity analysis of the summary odds ratio coefficients on the association for the ACE I/D polymorphism with DPN risk.
The Galbraith plot was used to find the source of the heterogeneity. As shown in Figure 4, two studies might be the source of the heterogeneity. After excluding these studies, the between-study heterogeneity effectively decreased and there was no obvious heterogeneity (I2=0%, p=0.70). Besides, the result was still statistically significant (OR=1.42; 95%CI, 1.14–1.76; p=0.002).
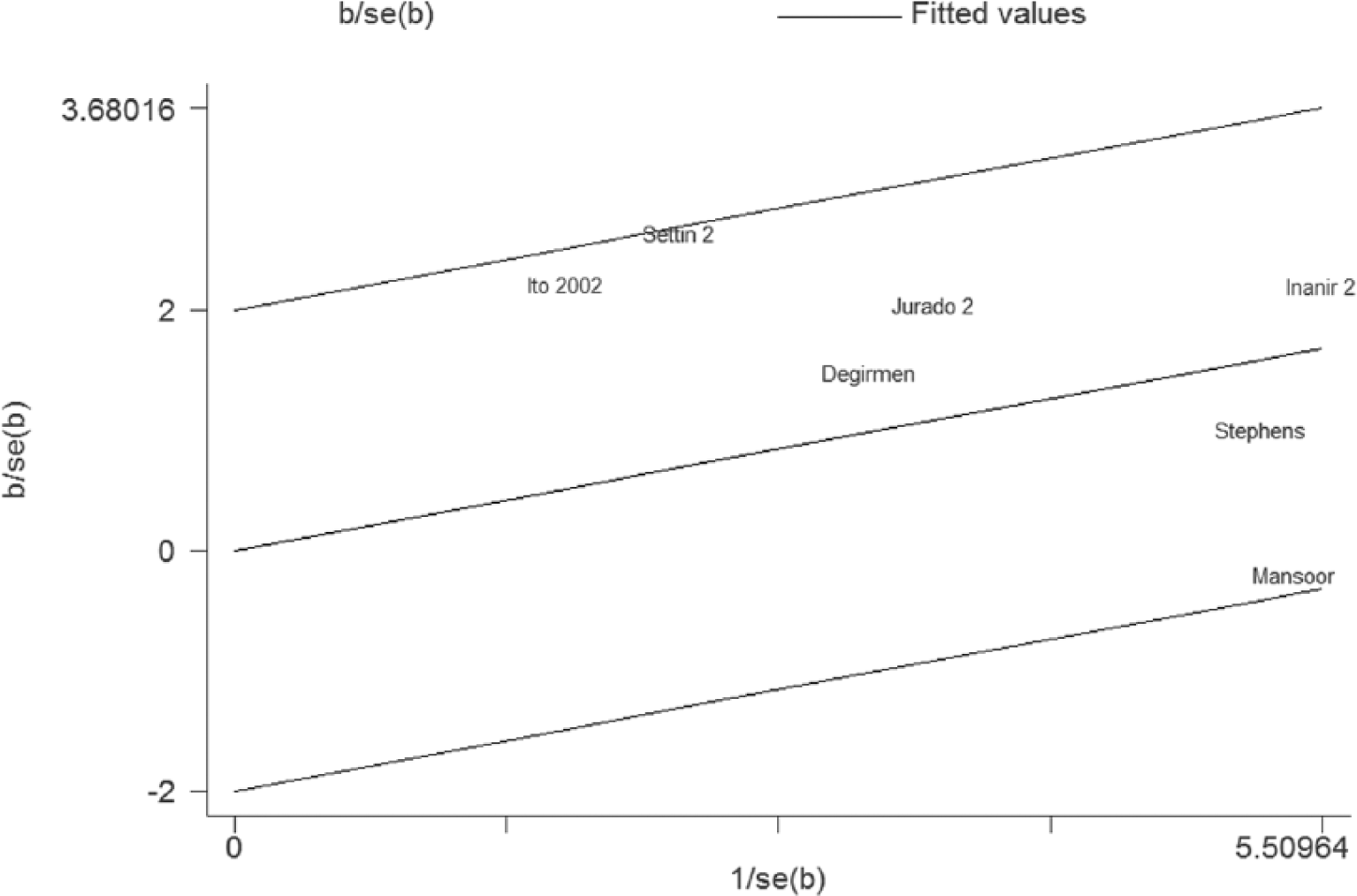
Galbraith plot of DPN risk and the ACE I/D polymorphism.
Discussion
This meta-analysis of seven case-control studies systematically evaluated the association between ACE I/D polymorphism and DPN risk. We found that ACE DD and DI genotypes were significantly associated with DPN risk. This result suggested that individuals with the DD or DI genotype had an increased DPN risk. In the subgroup analysis, we found that Caucasians with the DD or DI genotype had an increased DPN risk. However, only one study investigated the association between ACE I/D polymorphism and DPN risk in Asians. Thus, more studies are still needed to determine the association between ACE I/D polymorphism and DPN risk in Asians.
ACE catalyzes the generation of Ang II from Ang I as well as the cleavage of bradykinin. Ang II is a powerful circulating vasoconstrictor, which may contribute to vascular tone in nerve and muscle circulatory beds. It is clear that nerve blood flow deficits make an important contribution to the pathogenesis of diabetic neuropathy. 20 ACEI has been shown to protect against the loss of intraepidemal nerve fibers in a T2DM rat model. 21 In terms of clinical trials, ACEI produced some modest improvements in DPN. 22 In addition, a lack of bradykinin receptors exacerbated diabetic complications, including DPN. 23 These results suggested that high levels of ACE may play a critical role in the development of DPN. Since ACE I/D polymorphism was associated with plasma levels of ACE, it was possible that this polymorphism might influence DPN risk.
Heterogeneity is a potential problem that may affect the interpretation of the results. We found that significant heterogeneity existed in this meta-analysis. A Galbraith plot was used to find the sources of heterogeneity. We found that I2 value was decreased after excluding two studies. The result suggested that these two studies may be the major source of the heterogeneity. Moreover, heterogeneity did not influence the results, because the significance of the result was not altered after excluding the studies. Results from one-way sensitivity analysis suggested the stability of the result. In addition, the main result remained statistically significant when the adjusted ORs were combined. All together, these results suggest that the results of this meta-analysis are reliable.
However, some limitations should be noted. First, subgroup analyses were not performed by factors such as gender and age because insufficient data could be extracted from the primary articles. Second, there was only one case-control study that investigated the association of ACE I/D polymorphism with DPN risk in Asians. Therefore, more studies with large sample sizes are needed to further identify the association among Asians. Third, because of the complex nature of DPN, it is unlikely that a polymorphism in one single gene would be obviously associated with DPN risk. However, insufficient data could be extracted from the primary studies. Therefore, we did not perform the analysis of other polymorphisms in ACE and a haplotype analysis.
In conclusion, this meta-analysis suggested that ACE I/D polymorphism was associated with an increased DPN risk. Further studies with large sample sizes are needed to confirm our findings.
Footnotes
Conflict of interest
The authors declare that they have no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
