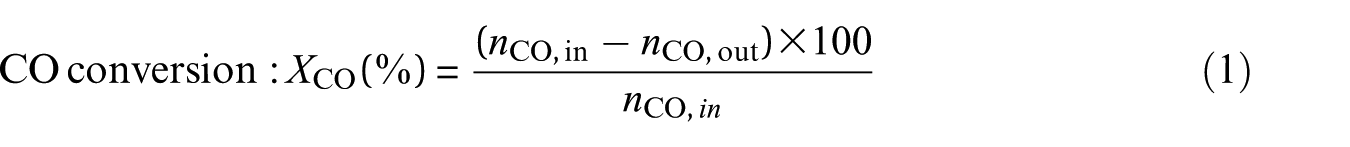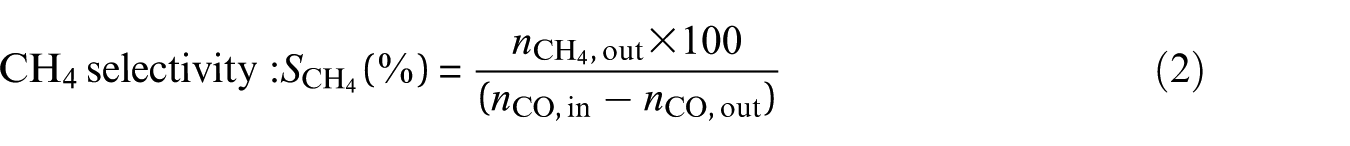Abstract
A series of Ni–Fe/MCM-41 bimetallic catalysts and also Ni/MCM-41 and Fe/MCM-41 catalysts were prepared by the incipient-wetness impregnation method and tested for their activity for CO methanation in a continuous-flow microreactor. The results showed that the catalytic activities of the Ni–Fe/MCM-41 bimetallic catalysts were much higher than those of the Ni/MCM-41 and Fe/MCM-41 catalysts at low temperatures (200°C–325°C). The 10%Ni–5%Fe/MCM-41 catalyst showed the best activity with a CO conversion of almost 100% and a CH4 selectivity of 98% at 350°C under a pressure of 1.5 MPa with a 3:1 molar ratio of H2 to CO and a weight hourly space velocity of 12,000 mL h−1 g−1. The catalysts were characterized by N2 physisorption measurements, X-ray diffraction, and H2-temperature-programmed reduction. The results showed that the addition of Fe will lead to the formation of finer Ni particles and Ni–Fe alloy, which were the main reasons for the activity increase in the Ni–Fe/MCM-41 catalysts.
Keywords
Introduction
Currently, oil and natural gas are used as raw materials for the production of fuel and chemicals by most countries. The reserves of petroleum and natural gas are expected to be available for only 40–60 years. 1 As one kind of clean, environment-friendly and efficient energy, natural gas is increasingly favored by the energy market. Therefore, development of alternative energy has become an urgent task in China and worldwide. 2 The energy structure characteristic of China is “rich in coal, lack of oil and natural gas,” and China has abundant biomass resources, such as coal. However, the utilization of coal and other biomass energy is simple and extensive in China, resulting in a great waste of carbon resources, and it also leads to environmental pollution problems, such as smog, the greenhouse effect, and so on. Coal and other biomass can be converted into clean energy with high quality, such as diesel, methanol, and hydrogen as well as synthetic natural gas (SNG), which is a more feasible way to produce an alternative to petrochemical energy. Due to its high conversion efficiency, mature synthetic conversion technology, and developed natural gas pipeline network, SNG has received increasing attention by research scholars. 3
As the most important step in the process of SNG preparation, methanation of syngas has attracted much attention. The methanation process is a strongly exothermic reaction, which easily leads to a rise in the transient temperature in the reaction system. The harsh reaction conditions of the traditional methanation process, such as high reaction pressure and high reaction temperature, lead to high cost of reaction equipment and sintering of traditional nickel-based methanation catalysts. 4 The development of new low-temperature methanation catalysts can effectively solve the problem of easy sintering of traditional methanation catalysts, and has important research significance.
Al2O3, SiO2, TiO2, and ZrO2 are the commonly used supports for methanation catalysts. 5 However, due to the harsh reaction conditions of methanation, the traditional support of Ni-based catalysts can easily lead to the sintering and deactivation of the catalyst. Mesoporous molecular sieve MCM-41 has excellent properties, such as a nanoscale pore structure, high specific surface area, and adjustable heteroatoms. MCM-41 is widely used as a hydrogenation catalyst, adsorbent, and carrier.6–9 Lensveld et al. 10 have successfully prepared a Ni-based MCM-41 catalyst with good dispersion by the incipient-wetness impregnation method. The Ni-based MCM-41 catalyst prepared by Wojcieszak et al. 11 showed good catalytic activity in the hydrogenation of benzene. Zhang et al. 12 prepared nickel-based catalysts with alumina, silica, and MCM-41 as carriers to catalyze the syngas methanation reaction. Thus, the 10%Ni/MCM-41 catalyst showed the best catalytic activity with a CO conversion of almost 100% at 350°C.
The addition of a second active component in a bimetallic catalyst can change the electron distribution and physical structure of the active component, leading to an improvement in the activity and selectivity of the catalyst.13,14 Wang et al. 15 prepared a Ni–Fe/Al2O3 catalyst and found that the addition of Fe could improve the dispersion of Ni and enhance the CO methanation activity of the catalyst. Bian et al. 16 found that the addition of Fe could lower the reduction temperature of the catalyst, resulting in the reduction of more active components and the improvement of catalyst activity. Ishihara et al. 17 found that the bimetallic catalyst showed higher catalytic activity in the Fischer–Tropsch reaction, and the formation of the alloy was an important reason for the improvement of its catalytic activity. Due to their fascinating properties, such as a large surface area and interconnected porous structures, metal/metal oxide functionalized mesoporous materials have received increasing attention, and Sahoo et al. 18 summarized the recent developments in design, preparation, and applications of transition metal/metal oxide–promoted MCM-41 for production of hydrogen energy and removal of aqueous pollutants.
In this article, a series of Ni–Fe bimetallic catalysts with mesoporous molecular sieve MCM-41 as the carrier and Ni and Fe as active components, and Ni/MCM-41 and Fe/MCM-41 catalysts were prepared by the incipient-wetness impregnation method. The CO methanation activities of the catalysts were evaluated in a continuous-flow fixed-bed reactor. The structural features and surface properties of the catalysts were studied by N2 physisorption measurements, X-ray diffraction (XRD), and H2-temperature-programmed reduction (H2-TPR), and the relationship between the structural features, surface properties of the catalysts, and their catalytic activities are discussed.
Experiment
Materials
Tetraethoxysilane (TEOS, AR), cetyltrimethylammonium bromide (CTAB, AR), nickel nitrate (Ni(NO3)2·6H2O, AR), and iron nitrate (Fe(NO3)3·9H2O, AR) were provided by Shanghai Lingfeng Chemical Reagents Co. Ltd. Carbon monoxide (CO, 99.9%), hydrogen (H2, 99.9%), nitrogen (N2, 99.9%), and argon (Ar, 99.9%) were provided by the Shijiazhuang Gas Supply Company. The experimental water was deionized water, which was provided by the Deionized Water Supply Center of the Shijiazhuang University of Applied Technology.
Preparation of catalyst
MCM-41 was synthesized with TEOS as the silicon source and CTAB as the template. The molar ratio was TEOS:CTAB:NaOH:H2O = 1:0.12:0.2:100. First, CTAB and NaOH were dissolved in deionized water at 40°C and TEOS was introduced to the solution dropwise with vigorous stirring. After stirring for 2 h, the solution was placed into a Teflon-lined reactor, maintained at 120°C for 24 h, cooled to room temperature and filtered. The residue was washed to neutral with deionized water and dried at 100°C to obtain MCM-41 raw powder. The raw powder was heated to 550°C at 1°C min−1 in a muffle furnace and calcined for 6 h to remove the template, to give the carrier MCM-41.
The carrier MCM-41 was impregnated with a measured solution of Ni(NO3)2·6H2O or Fe(NO3)3·9H2O, respectively, or a measured mixed solution of Ni(NO3)2·6H2O and Fe(NO3)3·9H2O by the incipient-wetness impregnation method at room temperature overnight to obtain the catalyst precursors. These were then dried in a vacuum oven at 60°C and calcined at 550°C for 4 h in air to obtain the catalysts. According to the Ni and Fe loadings of the catalysts, the catalysts were designated as 10%Ni–xFe/MCM-41, 10%Ni/MCM-41, and 10%Fe/MCM-41, respectively (where x = 1%, 3%, 5%, and 7% representing the molar content of iron). The active components were expressed in moles, with the total molar amount of catalyst as the benchmark.
Evaluation of catalysts
The catalytic experiments were performed in a fixed-bed reactor with a stainless steel tube (length: 30 cm and inner diameter: 8 mm). CO, H2, and N2 flows, controlled by mass flow controllers, were thoroughly mixed in a gas mixer before entering the fixed-bed reactor. For each experiment, 0.5 g of catalyst (sieve fraction 100 mesh) was placed in the middle of the tubular reactor and reduced in situ at 500°C for 2 h in a continuous flow of pure H2 (50 mL min−1). The mixed reactant gas consisting of H2/CO/N2 with a molar ratio of 3:1:1 (N2 was used as the internal standard) was fed into the reactor, and the weight hourly space velocity (WHSV) was selected as 12,000 mL h−1 g−1. The outlet gas was cooled by a cold trap at 5°C and analyzed online using a gas chromatograph (Techcomp, GC7900). The amounts of H2, N2, CH4, and CO in the gaseous products were analyzed using a thermal conductivity detector (TCD) with a TDX-01 column, while CO2, C2H4, C2H6, C3H6, and C3H8 were analyzed by another TCD with a Plot Q column. The outlet gases consisted of H2, N2, CO, CO2, CH4, H2O, and a negligible amount of C2H6 (<0.1 vol% in the outlet).
The experimental data for analysis were the averaged data of three repeated experiments. Conversion (XCO) of CO, selectivity (SCH4) for CH4, and yield (YCH4) of CH4 are defined as follows
where nCO,in is the number of moles of CO in the inlet gas, nCO,out is the number of moles of CO in the outlet gas, and nCH4,out is the number of moles of CH4 in the outlet gas.
Characterization of catalysts
Nitrogen adsorption–desorption isotherms of the catalysts were measured using an ASAP 2020 (Micromeritics) instrument, and the pore size distribution was calculated with the Barrett–Joyner–Halenda (BJH) method applied to the desorption branch. The specific surface area was calculated according to the Brunauer–Emmett–Teller (BET) method over the relative pressure (P/P0) range of 0.1–0.3. XRD patterns were recorded on a Rigaku Model D-MAX2500-PC instrument using CuKα radiation (λ = 1.54056 Å) operating at 50 kV and 100 mA. H2-TPR was carried out on a TP-5000-type multifunction adsorption instrument (Tianjin Xianquan) in a temperature range from room temperature to 900°C with a heating rate of 10°C min−1 in a binary gas flow (10 vol% H2/Ar) of 30 mL min−1, and the signal was recorded on a GC-4000A chromatograph together with a TCD.
Results and discussion
XRD analysis
The mesoporous structures of the MCM-41 carrier and the 10%Ni/MCM-41, 10%Fe/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts were characterized by small-angle XRD, and the results are shown in Figure 1. The small-angle XRD curves of the four samples showed (100), (110), and (200) characteristic diffraction peaks of MCM-41. 19 However, the characteristic diffraction peak intensities of the 10%Ni/MCM-41, 10%Fe/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts were weaker than those of MCM-41. This indicated that the long-range order of the MCM-41 carrier had been damaged to a certain extent.
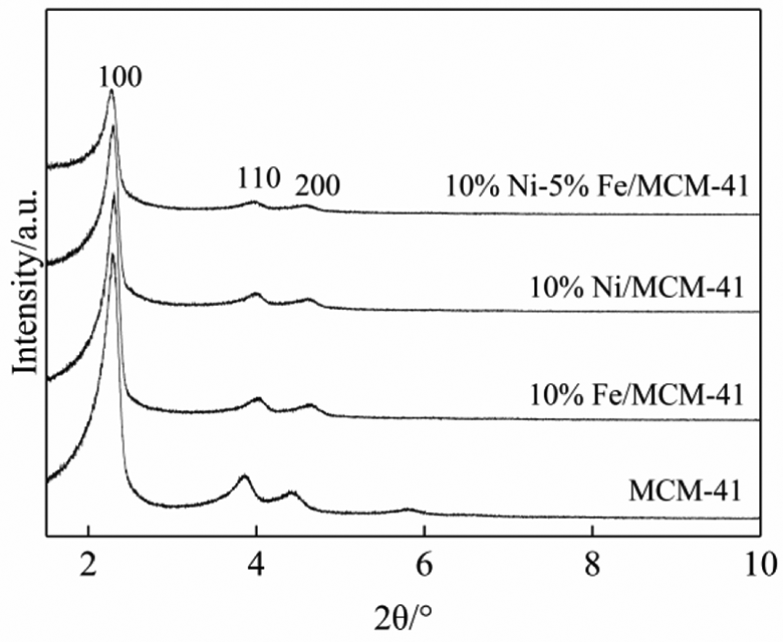
Small-angle XRD curves of 10%Ni/MCM-41, 10%Fe/MCM-41, 10%Ni–5%Fe/MCM-41 catalysts and MCM-41 carrier.
Figure 2 shows the wide-angle XRD curves of the 10%Ni/MCM-41, 10%Fe/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts after reduction for 2 h at 500°C with pure H2 (50 mL min−1). In the wide-angle diffraction curves of the reduced catalysts, the 10%Ni/MCM-41 and 10%Ni–5%Fe/MCM-41 catalysts showed obvious characteristic diffraction peaks of Ni particles (2θ = 44.28°, 52.52°, 76.84°), while no obvious characteristic diffraction peaks of NiO particles (2θ = 37.18°, 43.20°, 62.84°) could be observed. 19 This indicated that there were metallic Ni particles on the surfaces of the 10%Ni/MCM-41 and 10%Ni–5%Fe/MCM-41 catalysts. However, it was impossible to determine whether NiO particles existed on the catalysts’ surface. Meanwhile, the wide-angle XRD curve of the Fe/MCM-41 catalyst showed no obvious characteristic diffraction peaks of Fe particles, indicating that the high dispersion of the reduced metal Fe on the surface of the catalyst was as an amorphous form. 16 The particle size of Ni on the catalysts’ surface was calculated by Scherrer’s equation, and the results are shown in Table 1.
