Abstract
Background and aim:
Cardiovascular complications are one limitation of breast cancer treatment. The aim of the current study was to investigate whether the renin–angiotensin related genes could be altered by chemotherapy and radiotherapy, using a rat model.
Methods:
Female rats were divided into three groups: control, chemotherapy + irradiation (TC+IR) and irradiation (IR). Molecular analyses of the left ventricle were performed five months after the end of treatment. The analyses evaluated the changes in mRNA levels of some renin–angiotensin system (RAS) related genes: angiotensinogen, renin, angiotensin-converting enzyme (ACE) and angiotensin II type 1 receptor (AT1) and vascular endothelial growth factor (VEGF), which can be related to ACE production, by RT-PCR.
Results:
Renin was only observed in treated groups, TC+IR and IR, compared with the control group. ACE and VEGF levels were decreased in TC+IR (p<0.001) and in IR (p<0.001), and AT1 mRNA was higher in groups TC+IR (p<0.01) and IR (p<0.05) compared with the control group.
Conclusion:
Chemotherapy and irradiation can induce significant changes in some RAS related genes. These alterations are important to understand the pathways and consequences beyond cardiotoxicity induced by breast cancer treatments.
Introduction
Data from Global cancer statistics show that breast cancer (BC) is the most common type of cancer among women, leading the number of deaths caused by cancer. 1 Early detection through mammography allows more treatment options and consequently greater survival of these women. However, increased patient survival enhances their chances of suffering from the late side effects of cancer therapy.2,3 The therapy strategies today include combinations of drugs, radiation and surgery. Many of these therapies cause cardiac side effects that influence the success of treatment. 4
It is known that radiation-induced heart disease (RIHD) is a late effect caused by radiation in the thoracic region, when all or part of the heart is in the radiation field.3-5 Among the manifestations of RIHD are valvulopathy, pericarditis and myocardial fibrosis.6-8
In the literature it is possible find different forms related to cardiotoxicity caused by chemotherapeutic agents, including cellular damage caused by free radical formation, and induction of immunogenic reactions in the heart. The chemotherapy regimen choice depends on tumor stage, and also on the patient’s clinical condition. One of the schemes used for BC treatment is the association of taxanes and cyclophosphamide, known as the TC regimen.9,10 Taxanes such as docetaxel have been widely incorporated into the chemotherapy options for early stage BC treatment. The taxanes’ mechanism of action is the inhibition of cell division by interrupting the function of microtubules, but their use is also related to cardiotoxicity, and the exact way this occurs is still unknown.11,12 Alkylating agents such as cyclophosphamide act directly in the alkylation of the DNA molecule. The precise mechanisms of cyclophosphamide-induced cardiotoxicity are also unknown. One hypothesis is that cyclophosphamide causes direct damage to the endothelium followed by extravasations of toxic metabolites, resulting in damage to the cardiomyocytes, hemorrhage and edema. 13
The drugs that inhibit the renin–angiotensin system (RAS) are the major therapeutic strategy for treating hypertension and reducing cardiovascular damage, and they are being used in patients undergoing chemotherapy for BC treatment.14-16 Since its discovery in 1890, the RAS has been identified as an endocrine system, where the circulating renin, produced in the kidney, regulates cardiovascular function by angiotensin II, which exerts its biologic actions by binding to either the angiotensin type 1 or type 2 receptors on target tissues. Until recently only this conventional RAS was recognized. 17 However, it is now known that the kidneys are not the only source of renin production, and the angiotensin peptides may be produced in tissues and organs independently. In the heart, changes in some RAS components, such as angiotensin II type 1 receptor (AT1) and angiotensin-converting enzyme (ACE), are often observed in conditions leading to progression of heart failure.18-20 The local RAS functions are not yet fully explained, but it is believed that they are tissue-specific. It is known that some of the local cardiac RAS components are up-regulated in RIHD, but there are few studies that show its action on the association of both chemotherapy and irradiation treatments for BC.17,18,21
The present study evaluated the expression of some RAS components: angiotensinogen, renin, ACE and AT1 and vascular endothelial growth factor (VEGF), which can be related to ACE production, by RT-PCR, in female Wistar rats undergoing chemotherapy (TC) and/or irradiation. Also investigated was the presence of mast cells in the heart tissue of these animals, since they can be a major source of renin production. 22
Materials and methods
Animals
Female Wistar rats (n = 15), three months old, weighing approximately 200g, were kept in appropriate cages with four animals in each, on a 12-h light/dark cycle with food and water provided ad libitum. The animals were divided into three groups, five animals each: control (received no further treatment), TC + IR (group receiving a chemotherapy regimen and irradiation) and IR (group receiving only irradiation). The study protocol was approved by the local ethical council (No. CEUA/010/2012).
