Abstract
Objective:
The angiotensin-converting enzyme (ACE) may be involved in the pathogenesis of Parkinson’s disease (PD). There have been several studies investigating the association between ACE gene I/D polymorphism and PD risk, but they reported inconsistent findings. We performed a meta-analysis to investigate the association between ACE gene I/D polymorphism and PD risk.
Methods:
Published literature from PubMed and Embase databases were searched for eligible publications. Pooled odds ratios (ORs) with 95% confidence intervals (95% CIs) were calculated using random- or fixed-effects models based on between-study heterogeneity.
Results:
A total of five studies including 606 cases and 708 controls were finally included in the meta-analysis. Meta-analysis showed that there was no obvious association between ACE gene I/D polymorphism and PD risk under the homogeneous co-dominant model (OR = 1.14, 95% CI = 0.71–1.82), heterogeneous co-dominant model (OR = 0.92, 95% CI = 0.70–1.22), dominant model (OR = 0.99, 95% CI = 0.76–1.28) or recessive model (OR = 1.07, 95% CI = 0.83–1.37).
Conclusion:
The meta-analysis suggests that there is no evidence for the association between ACE gene I/D polymorphism and PD risk.
Introduction
Parkinson’s disease (PD) is a common neurodegenerative disorder clinically characterized by tremor, bradykinesia, rigidity and postural instability, and neuropathologically by the loss of dopaminergic neurons in the substantia nigra and the presence of Lewy bodies. 1 The disease is thought to result from a complex interaction between multiple predisposing genes and environmental effects. Recent studies have shown that genetic factors play an important role in contributing to the pathogenesis of PD.
The pivotal role of the angiotensin-converting enzyme (ACE) in the renin-angiotensin system lies in the conversion of angiotensin I to angiotensin II and degradation of bradykinin. 2 High concentrations of ACE have been detected in the nigrostriatal pathway and in basal ganglia. Thus, the ACE may be involved in the pathogenesis of PD. The ACE gene is located on chromosome 17q23 and consists of 26 exons and 25 introns. A functional polymorphism has been identified in intron 16 and consists of the presence (I, insertion) or absence (D, deletion) of a 287-base pair Alu repeat sequence. 3 To date, five studies have examined the association between ACE I/D polymorphism and PD.4 –8 However, the results have been discordant. Most studies indicated a nonsignificant association in Caucasians or Latin Americans,4,6 –8 while one study suggested significant association in a Chinese population. 5 Thus, the aim of the present study using meta-analysis was to clarify the relationship between ACE I/D polymorphism and PD across different ethnic populations.
Materials and methods
Literature and search strategy
We searched the literature databases including PubMed and Embase. The search strategy was to identify all possible studies that involved the use of the following key words: (angiotensin converting enzyme or ACE) and (polymorphism or variant or variation or genotype) and (Parkinson’s disease or PD). The language of publication was restricted to English. The reference lists of retrieved articles were curated manually. The literature search was last updated March 9, 2013.
Inclusion criteria and data extraction
We included a study in the meta-analysis if it met all the following inclusion criteria: (1) investigated the association of ACE I/D polymorphism with PD; (2) used case-control or cohort design and (3) provided odds ratio (OR) with 95% confidence interval (CI) or sufficient data (i.e. the genotype distribution of ACE I/D polymorphism in cases and controls) for calculation of this estimate. The following information was extracted from each study: (1) name of the first author; (2) year of publication; (3) country of origin; (4) ethnicity of the studied population; (5) number of cases and controls or total subjects; (6) genotype distribution in cases and controls; and (7) p for Hardy-Weinberg equilibrium test in controls. All the articles were independently accessed by two authors (Hengli Dou and Limei Zhao) to ensure their compliance with the inclusion/exclusion criteria. Any disagreements were resolved through discussion and a consistent decision was reached.
