Abstract
Objective:
Many studies have focused on the relationship between the angiotensin-converting enzyme gene (ACE) insertion/deletion (I/D) polymorphism and pancreatitis risk, but the results remain inconsistent. Thus, a meta-analysis was carried out to derive a more precise estimation of the association between ACE I/D polymorphism and pancreatitis risk.
Methods:
Relevant publications were searched in several widely used databases and seven studies from six eligible articles were included in the meta-analysis. Pooled odds ratios (ORs) and 95% confidence intervals (CIs) were calculated to evaluate the strength of the association between ACE I/D polymorphism and pancreatitis risk.
Results:
Significant associations between ACE I/D polymorphism and pancreatitis risk were observed in both overall meta-analysis for DI versus II (OR=0.80, 95% CI=0.67–0.96) and DD + DI versus II (OR=0.83, 95% CI=0.70–0.98), and acute pancreatitis subgroup for DI versus II (OR=0.65, 95% CI=0.44–0.95). However, no significant pancreatitis risk variation was detected for all genetic models in the subgroup meta-analysis based on ethnicity.
Conclusions:
According to the results of our meta-analysis, the ACE I/D polymorphism probably associates with pancreatitis risk, especially acute pancreatitis risk, with the I allele acting as a risk factor.
Introduction
The renin-angiotensin system (RAS) is essential for the regulation of blood pressure and serum electrolyte balance.1,2 Besides the circulating RAS, a local RAS in many tissues has been reported.3–5 Such a RAS functions to meet tissue specific needs 6 and is involved in the disease development of relevant tissues.7–10 The angiotensin-converting enzyme gene (ACE) encodes a key enzyme in the RAS, which converts angiotensin I to the potent vasoconstrictor angiotensin II.1,2,11 An insertion/deletion (I/D) polymorphism has been reported to be located in intron 16 of the ACE, and the angiotensin-converting enzyme (ACE) level is higher in subjects with D allele than those with I allele. 12
In this study, we investigated the association between ACE I/D polymorphism and pancreatitis risk. Pancreatitis is a complex disorder with both acute and chronic forms. Chronic pancreatitis is a chronic inflammatory disease which causes pancreatic fibrosis and destroys both of the exocrine and endocrine pancreas. 13 Acute pancreatitis is an acute inflammatory disease which is started by pancreatic injury and leads to autodigestion.14,15 An intrinsic RAS exists in the pancreas 5 and is associated with acute pancreatitis and chronic hypoxia in experimental animals.16–18 In the past decade, several studies have focused on the relationship between ACE I/D polymorphism and pancreatitis risk, but the results of those individual studies provided limited information and could not draw a convincing conclusion.19–24 Therefore, we performed a meta-analysis with a relatively large sample size of seven studies from six eligible articles (1335 cases and 1936 controls in all) to provide a more reliable conclusion about the relationship between ACE I/D polymorphism and pancreatitis risk.
Materials and methods
Literature search, selection, and data collection
In this study, we searched papers published before 26 December 2013 according to the keywords “angiotensin-converting enzyme”/”angiotensin I converting enzyme”/”ACE”, “pancreatitis”, and “polymorphism”/”polymorphisms”/”variation”/”variations”/ “genotype” in PubMed, Web of Science, EMBASE, and Google Scholar independently. The papers obtained were further selected for the meta-analysis and our selection criteria were: (a) full text English-written study; (b) study providing complete case and control data about the relationship between ACE I/D polymorphism and pancreatitis risk; (c) studies sharing the same sample of cases and controls were compared and the most complete study from them was included in our meta-analysis; (d) study investigated two or more populations was treated as two or more independent studies in this meta-analysis; (e) studies with control group genotypes in Hardy-Weinberg equilibrium (HWE). HWE was tested by χ2 test, and when χ2 test reported a p value of more than 0.05, the control group genotypes were consistent with HWE.
In this study, two investigators independently collected data from each eligible paper. The data were composed of first author, published year, country of origin, ethnicity, pancreatitis type, and numbers of cases and controls. Through checking between the two investigators, a final set of data was determined.
Meta-analysis methods
According to the data collected from each eligible paper, we performed both the overall meta-analysis and the subgroup meta-analysis based on ethnicity and pancreatitis type, to evaluate the relationship between ACE I/D polymorphism and pancreatitis risk. In the overall as well as the subgroup meta-analysis, pooled odds ratios (ORs) and 95% confidence intervals (CIs) for dominant, recessive, and codominant genetic models were all calculated by fixed effects model or random effects model. The model chosen was based on the heterogeneity test. For the heterogeneity test, we performed the χ2-based Q-test in this study. 25 When the Q-test reported a p value of more than 0.10, the fixed effects model was used to calculate the pooled ORs, 26 otherwise the random effects model was used. 27
Publication bias was also tested using Begg’s funnel plot and Egger’s test. 28 If the funnel plot was asymmetric and Egger’s test reported a p value of less than 0.05, a publication bias probably exists.
In this study, we used the software Stata version 12.0 (Stata Corporation, College Station, Texas, USA) to carry out the meta-analysis.
Results
Studies and data included in this meta-analysis
Through searching and selection, a final list of seven studies from six eligible articles 19–24 was collected for meta-analysis (see Figure 1). All seven studies collected were case-control studies with various ethnicities (one study of Asians, three studies of Caucasians, one study of Turkish, and two studies of unknown ethnicity), and pancreatitis types (two studies of acute pancreatitis, four studies of chronic pancreatitis, and one study of both acute and chronic pancreatitis). The control groups of the seven eligible studies were all in Hardy-Weinberg equilibrium (p>0.05). Information from these seven studies and the numbers of cases and controls with different genotypes reported in each study is presented in Table 1. In total, the seven eligible studies provided 1335 cases and 1936 controls about the relationship between ACE I/D polymorphism and pancreatitis risk.