Chemotherapy administration
In the TC+IR group, the multidrug TC was intraperitoneally administered to the animals. There were given four cycles, with an interval of seven days between them, and each animal received cyclophosphamide 50mg/kg dissolved in saline (Baxter, Brazil) and 12.5mg/kg docetaxel dissolved in 95% ethanol and diluted with 5% glucose solution (Eurofarma, Brazil). The dose for each drug, in each cycle, was calculated to be equivalent to a chemotherapy dose per cycle in humans by the Human Equivalent Dose Calculation. 23 Seven days after the last cycle of chemotherapy, the animals were irradiated (TC+IR). Animals from IR group received no chemotherapy, only irradiation.
Irradiation
Before irradiation rats were anesthetized with ketamine/xylazine intraperitoneally (0.1 mg/kg), and immobilized in a polystyrene support. This ensures the animals’ permanence in the same position during the procedure. Rats were irradiated with a single dose of 20 Gy to the heart, using a linear accelerator Varian Clinac ® 2100 C (Varian Medical Systems, CA, USA) with a 6 MV X-ray, and dose rate of 240 cGy/min. Single doses of 20 Gy are calculated to be approximately equivalent to total fractionated doses of 30–50 Gy (2 Gy per fraction), according to the linear quadratic model and an a/b ratio of 2:3 as described by Schultz-Hector et al. 24 Individual rats were irradiated in the supine position, using an anterior field size of 2 cm2 with bolus depth of 0.5 cm. The heart position was marked after computed tomography simulation (HiSpeed CT/Dual, GE Healthcare, USA). After irradiation, animals were returned to the cages and remained under close observation until euthanasia, five months after irradiation. These time points were chosen based on previous studies where, at four months after irradiation, cardiac lesions could be observed in rats exposed to single doses of 12.5 Gy or higher.25,26
Real-time PCR-RT
Total RNA was extracted from the left ventricle (LV) of animals from control, TC+IR and IR groups, by using Trizol Reagent (Invitrogen, NY, USA) according to the manufacturer’s protocol. Total RNA was treated with DNAse amplification grade I (Invitrogen, NY, USA) and first-strand cDNA was synthesized using 2µg from total RNA and ImProm-II™ Reverse Transcription System (Promega, WI, USA) following the manufacturer’s instructions. Oligonucleotide primers were designed based on the cDNA sequences reported in the GenBank database (www.ncbi.nlm.nih.gov). The sequences of primers are listed in Table 1. Real-time quantitative PCR analyses were performed using Applied Biosystems 7500 and SDS 7500 v.2.0.1 software (Applied Biosystems, CA, USA). PCR amplifications were performed using Gotaq qPCR Master Mix (Promega, WI, USA). PCR cycling conditions were as follows: initial 95°C for 10 min, then 40 cycles using 95°C for 20 s, and 60°C for 60 s. Amplification specificity was determined using melt curves. Each sample was run and analyzed in duplicate. Angiotensinogen, renin, ACE, AT1 and VEGF mRNA levels were normalized by the values relative to GAPDH (glyceraldehydes-3-phosphate dehydrogenase), and expression ratios were computed using the threshold cycle method (ΔΔCt).
Real time PCR-RT primer details for gene expression.

GAPDH: glyceraldehydes-3-phosphate dehydrogenase; ACE: angiotensin-converting enzyme; AT1: angiotensin II type 1 receptor; VEGF: vascular endothelial growth factor.