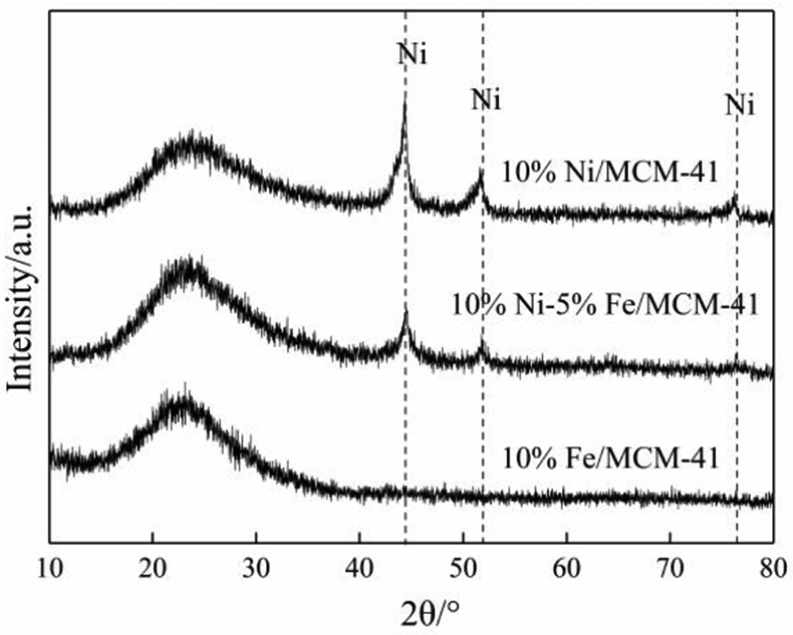
Wide-angle XRD spectra of 10%Ni/MCM-41, 10%Fe/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts.
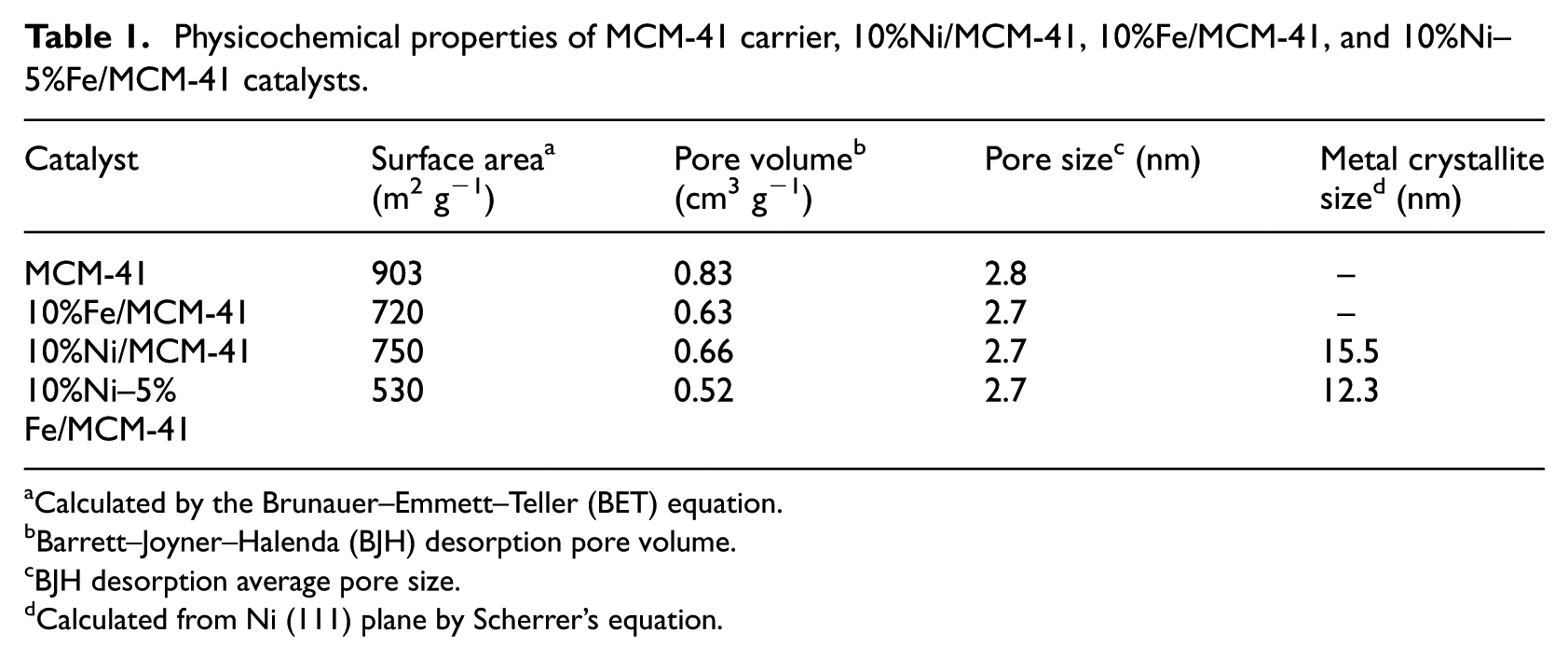
Physicochemical properties of MCM-41 carrier, 10%Ni/MCM-41, 10%Fe/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts.
Calculated by the Brunauer–Emmett–Teller (BET) equation.
Barrett–Joyner–Halenda (BJH) desorption pore volume.
BJH desorption average pore size.
Calculated from Ni (111) plane by Scherrer’s equation.
Physical properties of the catalysts
The physical properties of the MCM-41 carrier and the 10%Ni/MCM-41, 10%Fe/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts were characterized by N2 physical adsorption apparatus and the N2 physical adsorption–desorption isotherms are shown in Figure 3. The adsorption–desorption isotherm of MCM-41 was a typical type IV curve, and the sharp rise of adsorption isotherms reflected the regularity of the pore size of MCM-41. It can be seen from Figure 3 that the MCM-41 carrier and the 10%Ni/MCM-41, 10%Fe/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts all exhibited typical type IV isotherms, indicating that the MCM-41 carrier and the three catalysts were typical mesoporous materials. The detailed physical properties of the MCM-41 carrier and the 10%Ni/MCM-41, 10%Fe/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts are listed in Table 1.
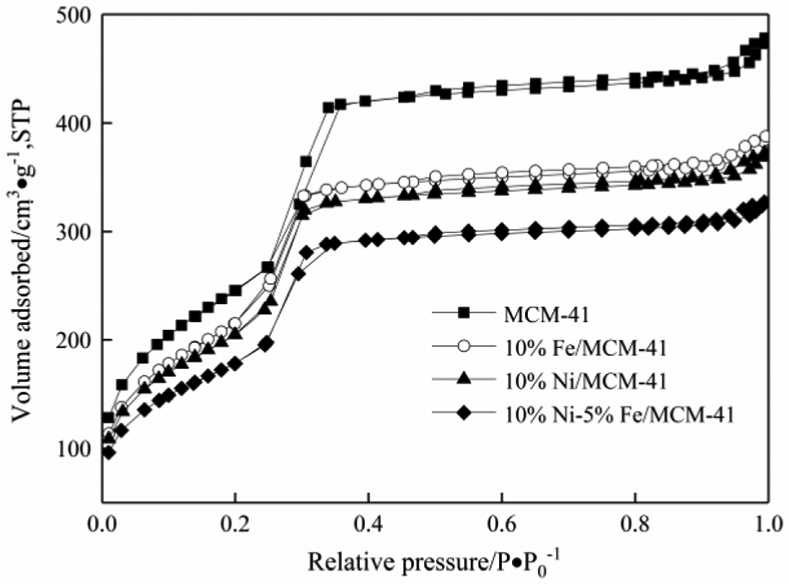
N2 adsorption–desorption curves of 10%Ni/MCM-41, 10%Fe/MCM-41, 10%Ni–5%Fe/MCM-41 catalysts and MCM-41 carrier.
From Table 1, the specific surface area of the MCM-41 carrier was 903 m2 g−1. The specific surface areas of the 10%Fe/MCM-41, 10%Ni/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts were reduced to 720, 750, and 530 m2 g−1, respectively, after the active metal components were loaded by the incipient-wetness impregnation method. This is due to the partial coverage of the surface and the partial blockage of the channel of the catalyst carrier by active components. The pore sizes of the catalysts showed no obvious change after the active metal component loading, while the pore volumes of the catalysts decreased with the active metal component loading, which was due to the partial blockage of the channel of the catalyst carrier by active metal components. As no obvious characteristic diffraction peaks of metallic Fe particles appeared in the XRD curve of the 10%Fe/MCM-41 catalyst, it was impossible to calculate the particle size of metallic Fe. The particle sizes of the active Ni components on the 10%Ni/MCM-41 and 10%Ni–5%Fe/MCM-41 catalysts by Scherrer’s equation were 15.5 and 12.3 nm, respectively, indicating that the addition of metal Fe could refine the Ni particles. This may be one of the reasons that the CO methanation activity of the 10%Ni–5%Fe/MCM-41 catalyst was higher than that of the 10%Ni/MCM-41 catalyst.
Reductive property of the catalysts
The reductive properties of the 10%Fe/MCM-41, 10%Ni/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts were characterized by H2-TPR analysis, and the results are shown in Figure 4. There were two reduction peaks in the H2-TPR curve of the 10%Ni/MCM-41 catalyst. The temperature ranges were 320°C–500°C and 550°C–800°C, respectively, which indicated that there were two different kinds of NiO particles on the surface of the 10%Ni/MCM-41 catalyst. The temperature range of the first reduction peak was basically the same as that of the pure NiO reduction peak. 12 Therefore, NiO particles on the surface of the 10%Ni/MCM-41 catalyst corresponding to the first reduction peak were pure NiO particles, which showed no interaction with the MCM-41 carrier. However, there was a strong interaction between the NiO particles corresponding to the second reduction peak and the MCM-41 carrier, which led to the difficult reduction of the 10%Ni/MCM-41 catalyst.
