Abstract
Introduction:
The purpose of this study was to examine whether the insertion (I) and deletion (D) polymorphism of the angiotensin-converting enzyme (ACE) confers susceptibility to schizophrenia and Parkinson’s disease (PD).
Materials and methods:
A meta-analysis was performed of the associations between the ACE I/D polymorphism and schizophrenia and PD.
Results:
Thirteen studies with 2024 cases and 2230 controls comprising eight studies on schizophrenia and five on PD were included in the meta-analysis. The meta-analysis revealed no association between the ACE D allele and schizophrenia (OR = 0.990, 95% CI = 0.889–1.102, p = 0.856) or PD (OR = 1.067, 95% CI = 0.907–1.255, p = 0.433). Stratification by ethnicity indicated no association between the ACE D allele and schizophrenia in European, Asian, or Turkish ethnic groups (OR = 0.896, 95% CI = 0.566–1.419, p = 0.640; OR = 1.057, 95% CI = 0.903–1.238, p = 0.492; OR = 1.111, 95% CI = 0.889–1.389, p = 0.354, respectively). Ethnicity-specific meta-analysis was not conducted for PD because only one ethnic PD study was available.
Conclusions:
This meta-analysis found no association between the ACE I/D polymorphism and schizophrenia or PD.
Introduction
Schizophrenia is a chronic relapsing psychic disorder characterized by the breakdown of thought processes and poor emotional responsiveness. Parkinson’s disease (PD) is a degenerative disorder of the central nervous system. The motor symptoms of PD result from the death of dopamine-generating cells in the substantia nigra region of the midbrain. Although the etiologies of schizophrenia and PD are not yet fully understood, both genetic and environmental factors and interactions between these factors are thought to contribute to these diseases.1,2
Angiotensin-converting enzyme (ACE) converts angiotensin I into angiotensin II and inactivates bradykinin via the kallikrein-kininogen system. 3 Angiotensin II is the main effector molecule of the renin-angiotensin system (RAS), is pleiotropic, and is a mediator of the development and progression of diseases. 4 Furthermore, angiotensin II is a multifunctional neuropeptide in the brain. ACE plays an important role in regulating the growth and differentiation of neuronal cells. 5 High concentrations of ACE have been observed in the nigrostrial pathway and basal ganglia and cerebrospinal fluid (CSF) of patients with psychiatric disorders.6,7 ACE hydrolysis of substance P in the brain and the primary loss of substance P have been associated with the pathogenesis of schizophrenia and PD.8,9 The ACE gene, which is located on chromosome 17q23, contains an insertion/deletion (I)/(D) polymorphism within intron 16 that involves the presence or absence of a 287-bp repeat sequence. 10 The DD genotype is associated with two-fold higher tissue and plasma concentrations of ACE than the II genotype. 10 Thus, it seems possible that the D allele could play a role in the pathogenesis of schizophrenia and PD.
The ACE I/D polymorphism has been studied in the context of schizophrenia and PD.11–23 However, the genetic associations of the ACE I/D polymorphism with schizophrenia and PD are unclear because inconclusive or contrasting results have been reported by different studies. This may be because of small sample sizes, low statistical power, and/or clinical heterogeneity. To overcome the limitations of individual studies, resolve inconsistencies, and reduce random errors responsible for false-positive or false-negative associations, we performed a meta-analysis24–26 to investigate whether the ACE I/D polymorphism contributes to susceptibility to schizophrenia or PD.
Materials and methods
Identification of eligible studies and data extraction
We searched the literature for studies that examined the association between the ACE I/D polymorphism and schizophrenia or PD. We utilized the MEDLINE and EMBASE citation indices to identify articles in which the ACE I/D genotype was determined in patients and controls (up to January 2013). In addition, all references mentioned in the identified articles were reviewed to identify studies not indexed by MEDLINE and EMBASE. The following key words and subject terms were used to search the databases: “angiotensin-converting enzyme,” “ACE,” “schizophrenia,” and “Parkinson’s disease.” Studies were included in the analysis if: (1) they were case-control studies; (2) contained original data; and (3) contained sufficient data to calculate odds ratios (ORs). No language restrictions were applied. We excluded the following: (1) studies containing overlapping data; (2) studies in which the number of null and wild genotypes or alleles could not be ascertained; and (3) studies in which family members had been studied because the analyses were based on linkage considerations. The following information was extracted from each identified study: author, year of publication, ethnicity of the study population, demographics, numbers of cases and controls, and frequencies of the genotypes and alleles of the ACE I/D polymorphism.
Evaluation of publication bias and study quality
Funnel plots are widely used to detect publication bias, but they require a range of studies of varying sizes and subjective judgments; thus, we evaluated publication bias using Egger’s linear regression test. 27 Egger’s linear regression test measures funnel plot asymmetry on a natural logarithmic scale of ORs. The chi square test was used to determine if observed genotype frequencies in controls conformed to Hardy-Weinberg (H-W) expectations.