Mast cell staining
Transverse sections of the LV were post-fixed in 10% buffered formalin and embedded in paraffin. Transverse LV paraffin (6 μm thick) sections were subjected to mast cell analysis using acidic toluidine blue staining solution. First, the sections were deparaffinized in xylene and hydrated through alcohol to distilled water. They were then incubated in an aqueous solution of 0.20% Toluidine Blue (Sigma-Aldrich, MA, USA) acidified to pH 2.3 for 2–3 min. Sections were washed three times in fresh distilled water. Then they were quickly dehydrated through 95% and two changes of 100% alcohol, cleared in xylene and mounted with Entellan (Merck Millipore, MA, USA). Mast cells stained a violet/red purple color with a blue background.
Statistical analysis
Statistical analysis was performed by one-way analysis of variance (ANOVA) and Tukey’s post-hoc test (GraphPad Prism 5.03; GraphPad Software, La Jolla, CA, USA). All results are means ± standard error of mean (SEM). Values of p< 0.05 were considered significant.
Results
Cardiac expression of RAS components in rats submitted to chemotherapy and/or radiotherapy
To ascertain whether RAS-related genes could be altered in treated rats, the expression levels of angiotensinogen, renin, ACE and AT1 were assessed. Compared with the control group no significant difference in angiotensinogen mRNA levels was observed (Figure 1). Renin was only observed in treated groups, TC+IR and IR, compared with the control group, and there was no significant difference between them (Figure 2). ACE levels were decreased in TC+IR (p<0.001) and in IR (p<0.001) groups compared with controls (Figure 3). AT1 mRNA was higher in groups TC+IR (p<0.01) and IR (p<0.05) when compared with the control group (Figure 4). These results suggest that some RAS-related genes could alter their mRNA levels in consequence of TC and/or IR treatment.
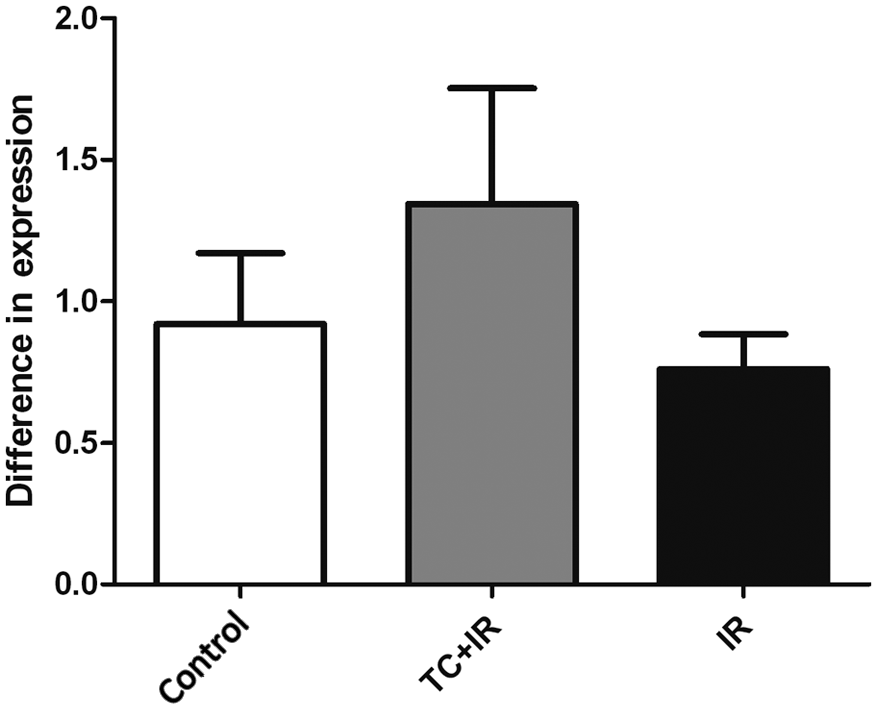
RT-PCR analysis of angiotensinogen expression. Groups: control, received no treatment; TC+IR, received chemotherapy and were irradiated; IR, were only irradiated. There was no significant difference between the groups. Values are given as mean ± SEM of five animals.
