Abstract
Introduction:
Hypokalemia in primary aldosteronism (PA) patients correlates with higher levels of cardiovascular events and altered left ventricular geometry. However, the influence of aldosterone on microvascular endothelial function and the effect of hypokalemia on the vascular structure still remain unclear.
Objectives:
We investigated the peripheral arterial functions, including the endothelial function of microvasculature and arterial stiffness in PA and essential hypertension (EH) patients, and the correlation between hypokalemia and peripheral arterial function among PA patients.
Methods:
Twenty patients diagnosed as EH and 37 patients with PA were enrolled in this study. Reactive hyperemia index (RHI) and the augmentation index (AI) were obtained by non-invasive peripheral arterial tonometry.
Results:
Twenty EH patients and a total of 37 PA patients, including 21 patients with normokalemia and 16 patients with hypokalemia, were enrolled and divided into groups 1, 2 and 3 respectively. PA patients had significantly higher AI (p=0.024) but not RHI than EH patients. RHI showed no difference between groups 1, 2 and 3. Group 3 had higher AI than either group 1 or group 2. In the whole study population, serum potassium level, after multivariate regression analysis testing, was the only factor associated with AI (ß= −0.102; p=0.002). In PA patients, serum potassium level was the only significant factor correlated with AI. (r= −0.458; p=0.004)
Conclusions:
PA patients had higher arterial stiffness but comparable microvascular endothelial function to EH patients. Hypokalemia correlated with arterial stiffness but not microvascular endothelial function in PA patients.
Keywords
Introduction
Primary aldosteronism (PA), one of the most frequent causes of secondary hypertension, is caused by over-secretion of aldosterone. It has attracted increased attention in recent studies because its prevalence among secondary hypertension subjects is greater than previous estimates, about 5–13% of hypertensive patients of any causes. 1 It has been suggested by several clinical studies that aldosterone itself could cause cardiovascular injuries by mechanisms other than sodium-water retention or hypertensive effects.2–4 Some investigations suggest that aldosterone is associated with arterial stiffness and endothelial dysfunction, which may lead to atherosclerosis or other cardiovascular complications.5–8 In our previous study, we demonstrated that PA patients have a higher degree of early atherosclerosis and vascular stiffness, and adrenalectomy reverses adverse vascular changes in PA patients. 8
Hypokalemia plays an important role in various cardiovascular diseases. 9 In a recent study, PA patients with hypokalemia had a higher cardiovascular morbidity than PA patients with normokalemia. 10 This implies that, in PA patients, hypokalemia plays a significant role in cardiovascular comorbidities. In our recent study, serum potassium level was significantly associated with left ventricular mass in PA patients. 11 However, the influence of hypokalemia on vascular function and stiffness remains unclear.
Flow-mediated dilation (FMD) is a common non-invasive method to measure endothelial function. 12 However, FMD is limited by its high operator dependence and wide difference between operators. 13 Pulse amplitude tonometry (PAT), a recent United States Food and Drug Administration (FDA)-approved technology, has become a good alternative choice to measure endothelial function without subjective measurements of FMD.12,14,15 PAT presents peripheral endothelial function as the reactive hyperemia index (RHI); furthermore, arterial stiffness can be also calculated from PAT waveform as the augmentation index (AI). PAT measures endothelial functions of microvasculature. In contrast, FMD measures the endothelial function of median size vessels. The associations between FMD and RHI are low, even displaying no association.16,17 Therefore, it is also of interest to study whether aldosterone influences the endothelial function of the microvasculature.
In this study, we investigate the differences in microvascular endothelial function and arterial stiffness between PA and essential hypertension (EH) patients, and the correlation between serum potassium level and peripheral arterial function among PA patients.