Statistical analysis
We analyzed the associations of ACE I/D polymorphism with PD by calculating pooled ORs and 95% CIs. We calculated the summary estimate under a co-dominant, dominant and recessive model, respectively. The significance of the OR was determined by a Z test (p < 0.05 was considered statistically significant), and Cochrane’s Q test was performed to test the between-study heterogeneity using a cut-off of p < 0.10 as statistically significant. We used a random- (DerSimonian-Laird method 9 ) or fixed- (Mantel-Haenszel method 10 ) effects model to calculate pooled OR in the presence (p ≤ 0.10) or absence (p > 0.10) of heterogeneity, respectively. We used Begg’s test 11 and Egger’s test 12 (p < 0.05 was considered statistically significant) to examine any publication bias. Statistical analyses for meta-analyses were performed using STATA version 11.0 (StataCorp LP, College Station, TX, USA).
Results
Characteristics of the studies
A flowchart describing the study inclusion/exclusion is presented in Figure 1. The literature search identified a total of 22 potentially relevant papers. Of them, 15 papers were excluded because of obvious irrelevance by reading the titles and abstracts (e.g. addressing Alzheimer disease, temporal lobe epilepsy, depression, coronary artery disease and hypertension). In addition, one paper was excluded because it was a family-based study. 13 Then, six papers met the primary inclusion criteria. However, one paper was excluded because it examined the association between ACE I/D polymorphism and L-dopa-induced adverse effects. 