Evaluation of statistical associations
We performed meta-analyses using (1) allelic contrast (D vs I); (2) recessive (DD vs ID + II); (3) dominant (DD + ID vs II), and (4) homozygote contrast (DD vs II) models. Point estimates of risks, ORs, and 95% confidence intervals (CIs) were estimated for each study. In addition, within- and between-study variation and heterogeneity were assessed using Cochran’s Q-statistic. Cochran’s Q-statistic test assesses the null hypothesis that all studies evaluated the same effect. The effect of heterogeneity was quantified using I2, with a range between 0 and 100%, which represents the proportion of between-study variability attributable to heterogeneity rather than chance. 28 I2 values of 25%, 50%, and 75% were considered low, moderate, and high estimates. The fixed-effects model assumes that a genetic factor has a similar effect on disease susceptibility across all studies investigated and that observed variations among studies are caused by chance alone. 29 The random-effects model assumes that different studies show substantial diversity and assesses both within-study sampling error and between-study variance. 30 When study groups are homogeneous, the two models are similar. If the study groups lack homogeneity, the random-effects model usually provides wider CIs than the fixed-effects model. The random-effects model is most appropriate in the presence of significant between-study heterogeneity. 30 Statistical manipulations were performed using Comprehensive Meta-Analysis software (Biosta, Englewood, NJ, USA). The power of each study was computed as the probability of detecting an association between the ACE I/D polymorphism and disease using a significance level of 0.05 and assuming an OR of 1.5 (small effect size). Power analysis was performed using the statistical program G*Power (http://www.psycho.uni-duesseldorf.de/aap/projects/gpower).
Results
Studies included in the meta-analysis
Thirty studies were identified by electronic and manual searches and 16 were selected for full-text review based on title and abstract details.11–23,31–33 Three studies were excluded because they contained no data.31–33 Thus, a total of 13 studies met our inclusion criteria:11–23 eight schizophrenia studies and five PD studies (Figure 1). With regard to ethnicity, three European, four Asian, and three Turkish studies, as well as one Jewish, one Australian, and one Latin American study, were analyzed. In total, the studies included 2024 cases and 2230 controls. Selected details of the individual studies are summarized in Table 1. The statistical power of these 13 studies ranged from 10.9% to 78.0%. None of the studies had a statistical power exceeding 80%.
Study flowchart.
Details of the individual studies included in the meta-analysis.

Citation reference; SZ: schizophrenia; PD: Parkinson’s disease; OR: odds ratio; CI: confidence interval; HWE: Hardy-Weinberg equilibrium. aPower calculations assumed α = 0.05, OR = 1.5.
Frequencies of the D allele of the ACE I/D polymorphism in different ethnic groups
The mean frequency of the D allele of the ACE I/D polymorphism was 44.9% among all normal controls, and subjects of Turkish origin had the lowest D allele prevalence among the ethnic groups tested in this study (29.0%). Among the normal controls, the frequencies of the D allele in the Turkish, Asian, Jewish, Latin American, European, and Australian populations were 29.0%, 34.0%, 39.1%, 45.6%, 53.0%, and 53.9%, respectively (Table 2).
Prevalence rates of the D allele of the ACE I/D polymorphism.

Meta-analysis of the association between the ACE I/D polymorphism and schizophrenia and PD
Meta-analysis was performed on each patient with schizophrenia or PD. A summary of the meta-analysis findings of the relation between the ACE I/D polymorphism and schizophrenia or PD are provided in Table 2. The meta-analysis revealed no association between the ACE D allele and schizophrenia (OR = 0.990, 95% CI = 0.889–1.102, p = 0.856) (Table 3). Analysis using the recessive model, dominant model, and homozygote contrast model showed the same pattern for the ACE D allele (Table 3, Figure 2). The meta-analysis also did not reveal an association between the ACE D allele and PD (OR = 1.067, 95% CI = 0.907–1.255, p = 0.433) (Table 3, Figure 2). Analysis using the recessive model, dominant model, and homozygote contrast model showed the same pattern for the ACE D allele (Table 3).
Meta-analysis of the association between the ACE I/D polymorphism and schizophrenia and PD in the overall group.

ACE I/D: angiotensin-converting enzyme insertion and deletion polymorphism; PD: Parkinson’s disease; OR: odds ratio; CI: confidence interval; F: fixed-effects model; R: random-effects model.