Materials and methods
Patients
This prospective study enrolled 37 patients diagnosed with primary aldosteronism after an initial evaluation and who were recorded in the Taiwan Primary Aldosteronism Investigation (TAIPAI) database from October 2006–October 2011. The database was constructed for quality assurance at one medical center (National Taiwan University Hospital, Taipei, Taiwan) one branch hospital (National Taiwan University Hospital Yun-Lin branch, Yun-Lin, southern Taiwan), and two cooperative hospitals (Far-Eastern Memorial Hospital, Taipei; Tao-Yuan General Hospital, Tao-Yuan, central Taiwan).8,11,18–25 The serum biochemistry studies (including potassium levels) were measured at the first evaluation of these patients at the National Taiwan University Hospital. Medical histories, including demographics and medication were carefully recorded. Patients with a hypokalemia history were defined as those who had known hypokalemia (K<3.5 mg/dl). In addition, another 20 patients with EH were enrolled as the control group. The diagnosis of EH was made by exclusion through appropriate clinical and biochemical investigations of all detectable forms of secondary hypertension, including having a post-captopril test of aldosterone-to-renin activity (ARR)<30. 26 This study was approved by the Institutional Review Board of National Taiwan University Hospital, Taipei, Taiwan, and all patients gave informed consent.
Diagnostic criteria of subtypes of PA
The diagnosis of aldosterone-producing adenoma (APA) was validated by the modified four corner criteria used after adrenalectomy as in our previously reports:24,27,28 (a) evidence of autonomous excess aldosterone production based on ARR>35 or urine ≥12 μg/24 h and TAIPAI score more than 60% 29 and post-saline loading plasma aldosterone concentration (PAC)>10 ng/dl; (b) lateralization of aldosterone secretion at adrenal vein sampling (AVS) or during dexamethasone suppression NP-59 single-photon emission computed tomography (SPECT)/computed tomography (CT); 30 (c) evidence of adenoma at CT scan; and (d) pathologically proven adenoma after an adrenalectomy if operated, and cure of hypertension without anti-hypertensive agents or improved hypertension, potassium, PAC, and plasma renin activity (PRA) as described.
Idiopathic hyperaldosteronism (IHA) was classified by the following criteria: (a) evidence of autonomous excess aldosterone production based on ARR>35 and TAIPAI score more than 60%; or urine ≥12 μg/24 h and post-saline loading PAC >10 ng/dl (b) non-lateralization of aldosterone secretion at AVS or during dexamethasone suppression NP-59 SPECT/CT; 30 (c) evidence of bilateral diffuse enlargement on CT scan; and/or (d) evidence of diffuse cell hyperplasia in the pathology studies.
Assessment of peripheral vascular function by PAT
Digital pulse amplitude was measured with a non-invasive PAT placed on the tip of both index fingers (Endo-PAT2000, Itamar Medical, Caesarea, Israel). The PAT generates a pneumatic plethysmo-graph that applies uniform pressure to the finger surface, which allows measurement of changes in pulse volume. The inflation pressure of PAT was set to either 10 mmHg below diastolic blood pressure (DBP) or 70 mmHg. The process initially took a two-minute measurement of baseline pulse amplitude. And then, the arterial flow was occluded for 5 min by a cuff placed on a proximal forearm with occlusion pressure higher than systolic blood pressure (SBP). Data was collected from both index fingers and analyzed by a computerized automated algorithm (Itamar Medical) that provided the average pulse amplitude for each 30 s interval after forearm cuff deflation up to 4 min.
The RHI was calculated as the ratio of the average amplitude of the PAT signal over a one-minute interval beginning one minute after cuff deflation divided by the baseline amplitude. Moreover, the AI was calculated by analyzing the waveform of the PAT signal divided by baseline and presented as percentage ratio. After natural logarithmic transformation, endothelial dysfunction was defined by ln (PAT ratio) <0.30. 31
Statistical analysis
Data was expressed as mean±standard deviation (SD). Student’s t-test was used to compare the continuous variable between two groups. One-way analysis of variance (ANOVA) test with post-hoc analysis by LSD method (Fisher’s least significant difference method) was used to compare continuous data of the three groups.To analyze categorical data, either the Chi-square or the Fisher’s exact test was used. The Pearson’s correlation test was used to analyze the association between AI and its determinants. Data of PAC, PRA, and ARR were log-transformed due to non-normality which was tested by the Kolmogorov-Smirnov test. The clinical factors showing significant Pearson’s correlation (p<0.05) with AI were further analyzed by multivariate regression test. A value of p<0.05 was considered to indicate statistical significance.
