Abstract
In this current work, two fresh transition metal coordination polymers (CPs) have been created with the reaction between terphenyl-3,3″,5,5″-tetracarboxylic acid (H4L), the symmetrical rigid carboxylic acid ligand and the relevant metal salts under the reaction conditions of solvothermal, and the chemical compositions of the compounds respectively are {[Cu2(TPTC) (DMF)2(H2O)]·2DMF·2H2O} (
Introduction
Postoperative infection is an important form of infection after surgery, which seriously affects the recovery progress of patients after surgery. 1 A large amount of data shows that surgical incision infections account for more than 10% of nosocomial infections, which seriously affects the quality of medical services and the recovery of patients. 2 As a result, in the current work, the novel candidates were designed and synthesized and their biological application values were evaluated.
In recent several years, the transition metal complexes involving organic ligands and inorganic metal ions are the focus of recent research on account of their versatility in structure and their extensive applications range for instance the antibacterial and antitumor activities, the performances of luminescence, catalytic activities, the adsorption of gas, DNA binding, and magnetic performances.3–6 As is known to all, for the metal–organic complexes, the diversity in structure and activity performances are decided by the selection of metal atoms and organic ligands applied to a great extent, together with the reaction mechanism for acquiring the complex.7–11 As a result, the choice of appropriate metal ions and the rational design of organic ligands possess a significant function in acquiring the complexes having ideal performances.12–15 The terphenyl-3,3″,5,5″-tetracarboxylic acid (H4TPTC) was employed in our investigation on the basis of these reasons: (1) the rigid skeleton for the ligand of H4TPTC is helpful to establish the stable CPs. ,2) the carboxyl groups of CPs can be coordinated in many ways, which makes it possible to establish the CPs having outstanding performances and fresh architectures, and (3) the conjugated aromatic rings of CPs are conducive to electron transport and the interactions of π-π with the bioactivity molecules, which might endow them with interesting bioactivity.16–20 In this current work, two fresh transition metal coordination