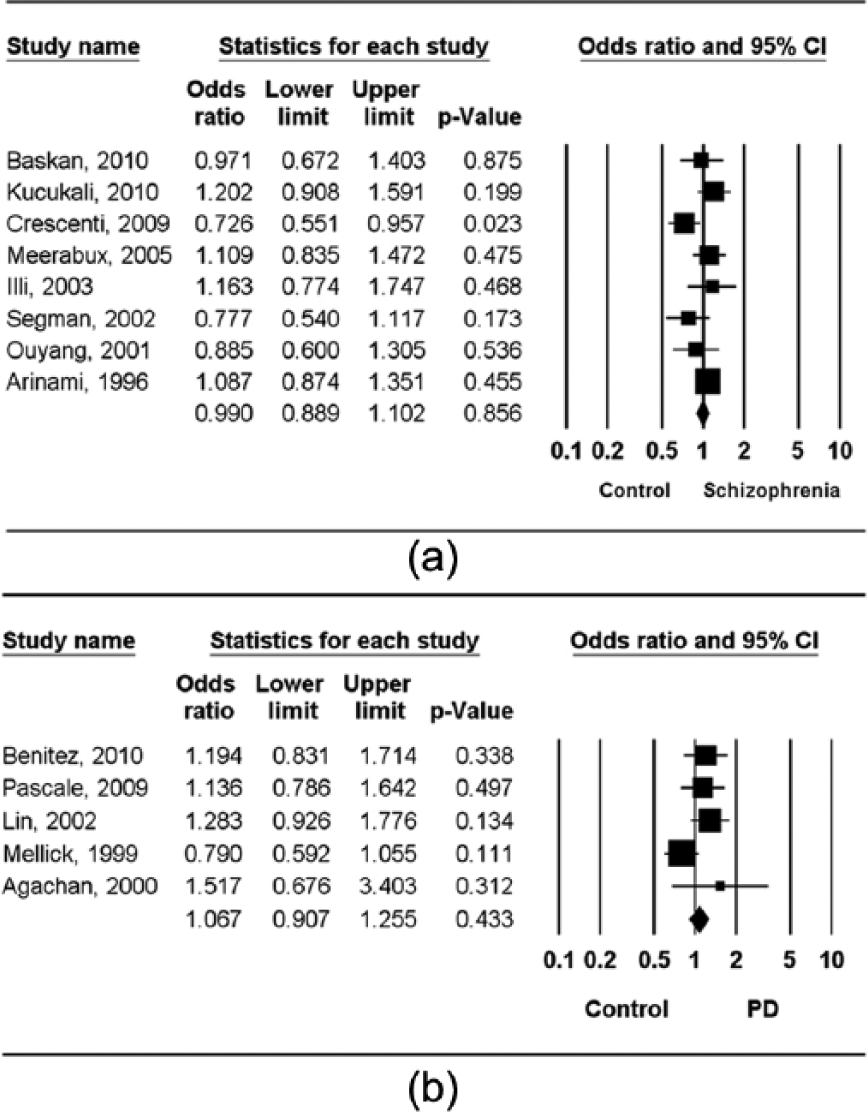
ORs and 95% CIs of individual studies and pooled data for the association between the D allele of the ACE I/D polymorphism and schizophrenia (a) and PD (b) in the overall group.
Meta-analysis of the association between the ACE I/D polymorphism and schizophrenia according to ethnicity
Stratification by ethnicity did not reveal an association between the ACE D allele and schizophrenia in European, Asian, or Turkish patients (OR = 0.896, 95% CI = 0.566–1.419, p = 0.640; OR = 1.057, 95% CI = 0.903–1.238, p = 0.492; OR = 1.111, 95% CI = 0.889–1.389, p = 0.354, respectively) (Table 3, Figures 3 and 4). Analysis using the recessive model, dominant model, and homozygote contrast showed the same pattern for the ACE D allele (Table 4). Ethnicity-specific meta-analysis for PD was not conducted because only one ethnic study of PD was available.

ORs and 95% CIs of individual studies and pooled data for the association between the D allele of the ACE I/D polymorphism and schizophrenia in each ethnic group studied.

Funnel plot of studies for the association between the D allele of the ACE I/D polymorphism and schizophrenia (a) and PD (b) in all subjects (Egger’s regression p values = 0.584, 0.335).
Meta-analysis of the association between the ACE I/D polymorphism and schizophrenia in the European, Asian, and Turkish populations.

ACE I/D: angiotensin-converting enzyme insertion and deletion polymorphism; OR: odds ratio; CI: confidence interval; F: fixed-effects model; R: random-effects model.
Heterogeneity and publication bias
The distribution of genotypes of the ACE I/D polymorphism in control groups was consistent with H-W equilibrium in all studies. Between-study heterogeneity was found during meta-analyses of schizophrenia, and PD, but no evidence of heterogeneity was found for meta-analyses of schizophrenia in Asians. Thus, the meta-analysis was performed using a random-effects model when heterogeneity was present in a population (Tables 3 and 4). Publication bias results in a disproportionate number of positive studies, which poses a problem for meta-analyses. Egger’s regression test showed no evidence of publication bias in this meta-analysis of the ACE I/D polymorphism for either of the two diseases studied (Egger’s regression test p values > 0.1) (Figure 4).