Results
Patients
Twenty EH patients (group 1) and 37 PA patients were enrolled in this study. PA patients were divided in two groups: 21 with normokalemia (group 2) and 16 with hypokalemia (group 3). The clinical data is shown in Table 1. Group 3 patients showed significant increases in both SBP and DBP, and a longer hypertensive history than group 1. Moreover, they also showed significant differences in PAC, PRA, log-transformed PAC and log-transformed PRA than group 1. Group 3 patients also had a higher SBP, DBP, PAC, log-transformed PAC, and longer hypertensive history than group 2.
Clinical data of the patients.
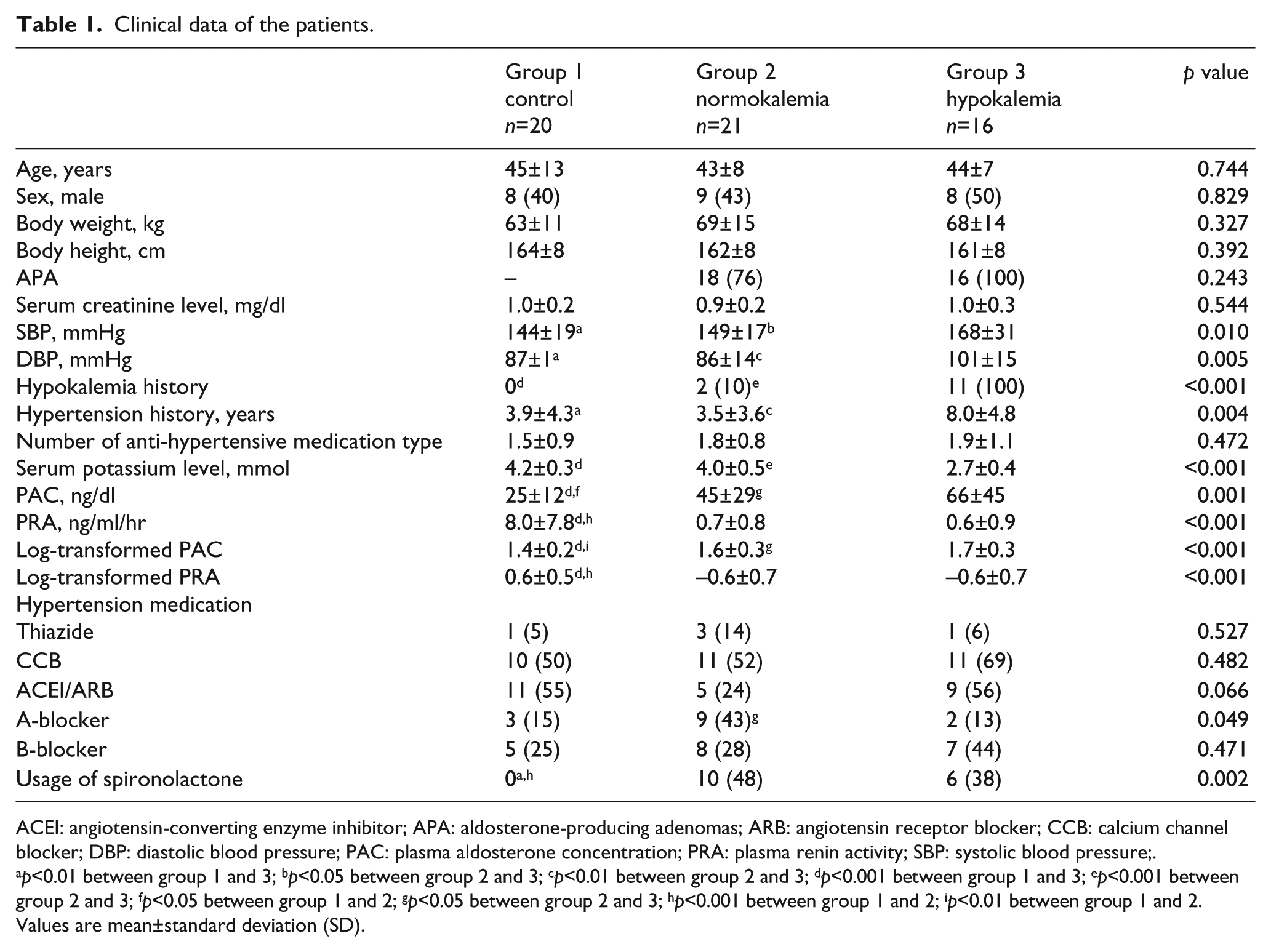
ACEI: angiotensin-converting enzyme inhibitor; APA: aldosterone-producing adenomas; ARB: angiotensin receptor blocker; CCB: calcium channel blocker; DBP: diastolic blood pressure; PAC: plasma aldosterone concentration; PRA: plasma renin activity; SBP: systolic blood pressure;.
p<0.01 between group 1 and 3; bp<0.05 between group 2 and 3; cp<0.01 between group 2 and 3; dp<0.001 between group 1 and 3; ep<0.001 between group 2 and 3; fp<0.05 between group 1 and 2; gp<0.05 between group 2 and 3; hp<0.001 between group 1 and 2; ip<0.01 between group 1 and 2.
Values are mean±standard deviation (SD).
PAT results
First, RHI and AI were tested and compared between EH and PA patients. The results showed that 37 PA and 20 EH patients had a significant difference in AI (0.298±0.206 vs 0.195±0.128; p=0.024) but not in RHI (2.209± 0.564 vs 2.106±0.445; p=0.484). Next, RHI and AI among subgroups of PA patients (normokalemia and hypokalemia) were further compared with the EH group. The results are shown in Figure 1. RHI again showed no differences between groups 1, 2 and 3 (2.0±0.7; 1.9±0.7; 2.2±0.9 in groups 1, 2, 3; p=0.498 by ANOVA). In AI analysis, group 3 had higher AI than either group 1 or group 2 (0.20±0.13; 0.22±0.14; 0.40±0.23 in group 1,2,3; p=0.001 by ANOVA; p<0.001 between groups 1, 3; p=0.001 between groups 2, 3).

Comparisons between groups for reactive hyperemia index and augmentation index. (a) Endo-pulse amplitude tonometry (PAT) results of patients, the reactive hyperemia index showed no difference between groups 1, 2 and 3; (b) group 3 patients had a higher augmentation index than either group 1 or group 2.
Factors associated with AI
Table 2 contains Pearson’s correlation coefficients between clinical factors and AI in all 57 patients. The coefficients showed significance in body height, serum potassium level, DBP, hypertension duration, and presence of PA. In multivariate regression analysis of AI in all patients, serum potassium level was the only factor associating with AI (ß= −0.102; p=0.002; Table 3).
Factors associated with augmentation index (AI) (all patients, n=57).

DBP: diastolic blood pressure; MBP: mean blood pressure; PAC: plasma aldosterone concentration; PRA: plasma renin activity; SBP: systolic blood pressure.
Multivariate regression analysis with augmentation index (AI) as the dependent variable (all patients, n=57, model adjusted R2=0.170).

CI: confidence interval.
Excluded parameters: diastolic blood pressure, hypertensive years, body height, presence of primary aldosteronism, and log-transformed plasma renin activity.
Furthermore, we tested the same clinical factors in 37 PA patients with AI (Table 4). Serum potassium level was the only factor showing a significant Pearson’s correlation coefficient with AI (r= −0.458; p=0.004).
Factors associated with augmentation index (AI) (primary aldosteronism (PA) patients, n=37).

APA: aldosterone-producing adenoma; DBP: diastolic blood pressure; MBP: mean blood pressure; PAC: plasma aldosterone concentration; PRA: plasma renin activity; SBP: systolic blood pressure.
Discussion
The major findings of this study are as follows: (a) PA patients have higher AI than EH patients; (b) hypokalemic PA patients have higher AI than either normokalemic PA or EH patients; (c) serum potassium level is associated with arterial stiffness; (d) no significant difference in RHI was found among 3 groups.
Serum aldosterone level has been recognized as a factor associated with arterial stiffness, which is an independent risk factor in cardiovascular morbidity and mortality in hypertensive patients.32,33 Previous studies have provided implications suggesting this correlation by showing a reduction in AI in hypertensive patients treated with aldosterone antagonists.33,34 Furthermore, the results of our previous studies not only supported this association in PA patients who received adrenalectomy, but also further suggested that a reduction in serum aldosterone in PA patients can improve myocardial fibrosis and carotid intima-media thickness.35,36 In this study, we provide further evidence that arterial stiffness in PA patients is still significantly higher than in EH patients by means of the non-invasive PAT method.
