Abstract
There is preliminary evidence that the local renin-angiotensin system (RAS) could affect neoplastic hematopoiesis. The aim of this study is to search messenger RNA (mRNA) expressions of the essential RAS elements in myeloid and lymphoid hematological neoplastic disorders. Forty-six patients with newly diagnosed myeloid (AML, biphenotypic leukemia, CML) or lymphoid (CLL, NHL, B-ALL, T-ALL) hematological disorders were included in the study. In the lymphoid group, the median expression values of
Introduction
The concept of a local hematopoietic renin-angiotensin system (RAS) in bone marrow (BM) was first suggested in 1996.1,2 Locally active BM RAS affects critical steps of physiological and pathological blood cell production in autocrine, paracrine and intracrine ways.3,4 The stimulation of the angiotensin type 1 and 2 receptors (AT1/AT2) by angiotensin II (Ang II), the principal effector molecule of the RAS, exerts an stimulatory/inhibitory action on the janus-kinase-signal transducer and activator of transcription (JAK-STAT) pathway, which is directly linked to activities of the erythropoietin, thrombopoietin and other hematopoietic cytokines during normal hematopoiesis and in myeloproliferative neoplasms.3,5,6 Local RAS is effective even at the stage of primitive embryonic hematopoiesis.7,8
There is preliminary evidence that local BM RAS could affect neoplastic hematopoiesis.9–16 Over-expression of the angiotensin-converting enzyme (ACE) (CD 143) surface antigen in leukemic myeloid blast cells have been detected by flow cytometric analyses. Moreover, a positive correlation has been found between the ACE and BM blast count.
11
ACE and p53 expressions were detected in the CD34+cells of patients with acute leukemia during and after induction chemotherapy.
17
ACE-expressing macrophages in lymph nodes of Hodgkin’s disease have been detected.
15
The renin system is present in the K562 leukemic cell line in vitro model. Multipotential, hematopoietic malignant K562 leukemic blast cells also exhibited significant expressions of
The purpose of this study is to research mRNA expressions of the essential RAS elements (
Materials and methods
Study population
The study group comprised 46 newly diagnosed myeloid (acute myeloid leukemia (AML), biphenotypic leukemia, chronic myeloid leukemia (CML)) or lymphoid (chronic lymphocytic leukemia (CLL), non-Hodgkin’s lymphoma (NHL), B cell acute lymphoblastic leukemia (B-ALL), T cell acute lymphoblastic leukemia (T-ALL)) hematological disorders. The diagnoses of hematological neoplasms were reached based on the criteria of the World Health Organization (WHO).21,22 Samples from BM were collected prior to the chemotherapy administered to each patient. The BM aspiration procedure from the iliac crest was performed only at the clinically indicated situation. At the time of the sample collection, all of the patients were in good health and well hydrated. Pediatric and adult patients receiving chemotherapy were excluded from this study. Written informed consent was obtained from all participants and the study protocol was approved by the local ethics committee of Hacettepe University with project number LUT 10/45.
Isolation of RNA and synthesis of cDNA
BM samples were collected in 2-ml ethylenediaminetetraacetic acid (EDTA) tubes. Total RNA was isolated according to the manufacturer’s instructions (Qiagen, Germany). RNA quality was measured by spectrophotometer (ND-1000, Nanodrop Technologies, Inc., USA), and one microgram of total RNA was used. Random primers (20 µM, Roche Diagnostics, Germany), dNTP set 10 mM, Fermentas UAB, Lithuania), RiboLock Rnase Inhibitor (20 U/µl, Fermentas) and Moloney murine leukemia virus (MMLV) reverse transcriptase (200 U/µl, Fermentas) were used for cDNA synthesis. cDNA samples were stored at −20°C.
Quantitative real-time polymerase chain reaction (PCR) analysis (qRT-PCR)
We searched for the gene expression of the major RAS components (
The primer sequences-probe sets.