14 At last, five studies for the association between ACE I/D polymorphism and PD were included in the final meta-analysis.4 –8 The characteristics of the included studies are listed in the Table.

Flowchart of inclusion/exclusion of the individual studies.
Characteristics of the included studies of the association between the ACE I/D variant and Parkinson’s disease.

ACE: angiotensin-converting enzyme; HWE: Hardy-Weinberg equilibrium.
Meta-analysis results
A total of 606 cases and 708 controls were identified for the analysis of the association between ACE I/D polymorphism and PD. The results indicated that ACE I/D polymorphism was not associated with PD under the homogeneous co-dominant model (OR = 1.14, 95% CI = 0.71–1.82, I2 = 50.1%, p for heterogeneity = 0.091, Figure 2), heterogeneous co-dominant model (OR = 0.92, 95% CI = 0.70–1.22, I2 = 0.0%, p for heterogeneity = 0.444, Figure 3), dominant model (OR = 0.99, 95% CI = 0.76–1.28, I2 = 17.4%, p for heterogeneity = 0.304, Figure 4) or recessive model (OR = 1.07, 95% CI = 0.83–1.37, I2 = 48.2%, p for heterogeneity = 0.102, Figure 5).
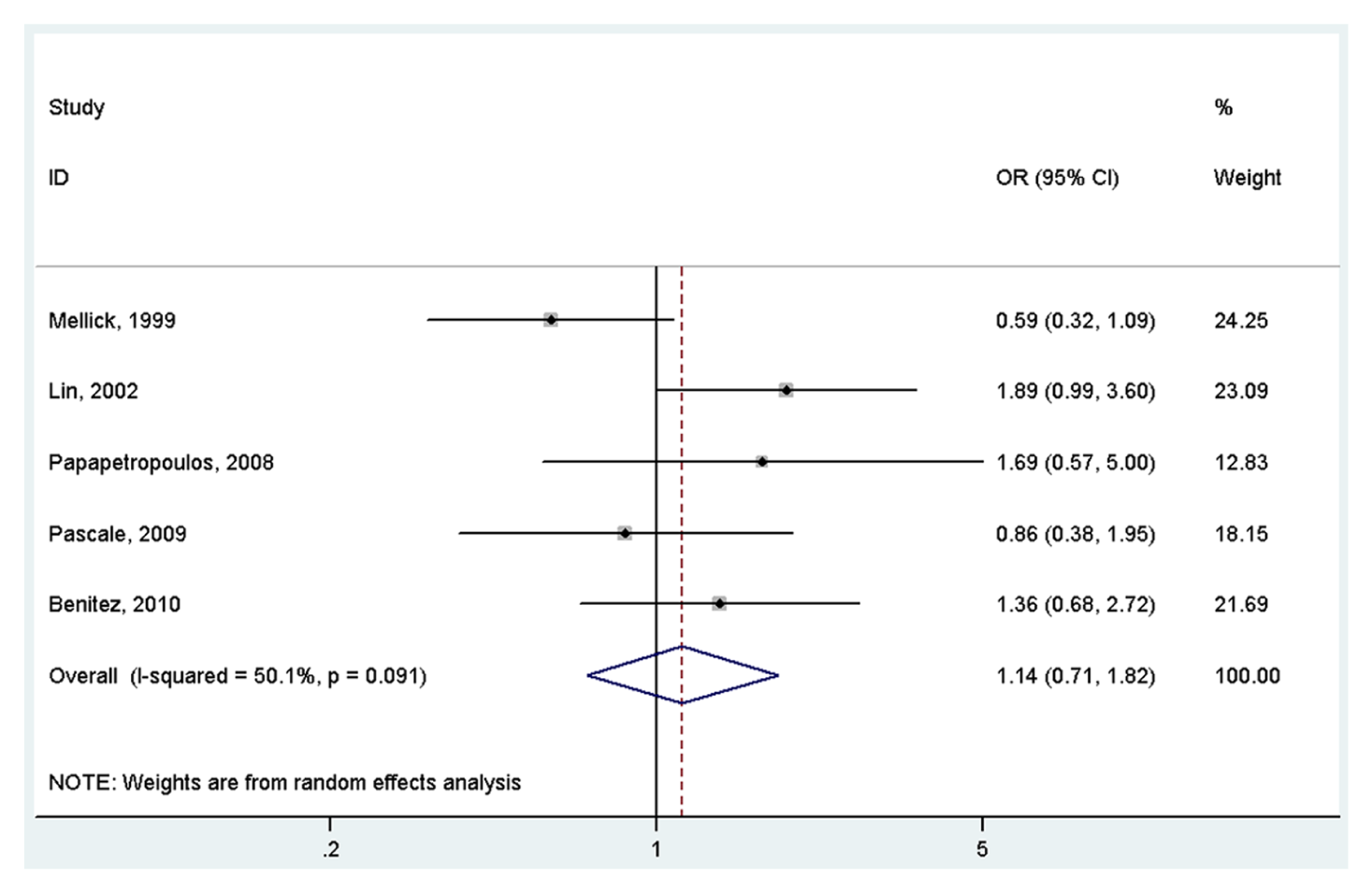
Meta-analysis of the association between the ACE I/D variant and Parkinson’s disease under the homogeneous co-dominant model (DD vs II) OR: odds ratio; CI: confidence interval.

Meta-analysis of the association between the ACE I/D variant and Parkinson’s disease under the heterogeneous co-dominant model (ID vs II) OR: odds ratio; CI: confidence interval.