Hypokalemia is associated with altered cardiac structure in PA patients, such as left ventricular hypertrophy.26,37 A recent study revealed that cardiovascular comorbidities, including angina pectoris and chronic cardiac insufficiency, are more prevalent in hypokalemic PA patients. 10 However, the effects of hypokalemia on peripheral blood vessels remain unclear. In our present study, serum potassium level is the only factor significantly associated with AI in PA patients. According to our best knowledge, this is the first study to demonstrate the relationship between arterial stiffness and hypokalemia in PA patients. Two possible mechanisms are the alternation of cytosolic calcium cycling and alternation of signal transduction through Na/K-ATPase. 38 In a cell culture system, reduction in extracellular potassium produces rapid and sustained increases of cytosolic calcium concentration and a hypertrophic response of myocytes in a dose dependent manner. 38 In addition, hypokalemia potentiates cardiac hypertrophy and decreases tissue Na/K-ATPase activity rates after aortic constriction surgery. 38 However, further studies are necessary to investigate the detailed mechanisms.
Recent studies have reported significant endothelial dysfunction observed in patients with high aldosterone level by measuring FMD.5,6,39 However, differences in endothelial dysfunction among EH or PA groups were insignificant in our study which measured RHI. In the Framingham Heart Study, Hamburg et al. have demonstrated that measuring brachial and digital vascular function by FMD and RHI respectively that there were different associations with cardiovascular risk factors and, interestingly, they were almost uncorrelated with each other. 16 In addition, Lee et al. reported no relationship between the reactive hyperemia-evoked digital PAT ratio and brachial FMD. 17 Furthermore, Schnabel et al. showed only modest correlations between conduit artery and peripheral arterial functions and also implied that they reflected different pathologies due to their different relationship to cardiovascular risk factors. 40 Therefore, the negative RHI results among the three groups in our study might imply an insignificant difference in endothelial functions in micro-vasculature.
Limitations of the study
This study is limited by the nature of a cross-section study, that is, it only shows the correlation between hypokalemia and AI. Whether hypokalemia is a factor influencing AI or only a marker of more severe PA is not clearly defined in this study setting. In addition, this study is also limited by the number of patients, and may be underpowered to detect the other possible factors which may be also associated with AI. A further study with a larger study population is required.
Conclusion
PA patients had higher arterial stiffness than, but comparable microvascular endothelial function to, EH patients. Hypokalemia correlated with arterial stiffness but not microvascular endothelial functions in PA patients.
Footnotes
Acknowledgements
The authors would like to thank the staff of the Second Core Lab of Department of Medical Research in National Taiwan University Hospital for technical assistance. The Taiwan Primary Aldosteronism Investigation (TAIPAI) Study Group includes: Vin-Cent Wu, Yen-Hung Lin, Yi-Luwn Ho, Hung-Wei Chang, Lian-Yu Lin, Fu-Chang Hu, Kao-Lang Liu, Shuo-Meng Wang, Kuo-How Huang, Yung-Ming Chen, Chin-Chi Kuo, Shih-Chieh Chueh, Ching-Chu Lu, Fang-Chi Chang, Shih-Cheng Liao, Ruoh-Fang Yen, Wei-Chou Lin, Bor-Sen Hsieh, and Kwan-Dun Wu.
Conflicts of interest
None declared.
Funding
This study was supported by the Department of Health, Executive Yuan, R.O.C. (DOH 100-PTH10002, DOH 101-HO-1013, DOH 102-PTH10204), National Taiwan University Hospital (NTUH 101-001974), National Taiwan University (National Taiwan University Cutting-Edge Steering Research 10R71608-1), NTU-NTUH MediaTek Innovative Medical Electronics Research Center (PC851), Taiwan National Science Council (NSC 100-2314-B-002-139, NSC 102-2314-B-002-078-MY3), and support from the Taiwan National Science Council for the Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan (NSC 101-2911-I-008-001).