UPL: Universal Probe Library, ACE: angiotensin-converting enzyme; ANGTS: angiotensinogen.
qRT-PCR analyses for
The 2–ΔΔCt method was used to calculate relative expression levels determined from the RT-PCR experiments. 23
Statistical analyses
Statistical Packages for the Social Sciences v13.0 (SPSS Inc., Chicago, IL, USA) software was used for all statistical analyses. The results were given as the mean ± standard error for the data with normal distribution, and as median (min-max) for the data without a normal distribution and ratio for the nominal data. Data distribution was tested with Kolmogorow-Smirnow’s test. Homogeneities of variances were evaluated with Levene’s test. Nonparametric tests were used since the sample sizes of the groups were small. Differences between two groups were assessed by the Mann-Whitney U test. A
Results
BM samples were obtained from 46 patients, with lymphoid (
The hematological diagnoses of patients with lymphoid and myeloid neoplasms.

CLL: chronic lymphoid leukemia; NHL: Non-Hodgkin’s lymphoma; ALL: acute lymphoblastic leukemia: AML: acute myeloid leukemia; APL: acute promyelocytic leukemia; CML: chronic myeloid leukemia.
RENIN, ACE1, ACE2 and ANGTS mRNA expressions
Specific mRNA expressions for the components of the RAS were detected in the BM samples by qRT-PCR analysis.

RNA: ribonucleic acid; ACE: angiotensin-converting enzyme; ANGTS: angiotensinogen.

ALL: acute lymphoblastic leukemia; RNA: ribonucleic acid; ACE: angiotensin-converting enzyme; ANGTS: angiotensinogen.
We also analyzed separately ALL and non-ALL patients versus those in the myeloid group.
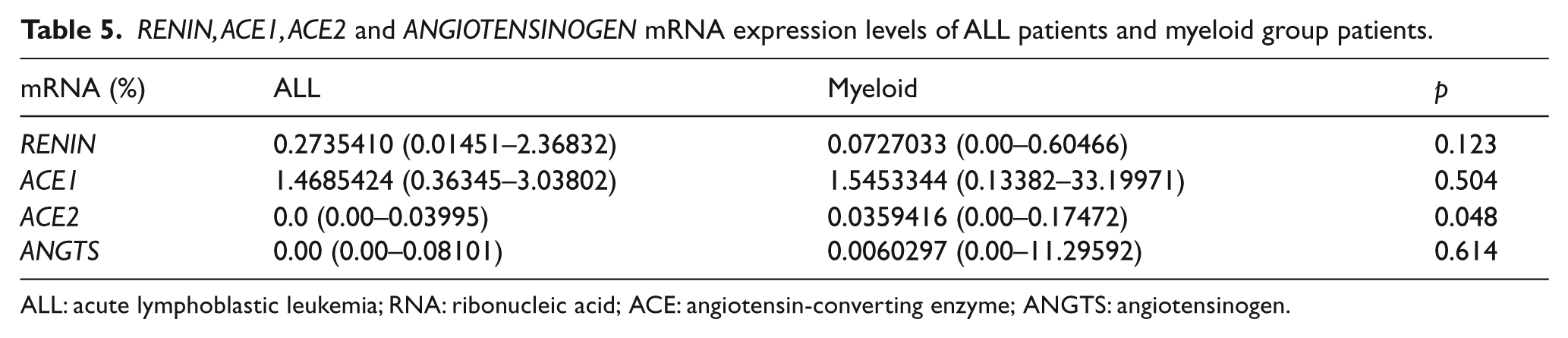
ALL: acute lymphoblastic leukemia; RNA: ribonucleic acid; ACE: angiotensin-converting enzyme; ANGTS: angiotensinogen.
Active disease status in all patients