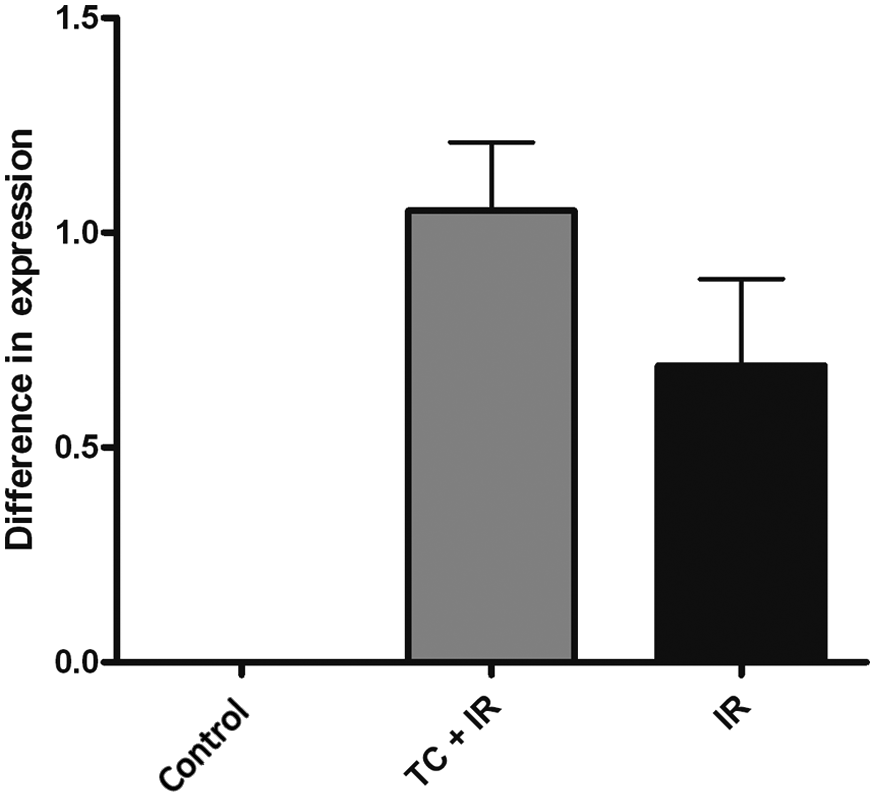
RT-PCR analysis of renin expression. Groups: control, received no treatment; TC+IR, received chemotherapy and were irradiated; IR, were only irradiated. The control group did not express renin. There was no significant difference between TC+IR and IR groups. Values are given as mean ± SEM of five animals.
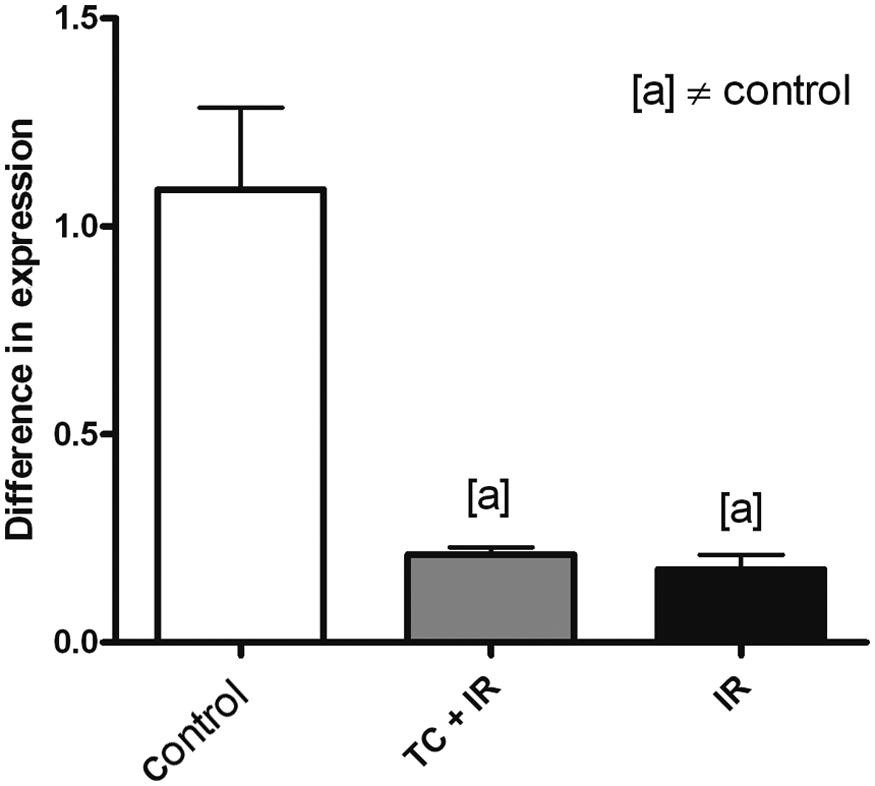
RT-PCR analysis of angiotensin-converting enzyme (ACE) expression. Groups: control, received no treatment; TC+IR, received chemotherapy and were irradiated; IR, were only irradiated. Symbol [a] means significant difference (p< 0.001) versus control group. Values are given as mean ± SEM of five animals.