polymers (CPs) have been created with the reaction between terphenyl-3.3″,5.5″-tetracarboxylic acid (H4L, Scheme 1), the symmetrical rigid carboxylic acid ligand and the relevant metal salts under the reaction conditions of solvothermal, and the chemical compositions of the compounds respectively are {[Cu2(TPTC) (DMF)2(H2O)]·2DMF·2H2O} ( The chemical drawings for the organic ligands.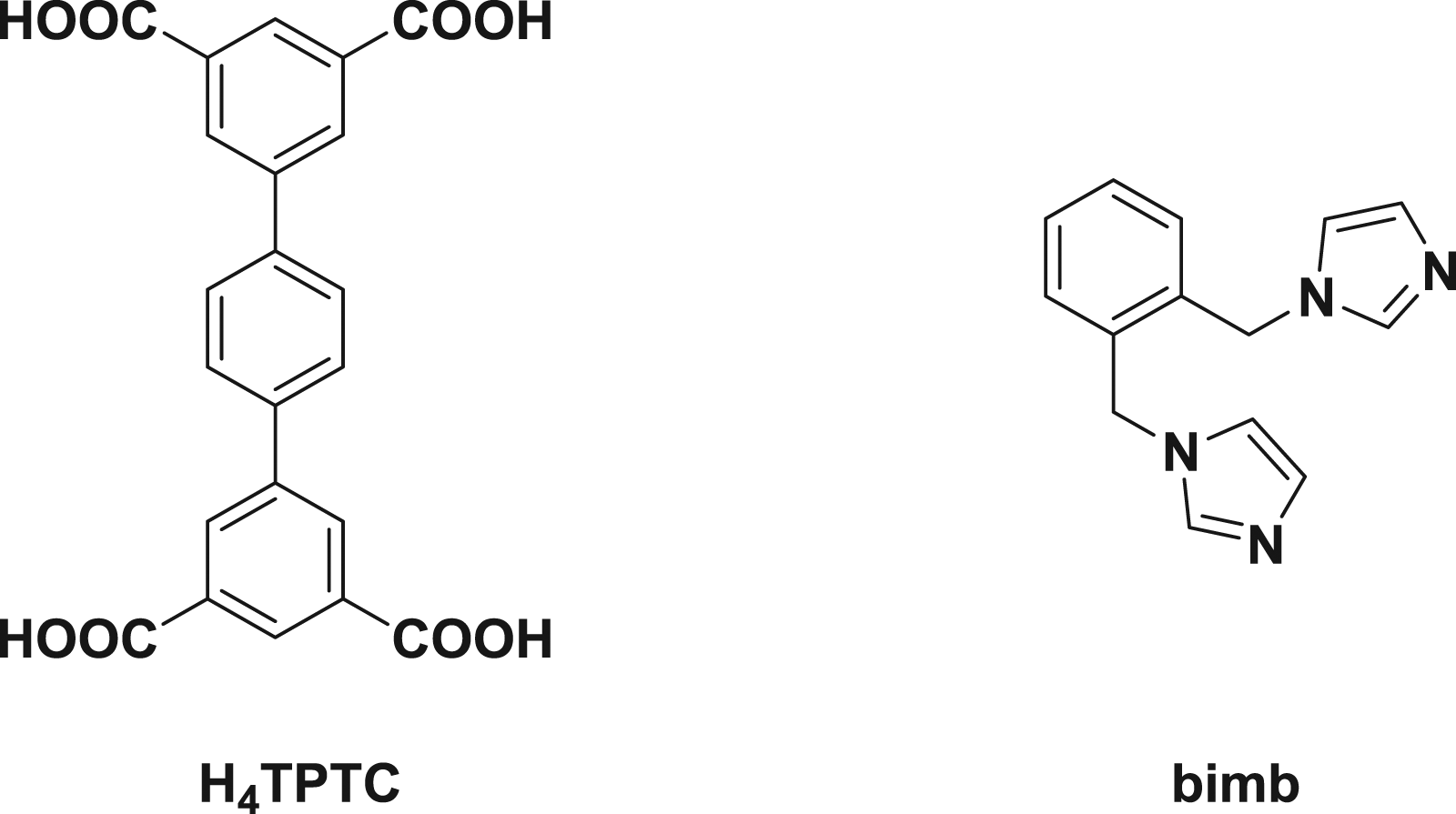
Experimental
Chemicals and measurements
All of the starting chemicals employed in our investigation could be acquired from the market, and they were exploited without processing. The FT-IR spectra could be performed utilizing KBr pellets and it was recorded through employing the spectrometer of Nicolet Impact 750 FTIR with the infrared spectra region form 400 cm−1 to 4000 cm−1. Through utilizing the analyzer of Perkin-Elmer 2400C, the analysis of Hydrogen, Nitrogen and Carbon elements was implemented.
Preparation and characterization for {[Zn(TPTC) (1,2-bimb)2]·4(H2O)}n (1) and {[Cu2(TPTC) (DMF)2(H2O)]·2DMF·2H2O} (2)
2.1 mg and 0.005 mmol of H4TPTC, 0.010 mmol and 2.4 mg of 1,2-bimb and 15 mg and 0.05 mmol of Zn(NO3)2·6H2O were dissolved into the mixed solution of CH3CN and H2O (4 mL, with 1:1 volume ratio). The product was kept in the reaction vessel and then it was heated for 72 hours to 130°C. The colorless needle crystals can be acquired after dropping to the environmental temperature slowly. (with 35% yield on the basis of Zn). Elemental analysis (%): calcd for the C50H47Zn2N8O12: N, 10.35%, C, 55.47% and H, 4.38%. Found: N, 10.68, C, 55.95 and H, 4.24%. IR (KBr pallet, cm−1): 3354 (s), 2836 (m), 1600 (s), 1551 (s), 1506 (m), 1490 (m), 1455 (s), 1433 (m), 1386 (m), 1366 (m), 1337 (s), 1312 (s), 1180 (w), 1149 (m), 1108 (s), 1008 (w), 947 (w), 907 (w), 843 (m), 769 (m), 737 (m), 707 (m), 687 (m), 613 (w), 598 (w), 562 (w), and 491 (m).
The mixture synthesized from 0.1 mmol and 0.025 g of terphenyl-3,3″,5,5″-tetracarboxylic acid, 0.048 g and 0.2 mmol of Cu(NO3)2·3H2O and 3 mL of DMF was stirred in the air for half an hour. The obtaining product was kept in the vial container (25 mL) and then this solution was heated for 3 days to 105°C. Subsequently, the mixture of reaction was dropped at 2°Ch−1 rate to environmental temperature. The complex
The complexes’ details of optimization and their parameters of crystallography.