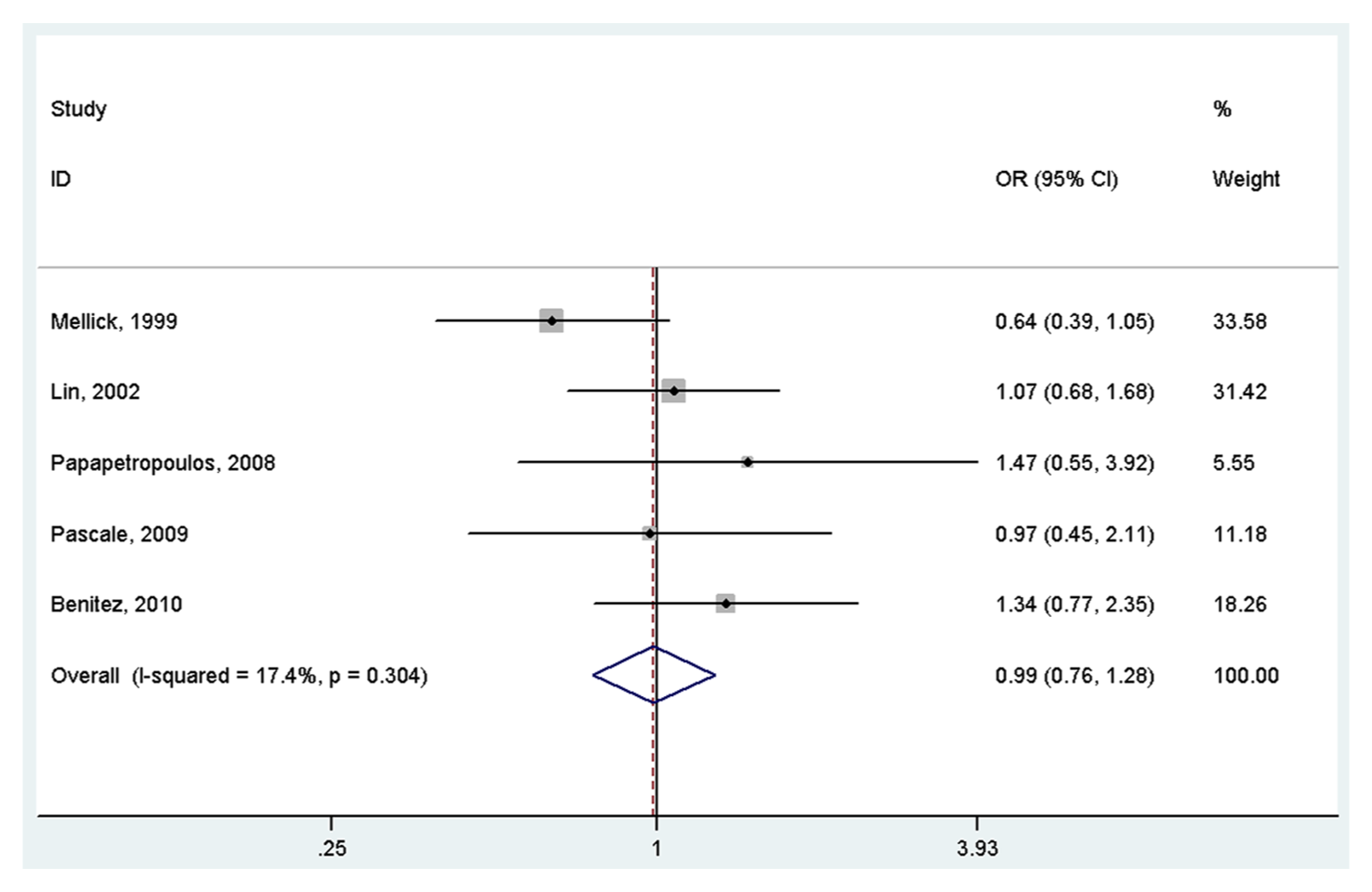
Meta-analysis of the association between the ACE I/D variant and Parkinson’s disease under the dominant model (DD+ID vs II) OR: odds ratio; CI: confidence interval.
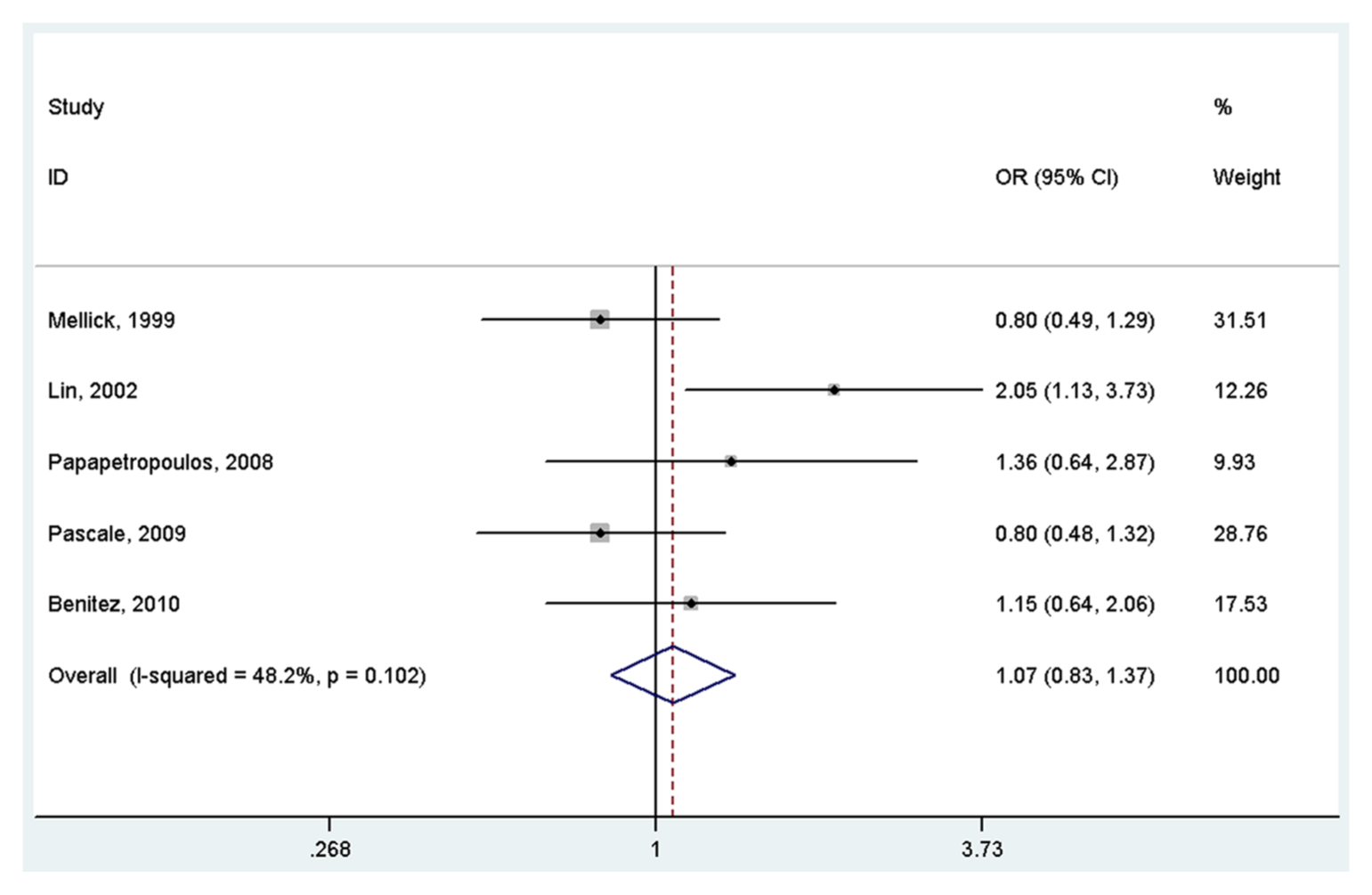
Meta-analysis of the association between the ACE I/D variant and Parkinson’s disease under the recessive model (DD vs ID+II) OR: odds ratio; CI: confidence interval.
Publication bias
No publication bias was detected under the homogeneous co-dominant model (Egger’s test, p = 0.806; Begg’s test, p = 0.647), heterogeneous co-dominant model (Egger’s test, p = 0.462; Begg’s test, p = 0.260), dominant model (Egger’s test, p = 0.806; Begg’s test, p = 0.547) and recessive model (Egger’s test, p = 0.086; Begg’s test, p = 0.198).
Discussion
To our knowledge, this is the first meta-analysis examining the association between ACE I/D polymorphism and PD. The results suggested that ACE I/D polymorphism was not associated with PD. However, the association was significant in one Chinese population.
In 1999, Mellick et al. 4 first hypothesized that the ACE gene might play an important role in the development of PD. However, they reported null association between the ACE I/D polymorphism and PD in one Caucasian population. The subsequent studies on other Caucasian populations also revealed that ACE I/D polymorphism was not associated with PD. However, the study by Lin et al. 5 demonstrated that the D allele of I/D polymorphism is a risk factor for PD in a Chinese population. The discrepancy might be due to various reasons like differences in ethnicity, sample size and different recruitment methods used for the control population between studies. In fact, the frequency of the D allele in normal Caucasians is 50%-58% and 35%-39% in normal Chinese. Thus, it is not surprising that an association study of the ACE gene and PD between Chinese and Caucasian might have different results. It is important to extend the association study between the ACE I/D polymorphism to different populations to discover a significant contribution to the pathogenesis of PD.
Several limitations of our study should be considered. First, our meta-analysis was based on unadjusted estimates, and the confounding factors could not be controlled for because most studies did not provide these data. Second, gene-gene and gene-environment interactions were not addressed in our meta-analysis. Third, although five studies including 606 PD cases and 708 controls were included in the present meta-analysis, it should be noted that the statistical power was still not sufficient. Thus, the results should be interpreted with caution.
In conclusion, our meta-analysis did not support the association between ACE I/D polymorphism and PD risk. Further large-scale studies with consideration for gene-gene/gene-environment interactions should be conducted to investigate the association.
Footnotes
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors