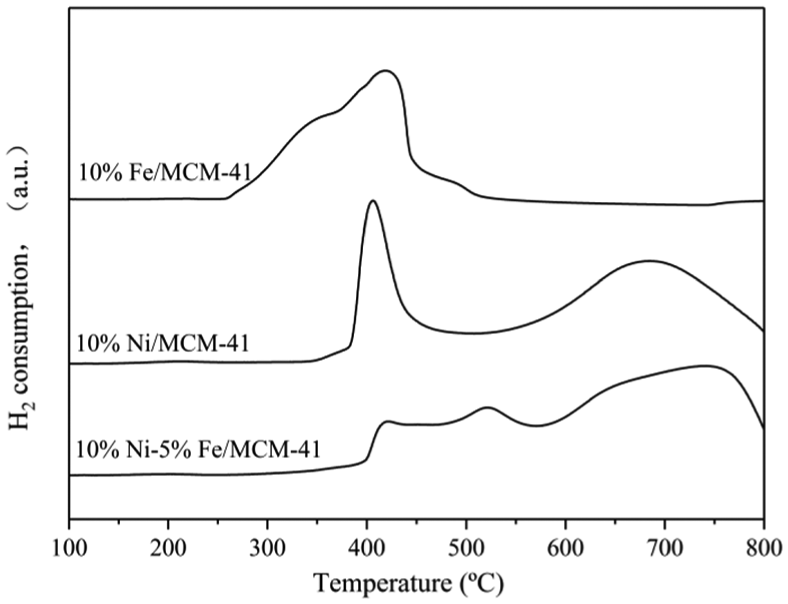
H2-TPR spectra of 10%Fe/MCM-41, 10%Ni/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts.
The H2-TPR curve of the 10%Fe/MCM-41 catalyst showed only one reduction peak with a temperature range of 250°C–500°C, indicating that there was only one kind of iron oxide species on the catalyst surface, and there was no obvious interaction between iron oxide species and the MCM-41 carrier. There were three reduction peaks appearing in the H2-TPR curve of the 10%Ni–5%Fe/MCM-41 catalyst. Compared with the 10%Ni/MCM-41 catalyst, there was one more reduction peak with a temperature range of 450°C–550°C, which indicated that Ni–Fe alloy was formed on the surface of the 10%Ni–5%Fe/MCM-41 catalyst. 20 The formation of Ni–Fe alloy was also one of the reasons for the great improvement of 10%Ni–5%Fe/MCM-41 catalyst activity. 21
Catalytic activity
Effect of Fe content
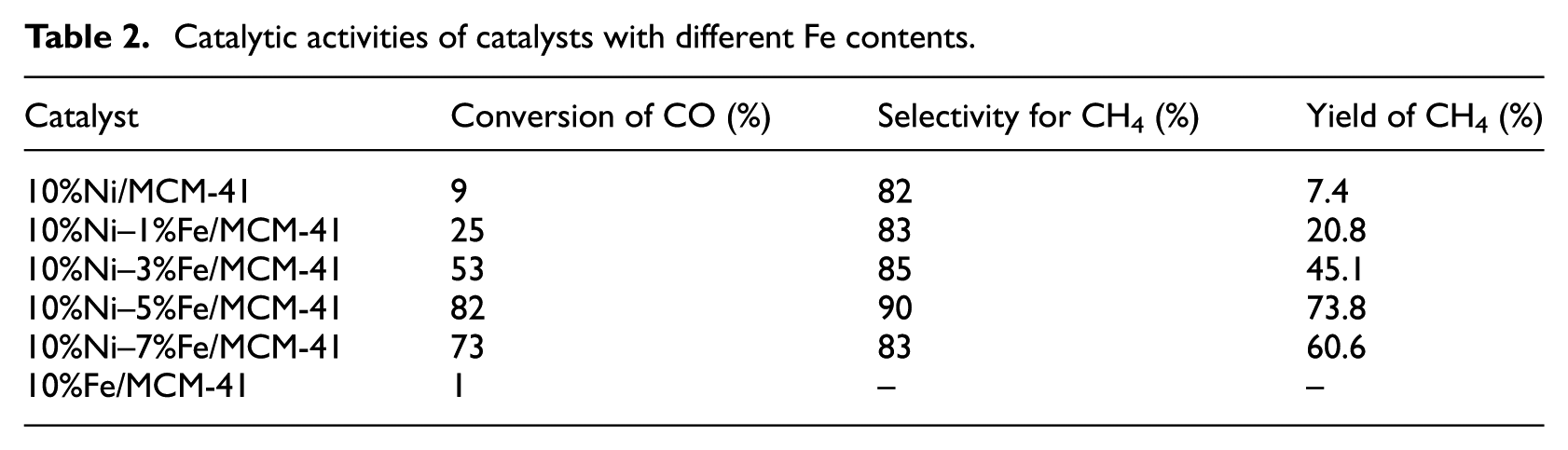
Table 2 shows the effect of Fe content on the catalytic activities of the Ni–Fe/MCM-41 catalysts at 300°C at atmospheric pressure. It can be seen from Table 2 that CO conversion and CH4 selectivity for the 10%Ni/MCM-41 catalyst were 9% and 82%, respectively. Under the same reaction conditions, the 10%Fe/MCM-41 catalyst showed almost no catalytic activity for CO methanation. The catalytic activities of the Ni–Fe/MCM-41 catalysts gradually increased with increasing Fe molar content, and the 10%Ni–5%Fe/MCM-41 catalyst showed the best catalytic activity with CO conversion and CH4 selectivity of 82% and 90%, respectively. Compared with the 10%Ni/MCM-41 catalyst, CO conversion and CH4 selectivity of the 10%Ni–5%Fe/MCM-41 catalyst increased by 811% and 10% respectively. However, compared with the 10%Ni–5%Fe/MCM-41 catalyst, the activity of the 10%Ni–7%Fe/MCM-41 catalyst decreased significantly with CO conversion and CH4 selectivity of 73% and 83%, respectively. This was probably due to partially active component Ni being covered by excessive metal Fe.
Catalytic activities of catalysts with different Fe contents.
Effect of reaction temperature
The CO methanation activities of the 10%Fe/MCM-41, 10%Ni/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts at different reaction temperatures were studied with reaction temperature ranging from 100°C to 450°C, and the results of activity evaluation are shown in Figure 5. It can be seen from Figure 5 that the initial temperatures of the methanation reaction for the 10%Ni–5%Fe/MCM-41, 10%Ni/MCM-41, and 10%Fe/MCM-41 catalysts were 125°C, 200°C, and 300°C, respectively. In the range of reaction temperature, the 10%Fe/MCM-41 catalyst showed very low activity with a CO conversion less than 5%. The catalytic activities of the 10%Ni/MCM-41 and 10%Ni–5%Fe/MCM-41 catalysts increased first and then decreased with the increase in reaction temperature.
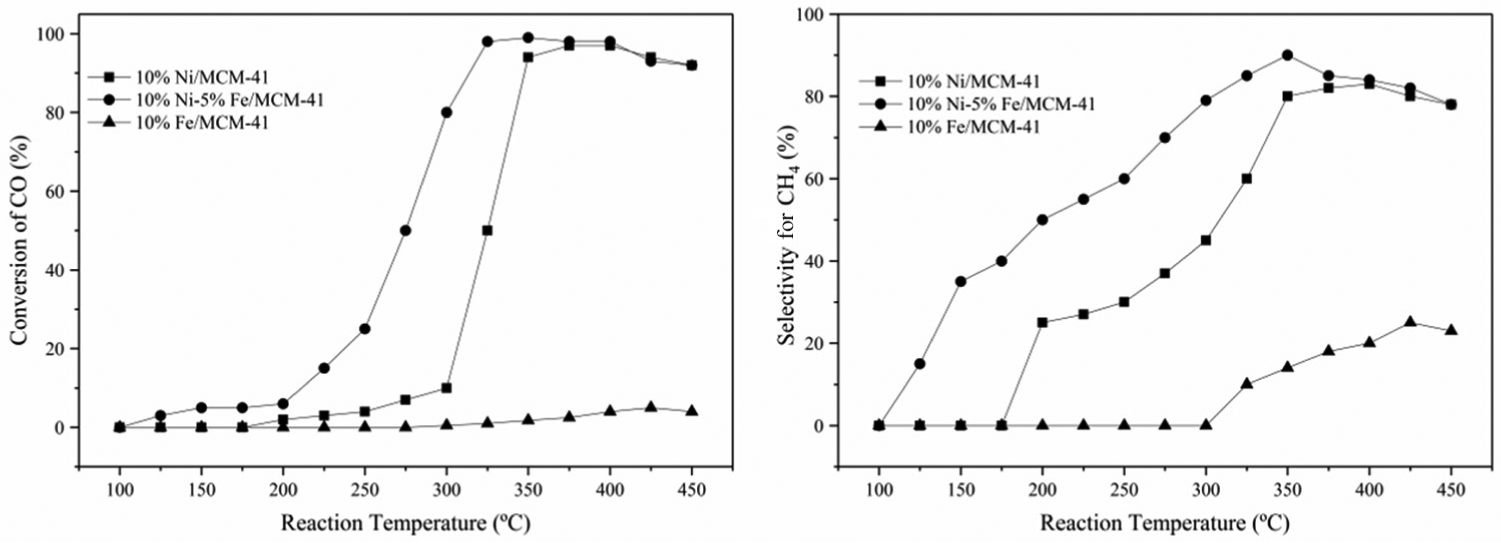
Effect of reaction temperature on activities of 10%Fe/MCM-41, 10%Ni/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts.
The addition of Fe promoter greatly enhanced the low-temperature activity of the 10%Ni/MCM-41 catalyst. CO conversion of the 10%Ni–5%Fe/MCM-41 catalyst reached more than 95% at 325°C, while that of the 10%Ni/MCM-41 catalyst was only about 50%. The addition of Fe increased the activity of the 10%Ni/MCM-41 catalyst by almost 100% at low temperature. However, the CO conversions of the 10%Ni–5%Fe/MCM-41 and 10%Ni/MCM-41 catalysts at 350°C were 99% and 92%, respectively. This indicated that the enhancement effect of Fe promoter on the activity of the Ni–Fe/MCM-41 catalyst at high temperature was gradually weakened. Under the same reaction conditions, the CO methanation activity of the Ni–Fe/MCM-41 catalysts was higher than those of the monometallic catalysts, and the order of catalytic activity was 10%Ni–5%Fe/MCM-41 > 10%Ni/MCM-41 > 10%Fe/MCM-41.
Effect of reaction pressure
Figure 6 shows the effect of reaction pressure on the catalytic activities of the 10%Fe/MCM-41, 10%Ni/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts at 350°C. It can be seen that the catalytic activities of all the catalysts increased with the increase in reaction pressure. For all the catalysts, the best catalytic activities were obtained at 1.5 MPa and kept steady at higher pressure. For the 10%Ni–5%Fe/MCM-41 catalyst, the CO conversion was maintained at almost 100%, and CH4 selectivity gradually increased with the increase in reaction pressure. With the increase in reaction pressure from atmospheric pressure to 2.0 MPa, CH4 selectivity increased from 90% to 98%. Because CO methanation is a reaction with a decreasing number of gas molecules, high pressure is beneficial to the positive reaction, resulting in the increase in CH4 selectivity.
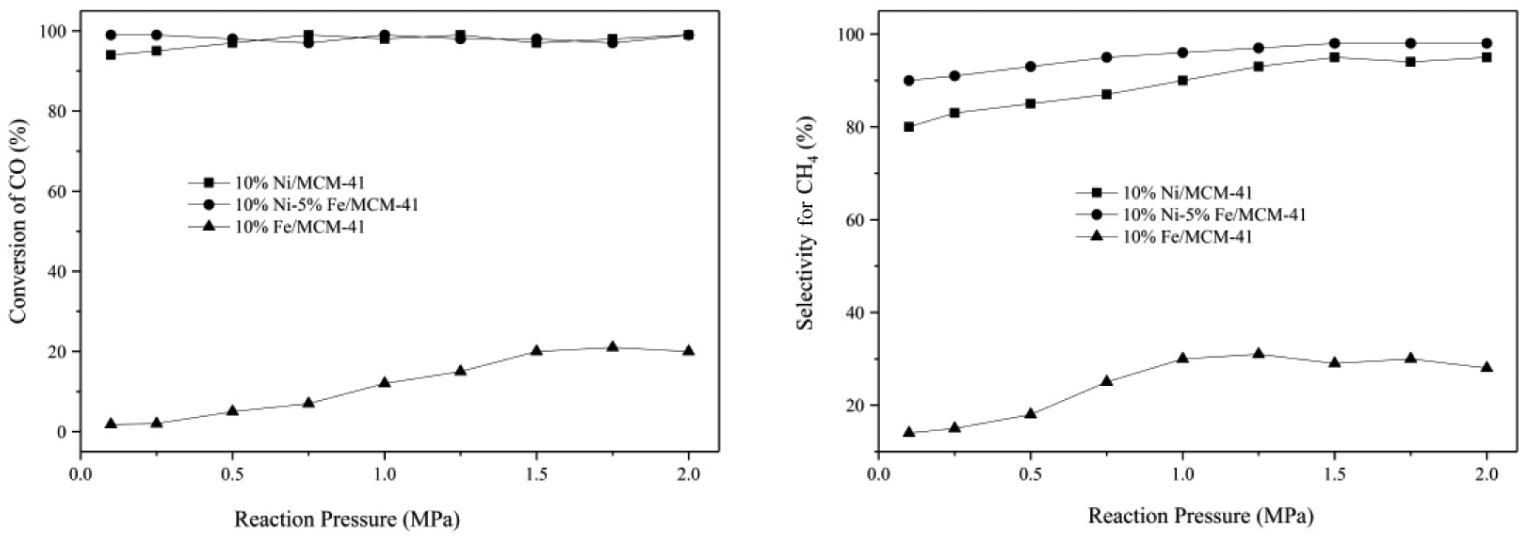
Effect of reaction pressure on activities of 10%Fe/MCM-41, 10%Ni/MCM-41, and 10%Ni–5%Fe/MCM-41 catalysts.
Conclusion
The catalytic activities of the Ni–Fe/MCM-41 bimetallic catalysts were much higher than those of the Ni/MCM-41 and Fe/MCM-41 monometallic catalysts at low temperatures (200°C–325°C). The addition of Fe made obvious interaction between active metal component Ni and the promoter Fe of bimetallic Ni–Fe/MCM-41 catalysts, and Ni–Fe alloy was formed during the reduction process. The addition of Fe could lead to the refinement of active component Ni particles on the surface of the catalysts, which was one of the main reasons for the good catalytic activity of bimetallic Ni–Fe/MCM-41 catalysts. The 10%Ni–5%Fe/MCM-41 catalyst showed the best activity with a CO conversion of almost 100% and a CH4 selectivity of 98% at 350°C under a pressure of 1.5 MPa with a 3:1 molar ratio of H2 to CO and a WHSV of 12,000 mL h−1 g−1.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support from the Doctoral Foundation of Shijiazhuang University of Applied Technology (15YB1001).