Flow chart of study selection.
Studies and data included in this meta-analysis.

D: deletion; I: insertion; NA: not available.
p value for Hardy-Weinberg equilibrium (HWE) test in each control group.
The article Hucl et al., 2009 investigated two populations (German and Finnish) respectively and was treated as two independent studies in this meta-analysis. In the German population, this article further investigated both chronic and acute pancreatitis types and therefore was treated as two independent studies in the subgroup analysis based on pancreatitis type.
Overall and subgroup meta-analysis results
In this study we performed both the overall meta-analysis and the subgroup meta-analysis based on ethnicity and pancreatitis type. The detailed results of our meta-analysis are shown in Table 2. The results of the overall meta-analysis provided evidence of the association between ACE I/D polymorphism and pancreatitis risk (OR=0.80, 95% CI=0.67–0.96 for DI versus II; OR=0.83, 95% CI=0.70–0.98 for DD + DI versus II, see Table 2, Figures 2 and 3). The subgroup meta-analysis based on pancreatitis type further showed that ACE I/D polymorphism was significantly associated with acute pancreatitis risk (OR=0.65, 95% CI=0.44–0.95 for DI versus II, see Table 2 and Figure 4), while no such significant association was detected in the chronic pancreatitis subgroup (see Table 2). In the stratified meta-analysis based on ethnicity, no obvious association existed in either Asians or Caucasians (see Table 2). In summary, according to the results of our meta-analysis, the ACE I/D polymorphism probably associates with pancreatitis risk, especially acute pancreatitis risk, with the I allele acting as a risk factor.
Detailed results of the meta-analysis.

CI: confidence interval; D: deletion; I: insertion; OR: odds ratio.
p value for heterogeneity test. If p>0.1, ORs were calculated using fixed effects model, otherwise the random effects model was used.
ORs calculated using random effects model.
Results which are statistically significant.
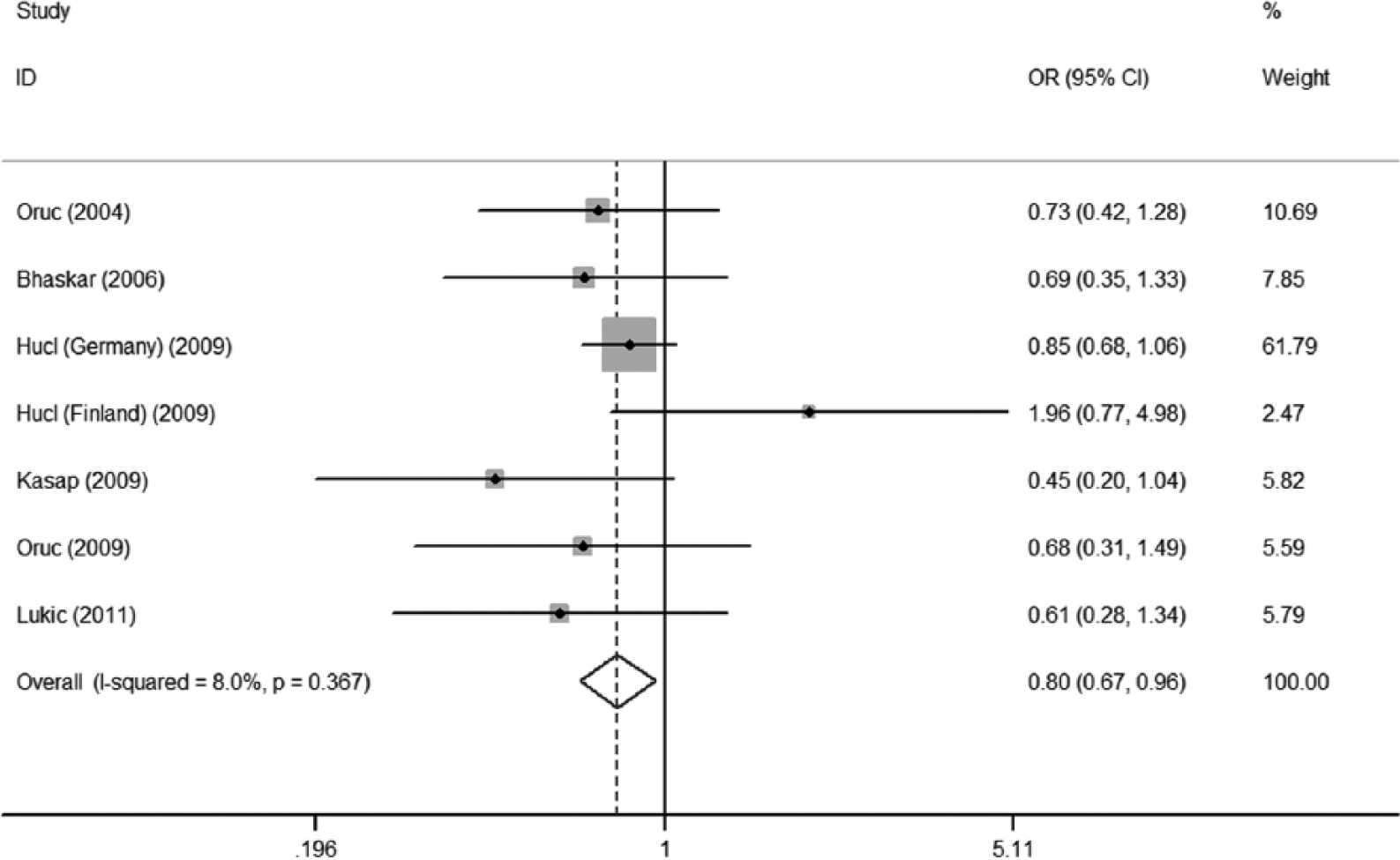
Forest plot for DI versus II of the overall meta-analysis using fixed-effects model.