Inflammatory cytokines determination
After the establishment of model and the treatment of compound, the inflammatory cytokines releasing into plasma in diverse groups was determined by utilizing the ELISA detection kit. This implementation was completed fully in accordance with the protocols of manufactures with minor change. Afterward, 40 BALB/c mice applied in this work were provided via the Shanghai Southern Model Biotechnology Co, Ltd and in this research, all of the conduction were granted through the Animal Ethics Committee of China. The Staphylococcus aureus was used to infect the animal to induce the postoperative infection. Then, the treatment was completed via utilizing the compounds with 5 mg/kg. The animal plasma was collected and the inflammatory cytokines content was detected.
Bacterial survival genes expression
The real time RT-PCR was further accomplished in the current investigation for the detection of the bacterial survival genes expression after the incubation of compound. This study was implemented strictly in the light of instructions with minor modifications. Shortly, the S. aureus were harvested and then they were inoculated into plates of 96 well, and the treatment was conducted after adding the compounds at 10 ng/mL, 20 ng/mL and 50 ng/mL concentration. Next, the S. aureus were harvested and in cells, the overall RNA could be extracted through utilizing the reagent of TRIZOL. After the determination of the entire RNA concentration, the concentration was reverse transcript into the cDNA. In the end, the real time RT-PCR was performed and the S. aureus survival genes relative expression was detected.
Results and discussion
Crystal structures
In accordance with the analysis of the diffraction of X-ray, the complex (a) The asymmetry unit for the compound 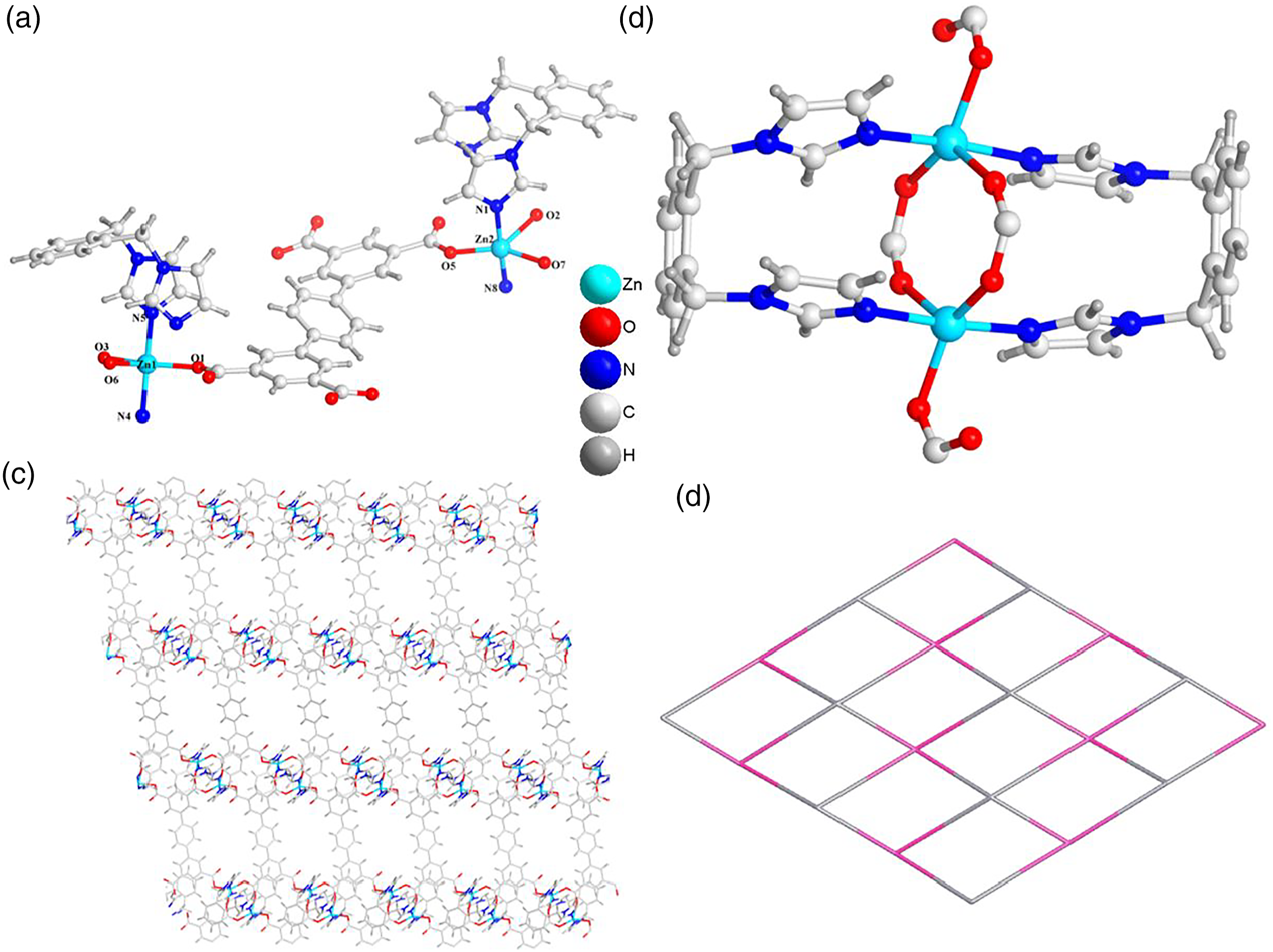
The complex (a) The asymmetry unit for the complex 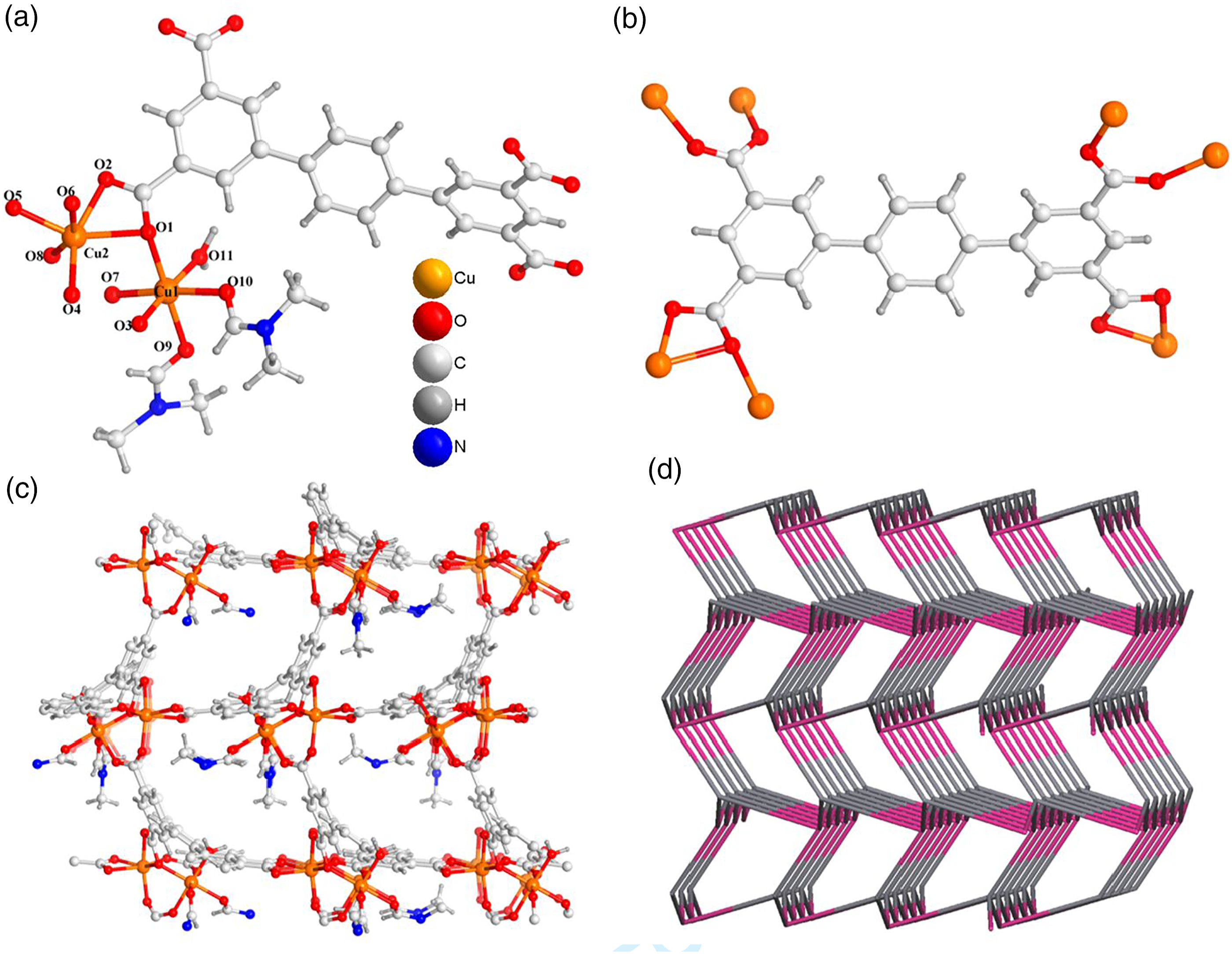
To check the phase purity of the products, powder X-ray diffraction (PXRD) experiments have been carried out for these complexes (Figure 3(a) and (b)). The peak positions of the experimental and simulated PXRD patterns are in good agreement with each