RT-PCR analysis of angiotensin II type 1 receptor (AT1). Groups: control, received no treatment; TC+IR, received chemotherapy and were irradiated; IR, were only irradiated. Symbol [a] means significant difference versus control group (p< 0.05 for TC+IR group and p< 0.01 for IR group). Values are given as mean ± SEM of five animals.
Cardiac expression of VEGF
As a decreased level of ACE mRNA was observed in treated groups, and ACE is mainly produced by endothelial cells, the expression levels of VEGF were assessed. It was observed that VEGF mRNA levels were decreased in the TC+IR group (p<0.001) and in the IR group (p<0.001) when compared with controls (Figure 5).
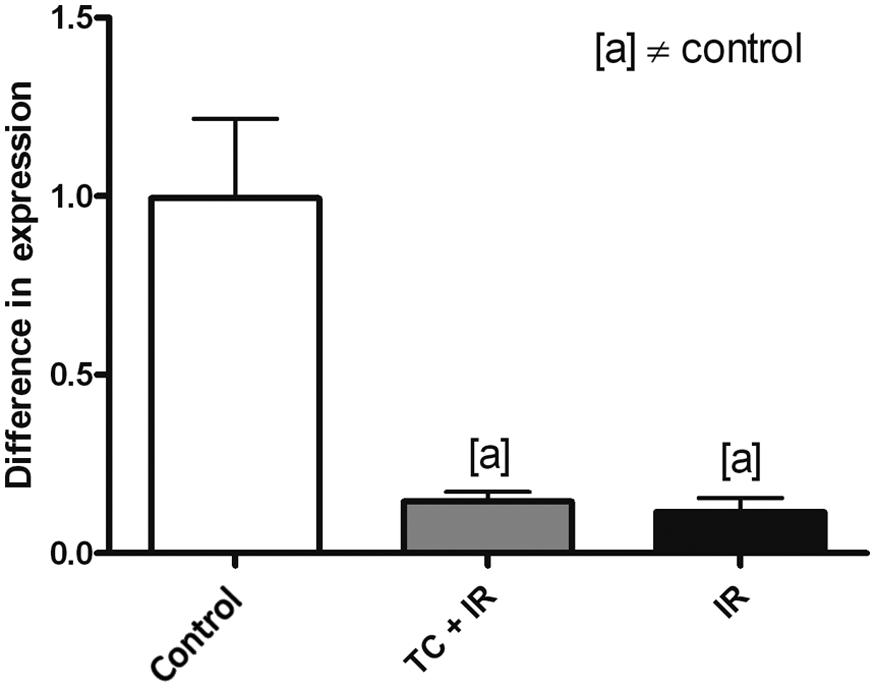
RT-PCR analysis of vascular endothelial growth factor (VEGF) expression. Groups: control, received no treatment; TC+IR, received chemotherapy and were irradiated; IR, were only irradiated. Symbol [a] means significant difference (p < 0.001) versus control group. Values are given as mean ± SEM of five animals.
Mast cell staining
The presence of mast cells in the LV tissue of all groups was investigated using a toluidine blue solution. Mast cells were identified in treated groups, TC+IR and IR, but not in controls (Figure 6).

Toluidine blue-stained photomicrographs (magnification of 60×) showing the mast cells in the rats’ left ventricle. (a) Control, received no treatment; (b) TC+IR, received chemotherapy and were irradiated; (c) IR, were only irradiated.