Forest plot for DD+DI versus II of the overall meta-analysis using fixed-effects model.

Forest plot for DI versus II of the acute pancreatitis subgroup using fixed-effects model.
Publication bias test results
The results of Begg’s funnel plot (see Figure 5) and Egger’s test showed no publication bias for DD versus II (p=0.708), for DI versus II (p=0.627), for DD + DI versus II (p=0.623), and for DD versus DI + II (p=0.869) in the overall meta-analysis.

Begg’s funnel plots for angiotensin-converting enzyme gene (ACE) insertion/deletion (I/D) polymorphism and pancreatitis risk ((a): DD versus II, (b): DI versus II, (c): DD+DI versus II, (d): DD versus DI+II); logor: logarithm of odds ratios; s.e.: standard error.
Discussion
In this study, the results of our overall meta-analysis and subgroup meta-analysis based on pancreatitis type suggest that the ACE I/D polymorphism probably associates with pancreatitis risk, especially acute pancreatitis risk, with the I allele acting as a risk factor. It is reported that a local RAS exists in the pancreas, 5 which plays an important role in the maintaining of pancreatic homeostasis. 29 Increase of ACE (the key enzyme in RAS) activity in chronic hypoxia and acute pancreatitis has been detected in experimental animals. 17 The ACE I/D polymorphism accounts for 47% of the variance of serum ACE concentration, 12 hence it may not function independently in pancreatitis development. Instead, combined effects of this polymorphism with other variants involved in the modulation of ACE levels and environmental factors probably exist. Further research on the exact molecular mechanism is required in the future.
In the stratified meta-analysis based on ethnicity, however, no significant association was detected in all the subgroups. This result is not that convincing because of the insufficient data available at present for each subgroup, and the exact roles of ACE I/D polymorphism in different ethnicities will require further research.
In addition, all the results of our meta-analysis should be considered prudently due to the existence of several limitations. One limitation is the insufficient sample size used in our meta-analysis, especially in the subgroup analysis based on ethnicity and pancreatitis type. A second limitation is the lack of case-control data adjustment according to detailed individual information such as age, sex, and lifestyle in our meta-analysis. The third limitation is that the exact molecular basis of the association between the ACE I/D polymorphism and pancreatitis risk is still not clear enough at present and needs further investigation. Hence, in order to achieve a more convincing conclusion, further analysis using larger sample size and adjusted individual data is required, and further experimental research on molecular mechanism should also be performed.
In conclusion, supported by a meta-analysis with a total of seven studies from six eligible articles (1335 cases and 1936 controls in all), our study indicates that the ACE I/D polymorphism probably associates with pancreatitis risk, especially acute pancreatitis risk, with the I allele acting as a risk factor. Although there are some limitations, our meta-analysis still provides valuable information for studying the relationship between the ACE I/D polymorphism and pancreatitis risk.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by grants from key medical subjects of Jiangsu province (grant number XK201120); innovative team of Jiangsu Province (grant number LJ201114); special clinical medical science and technology of Jiangsu province (grant numbers BL2012050, BL2013014); key laboratory of Suzhou (grant numbers SZS201108, SZS201307); and National Natural Science Foundation (grant numbers 81100371, 81370627, 81300423, 81272143).