other, indicating that the crystal structures are truly representative of the bulk crystal products. The differences in intensity may be owing to the preferred orientation of the crystal samples. The PXRD patterns for complex 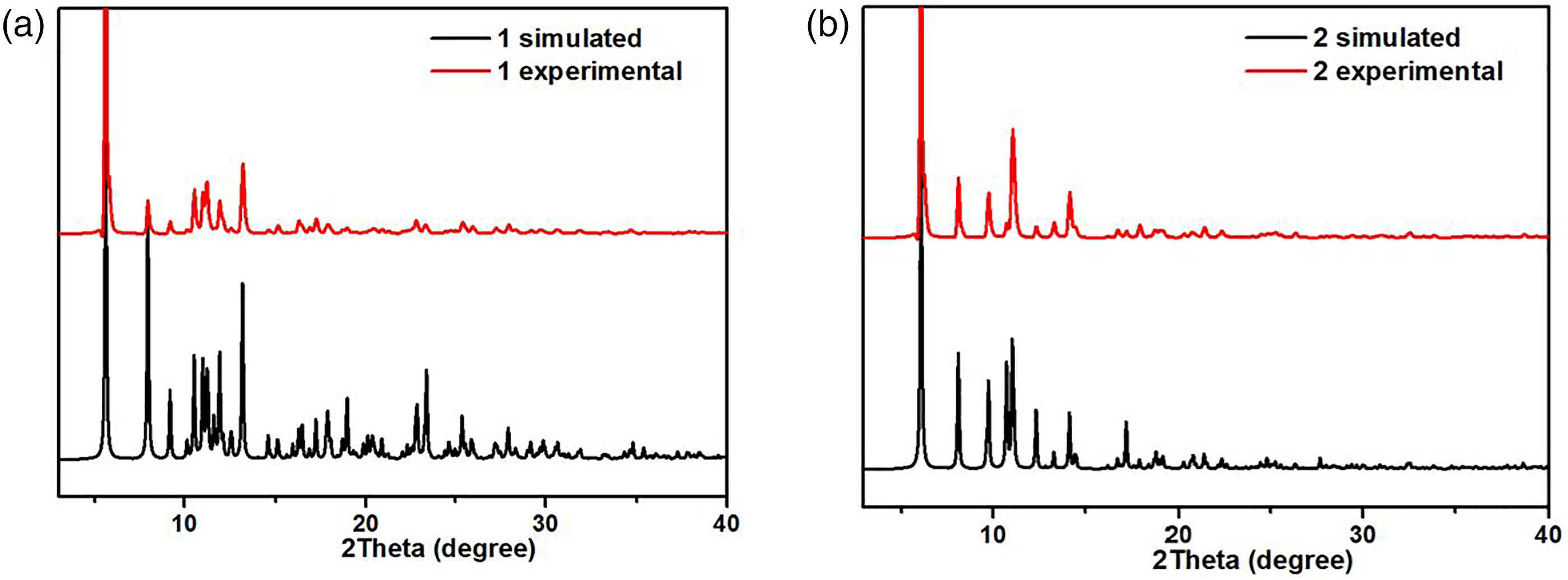
Compound significantly inhibited the releasing of the inflammatory cytokines into the plasma
The compounds Significantly inhibited inflammatory cytokines releasing into plasma after treating with the compound. The Staphylococcus aureus was used to infect the animal to induce the postoperative infection. Then, the treatment was completed by utilizing the compounds with 5 mg/kg. The animal plasma was harvested and the inflammatory cytokines content was determined.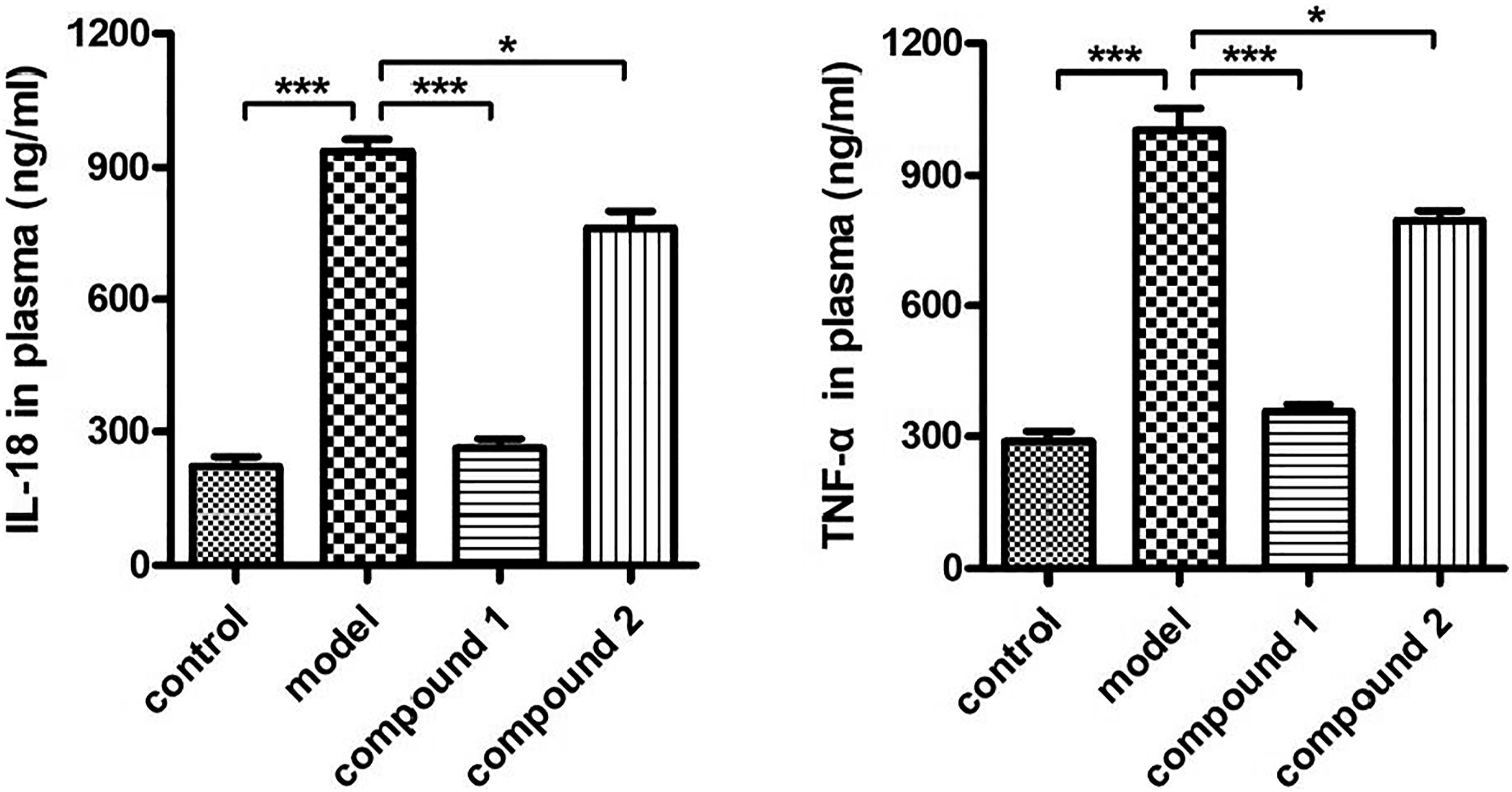
Compound obviously reduced the expression of the bacterial survival genes
As previous described, the Obviously reduced expression of the bacterial survival genes after compound treatment. The Staphylococcus aureus were collected and incubated with compounds 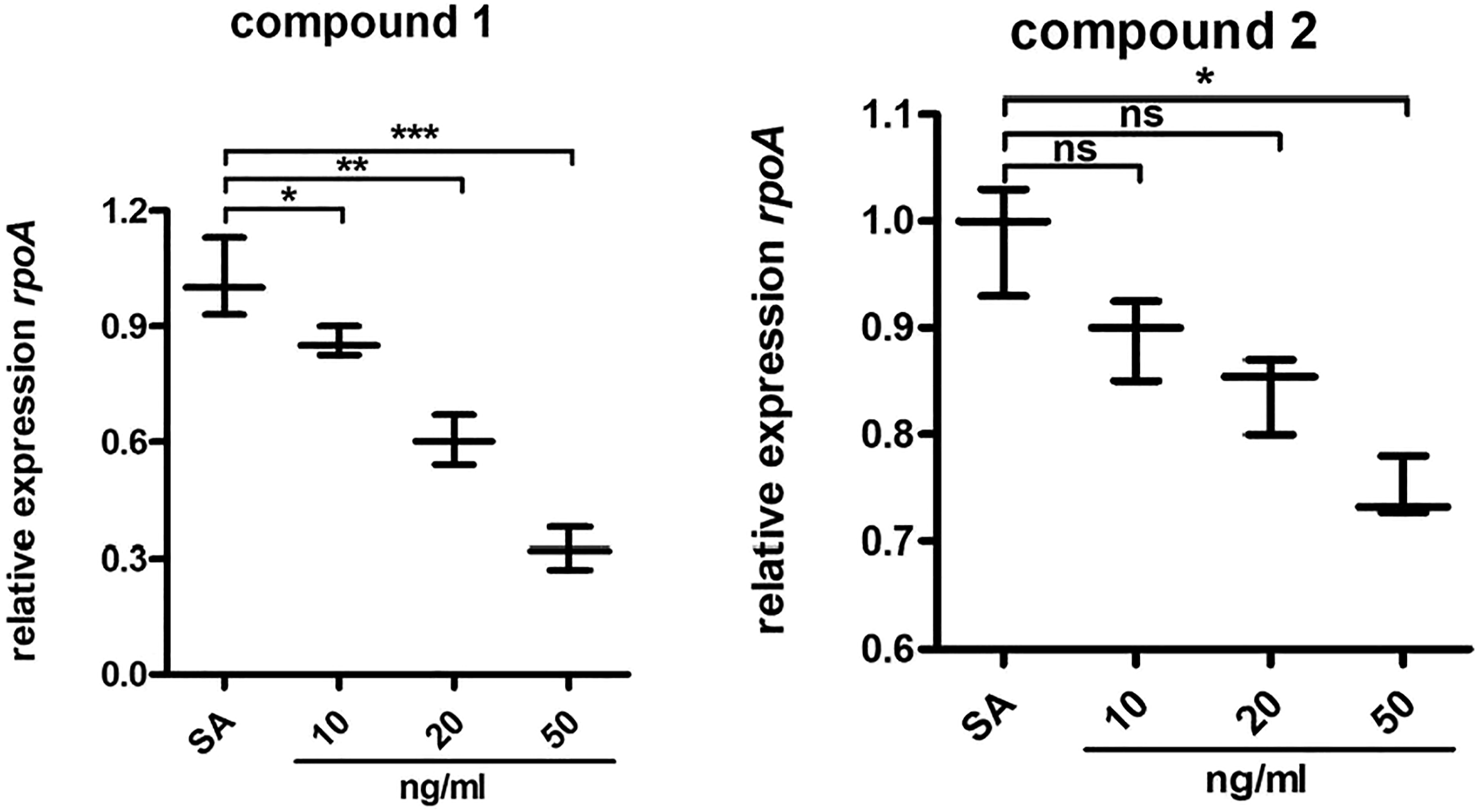
Conclusion
On the whole, we have produced two fresh transition metal coordination polymers triumphantly with the reaction between terphenyl-3.3″,5.5″-tetracarboxylic acid (H4L), the symmetrical rigid carboxylic acid ligand and the relevant metal salts under the reaction conditions of solvothermal. The analysis of structure suggest that the
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