Discussion
ACE is expressed in a number of tissues, including kidney, heart, lung, vascular endothelium, skin, joint, brain, and testes tissues. ACE plays an important role in RAS and angiotensin II conversion, and directly increases vascular smooth muscle cell contraction. It also affects smooth muscle proliferation, monocyte adhesion, platelet adhesion and aggregation, and acts as a potent pro-inflammatory modulator and neurotransmitter.4,34,35 The ACE I/D polymorphism is located within an intron of the ACE gene, and is in strong linkage disequilibrium with genetic factors that influence serum ACE levels. 10 In fact, the ACE I/D polymorphism accounts for approximately half of the variance in ACE plasma levels in humans. ACE activity is higher in patients with the D allele, which results in an increase in angiotensin II levels. 10 Furthermore, this polymorphism has been studied in several diseases, including Alzheimer’s disease, depression, myocardial infarction, rheumatoid arthritis, and psoriasis. These studies all reported associations between susceptibilities to these diseases and the DD genotype and the D allele of the ACE I/D polymorphism.36,37
In this meta-analysis, we combined evidence of associations between the ACE I/D polymorphism and susceptibility to schizophrenia or PD. Our results revealed no association between the ACE I/D polymorphism and susceptibility to schizophrenia or PD. Furthermore, subsequent meta-analysis after stratification by ethnicity revealed no significant associations between the ACE I/D polymorphism and schizophrenia or PD. The prevalence of the D allele was found to vary among ethnic controls from 29.0% to 53.9% for the ACE I/D polymorphism. The frequency of the ACE D allele was lowest among Turkish subjects and highest among Europeans and Australians. However, meta-analysis failed to reveal any association between the ACE I/D polymorphism and schizophrenia or PD in European, Asian, and Turkish populations.
Our meta-analysis results are not consistent with functional studies of ACE that have suggested ACE involvement in schizophrenia and PD pathogenesis. ACE levels are increased in the brain CSF of patients with psychiatric disorders including schizophrenia and PD.6,7 Inflammatory processes play a role in schizophrenia and PD, and angiotensin II is known to be a potent pro-inflammatory modulator. 4 Schizophrenia causes an increase in the dopaminergic activity of the limbic structure of brain, and angiotensin II is a neurotransmitter that interacts with dopamine in mesocorticolimbic areas, thereby increasing dopamine release in the striatum. 35 Loss of substance P in the brain is associated with the pathogenesis of schizophrenia and PD, and ACE has been shown to inactivate substance P in the basal ganglia.8,9 However, because schizophrenia and PD are complex diseases, epidemiologic results sometimes do not coincide with the results of functional studies. For example, multiple genes, genetic backgrounds, and environmental factors contribute to glaucoma development. The lack of any association between the ACE I/D polymorphism and schizophrenia and PD could be explained in four ways. First, genetic heterogeneity for the ACE I/D polymorphism and schizophrenia and PD may exist in different populations. In fact, genetic association studies of the ACE I/D polymorphism and schizophrenia and PD have demonstrated genetic heterogeneity. Second, clinical heterogeneities and differences between patient populations may be responsible. Third, discrepancies may be caused by different linkage disequilibrium (LD) patterns; for example, these polymorphisms may be in LD with a nearby causal variant in one ethnic group but not in another. Fourth, our meta-analysis results might be skewed by Type II error.
The present study has some limitations that should be considered. First, heterogeneity, confounding factors, and publication bias may have distorted our analyses. Second, another polymorphism located in the ACE gene that may affect ACE activity could have been associated with schizophrenia and PD. However, the limited amount of data available prevented further meta-analysis. Third, there are varying levels of severity of schizophrenia and PD, and the severity of the schizophrenia and PD cases was not taken into account in this study. Further research is required to examine whether an association exists between the ACE I/D polymorphism and the severity and clinical features of schizophrenia and PD. Fourth, we included data from European, Asian, and Turkish patients in our ethnic group-specific meta-analysis, thus our ethnicity-associated results are applicable only to these ethnic groups. In addition, the number of studies in the ethnic group-specific meta-analysis was too low to obtain conclusive results.
In conclusion, a meta-analysis demonstrated no association between the ACE I/D polymorphism and schizophrenia or PD, indicating that the ACE I/D polymorphism does not play an important role in schizophrenia or PD. However, we did find that the prevalence of the ACE D allele is ethnicity dependent. Thus, larger-scale studies in populations with different ethnicities are required to further explore the relationship between polymorphisms in the ACE gene and the pathogenesis of schizophrenia and PD.
Footnotes
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