RNA: ribonucleic acid; ACE: angiotensin converting enzyme; ANGTS: angiotensinogen
Active disease status in lymphoid group
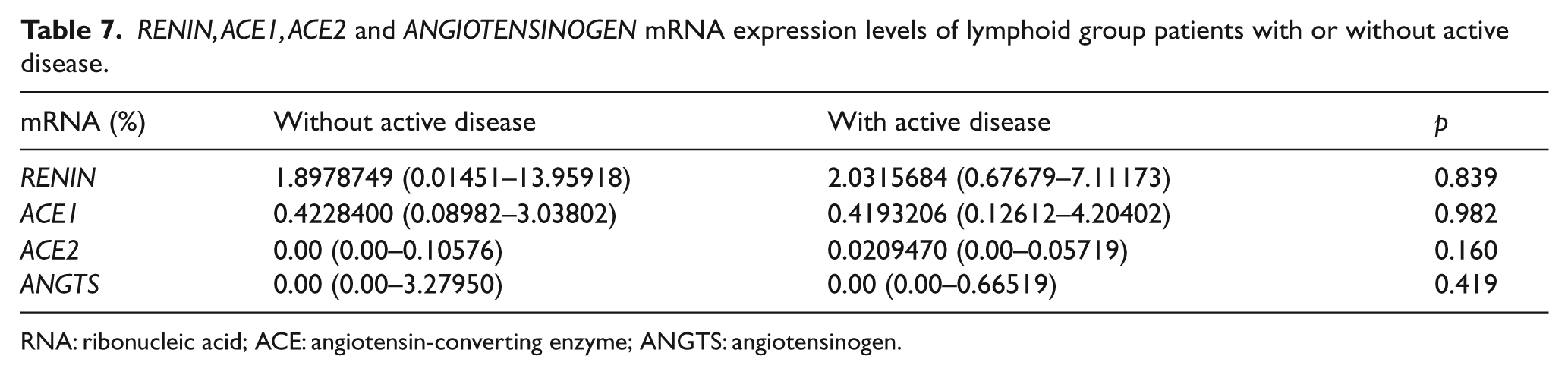
RNA: ribonucleic acid; ACE: angiotensin-converting enzyme; ANGTS: angiotensinogen.
Discussion
In this study, specific mRNA expressions of the components of the RAS (
Renin, as an aberrant leukemic marker of acute leukemia, was expressed in leukemia cell lines such as K562, KU812 and MEG-01.14,25,26 Renin-like enzyme activity due to specific immunoreactive renin-like peptide of 47 kDa converting angiotensinogen to Ang-I had been detected in leukemic myeloid blast cells.27,28 Increased activity of the renin gene was detected during the NUP98-HOXA9 enhanced blast formation.29,30 Gómez-Casares et al. previously disclosed renin expressions in cells from AML, CML and ALL. The highest frequency was observed in AML patients (47.2% of the cases).
25
Renin expression disappeared during the complete remission of AML.25,26 The same investigation team has analyzed 76 samples taken from patients with AML with follow-up of the positive patients. Thirty-one patients (41%) were positive for renin gene expression at diagnosis in their study. All of the renin-positive patients at diagnosis showed no expression during complete remission (CR), but expression recurred in those experiencing relapse and persisted when the disease was refractory to treatment.
26
In our study,
ACE existence throughout hematopoietic ontogeny7,8 indicates primitive hematopoiesis casts attention on the effects of RAS on neoplastic tissues. Immunohistochemical studies have shown the possible role of ACE/RAS in BM by evaluating
Deficiency of ACE2 in BM-derived cells increased mRNA abundance of a macrophage marker (F4/80) and tumor necrosis factor-alpha (TNF-α) in the stromal vascular fraction of retroperitoneal adipose tissue, suggesting increased infiltration of macrophages into adipose tissue of chimeric mice lacking ACE2 in leukocytes.
36
In our study,
Human umbilical cord blood cells express
The contributions of the RAS components to primitive and neoplastic hematopoiesis are evident.3,4 AT type 1a (AT1a) receptors are present on human CD34+CD38– cells, CD34+CD38+ cells, lymphocytes and stromal cells. Ang-II, the main effector peptide of RAS, increased hematopoietic progenitor cell proliferation by acting on AT1a receptors present on the CD34+ HSCs. 42 Ang-II was found to be acting as an autocrine growth factor for AML. 43 BM AT1 expression levels of the myeloma patients disclosed positive correlation with their BM infiltration pattern and tumor load indicated by serum beta 2 microglobulin levels. 33 The pharmacologically developed drug Ang-1-7 is already in phase I/II clinical trials for the modulation of BM RAS in distinct disease states.44,45 However, since most of the cellular effects of the local RAS are in an autocrine, paracrine and intracrine fashion, future drugs intended to modulate local RAS functions shall be prepared to have local targeted actions in the tissue microenvironment, such as inside BM.
Conclusions
The results of our present study indicate that the activities of local RAS may differ in distinct disease states such as leukemia and lymphomas. These data further support the original hypothesis that there is a local RAS in the BM affecting physiological and pathological hematopoiesis.
Footnotes
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