Discussion
The new detection methods and treatment for BC have improved life expectancy of survivors; however, they allow the development of cardiac side effects. It is therefore essential to understand the causes of this complication to achieve success in patient treatment.4,10,12,27
Knowledge of the RAS has advanced in recent years since the classic endocrine system to a new concept that includes a series of local RASs, which operate independently in several organs, especially in the heart.28-31
In this study an animal model was developed, using female Wistar rats submitted to four cycles of chemotherapy and/or irradiation, mimicking a regular BC treatment in women. The purpose was to evaluate the expression of some local cardiac RAS components, using the quantitative RT-PCR technique, focusing on late cardiac treatment complications.
In the literature the control of angiotensinogen expression is controversial. There are no supporting evidences that correlate its expression and radiation or chemotherapy regimens. The obtained results showed no significant alterations in angiotensinogen expression between groups. Interestingly, we identified that renin was not ubiquitously expressed in cardiac tissue, but it is strongly induced by TC+IR and IR treatments. These findings corroborate previous studies that show angiotensinogen in cardiac tissue is produced by cardiomyocytes and fibroblasts, but that the renin found in this tissue is produced by the kidneys.17,21,32,33 It is known that mast cells can synthesize, store and secrete renin, being an important source of this enzyme. 22 A study made by Boerma and Hauer-Jensen 5 revealed the presence of mast cells in cardiac tissue after exposure to localized heart irradiation with a single dose of 18 Gy. As in the present study the presence of renin was detected in treated groups; it was suggested that this renin could be produced by mast cells present on the tissue. In order to confirm this hypothesis our study performed a toluidine blue staining to visualize the presence of mast cells in cardiac tissue. Mast cells were found only in treated groups, supporting our hypothesis that these cells are responsible for the renin mRNA levels in TC+IR and IR cardiac tissue.
The expression of ACE in cardiac tissue was also assessed, and the treated groups had decreased mRNA levels of ACE compared with controls. It is known that the ACE is mainly produced by endothelial cells,34-36 and that irradiation primarily affects these cells, causing apoptosis of the same, and subsequent decrease in capillary density.37-39 Previous studies demonstrated that several chemotherapeutic agents, like docetaxel and cyclophosphamide, have an antiangiogenic activity by increasing endothelial cells apoptosis and decreasing the expression of VEGF levels.40-43 Thus, in this study heart VEGF mRNA levels were evaluated and results showed that, similarly to ACE levels, treated groups had decreased levels of VEGF compared with controls. These findings taken together suggest that irradiation and chemotherapy cause an angiogenesis reduction, and as ACE is mainly produced by endothelial cells, it may cause a reduction in ACE levels in the cardiac tissue.
The mRNA AT1 levels were assessed in this study and results showed that their levels were higher in treated groups compared with controls. AT1 receptors are expressed in cardiomyocytes, fibroblasts and, recently, they are known to be expressed in cardiac mast cells.44,45 Many studies showed that this receptor is up-regulated in the cardiac remodeling process after several pathologic conditions.46,47
In this present study the ACE expression was reduced and AT1 expression was higher, which seems to be controversial. This can be associated with the novel and more complex aspects of the RAS role in cardiovascular function. Evidence is available that angiotensin II is not only generated by ACE, but other enzymes can participate in this process, for example chymase.48,49 Furthermore, recent studies show that the AT1 receptors can exert their function and can be over expressed in the absence of angiotensin II. 50
The participation of cardiac local RAS components in cardiovascular complications induced by cancer treatments is a novel and controversial area, and additional studies are needed to better understand the pathways and consequences beyond this mechanism. Such studies intend to improve breast cancer treatment strategies, limiting late cardiovascular complications.
Conclusion
The local RAS plays an important role in cardiovascular disorders. This study aimed to evaluate cardiac RAS components using the RT-PCR technique. The main results were the gene expression of renin only in treated groups, decreased expression of ACE and increased expression of AT1 receptor in the same groups. The latter observations indicate that therapeutic strategies for BC must consider these RAS component alterations in order to avoid the known cardiac complications.
Footnotes
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors
